Abstract
The investigation was inspired by the declaration of critically polluted industrial zones in India. It was also reported that most of the Common Effluent Treatment Plants (CETPs) are non-compliant due to higher COD values in discharge than regulatory norms. Furthermore, most of the literature has not reported the efficacy of advanced wastewater technologies for real industrial effluent. Thus, this research focused on field trials of scalable advanced wastewater technologies in Vatva Industrial Estate (VIE), Ahmedabad. The potential of physico-chemical and advanced treatment processes are explored for applicability in industrial wastewater treatment. Thus, for this research, three industries from each sector, i.e., textile processing, dyes, and dye intermediates, a total of nine different industries were chosen. A sufficient volume of effluent was collected from each industry for detailed investigations. The initial COD loading observed was in the range of 68 to 7074, 30 to 896, and 100 to 1541 kg COD/day for textile processing, dyes, and dye intermediates, respectively, with significant variation in the qualitative profiles. The collected effluent was treated by electrocoagulation and Fenton's reagent in batch scale reactors. The batch scale reactors for both the technologies were of 1L size. The results of treatability studies represented that Fenton's reagent performed better than the electrocoagulation process for most of the industries selected in this study. This research provides insight for improvement in the environmental performance of the individual industries in general and CETPs in particular.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The pollution problem precisely due to industrial effluents has become severe in the last few decades in developing nations. Rapid industrialization adversely impacts the environment in general and water quality in particular. Water quality is central to all ecosystem functions it mediates. Pollutants from industries contaminate aquatic habitats and thus exhibit potential toxicity towards aquatic organisms and may enter the food chain [1, 2]. Further, it is well known that industrial wastewaters can be very different from sewage in terms of characteristics, related treatments, and discharge patterns. Nevertheless, many industrial wastewater treatment plants use biological processes as key unit processes. They, however, pose unique challenges in terms of performance and treatment outcomes [3,4,5].
In India, a gap exists between the capacity of industrial effluent treatment and industrial effluent generated, mainly for micro, small and medium enterprises (MSMEs) [6]. This gap may lead to pollution of water bodies. In addition, population growth and mushrooming of MSMEs with the absence of proper domestic and industrial wastewater treatment may create serious water quality problems for India [7]. Typically, the best way to address pollution emanating from industries is an implementation of cleaner production practices/technologies and waste minimization initiatives, supported with end-of-the-pipe treatments. The various environmental agencies, researchers, and scientists worldwide are under increasing pressure to reduce the negative environmental impacts through scientific innovations and managerial approaches [8,9,10,11,12]. To control the menace of pollution, regulatory authorities across the globe have set effluents discharge standards and approaches to achieve these standards. The interplay of the volume of effluents, their qualitative profiles, production and treatment process efficiency, and the overall regulatory framework determine the outcome of such systems. It is further likely that their individual and synergistic impacts could vary across and within sectors. In India, for almost all MSMEs, diversity in the treatability of effluent exists, which creates complex issues for wastewater treatment in Common Effluent Treatment Plants (CETPs). The concept of CETPs in India was adopted to achieve end-of-pipe treatment of combined wastewater to benefit from the scale of operation. In addition, the CETPs also reduce the number of discharge points in an industrial estate for better enforcement by environmental regulatory agencies [13, 14].
Usually, authorities of CETPs provide standard guidelines to all MSMEs with uniform loading of COD. These guidelines are usually as per the inlet norms of CETPs. Effluents released from MSMEs are finally fed to CETP for further treatment. Typically, conventional biological treatment fails to treat industrial effluents because the effluents from several industries are mixed, and as a result, it possesses complex characteristics. Biological processes are widely adopted for most organic wastewaters yet do not always give satisfactory results [8, 13, 14]. Especially for the treatment of industrial wastewaters, many organic substances produced by the chemical and related industries are inhibitory, toxic, or resistant to biological treatment. Therefore, wastewater treatment depends on biodegradability, as a ratio of COD (Chemical Oxygen Demand) and BOD5 (Biochemical Oxygen Demand for 5 days test), i.e., COD: BOD5. This ratio would typically be about 1.5:1 for domestic wastewater [15]. However, for industrial wastewater, the ratio is much higher and depends on the type of industrial sector [5, 10, 16]. Therefore, advanced technologies based on oxidation may be the only viable options for treating biologically recalcitrant wastewater. However, treatment efficiency varies based on the characteristics of wastewater to be treated [14]. Overall, it has created a need for detailed treatability study for the critically polluted industrial cluster.
