Abstract
Purpose of Review
Although the molecular mechanism of insulin resistance involves multiple factors and several intrinsic and extrinsic mechanisms have been identified, this comprehensive review provides key information on some of the core mechanisms and complex interactions of the molecules involved in the signaling pathways of insulin resistance.
Recent Findings
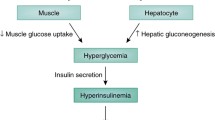
Diabetes Mellitus, the most common metabolic disorder, is one of the greatest global medical challenges at present. There has been a significant increase in complications associated with diabetes such as heart disorders, stroke, neuropathy, dyslipidemia, metabolic dysfunction-associated steatotic liver disease, and nephropathy. This calls for immediate strategic action to combat this complex metabolic disorder. Insulin resistance, a characteristic marker of type 2 diabetes is a condition in which the regulation of glucose metabolism in body tissues, such as the liver, adipose tissue, and skeletal muscle, becomes disrupted. It is generally associated with hyperglycemia, hyperinsulinemia, hyperlipidemia, and impaired glucose homeostasis.
Summary
Understanding the pathophysiological molecular mechanisms involved in insulin resistance is critical for developing new therapeutic strategies to treat this polygenic multifactorial condition. Impairment of insulin action is caused by several factors such as lipotoxicity, increased adiposity, enhanced inflammatory signaling, endoplasmic reticulum stress, adipokines, mitochondrial dysfunction, increased free fatty acids, and dysfunctional insulin signaling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The growing burden of diabetes, amplified by unhealthy eating habits, obesity, physical inactivity, and genetic mutations, poses the biggest challenge to the healthcare sector. In 2021, the prevalence of impaired glucose tolerance among adults aged 20–79 worldwide was around 9.1%. This figure is projected to increase by 10%, to around 638 million by 2045 [1••]. Due to the wide spectrum of signaling cascades, crosstalk, and complex molecular mechanisms involved in the progression of insulin resistance (IR), it is critical to gain a deeper understanding of the mechanistic insights to uncover novel, comprehensive therapeutic strategies to prevent and manage this multifaceted condition. Clinically, the term “insulin resistance” necessitates a higher concentration of insulin to maintain normal glucose levels. On a cellular level, it is defined as the insufficient strength of insulin signaling from the insulin receptor (INSR) downstream to the final substrates of insulin action involved in various metabolic aspects of cellular function and crosstalk [2•].
The regulation of glucose homeostasis is accomplished through the production of insulin by beta cells in response to increased blood glucose levels after food intake. The driving force behind this homeostasis is the insulin sensitivity of various organs, including the liver, skeletal muscle, and adipose tissue. A tightly regulated cascade of insulin-dependent signaling controls multiple metabolic processes, such as reducing glucose production by the liver, increasing glucose uptake by adipose tissue and skeletal muscle, and suppressing the release of free fatty acid (FFA) release from adipocytes. Insulin actions are carefully regulated to ensure proper metabolic function and maintain energy balance [3]. IR, which is characterized by a dysregulation in any of these metabolic processes and a failure of target cells to respond to normal levels of circulating insulin, is marked by both intrinsic and extrinsic cell signaling pathways. The intrinsic pathway controls mitochondrial function and ER stress, while the extrinsic mechanisms impact adipokines, FFAs, and inflammation in the metabolic tissues [4]. The diversity of physiological insulin responses in the liver, white adipose tissue, and skeletal muscle is largely due to distinct distal effectors, while the proximal components involved in insulin signal transduction are similar in all insulin-responsive cells.
A widely accepted notion is that obesity, which involved the accumulation of toxic metabolic by-products and increased FFAs, directly contributes to IR. However, recent studies revealed other mechanisms that also play a role. These include the activation of inflammatory pathways and intracellular stress response pathways, altered metabolic functions caused by lipid accumulation and lipid-derived metabolites, metabolic overload in muscle, mitochondrial stress, and dysfunction.
This review provides essential information and an overview of the role that these core mechanisms play in the development of IR. Additionally, it presents a unified framework for comprehending this intricate network of signaling axes.
Normal Insulin Action and Signaling
To better understand the complex mechanisms and interactions involved in IR, it is essential to shed light on normal insulin action and the insulin signaling mechanism. In a fasted state, the liver secretes glucose into the bloodstream to maintain euglycemia and provide fuel for glucose-consuming tissues. The hepatic glucose production involves glycogenolysis and the de novo synthesis of glucose, in a process known as gluconeogenesis [5]. After food intake, insulin secretion by the pancreatic cells suppresses catabolic programs and stimulates skeletal muscle and adipose tissue to take up glucose. Insulin secretion also promotes glycogen and lipid synthesis in the liver, skeletal muscle, and adipose tissue by suppressing hepatic glucose production. This is achieved by decreasing the expression of genes involved in gluconeogenesis and lipolysis in adipose tissue [6].
Insulin receptor tyrosine kinase (IRTK) mediates the intracellular functions of insulin. When insulin binds to the extracellular domain of IRTK, it induces a conformational change and leads to autophosphorylation of tyrosine residues in IRTK. This activation of IRTK results in the activation of insulin receptor substrate (IRS), growth factor receptor-bound protein (GRB) 2, GRB10, and SHC, which are phosphotyrosine binding proteins [7], which are known to play critical regulatory roles. For example, GRB10 phosphorylation and stabilization by mTORC1, activated by insulin signaling, provide feedback inhibition of INSR activity. Other substrates, such as the SH2B adapter protein 2 (SH2B2), also known as adapter protein with a pleckstrin homology and a Src homology 2 domain (APS), help initiate the metabolic insulin response in certain cell types [8, 9]. The proximal insulin signaling pathway consists of two major arms: the mitogenic arm (initiated by GRB2 and SHC) and the metabolic arm (initiated by IRS proteins and SH2B2/APS). The characteristic positive feedback mechanism of insulin action involves the Gα-interacting vesicle-associated protein (GIV) potentiating of PI3K/AKT signaling and phosphatase inhibition by NAD(P)H oxidase 4. Negative feedback mechanisms involve the stabilization and recruitment of GRB10 to INSR and the activation of S6K1 to phosphorylate and inhibit IRS proteins [10].
To summarize, the proximal signaling events include IR activation and phosphorylation of signaling proteins, particularly IRS, PI3K, and AKT isoforms. These events are highly conserved in insulin target tissues and initiate the insulin response at the plasma membrane. This phosphorylation forms the basis for substrate association with downstream signaling proteins, leading to three pathways. The PI3K-dependent pathway mediates glucose/lipid/protein metabolism and insulin-stimulated glucose uptake. The CAP/Cb1 pathway is necessary for GLUT4 translocation, and the MAP kinase pathway controls cell proliferation and differentiation. The phosphorylation of IRS, induced by IRTK, mediates the effects of insulin action on glucose and lipid metabolism by recruiting PI3K and catalyzing the production of PIP3 from PIP2. AKT is then activated by PKD1 and the mechanistic target of rapamycin complex 2 (mTORC2) after being recruited to the plasma membrane by PIP3. AKT activation is followed by the phosphorylation of various downstream substrates in skeletal muscle, adipose tissue, and the liver, enabling insulin-mediated nutrient reservation in these tissues [11].
In skeletal muscle, the primary substrate for INSR is IRS1. INSR activation in skeletal muscle has two major metabolic functions: glucose uptake and glycogen storage. Insulin stimulates the translocation of GLUT4-containing vesicles to the plasma membrane, which is important for glucose uptake and leads to increased production of intracellular glucose 6-phosphate. Additionally, the dephosphorylation of glycogen metabolic proteins promoted by insulin enables net glycogen synthesis. The core event occurring in skeletal muscle IR is the increase in glucose transport, facilitated by the anchoring and fusion of the plasma membrane with GLUT4-containing vesicles. This process is mediated by the GTPase-activating protein AKT substrate. Furthermore, the promotion of the GTP-bound form of RAS-related C3 botulinum toxin substrate 1 (RAC1) is also involved in this event. The insulin stimulation of glycogen synthesis is achieved through the inhibition of glycogen kinase synthase 3 (GSK3), which subsequently activates glycogen synthase and deactivates glycogen phosphorylase [9, 12, 13].
The main mechanism involved in hepatic insulin signaling includes AKT signaling, which activates the machinery responsible for glycogen and protein synthesis. Other transcriptionally mediated effects include upregulation of glucokinase, reduction of gluconeogenesis, and stimulation of de novo lipogenesis capacity [14]. In the liver, AKT decreases gluconeogenesis by suppressing of forkhead box O1 (FOXO1)-mediated expression of gluconeogenic genes. Hepatic glycogen synthesis is increased through regulation of GYS2 and glycogen phosphorylase via GSK3 and protein phosphatase 1 [15, 16].
The key somatic function of insulin action in white adipose tissue is to inhibit lipolysis. This inhibition requires phosphodiesterase 3B (PDE3B), which weakens cAMP-stimulated PLIN and hormone-sensitive lipase phosphorylation. Insulin stimulates the uptake of glucose through two pathways: one insulin receptor, which is PI3K dependent, and a second insulin receptor, which is PI3K independent. The PI3K-independent pathway uses several effectors to regulate the translocation and fusion of glucose transporter storage vesicles and the plasma membrane [17]. Additionally, insulin promotes lipogenesis in white adipose tissue by activating SREBP1c, which stimulates the esterification of fatty acids and enhances the translocation of glucose or fatty acid transport proteins [18, 19].
Collectively, members of the IRS family play a major role in controlling metabolic fuel homeostasis. IRS1 serves as a key mediator of insulin-mediated glucose uptake and activation of anabolic pathways in muscle and adipose tissue. In contrast, the liver is primarily influenced by IRS2 [20]. The insulin signaling cascade can be regulated by various factors, including the reversal of IRS phosphorylation by Tyr-phosphatase 1β and the inhibition of tyrosine kinase activity by the suppressor of cytokine signaling (SOCS)1 and SOCS2 [20, 21•]. Understanding these core elements of the insulin signaling cascade, the complex network of phosphorylation, and their modulation could lead to improved therapies. Figure 1 provides an overview of proximal insulin signaling and some of the most important effects associated with it.
The insulin signaling cascade. When insulin binds to its receptor, it leads to autophosphorylation and recruitment of insulin receptor substrates (IRS) that become phosphorylated. This, in turn, enhances the phosphoinositide-3-kinase (PI3K)-AKT signaling cascade, which is crucial for glucose metabolism. It promotes glycolysis and prevents gluconeogenesis. AKT activity is further regulated by protein phosphatase 2A (PP2A), which is activated by ceramides. The levels of ceramides increase due to mitochondrial dysfunction, decreased lipid oxidation, and accumulation. These factors also cause oxidative stress and the production of reactive oxygen species (ROS). This further impacts the insulin cascade, leading to mitophagy and apoptosis. Additionally, elevated lipids result in higher levels of diacylglycerols (DAG), which excessively activate protein kinase C (PKC) and inhibit insulin signaling, causing induce insulin resistance
Upon insulin binding, the insulin receptor undergoes autophosphorylion, which subsequently leads to the recruitment of multiple substrates. There are major branches of insulin signaling: mitogenic (signaling, which is initiated by GRB2 and SHC), and metabolic (signaling, which is initiated by IRS proteins and SH2B2/APS). Insulin signaling also involves feedback through Gα-interacting vesicle-associated protein (GIV), which enhances phosphoinositide-3-kinase (PI3K)-AKT signaling, and inhibition of phosphatase by NAD(P)H oxidase 4 (NOX4)-derived H2O2. Additionally, there is a negative feedback through the stabilization and recruitment of GRB10 to the INSR, as well as the activation of S6 kinase 1 (S6K1) to phosphorylate and inhibit IRS proteins.
Mechanisms of Insulin Resistance
Defects in Insulin Signaling and Insulin Resistance
The proximal insulin signaling abnormalities are rarely associated with IR in most forms of type 2 diabetes mellitus. It has been discovered that only 2.4% of the total INSRs are necessary for a complete biological response in insulin-binding experiments of rat adipocytes [22]. However, defects at the receptor or post-receptor level can lead to the inactivation of insulin during circulation. Over 60 mutations have been identified in the INSR gene, with type A INSR being associated with a heterozygous mutation state that results in decreased tyrosine phosphorylation of the β subunit after insulin binding [23]. Abnormal production of anti-INSR antibodies can also cause this [24]. Another finding showed decreased INSR activity in hyperinsulinemic ob/ob mice, which was due to a decreased tyrosine phosphorylation state of IRS1 [25].
Dysregulation of insulin at the post-receptor level has recently gained attention. An increased TNFα level causes an increase in Ser/Thr phosphorylation of IRS1, which is a crucial factor in IR. Additionally, hyperinsulinemia also affects the concentration of IRS2, causing downstream changes in insulin signaling [26]. Interestingly, glycated proteins resulting from hyperglycemia have been shown to decrease intracellular PI3K, PKB, and GSK-3 activity, thus contributing to IR. Similarly, glucosamine, produced from glucose which is a major substrate for cellular glycosylation, enhances IRS1 glycosylation, reducing its activity and insulin responsiveness [26]. Among the three isoforms of AKT, only defects in AKT2 and AKT3 are associated with impaired insulin-stimulated glucose transport in IR, not AKT1 [27, 28].
Insulin Resistance in Skeletal Muscle
The skeletal muscle plays an important role in insulin-stimulated glucose consumption, which means that muscular IR can have various effects in overall body metabolism [29]. Early studies on the skeletal muscle of obese and diabetic individuals showed a decrease in surface INSR content and a decrease in the activity of purified receptors [30••].
Multiple studies have found that impaired translocation of GLUT4 protein is responsible for IR in skeletal muscle [31]. Additionally, defects in proximal insulin signaling, specifically in the activities of IRTK, IRS1, PI3K, and AKT, could also contribute to IR in skeletal muscle. This is supported by the reduced activity in the skeletal muscle of obese/diabetic mice [32]. In 1963, Randle et al. proposed that lipid-induced IR in skeletal muscle is caused by a decrease in glucose utilization due to an increase in fatty acid oxidation. This increase in fatty acid oxidation leads to an accumulation of G6P and intramyocellular glucose, as well as a decrease in glucose uptake [33]. Some models have challenged the relevance of the glucose-fatty acid cycle in lipid-induced IR. For example, one study showed that the glucose-fatty acid cycle is inoperative in pyruvate dehydrogenase kinase 2/4 (Pdk2/4)-double knockout mice, as these mice preferentially oxidize glucose in an inflexible manner [34]. However, Randle’s proposal is still physiologically relevant for the control of oxidative substrate selection and can be seen as a cell-autonomous response to the overall regulation of substrate availability, promoting efficient glucose oxidation in the fed state and fatty acid oxidation in the fasted state [35]. For instance, in lipid infusion models, there is an increase in intramyocellular G6P concentration and a decrease in glycolytic flux, followed by the development of significant IR after 3 h of acute lipid infusion. This aligns with Randle’s glucose-fatty acid hypothesis [36].