1.1 Selection of study area for field visits
Literature search suggested comparative studies with appropriate advanced technologies are justified to meet regulatory norms for industrial effluents. Treatment of industrial wastewater is an excellent challenge in CETPs due to the complex nature of wastewater from different industries. However, research related to MSMEs and CETPs is not much explored [13]. Moreover, literature related to comparative studies on the treatment of industrial effluents is not widely reported in scientific documents to provide a proper understanding of the treatment efficacy of advanced wastewater technologies. Therefore, an attempt has been made in this research to carry out field trials to assess the applicability of scalable advanced wastewater technologies for industrial effluents.
Central Pollution Control Board (CPCB) of India has released the Comprehensive Environmental Pollution Index (CEPI) monitoring for Polluted Industrial Areas (PIAs) of India. The report stated that Vatva Industrial Estate (VIE) is a critically polluted zone with an overall CEPI score of 74.77 in 2009, 87.46 in 2011, and 83.44 in 2013. These values are entirely above the criterion for critically polluted CEPI score by the Government of India [17]. In addition, Vatva is located in the neighborhood of metro city Ahmedabad. Thus, Vatva emerged as a good candidate for the present study. However, the matter of caution about Vatva is that it has groups of heterogeneous MSMEs, primarily divided into three major types of MSMEs: textile processing, dyes, and dye intermediates. Therefore, CETP Vatva, with a group of heterogeneous MSMEs releasing their wastewater, provides an excellent research opportunity.
It was reported that except for COD, all other parameters of treated effluent released from CETP Vatva are as per the legal guidelines. Therefore, it is implicit that installing a tertiary treatment unit is essential—this unit is to be installed either by CETP or by more polluting member industries. However, if a tertiary treatment unit is to be installed by CETP, it is of large capacity, and thus it requires enormous investment. Instead, if a specific industry / industrial sector is chosen, then treatment cost for tertiary treatment is much less. Therefore, it is essential to explore treatability studies for each industrial sector. Thus, treatability studies are needed for the industrial sector by employing easy to operate yet efficient technologies.
1.2 Selection of technologies for the treatment of industrial effluents for the study area
Industrial wastewater contains complex and slowly biodegradable compounds often ineffectively treated by conventional CETPs, which usually employ activated sludge systems. As reported in the literature, many advanced technologies are available. However, in the context of MSMEs in India, one has to choose low cost and effective alternative treatments which are capable to remove recalcitrant organic compunds from large volumes of effluents [18, 19]. Therefore, based on technologies reviewed in literature for the treatment of dyes in the effluent, two efficient treatment technologies were chosen for the research, namely, Electrocoagulation and Fenton's reagent [20,21,22]. Table 1 depicts a handy and straightforward comparison of both technologies.
Thus, this research aims to fill the gap related to comparing industrial effluent treatment from textile processing, dyes, and dye intermediates employing electrocoagulation and Fenton's reagent.
2 Data collection and field visits
The preliminary survey and exhaustive field visits of Vatva Industrial Estate and CETP had been conducted with CETP monitoring representatives. Field visits helped to understand the basic infrastructure available at individual industries and CETP.
2.1 Classification of industries for the research from the study area
It is crucial to choose specific industries in the heterogeneous industrial cluster, i.e., for VIE, because probably effluent from all member industries may be of different characteristics. In the case of the VIE, dual heterogeneity exists. First related to the ‘manufacturing sector’, i.e., textile processing, dyes manufacturing, and dye intermediates manufacturing. The second type of heterogeneity in each manufacturing sector is associated with the ‘scale of production’, i.e., small scale, medium scale, and large scale. During this research, it was attempted to identify the basis of the classification of industry. It was observed that government agencies classify ‘scale of production’ξ based on investment by industrial unit. However, in this research, classification based on investment is useless because the prime focus of the research is around treatability of wastewater which depends on the characteristic of wastewater and quantity of wastewater.
Further, it is well understood that the characteristic of wastewater depends on specific factors such as type of product, type of manufacturing process, control of the process, and workforce skills. Thus, based on these parameters, the classification of industrial units is not possible. Another critical parameter is the quantity of wastewater generated, which usually correlates rate of production in the industrial unit. Thus, in this research, based on data collected from VIE, classification of small, medium, and large scale industries were made based on effluent generated to avoid any bias in the selection of industries. Table 2 depicts the classification of industries for this research. It is well-known that textile processing units consume excessive water and generate more wastewater than dyes and dye intermediates industries [23, 24]. Therefore, to serve the purpose of this research, the classification of industries of each sector is carried out based on wastewater generated.
One cautionary remark related to classification is needed to be mentioned that the range of wastewater generated by industrial units in a particular sector may vary in different industrial zones. For example, in VIE, small-scale textile processing industries are classified as effluent generated less than 100 m3/day. In contrast, it is possible that in other industrial zone minimum effluent generated by any industry is 200 m3/d. Thus, in that particular industrial zone, the classification of industries needs to be done accordingly. Therefore, the classification used in the research may not be adopted as it is.
2.2 Selection of industries for the research from the study area
After the classification of industries of VIE, industries in different sectors and scales were selected for further sampling and analysis for this research. Nine industries were chosen for further data analysis from three industrial sectors—Textile, Dyes, and Intermediates as mentioned in Table 3
3 Materials and methods
3.1 Protocol for wastewater collection
The untreated wastewater was collected from nine identified industries of VIE. The names of industries are not revealed in the research, but for the identification of industries, codes are mentioned in Table 3. The wastewater from each industry was collected from the equalization tank of the Effluent Treatment Plant (ETP). It was decided to collect 1000 L of wastewater in polypropylene (PP) intermediate bulk container (IBC) tanks (Times Technoplast Limited, India) of 1000 L volume from each industrial unit. Two such IBC are used for the entire experimental work. The first sample of homogeneous untreated wastewater was drawn to analyze common parameters, such as pH and COD. In all cases, the experiments were conducted within three days from the date of collection of the sample. The treatability studies were conducted in batch scale reactors for both the methods, i.e., Fenton's reagent and electrocoagulation.
3.2 Protocols for treatability studies
3.2.1 Treatability studies with electrocoagulation
All batch experiments of electrocoagulation were conducted as per the standard protocol [25]. The experiments were carried out with 1L of wastewater in an acrylic cell equipped with iron anode & cathode and magnetic stirrer apparatus, which operated at a constant 150 rpm for homogeneous mixing. There were two monopolar electrodes, one anode and one cathode of similar dimensions. All cathode and anode were made from iron plates with dimensions of 70 mm × 65 mm × 3 mm. The total effective electrode surface area was 45.5 cm2, and the distance between electrodes was 10 mm in all experiments. The current density was maintained constant employing a precision DC power supply (LD-3205, Aplab India). For each experiment, the pH of wastewater at different time intervals was measured with a calibrated digital pH meter (Systronics-335, India) and pH adjusted by adding 1 N H2SO4 or lime solution. For batch studies, 10 mL samples were drawn (at 0, 1, 2, 3 h) for COD analysis, and the remaining wastewater was discarded at the end of the experiment. All samples were individually filtered with ordinary filter paper before conducting COD analysis.