Insulin Resistance in Liver and Adipose Tissue
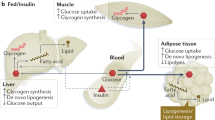
The liver controls the post-prandial glucose level by suppressing hepatic glucose production and promoting the storage of glucose as glycogen [37•]. In individuals with type 2 diabetes mellitus, the main cause of fasting hyperglycemia is increased hepatic gluconeogenesis. This is primarily linked to impaired lipolysis in adipose tissue and the failure to suppress the FOXO1 transcription factor in the liver [37•]. Additionally, IR is associated with defects in the ability of insulin to stimulate glycogen synthesis. It is important to note that hepatic IR reduced the extent of hepatic glycogen metabolism during fasting and feeding [38]. Similarly, to the liver and muscle, individuals with type 2 diabetes mellitus experience diminished tyrosine kinase activity. This, along with a decrease in plasma membrane INSR content, leads to IR in the adipose tissue [39]. Figure 2 illustrates the inter-organ communication between muscle, liver, adipose tissue, pancreas, gut, and adipose tissue in obesity-induced IR that leads to ectopic lipid storage in different organs [40].
Inter-organ communication in obesity-induced insulin resistance. Lipid overload and overnutrition provoke an increased uptake of triacylglycerol (TAG) through chylomicron triacylglycerol particles (CM-TAG) from the gut. If the adipose tissue’s absorption and storage capacity is overwhelmed, elevated TAG is then transferred into different organs, such as the liver, muscles, and pancreas, via very low-density lipoprotein-TAG and the lipoprotein lipase (LPL). Additionally, this leads to higher concentrations of circulating non-esterified fatty acids (NEFA) and triacylglycerol. In the long term, impaired lipid oxidation leads to fat accumulation in different organs, the development of insulin resistance, and significantly higher levels of circulating glucose
Ectopic Lipid Accumulation
As suggested by numerous studies, ectopic fat accumulation in peripheral tissues such as the liver and skeletal muscle can lead to severe IR. It has been demonstrated that leptin treatment or moderate weight loss can dramatically reverse hepatic IR in patient with type 2 diabetes mellitus and non-alcoholic fatty liver disease [41]. Furthermore, additional evidence suggests that the knockout of fat transport proteins, such as CD36, increases insulin-mediated glucose uptake in skeletal muscle, while liver-specific knockdown of FATP2 reduces hepatic lipid accumulation and improves glucose tolerance [42, 43].
Several lipid metabolites such as diacylglycerol, lysophosphatidic acid, ceramides, and acylcarnitine are involved in the ectopic lipid accumulation-induced IR in the liver and skeletal muscle [44]. This popular theory suggests that lipid-induced hepatic IR occurs due to the accumulation of lipids caused by impaired fatty acid oxidation. This impairment leads to the redirection of long-chain acyl CoA molecules into endoplasmic reticulum (ER)-localized and cytosolic lipid species, such as diacylglycerols, ceramides, and triglycerides. In the liver and skeletal muscle, elevated levels of diacylglycerol trigger the movement of nuclear protein kinase C (PKC) to the plasma membrane. This, in turn, hinders IRTK activity by phosphorylating Thr1160, ultimately leading to the deactivation of IRS2, PI3K, and AKT2 [45]. Numerous studies provide evidence supporting the diacylglycerol hypothesis of IR. For instance, an early study revealed increased PKC levels in the soleus muscles of diabetic rats and livers of obese rats [46].
Ceramides have been shown to mediate lipid-induced IR, but the exact mechanism has not yet been demonstrated. Ceramide is a crucial bioactive sphingolipid that is produced from an intracellular fatty acid and sphingosine [47]. In one study, it was found that liver-specific knockout of Cers6 led to a decrease in hepatic ceramide levels, which protected against high-fat diet-induced obesity and improved glucose tolerance [48]. Although the precise molecular mechanisms of ceramide-induced IR are still unclear, a few theories have been proposed. One theory suggests that ceramide negatively regulates insulin action by inhibiting AKT/PKB, a serine/threonine kinase that plays a central role in glucose uptake and metabolism [49]. Ectopic fat accumulation in obesity is primarily caused by the action of inflammatory chemokines and adipocytokines from macrophages and other cells that infiltrate the adipose tissue [50•]. Figure 3provides a brief illustration of the pathophysiology behind ectopic fat accumulation that is promoted by adipocyte dysfunction, IR in the muscle, heart, and liver, and elevated secretion of insulin and β-cell apoptosis.
Adipocyte dysfunction in ectopic fat accumulation and associated consequences. Elevated fat uptake leads to adipocyte hypertrophy, which in turn increases the number of infiltrating macrophages that release inflammatory adipokines. Over time, this dysfunction in adipose tissue impairs the uptake of free fatty acid (FFA). The liver, muscles, heart, and pancreas then take up higher concentrations of FFA. Ectopic fat induced in these organs is associated with insulin resistance, mitochondrial dysfunction, and key features typical of type 2 diabetes. Examples include increased secretion of VLDL, hepatic steatosis, and the development of non-alcoholic fatty liver disease (NAFLD)
Role of Inflammation in Insulin Resistance
The most important contributor to obesity-induced IR is the systemic chronic inflammatory response caused by altered cytokine production and activation of inflammatory signaling pathways [51]. In obesity, there is an increase in fat accumulation in the adipose tissue, leading to larger adipocyte sizes, expansion of adipose tissue, and changes in pro-inflammatory cytokines. Additionally, obesity leads to an increase in the infiltration of macrophages, which contributes to cytokine production [51]. The inflammatory response is connected to IR in two ways. Firstly, the stimulation of inflammatory signaling intermediates directly leads to serine phosphorylation of IRS1 in hepatocytes and myocytes, inducing IR. Secondly, within the adipose tissue, inflammatory cell infiltration may alter adipocyte lipid metabolism, with TNF-α promoting lipolysis and changes in cytokine production, such as IL-6 [52]. This is supported by studies showing reduced IR in rodents and humans when TNF-α is knocked out [53]. The role of TNF-α in IR is mediated by JNK1 which phosphorylates serine 307 of IRS1 [54].
Strikingly, it has been reported that the heterogeneous deletion of another inflammatory signaling intermediate IKK-β alleviates IR in obese rodents on a high-fat diet [55]. The trademark identification of the inflammatory serine kinase involved in IR is Jun kinase 1. Improved insulin sensitivity has been demonstrated in JNK1 knockout mice and increased JNK activity has been reported in obese rodents and humans [56].
A fascinating study suggests that SOCS3, which participates in negative feedback loops in cytokine signaling, is reported to be increased in obese rodents [57]. In the same context, in vitro studies suggest that SOCS3 directly interacts with the INSR, inhibiting IRS1 tyrosine phosphorylation and decreasing insulin-stimulated glycogen synthesis in cultured myotubes [58]. In rodents on a high-fat diet, it has been observed that there is an increase in macrophage infiltration of adipose tissue and expression of MCP-1, which recruits monocytes to sites of injury. This supports the fact that IR is due to infiltrating immune cells and changes in cytokine production by the liver in response to chronic lipid exposure [59, 60].
It is worth noting that the pathophysiological adipose tissue microenvironment induces a significantly increased expression of miR-210-3p. This miRNA promotes NF-κB activation-dependent proinflammatory cytokine expression and down-regulates anti-inflammatory cytokines [61••]. In the respective study, the authors demonstrated that miR-210-3p binds to the 3′-UTR of SOCS1 mRNA, leading to the silencing its expression. As a result, the proteosomal degradation of NF-κB is prevented, supporting the notion that specific miRNAs could be targeted to interfere with obesity-induced type 2 diabetes [61••]. Interestingly, several other miRNAs, including miR-126, miR-222-3p, miR-182, let-7b-5p, and miR-1-3p, showed reduced expression in samples taken from patients with type 2 diabetes. On the other hand, miRNAs such as miR-21, miR-30d, miR-148a-3p, miR-146b, and miR-486 showed increased expression [62•]. These findings suggest that several miRNAs have the potential to impact the pathogenesis of type 2 diabetes.
Adipokines and Insulin Resistance
Adipocytes play a major role in the development of IR due to their capacity to store excess lipids in obese individuals. This storage leads to an abnormal redistribution of lipids to other organs and tissues [63]. Pro-inflammatory factors such as IL-6, TNF-α, ROS, and hypoxia reduce plasma adiponectin levels in obese and diabetic animal models [64]. Adiponectin activates AMPK and PPAR-α signaling through Adiponectin receptors 1 and 2, respectively. This promotes enhanced fatty acid oxidation and glucose uptake in muscle, while suppressing gluconeogenesis in liver tissues. Therefore, the deletion of adipoR1 induces IR by increasing endogenous glucose production, and the deletion of Adiponectin receptor 2 induces IR by decreasing PPAR-α signaling pathway activity [65••].
A milestone discovery was made regarding the mutated gene “leptin” found in ob/ob mice, which exhibit hyperlipidemia and IR. Another discovery was made regarding adiponectin, which modulates glucose homeostasis and cell insulin sensitivity. Both leptin and adiponectin are categorized as “anti-diabetogenic” due to their capability to decrease triglyceride synthesis and activate beta-oxidation. This leads to enhanced insulin action in the liver and skeletal muscle by activating 5′-AMP-activated protein kinase [66].
It is worth noting that adiponectin levels are decreased, and leptin levels are increased in obese animals and humans with IR. This suggests that obesity leads to deficiency in adiponectin deficient and resistance to leptin [67]. Additionally, leptin has been found to play a crucial role in rescuing insulin action in lipodystrophic mouse models, improving IR in these mice [68]. Leptin binds to the leptin receptor (LepRb) and activates the JAK/STAT3 pathway, directly affecting insulin action [69]. Leptin and insulin both play important roles in regulating glucose homeostasis. Leptin limits insulin secretion and increases insulin sensitivity, while also decreasing hepatic glucose production. In turn, insulin stimulates the production of leptin in adipose tissue [70]. Interestingly, high levels of leptin associated with IR promote the release of pro-inflammatory components such as IL-6, TNF-α, and IL-12 by monocytes and macrophages [71].
Interestingly, recent data emphasizes the role of dysfunctional circulating mediators such as resistin in the onset of obesity, IR, and cardiovascular diseases [72]. The genes responsible for regulating lipid and glucose homeostasis are controlled by chemerin, a highly expressed chemokine in the liver and white adipose tissue. The regulatory functions of this chemokine include IRS1 tyrosine phosphorylation, enhancing GLUT4 expression, fatty acid synthase, and adiponectin, ultimately leading to insulin sensitivity in adipose tissue [73]. In summary, adipose tissue acts as a central hub for various adipokines and bioactive lipid mediators in the molecular mechanisms involved in the pathophysiology of IR.
The Indispensable Role of Mitochondrial Dysfunction in Insulin Resistance
Multiple studies have shown that IR can stem from mitochondrial dysfunction. One study suggests that the loss of mitochondrial function due to aging causes the accumulation of intramyocellular lipids, resulting in IR in elderly subjects [74]. In the same context, young individuals who are offspring of parents with type 2 diabetes were found to have a 60% increase in intramyocellular content, a 38% reduction in mitochondrial density, and a 50% increase in serine phosphorylation of IRS1 [75]. These pieces of evidence collectively suggest that severe mitochondrial dysfunction is linked to IR and diabetes.
Recently, it has been suggested that chronic exposure of skeletal muscle to lipids increases the expression of genes involved in the fatty acid β-oxidation pathway. Interestingly, there is a lack of coordination between the upregulation of β-oxidation enzymatic machinery and downstream metabolic pathways such as the tricarboxylic acid cycle and the electron transport chain. This lack of coordination leads to the incomplete metabolism of fatty acids in β-oxidation pathways and the accumulation of lipid metabolites in mitochondria [76].
This is evidenced by increased rates of incomplete fatty acid oxidation in isolated mitochondria of insulin-resistant rat skeletal muscle and accumulation of several long- and medium-chain acylcarnitine in the muscle of obese rats compared to lean rats [77]. Furthermore, insulin sensitivity and glucose tolerance were restored in association with increased tricarboxylic acid cycle activity in these animals on a high-fat diet through exercise intervention [77].
Another phenomenon known as “metabolic inflexibility” in isolated and whole-body muscle mitochondria demonstrates that obesity causes impaired switching from fatty acid to carbohydrate substrates during the fasting-fed transition and a decrease in several tricarboxylic acid cycle intermediates [78]. In the case of muscle-specific IR, mitochondrial dysfunction is characterized by metabolic abnormalities and diminished oxidative phosphorylation, decreased electron transport chain activity, and low expression levels of PGC1α, a master transcriptional regulator of mitochondrial biogenesis [79].
Another interesting finding is the relevance of reduced coenzyme Q associated with IR in humans. Coenzyme Q is a crucial component of the electron transport chain, responsible for transferring electrons from complex 1 or 2 to complex 3. Reduced coenzyme Q leads to reductive stress in complexes 1 and 2, resulting in increased production of reactive oxygen species and exacerbating IR [80]. In summary, mitochondrial dysfunction leads to the accumulation of FFAs and lipids, promotion formation of diacylglycerols, ceramide, and reactive oxygen species (ROS), and mitochondrial damage, ultimately inhibiting insulin signaling and inducing the development of IR [81].
Endoplasmic Reticulum Stress and Insulin Resistance
Several interesting mechanisms explain the development of IR in obese or dyslipidemic individuals. One such mechanism is ER stress, which is enhanced in obesity and causing IR in the liver and pancreatic β-cells [80]. In the scenario of metabolic overload or overnutrition, the liver produces an excess of enzymes to process these, which stimulates the unfolded protein response (UPR) by accumulating unfolded proteins in the ER [82••]. This is supported by a finding that showed oral administration of chemical chaperones like 4-phenyl butyric acid alleviates ER stress and increases insulin signaling and sensitivity in ob/ob mice [83].