3.2.2 Treatability studies with Fenton's reagent
All batch experiments with Fenton's reagent were conducted as per the standard protocol [26]. Identical to electrocoagulation, all batch experiments with Fenton's reagent were conducted in a 1L acrylic cell filled up with collected wastewater equipped with a magnetic stirrer which operated at constant 150 rpm for homogeneous mixing. For raw samples, 10 mL samples were drawn for all industries before pH was adjusted. Then, the pH of the solution was measured by a pH meter and adjusted by adding 1 N H2SO4 or lime solution. The regular dose of FeSO4 solution (30% solution w/V) and H2O2 (50% solution w/w) was applied as per the required quantity as suggested by Gulkaya et al. [27]. Experiments were conducted with the different quantities for trial and error to derive the most effective quantity of H2O2 for COD treatment. Based on the most effective dosage of H2O2, the concentration of H2O2 in the studies for each industry was identified and applied. For each experiment, the pH of wastewater at different time intervals was measured with a calibrated digital pH meter and pH adjusted by adding 1 N H2SO4 or lime solution. For all experimentation, 10 mL samples were drawn at every 30 min, and characterization was carried out. All samples were filtered with the ordinary filter paper individually before characterization.
4 Results and discussions
4.1 Key observations from CETP Vatva field visits
Figures 1 and 2 depict the location and site plan of Vatva. About 90% of the total industries in the estate are mainly engaged in the manufacturing of dyes, dye intermediates, pigments, fine chemicals, pharmaceutical products, and textile process houses, which contribute to wastewater generation. However, the small and medium scale industrial units having pollution potential located in the VIE faced constraints of space, finance, and appropriate technology for the treatment and disposal of wastewater in compliance with the provisions of the pollution control laws. To resolve this problem and prevent environmental pollution, a common effluent treatment plant (CETP) was established and operated by The Green Environment Services Co-operative Society Limited (GESCSL) with the support of the Vatva Industrial Association (VIA) in May 1998.
Each industry located in Vatva Industrial Estate (VIE) has to provide primary treatment to their wastewater and then send wastewater to CETP Vatva, operated by GESCSL. The CETP was designed for 16 MLD of wastewater to treat 3000 mg/L of COD to 250 mg/L of COD. Ministry of Environment, Forest and Climate Change (MoEFCC), Government of India (GoI) has declared Vatva as one of the critically polluted area of India. Therefore, further activities such as establishing new industries or expansion in industries are banned in the area.
Usually, the significant issue of noncompliance of CETPs may be attributed to undisciplined industries of the particular industrial estate. The industries can discharge their effluent directly in pipelines connected with CETP. This system enables industries to release effluent uncontrollably. As a result, the discharged effluent may be more in quantity or pollutant loading. Uncontrolled release of effluent with variation in quantity and inconsistent characteristics creates influent pollutant loading level beyond to design criteria of the CETP. Thus, uncontrolled effluent release from individual industries serves as the starting point for noncompliance of the CETP.
The VIE has taken appropriate precautions to avoid all such problems associated with noncompliance of CETP. The VIE's Effluent Collection System (ECS) is depicted in Fig. 3. This ECS is one of the best and exceptional among all industrial estates in India. The VIE has introduced 'Sump Room' (SR), through which the effluent from industry is discharged in a controlled manner. The primary purpose of SR is to regulate effluent discharge from individual industries using a flow meter and control valve. In addition, the effluent samples are collected before releasing it into a pipeline. If any irregularities are observed, a penalty will be imposed on the industry, or CETP authorities can deny accepting effluent. Thus, this ECS ensures that individual industries are not empowered to release effluent in the pipeline. Instead, the authorities of CETP have complete control over the release of effluents. None of the individual industries can discharge effluent without permission and knowledge of CETP authorities. This ECS confirms minimal variation in the influent of CETP.
4.2 Treatability studies of effluent from the Textile Processing (TP) sector
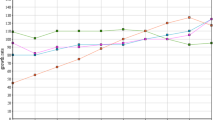
The treatability experiments were conducted for all three industries selected from the Textile Processing sector by employing a laboratory scale set-up of Fenton's reagent (FE) and electrocoagulation (EC). It is reported that the main problem of textile wastewater is intense color and COD, which must be removed before discharge to the environment. Generally, color removal is achieved rapidly with advanced treatments. However, COD removal requires more time, even with advanced treatments [28]. The results obtained for all three industries are depicted in Fig. 4. Fenton's reagent provides significantly better removal of COD for small and medium scale industries. Electrocoagulation provides marginally better results for the large-scale industry, as depicted in Table 4. Although inlet COD of industries 1 and 3 are almost comparable, the treatment efficiency of both technologies varies a lot. This variation was speculated initially, and this may be attributed to specific chemical compounds present in the wastewater. This variation is not reported in the literature because most studies have employed synthetic wastewater for their research. The variation in results proves the need for this research before adopting any specific treatment technologies for the industrial effluent.