Similarly, pancreatic β-cells develop ER stress in chronic hyperglycemic states [84]. In diet-induced obesity models, elevated ER stress markers in the liver and adipose tissue are linked to activation of c-Jun-N terminal kinase (JNKs), which phosphorylates IRS1 and interferes with insulin action. Overexpression of XBP1, a downstream transcription factor modulating the URP, prevents JNK activation in liver cells treated with agents that cause ER stress. Additionally, XBP1 deletion increased susceptibility of mice to diet-induced IR. This IR is partly controlled by induction of the ER stress response, which connects to core insulin signaling pathways [80]. In conjunction, ER stress has emerged as a key player in IR and could be a potential therapeutic target for diabetes.
Increased Concentration of Free Fatty Acids
Several studies have established associations between IR and the increased availability of FFAs resulting from obesity. In obese individuals, adipose tissue releases higher levels of TNF-α, which triggers the secretion of FFA’s into circulation [85]. TNFα mediates the repression of multiple genes involved in glucose and FFA uptake and storage. For example, in 3T3-L1 adipocytes, it has been shown that TNF-α enhances lipolysis, leading to increased release of FFAs and cytokines. Additionally, TNF-α-dependent gene upregulation or downregulation occurs through obligatory activation of NF-κB [86]. In muscle tissue, TNF-α and FFAs impair insulin signaling by promoting increased NADH/NAD+ and acetyl-CoA/CoA ratios. Furthermore, β-oxidation is insufficient to effectively remove these elevated fatty acids from circulation, resulting in the storage of excess FFAs as triglyceride droplets in the muscle [87]. In adipose tissue, FFAs inhibit the lipoprotein lipase, leading to impaired clearance of FFAs from circulation. The excess FFAs are then transported as triglycerides by VLDL to non-adipose tissues [88].
A significant relationship exists between glucocorticoids and FFAs. Glucocorticoids impair peripheral glucose uptake and increase the availability of FFAs. This, in turn, reduces glucose oxidation and uptake by peripheral tissues. Glucocorticoids directly enhance hormone-sensitive lipase, inhibit lipoprotein lipase, and increase the conversion of noradrenaline. These actions promote lipolysis in adipose tissue [89]. In summary, it is widely accepted that the increase in FFAs caused by obesity leads to IR and hyperglycemia.
In summary, diabetes mellitus is a chronic disorder that affects over 170 million people worldwide. It is projected to affect over 365 million people by 2030 [90]. Additionally, the global prevalence of diabetes is expected to increase to 700 million by 2045, making it a global health emergency [91•]. There is growing evidence suggesting that several mechanisms, such as ectopic lipid accumulation in peripheral tissues, are strongly associated with IR. Other factors include ER stress, dysregulation of adipokine, and inflammation. These mechanistic pathways involved in IR often run parallel to each other, meaning that the involvement of one mediator does not exclude the involvement of another. Therefore, a multidisciplinary strategy should be developed to treat this complex condition, targeting both physiological and metabolic impairments.
Conclusions
The major target tissues of insulin are skeletal muscle, the liver, and adipose tissue. It has been shown that increased plasma FFAs reduce glucose uptake, thereby decreasing plasma lipids and improving insulin activity in skeletal muscle cells, adipocytes, and the liver.
Insulin controls glucose homeostasis by stimulating glucose uptake into peripheral tissues and suppressing the release of stored lipids from adipose tissue.
Impairment in the secretion or action of insulin leads to IR, which results in a loss of metabolic control, dysregulated glucose homeostasis, and increased circulating lipids.
Several factors contribute to IR including the activation of inflammatory pathways, ER stress, mitochondrial dysfunction, and the accumulation of by-products in insulin-sensing tissues due to nutritional overload.
In the liver, increased glucose levels and excess lipids activate serine kinases, leading to the phosphorylation and inactivation of key insulin signaling molecules.
In muscle tissue, excess lipids accumulate due to incomplete metabolism in the mitochondria, resulting in impaired insulin signaling.
Given the diversity of molecular mechanisms involved in the development of IR, a holistic approach is necessary. This approach should consider several factors such as overnutrition, the inflammatory response, and energy imbalance, to effectively combat this complex condition.
Abbreviations
- ER:
-
Endoplasmic reticulum
- FFA(s):
-
Free fatty acid(s)
- INSR:
-
Insulin receptor
- IR:
-
Insulin resistance
- IRS:
-
Insulin receptor substrate
- IRTK:
-
Insulin receptor tyrosine kinase
- SOCS:
-
Suppressor of cytokine signaling
- UPR:
-
Unfolded protein response
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
•• Rooney MR, Fang M, Ogurtsova K, Ozkan B, Echouffo-Tcheugui JB, Boyko EJ, et al. Global prevalence of prediabetes. Diabetes Care 2023;46:1388-94. https://doi.org/10.2337/dc22-2376. This study presents accurate information on the worldwide, regional, and national rates of prediabetes. The data is derived from studies that report impaired glucose tolerance from 43 countries and impaired fasting glucose from 40 countries.
• Kosmas CE, Bousvarou MD, Kostara CE, Papakonstantinou EJ, Salamou E, Guzman E. Insulin resistance and cardiovascular disease. J Int Med Res 2023; 51:3000605231164548. This review summarizes the current scientific and clinical evidence pertaining to insulin resistance and the mechanisms connecting insulin resistance with cardivascular disease.
Himsworth H. Diabetes mellitus: its differentiation into insulin-sensitive and insulin-insensitive types. Int J Epidemiol. 2013;42:1594–8. https://doi.org/10.1093/ije/dyt203.
Odegaard JI, Chawla A. Pleiotropic actions of insulin resistance and inflammation in metabolic homeostasis. Science. 2013;339:172–7. https://doi.org/10.1126/science.1230721.
Rizza RA. Pathogenesis of fasting and postprandial hyperglycemia in type 2 diabetes: implications for therapy. Diabetes. 2010;59:2697–707. https://doi.org/10.2337/db10-1032.
Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev. 2018;98:2133–223. https://doi.org/10.1152/physrev.00063.2017.
Youngren JF. Regulation of insulin receptor function. Cell Mol Life Sci. 2007;64:873–91. https://doi.org/10.1007/s00018-007-6359-9.
Hsu PP, Kang SA, Rameseder J, Zhang Y, Ottina KA, Lim D, et al. The mTOR-regulated phosphoproteome reveals a mechanism of mTORC1-mediated inhibition of growth factor signaling. Science. 2011;332:1317–22. https://doi.org/10.1126/science.1199498.
Leto D, Saltiel AR. Regulation of glucose transport by insulin: traffic control of GLUT4. Nat Rev Mol Cell Biol. 2012;13:383–96. https://doi.org/10.1038/nrm3351.
Pessin JE, Saltiel AR. Signaling pathways in insulin action: molecular targets of insulin resistance. J Clin Invest. 2000;106:165–9. https://doi.org/10.1172/JCI10582.
Chang L, Chiang SH, Saltiel AR. Insulin signaling and the regulation of glucose transport. Mol Med. 2004;10:65–71. https://doi.org/10.2119/2005-00029.Saltiel.
Chiu TT, Jensen TE, Sylow L, Richter EA, Klip A. Rac1 signalling towards GLUT4/glucose uptake in skeletal muscle. Cell Signal. 2011;23:1546–54. https://doi.org/10.1016/j.cellsig.2011.05.022.
Agius L. Role of glycogen phosphorylase in liver glycogen metabolism. Mol Aspects Med. 2015;46:34–45. https://doi.org/10.1016/j.mam.2015.09.002.
Leavens KF, Birnbaum MJ. Insulin signaling to hepatic lipid metabolism in health and disease. Crit Rev Biochem Mol Biol. 2011;46:200–15. https://doi.org/10.3109/10409238.2011.562481.
Dong XC, Copps KD, Guo S, Li Y, Kollipara R, DePinho RA, et al. Inactivation of hepatic Foxo1 by insulin signaling is required for adaptive nutrient homeostasis and endocrine growth regulation. Cell Metab. 2008;8:65–76. https://doi.org/10.1016/j.cmet.2008.06.006.
Ros S, García-Rocha M, Domínguez J, Ferrer JC, Guinovart JJ. Control of liver glycogen synthase activity and intracellular distribution by phosphorylation. J Biol Chem. 2009;284:6370–8. https://doi.org/10.1074/jbc.M808576200.
Zechner R. FAT FLUX: enzymes, regulators, and pathophysiology of intracellular lipolysis. EMBO Mol Med. 2015;7:359–62. https://doi.org/10.15252/emmm.201404846.
Choi YH, Park S, Hockman S, Zmuda-Trzebiatowska E, Svennelid F, Haluzik M, et al. Alterations in regulation of energy homeostasis in cyclic nucleotide phosphodiesterase 3B-null mice. J Clin Invest. 2006;116:3240–51. https://doi.org/10.1172/JCI24867.
Kersten S. Mechanisms of nutritional and hormonal regulation of lipogenesis. EMBO Rep. 2001;2:282–6. https://doi.org/10.1093/embo-reports/kve071.
Cohen P. The twentieth century struggle to decipher insulin signalling. Nat Rev Mol Cell Biol. 2006;7:867–73. https://doi.org/10.1038/nrm2043.
• White MF, Kahn CR. Insulin action at a molecular level - 100 years of progress. Mol Metab 2021;52:101304. This review provides information on both cell-extrinsic and cell-intrinsic factors that contribute to insulin resistance in type 2 diabetes mellitus.
Copps KD, White MF. Regulation of insulin sensitivity by serine/threonine phosphorylation of insulin receptor substrate proteins IRS1 and IRS2. Diabetologia. 2012;55:2565–82. https://doi.org/10.1007/s00125-012-2644-8.
McIntyre EA, Walker M. Genetics of type 2 diabetes and insulin resistance: knowledge from human studies. Clin Endocrinol (Oxf). 2002;57:303–11. https://doi.org/10.1046/j.1365-2265.2002.01610.x.
O’Rahilly S. Insights into obesity and insulin resistance from the study of extreme human phenotypes. Eur J Endocrinol. 2002;147:435–41. https://doi.org/10.1530/eje.0.1470435.
Lei C, Wang J, Li X, Mao YY, Yan JQ. Changes of insulin receptors in high fat and high glucose diet mice with insulin resistance. Adipocyte. 2023;12:2264444. https://doi.org/10.1080/21623945.2023.2264444.
Pirola L, Johnston AM, Van Obberghen E. Modulation of insulin action. Diabetologia. 2004;47:170–84. https://doi.org/10.1007/s00125-003-1313-3.
Brozinick JT, Roberts BR, Dohm GL. Defective signaling through Akt-2 and -3 but not Akt-1 in insulin-resistant human skeletal muscle: potential role in insulin resistance. Diabetes. 2003;52:935–41. https://doi.org/10.2337/diabetes.52.4.935.
Batista T, Haider N, Kahn C. Defining the underlying defect in insulin action in type 2 diabetes. Diabetologia. 2021;64:994–1006. https://doi.org/10.1007/s00125-021-05415-5.
DeFronzo RA, Tripathy D. Skeletal muscle insulin resistance is the primary defect in type 2 diabetes. Diabetes Care. 2009;32:S157–63. https://doi.org/10.2337/dc09-S302.
•• Wang Y Zhou H, Palyha O, Mu J. Restoration of insulin receptor improves diabetic phenotype in T2DM mice. JCI Insight 2019;4:e124945. https://doi.org/10.1172/jci.insight.124945. This study demonstrates that a single-dose adeno-associated virus vector, driving hepatic expression of human insulin receptor, significantly improves the diabetic phenotype in inducible insulin receptor defcient mice.
Kim JK, Zisman A, Fillmore JJ, Peroni OD, Kotani K, Perret P, Z et al. Glucose toxicity and the development of diabetes in mice with muscle-specific inactivation of GLUT4. J Clin Invest 2001;108:153–60. doi:https://doi.org/10.1172/JCI10294
Li M, Chi X, Wang Y, Setrerrahmane S, Xie W, Xu H. Trends in insulin resistance: insights into mechanisms and therapeutic strategy. Signal Transduct Targeted Ther. 2022;7:216. https://doi.org/10.1038/s41392-022-01073-0.
Hue L, Taegtmeyer H. The Randle cycle revisited: a new head for an old hat. Am J Physiol Endocrinol Metab. 2009;297:E578–91. https://doi.org/10.1152/ajpendo.00093.2009.
Rahimi Y, Camporez JP, Petersen MC, Pesta D, Perry RJ, Jurczak MJ, et al. Genetic activation of pyruvate dehydrogenase alters oxidative substrate selection to induce skeletal muscle insulin resistance. Proc Natl Acad Sci U S A. 2014;111:16508–13. https://doi.org/10.1073/pnas.1419104111.
Frayn KN. The glucose-fatty acid cycle: a physiological perspective. Biochem Soc Trans. 2003;31:1115–9. https://doi.org/10.1042/bst0311115.
Samuel VT, Petersen KF, Shulman GI. Lipid-induced insulin resistance: unravelling the mechanism. Lancet. 2010;375:2267–77. https://doi.org/10.1016/S0140-6736(10)60408-4.
• Lewis GF, Carpentier AC, Pereira S, Hahn M, Giacca A. Direct and indirect control of hepatic glucose production by insulin. Cell Metab 2021;33:709-20. https://doi.org/10.1016/j.cmet.2021.03.007. This review article provides a comprehensive overview of the experimental evidence that demonstrates how insulin acts on glucose production through both extrahepatic mechanisms and a direct hepatic effect.
Krssak M, Brehm A, Bernroider E, Anderwald C, Nowotny P, Dalla Man C, et al. Alterations in postprandial hepatic glycogen metabolism in type 2 diabetes. Diabetes. 2004;53:3048–56. https://doi.org/10.2337/diabetes.53.12.3048.
Boucher J, Kleinridders A, Kahn CR. Insulin receptor signaling in normal and insulin-resistant states. Cold Spring Harb Perspect Biol. 2014;6:a009191. https://doi.org/10.1101/cshperspect.a009191.
van der Kolk B. Tissue-specific insulin resistance in human obesity: A physiological approach to lipid metabolism. 2018. [Doctoral Thesis, Maastricht University]. Gildeprint Drukkerijen. https://doi.org/10.26481/dis.20180503bk
Petersen KF, Dufour S, Befroy D, Lehrke M, Hendler RE, Shulman GI. Reversal of nonalcoholic hepatic steatosis, hepatic insulin resistance, and hyperglycemia by moderate weight reduction in patients with type 2 diabetes. Diabetes. 2005;54:603–8. https://doi.org/10.2337/diabetes.54.3.603.