Furthermore, laboratory-scale studies concluded that the steady-state condition was attained within 1 h of study for all the experiments employing EC and within 40 min by employing FE. Singh et al., [29] reported a similar observation for electrocoagulation. Ghanbari et al., [28] reported around 78% COD removal for TP sector effluent by employing EC, whereas, in this research, it is in the range of 61 to 75% removal. Similarly, for FE, literature reported around 60% removal for TP sector [30], whereas, for this research, the results were obtained in the range of 66 to 86% removal.
4.3 Treatability studies of effluent from the Dyes (DY) sector
Dyes in wastewater usually present high stability under sunlight and resistance to microbial attack and temperature, thus need proper scrutiny for identification of suitable advanced treatment technologies [31]. Therefore, the treatability experiments were conducted for all three industries selected from the Dyes sector by employing a laboratory scale set-up of Fenton's reagent (FE) and electrocoagulation (EC). The results obtained for all three industries are depicted in Fig. 5. Fenton's reagent provides significantly better removal of COD than electrocoagulation for small, medium, and large scale industries, as depicted in Table 5. Although inlet COD of industries 4 and 5 are almost comparable, the treatment efficiency of both technologies varies a lot. It is also observed that inlet COD for industry 6 is almost 3 times higher than industries 4 and 5. However, both technologies provide better COD removal efficiency for industry 6. Again, these results emphasize that merely COD values are not deciding factors for selecting treatment technologies.
It is reported in the literature that color removal by electrocoagulation is taking place by floc formation, and it is efficient [32]. Thus for synthetic wastewater, the technology looks appropriate. However, the results are sometimes not encouraging for industrial wastewater, especially COD removal [33, 34]. In contrast, Fenton's reagent directly reacts with organic pollutants and destroys them, thus better COD removal was obtained [35]. Malik and Saha [36] reported that dyes are decomposed in a two-stage reaction, and around 70% COD removal was observed for synthetic wastewater prepared using Direct Blue 2B and Direct Red 12B dyes. This research COD removal studies were carried out for effluents from dyes industries, and COD removal was observed in the range 58 to 74%
4.4 Treatability studies of effluent from the Dye Intermediates (DI) sector
Dye intermediates are bioresistant and refractory pollutants. Therefore, conventional biotreatment fails to remove these chemical compounds [37]. Therefore, the treatability experiments were conducted for all three industries selected from the Dye Intermediates sector by employing a laboratory scale set-up of Fenton's reagent (FE) and electrocoagulation (EC). The results obtained for all three industries are depicted in Fig. 6. Fenton's reagent provides significantly better removal of COD than electrocoagulation for small, medium, and large scale industries, as depicted in Table 6. Although inlet COD of industries 7 and 9 are almost comparable, and industry 8 is 1.5 times higher than both, nevertheless the treatment efficiency of Fenton's reagent is almost similar for all three industries.
Strikingly, electrocoagulation fails to remove COD for all the industries selected from the DI sector. The reason may be attributed to the COD removal mechanism while electrocoagulation is employed. As per the literature, when electrocoagulation is employed, floc formation occurs. Once the insoluble flocs of Fe(OH)3 are produced, they can remove dissolved dyes by surface complexation or electrostatic attraction. The dye can act as a ligand to bind a hydrous iron moiety of the floc yielding a surface complex, and removal of dye can be achieved [38, 39]. As the dye removal takes place, simultaneously COD removal is also attained. Now, for the wastewater from DI sector, such floc formation is ineffective for removing dye intermediates, and thus, COD removal efficiency is trivial. In contrast, the percentage removal efficiency of Fenton's reagent for all three industries is almost similar, in the range of 55 to 60%, and better than electrocoagulation.
In all the experiments, the reaction time is around 60 min or less to achieve a steady-state condition. Similar observations are reported in the literature. The literature also reported that Fenton's reaction has a relatively short reaction time and can degrade wastes with high COD concentrations (< 5000 mg/L). However, sometimes it requires a lot of hydrogen peroxide to achieve complete mineralization [40]. Thus, this research provided important information for operability parameters for industrial wastewater treatment using Fenton's reagent and electrocoagulation.
The summary of comparative treatability studies is depicted in Fig. 7. For the wastewater from industries 1 to 9, the removal of COD seems much better for TP sector than the other two sectors for both treatment technologies. Interestingly, except for industry 3 for all other industries, Fenton's reagent depicted better performance than electrocoagulation. The data from three industries of the DY sector represented variation in both inlet COD and outlet COD by both the technologies. No doubt that Fenton's reagent represented better treatability results than electrocoagulation. However, treatment up to the environmental compliance level is not achieved. Finally, all three industries from the DI sector represented higher inlet COD values. Therefore, It was also experienced that treatability was most difficult for both the technologies employed for DI sector. Thus, the best suitable technology for wastewater treatment is not generalized for the specific industrial sector. It needs to be identified based on treatability studies of individual industries. In addition, no correlation can be established among industry type or industrial sector or for the suitable treatment.
Thus, the time required and efficiency attained for the removal of COD from industrial effluent are provided by this research. These parameters are essential for finalizing the dimensions of a large-scale reactor for industrial effluent. In addition, the study proves variation in efficiency across the treatments and across the industries, which is essential understating before adopting technologies for any specific industry for the treatment of effluents.
5 Conclusions
The Effluent Collection System (ECS) for CETP Vatva is one of the best and exceptional across India. This ECS provides complete control to CETP authorities over the collection of effluents from industries. The ECS ensured that all member MSMEs must control the quantity and characteristic of their effluent before discharge, and thus minimal variation in the influent of CETP was observed. Further, treatability studies were carried out by electrocoagulation and Fenton's reagent for selected nine industries from the three industrial sectors, i.e., textile processing, dyes, and dye intermediates, in batch scale reactors. The results depicted COD removal in the range of 61% to 86%, 27% to 73% and 9% to 59% for textile processing, dyes, and dye intermediates, respectively. It is also concluded that no emphatic correlation exists between the scale of industries and/or treatment technologies and COD removal efficiency. The results of treatability studies by batch reactors represented that Fenton's reagent performed better than the electrocoagulation process for most of the industries selected in this study. Thus, Fenton's reagent provided improved COD removal performance from both scalable advanced wastewater technologies selected in this research for various industrial effluents. This study provides significant insight into the possibilities of industrial effluents' treatment by employing a non-biological yet straightforward treatment process.
Availability of data and materials
All data generated or analyzed during this study are included in this article.
References
Alvares ABC, Diaper C, Parsons SA. Partial oxidation by ozone to remove recalcitrance from wastewaters—a review. Environ Technol. 2001;22(4):409–27.
Babuponnusami A, Muthukumar K. A review on Fenton and improvements to the Fenton process for wastewater treatment. J Environ Chem Eng. 2014;2:557–72.
Bautista P, Mohedano AF, Casas JA, Zazo JA, Rodriguez JJ. An overview of the application of Fenton oxidation to industrial wastewaters treatment. J Chem Technol Biotechnol. 2008;83(10):1323–38.
Bolong N, Ismail AF, Salim MR, Matsuura T. A review of the effects of emerging contaminants in wastewater and options for their removal. Desalination. 2009;239(1–3):229–46.
Can OT, Bayramoglu M, Kobya M. Decolorization of reactive dye solutions by electrocoagulation using aluminum electrodes. Ind Eng Chem Res. 2003;42(14):3391–6.
CPCB. Assessment of the need for common effluent treatment plants. New Delhi: Central Pollution Control Board, Government of India; 2015.