Goudriaan JR, Dahlmans VE, Teusink B, Ouwens DM, Febbraio M, Maassen JA, et al. CD36 deficiency increases insulin sensitivity in muscle, but induces insulin resistance in the liver in mice. J Lipid Res. 2003;44:2270–7. https://doi.org/10.1194/jlr.M300143-JLR200.
Falcon A, Doege H, Fluitt A, Tsang B, Watson N, Kay MA, et al. FATP2 is a hepatic fatty acid transporter and peroxisomal very long-chain acyl-CoA synthetase. Am J Physiol Endocrinol Metab. 2010;299:E384–93. https://doi.org/10.1152/ajpendo.00226.2010.
Chen Y, Huang L, Qi X, Chen C. Insulin receptor trafficking: consequences for insulin sensitivity and diabetes. Int J Mol Sci. 2019;20:5007. https://doi.org/10.3390/ijms20205007.
Petersen MC, Madiraju AK, Gassaway BM, Marcel M, Nasiri AR, Butrico G, et al. Insulin receptor Thr1160 phosphorylation mediates lipid-induced hepatic insulin resistance. J Clin Invest. 2016;126:4361–71. https://doi.org/10.1172/JCI86013.
Schmitz-Peiffer C, Biden TJ. Protein kinase C function in muscle, liver, and β-cells and its therapeutic implications for type 2 diabetes. Diabetes. 2008;57:1774–83. https://doi.org/10.2337/db07-1769.
Chavez JA, Summers SA. A ceramide-centric view of insulin resistance. Cell Metab. 2012;15:585–94. https://doi.org/10.1016/j.cmet.2012.04.002.
Turpin SM, Nicholls HT, Willmes DM, Mourier A, Brodesser S, Wunderlich CM, et al. Obesity-induced CerS6-dependent C16:0 ceramide production promotes weight gain and glucose intolerance. Cell Metab. 2014;20:678–86. https://doi.org/10.1016/j.cmet.2014.08.002.
Stratford S, Hoehn KL, Liu F, Summers SA. Regulation of insulin action by ceramide: dual mechanisms linking ceramide accumulation to the inhibition of Akt/protein kinase B. J Biol Chem. 2004;279:36608–15. https://doi.org/10.1074/jbc.M406499200.
• Al-Mansoori L, Al-Jaber H, Prince MS, Elrayess MA. Role of inflammatory cytokines, growth factors and adipokines in adipogenesis and insulin resistance. Inflammation 2022;45:31-44. https://doi.org/10.1007/s10753-021-01559-z. This article highlights the most studied factors related to the inflammatory response and insulin resistance associated with obesity, and their connection to the resulting metabolic consequences.
Wellen KE, Hotamisligil GS. Obesity-induced inflammatory changes in adipose tissue. J Clin Invest. 2003;112:1785–8. https://doi.org/10.1172/JCI20514.
Hotamisligil GS. Inflammatory pathways and insulin action. Int J Obes Relat Metab Disord. 2003;27(Suppl 3):S53–5. https://doi.org/10.1038/sj.ijo.0802502.
Salles J, Tardif N, Landrier JF, Mothe-Satney I, Guillet C, Boue-Vaysse C, et al. TNFα gene knockout differentially affects lipid deposition in liver and skeletal muscle of high-fat-diet mice. J Nutr Biochem. 2012;23:1685–93. https://doi.org/10.1016/j.jnutbio.2011.12.001.
Aguirre V, Uchida T, Yenush L, Davis R, White MF. The c-Jun NH(2)-terminal kinase promotes insulin resistance during association with insulin receptor substrate-1 and phosphorylation of Ser(307). J Biol Chem. 2000;275:9047–54. https://doi.org/10.1074/jbc.275.12.9047.
Kim JK, Kim YJ, Fillmore JJ, Chen Y, Moore I, Lee J, Y et al. Prevention of fat-induced insulin resistance by salicylate. J Clin Invest 2001;108:437–46. https://doi.org/10.1172/JCI11559
Hirosumi J, Tuncman G, Chang L, Görgün CZ, Uysal KT, Maeda K, et al. A central role for JNK in obesity and insulin resistance. Nature. 2002;420:333–6. https://doi.org/10.1038/nature01137.
Ueki K, Kondo T, Kahn CR. Suppressor of cytokine signaling 1 (SOCS-1) and SOCS-3 cause insulin resistance through inhibition of tyrosine phosphorylation of insulin receptor substrate proteins by discrete mechanisms. Mol Cell Biol. 2004;24:5434–46. https://doi.org/10.1128/MCB.24.12.5434-5446.2004.
Shi H, Tzameli I, Bjørbaek C, Flier JS. Suppressor of cytokine signaling 3 is a physiological regulator of adipocyte insulin signaling. J Biol Chem. 2004;279:34733–40. https://doi.org/10.1074/jbc.M403886200.
Chen A, Mumick S, Zhang C, Lamb J, Dai H, Weingarth D, et al. Diet induction of monocyte chemoattractant protein-1 and its impact on obesity. Obes Res. 2005;13:1311–20. https://doi.org/10.1038/oby.2005.159.
Suganami T, Tanaka M, Ogawa Y. Adipose tissue inflammation and ectopic lipid accumulation. Endocr J. 2012;59:849–57. https://doi.org/10.1507/endocrj.EJ12-0271.
•• Patra D, Roy S, Arora L, Kabeer SW, Singh S, Dey U, et al. miR-210-3p promotes obesity-induced adipose tissue inflammation and insulin resistance by targeting SOCS1-mediated NF-κB pathway. Diabetes 2023;72:375-88. https://doi.org/10.2337/db22-0284. This study has shown that delivering anti-miR-210-3p-locked nucleic acid into the adipose tissue microenvironment significantly rescued mice from obesity-induced adipose tissue inflammation and insulin resistance. This supports the idea that miRNAs could be effective targets for intervening in obesity-induced type 2 diabetes.
• Taheri M, Eghtedarian R, Ghafouri-Fard S, Omrani MD. Non-coding RNAs and type 2 diabetes mellitus. Arch Physiol Biochem. 2023;129:526-35. https://doi.org/10.1080/13813455.2020.1843498. This article summarizes the significant advances in understanding the role of miRNAs in the pathophysiology of type 2 diabetes and their potential application as biomarkers.
Stumvoll M, Häring H. Resistin and adiponectin–of mice and men. Obes Res. 2002;10:1197–9. https://doi.org/10.1038/oby.2002.162.
Li S, Shin HJ, Ding EL, van Dam RM. Adiponectin levels and risk of type 2 diabetes: a systematic review and meta-analysis. JAMA. 2009;302:179–88. https://doi.org/10.1001/jama.2009.976.
•• Yamauchi T, Nio Y, Maki T, Kobayashi M, Takazawa T, Iwabu M, et al. Targeted disruption of AdipoR1 and AdipoR2 causes abrogation of adiponectin binding and metabolic actions. Nat Med 2007;13:332-9. https://doi.org/10.1038/nm1557. This landmark paper demonstrates that adenovirus-mediated expression of adiponectin receptor 1 (AdipoR1) and adiponectin receptor 2 (AdipoR2) in the liver of leptin receptor-disrupted mice is effective in reducing gluconeogenesis and increasing fatty acid oxidation, which leads to the amelioration of diabetes.
Moonishaa TM, Nanda SK, Shamraj M, Sivaa R, Sivakumar P, Ravichandran K. Evaluation of leptin as a marker of insulin resistance in type 2 diabetes mellitus. Int J Appl Basic Med Res. 2017;7:176–80. https://doi.org/10.4103/ijabmr.IJABMR_278_16.
Sethi JK, Vidal-Puig AJ. Thematic review series: adipocyte biology. Adipose tissue function and plasticity orchestrate nutritional adaptation. J Lipid Res 2007;48:1253–62. https://doi.org/10.1194/jlr.R700005-JLR200
Petersen KF, Oral EA, Dufour S, Befroy D, Ariyan C, Yu C, et al. Leptin reverses insulin resistance and hepatic steatosis in patients with severe lipodystrophy. J Clin Invest. 2002;109(10):1345–50. https://doi.org/10.1172/JCI0215001.
Ceddia RB, Koistinen HA, Zierath JR, Sweeney G. Analysis of paradoxical observations on the association between leptin and insulin resistance. FASEB J. 2002;16:1163–76. https://doi.org/10.1096/fj.02-0158rev.
Seufert J. Leptin effects on pancreatic beta-cell gene expression and function. Diabetes. 2004;53(Suppl 1):S152–8. https://doi.org/10.2337/diabetes.53.2007.S152.
Iikuni N, Lam QL, Lu L, Matarese G, La Cava A. Leptin and inflammation. Curr. Immunol Rev. 2008;4:70–9. https://doi.org/10.2174/157339508784325046.
Rabe K, Lehrke M, Parhofer KG, Broedl UC. Adipokines and insulin resistance. Mol Med. 2008;14:741–51. https://doi.org/10.2119/2008-00058.Rabe.
Takahashi M, Takahashi Y, Takahashi K, Zolotaryov FN, Hong KS, Kitazawa R, et al. Chemerin enhances insulin signaling and potentiates insulin-stimulated glucose uptake in 3T3-L1 adipocytes. FEBS Lett. 2008;582:573–8. https://doi.org/10.1016/j.febslet.2008.01.023.
Morino K, Petersen KF, Shulman GI. Molecular mechanisms of insulin resistance in humans and their potential links with mitochondrial dysfunction. Diabetes. 2006;55(Suppl 2):S9–15. https://doi.org/10.2337/db06-S002.
Morino K, Petersen KF, Dufour S, Befroy D, Frattini J, Shatzkes N, et al. Reduced mitochondrial density and increased IRS-1 serine phosphorylation in muscle of insulin-resistant offspring of type 2 diabetic parents. J Clin Invest. 2005;115(12):3587–93. https://doi.org/10.1172/JCI25151.
Muoio DM, Newgard CB. Obesity-related derangements in metabolic regulation. Annu Rev Biochem. 2006;75:367–401. https://doi.org/10.1146/annurev.biochem.75.103004.142512.
Koves TR, Li P, An J, Akimoto T, Slentz D, Ilkayeva O, et al. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency. J Biol Chem. 2005;280:33588–98. https://doi.org/10.1074/jbc.M507621200.
Galgani JE, Moro C, Ravussin E. Metabolic flexibility and insulin resistance. Am J Physiol Endocrinol Metab. 2008;295:E1009–17. https://doi.org/10.1152/ajpendo.90558.2008.
Mootha VK, Lindgren CM, Eriksson KF, Subramanian A, Sihag S, Lehar J, et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat Genet. 2003;34:267–73. https://doi.org/10.1038/ng1180.
Yoo JY, Yum KS. Effect of coenzyme Q(10) on insulin resistance in Korean patients with prediabetes: a pilot single-center, randomized, double-blind, placebo-controlled study. Biomed Res Int. 2018;2018:1613247. https://doi.org/10.1155/2018/1613247.
Gutiérrez-Rodelo C, Roura-Guiberna A. Olivares-Reyes JA [Molecular mechanisms of insulin resistance: an update]. Gac Med Mex. 2017;153:214–28 (PMID: 28474708).
•• Ozcan U, Cao Q, Yilmaz E, Lee AH, Iwakoshi NN, Ozdelen E, et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 2004;306:457-61. https://doi.org/10.1126/science.1103160. This groundbreaking study was the first to demonstrate that obesity causes endoplasmic reticulum stress, which in turn suppresses insulin receptor signaling. This suppression occurs due to the hyperactivation of c-Jun N-terminal kinase and subsequent phosphorylation of insulin receptor substrate-1.
Ozcan U, Yilmaz E, Ozcan L, Furuhashi M, Vaillancourt E, Smith RO, et al. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science. 2006;313:1137–40. https://doi.org/10.1126/science.1128294.
Marchetti P, Bugliani M, Lupi R, Marselli L, Masini M, Boggi U, et al. The endoplasmic reticulum in pancreatic beta cells of type 2 diabetes patients. Diabetologia. 2007;50:2486–94. https://doi.org/10.1007/s00125-007-0816-8.
Ruan H, Lodish HF. Insulin resistance in adipose tissue: direct and indirect effects of tumor necrosis factor-alpha. Cytokine Growth Factor Rev. 2003;14:447–55. https://doi.org/10.1016/S1359-6101(03)00052-2.
Ruan H, Hacohen N, Golub TR, Van Parijs L, Lodish HF. Tumor necrosis factor-alpha suppresses adipocyte-specific genes and activates expression of preadipocyte genes in 3T3-L1 adipocytes: nuclear factor-kappaB activation by TNF-alpha is obligatory. Diabetes. 2002;51:1319–36. https://doi.org/10.2337/diabetes.51.5.1319.
Jenkins AB, Campbell LV. The genetics and pathophysiology of diabetes mellitus type II. J Inherit Metab Dis. 2004;27:331–47. https://doi.org/10.1023/B:BOLI.0000031097.31587.81.
Bloomgarden ZT. Obesity, hypertension, and insulin resistance. Diabetes Care. 2002;25:2088–97. https://doi.org/10.2337/diacare.25.11.2088.
Althaher AR. An overview of hormone-sensitive lipase (HSL). ScientificWorldJournal. 2022;2022:1964684. https://doi.org/10.1155/2022/1964684.
Wild S, Roglic G, Green A, Sicree R, King H. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047–53. https://doi.org/10.2337/diacare.27.5.1047.
• Guerra JVS, Dias MMG, Brilhante A, Terra MF, García-Arévalo M, Figueira ACM. Multifactorial basis and therapeutic strategies in metabolism-related diseases. Nutrients 2021;13:2830. https://doi.org/10.3390/nu13082830. This article provides a summary of the therapeutic strategies currently available for the treatment of obesity, diabetes, metabolic-associated fatty liver disease, and cardiovascular diseases.
Funding
Open Access funding enabled and organized by Projekt DEAL. RW is supported by the German Research Foundation (grants WE2554/13–1, WE2554/15–1, and WE 2554/17–1), the Deutsche Krebshilfe (grant 70115581), and the Interdisciplinary Centre for Clinical Research within the Faculty of Medicine at the RWTH Aachen University (grant PTD 1–5). The funders had no role in the design of this article or in the decision to publish it.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chandrasekaran, P., Weiskirchen, R. Cellular and Molecular Mechanisms of Insulin Resistance. Curr. Tissue Microenviron. Rep. 5, 79–90 (2024). https://doi.org/10.1007/s43152-024-00056-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43152-024-00056-3