CPCB. Industrial pollution. 18 April 2019. http://cpcbenvis.nic.in/industrial_pollution.html#.
Crini G, Lichtfouse E. Advantages and disadvantages of techniques used for wastewater treatment. Environ Chem Lett. 2019;17(1):145–55.
Daneshvar N, Oladegaragoze A, Djafarzadeh N. Decolorization of basic dye solutions by electrocoagulation: an investigation of the effect of operational parameters. J Hazard Mater. 2006;129(1–3):116–22.
de Oliveira Neto GC, Correia JMF, Silva PC, de Oliveira Sanches AG, Lucato WC. Cleaner production in the textile industry and its relationship to sustainable development goals. J Clean Prod. 2019;228:1514–25.
Deblonde T, Cossu-Leguille C, Hartemann P. Emerging pollutants in wastewater: a review of the literature. Int J Hyg Environ Health. 2011;214(6):442–8.
Ghanbari F, Moradi M. A comparative study of electrocoagulation, electrochemical Fenton, electro-Fenton and peroxi-coagulation for decolorization of real textile wastewater: electrical energy consumption and biodegradability improvement. J Environ Chem Eng. 2015;3(1):499–506.
Ghumra DP, Agarkoti C, Gogate PR. Improvements in effluent treatment technologies in Common Effluent Treatment Plants (CETPs): review and recent advances. Process Safety and Environmental Protection 2021.
Gogate PR, Pandit AB. A review of imperative technologies for wastewater treatment I: oxidation technologies at ambient conditions. Adv Environ Res. 2004;8(3–4):501–51.
Gulkaya I, Surucu GA, Dilek FB. Importance of H2O2/Fe2+ ratio in Fenton’s treatment of a carpet dyeing wastewater. J Hazard Mater. 2006;136(3):763–9.
Höfl C, Sigl G, Specht O, Wurdack I, Wabner D. Oxidative degradation of AOX and COD by different advanced oxidation processes: a comparative study with two samples of a pharmaceutical wastewater. Water Sci Technol. 1997;35(4):257.
Holkar CR, Jadhav AJ, Pinjari DV, Mahamuni NM, Pandit AB. A critical review on textile wastewater treatments: possible approaches. J Environ Manage. 2016;182:351–66.
Kabdaşlı I, Arslan-Alaton I, Ölmez-Hancı T, Tünay O. Electrocoagulation applications for industrial wastewaters: a critical review. Environ Technol Rev. 2012;1(1):2–45.
Kumar MD, Tortajada C. Assessing wastewater management in India. Singapore: Springer; 2020.
Lal H, Delgado JA, Gross CM, Hesketh E, McKinney SP, Cover H, Shaffer M. Market-based approaches and tools for improving water and air quality. Environ Sci Policy. 2009;12(7):1028–39.
Lin SH, Lo CC. Fenton process for treatment of desizing wastewater. Water Res. 1997;31(8):2050–6.
Lopez A, Ricco G, Mascolo G, Tiravanti G, Di Pinto AC, Passino R. Biodegradability enhancement of refractory pollutants by ozonation: a laboratory investigation on an azo-dyes intermediate. Water Sci Technol. 1998;384(4–5):239–45.
Malik PK, Saha SK. Oxidation of direct dyes with hydrogen peroxide using ferrous ion as catalyst. Sep Purif Technol. 2003;31(3):241–50.
Martınez-Huitle CA, Brillas E. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods: a general review. Appl Catal B. 2009;87:105–45.
Moussa DT, El-Naas MH, Nasser M, Al-Marri MJ. A comprehensive review of electrocoagulation for water treatment: potentials and challenges. J Environ Manage. 2017;186:24–41.
Ng WJ. Industrial wastewater treatment. World Scientific; 2006.
Nidheesh PV, Ravindran V, Gopinath A, Kumar MS. Emerging technologies for mixed industrial wastewater treatment in developing countries: an overview. Environmental Quality Management 2021.
Oller I, Malato S, Sánchez-Pérez J. Combination of advanced oxidation processes and biological treatments for wastewater decontamination—a review. Sci Total Environ. 2011;409(20):4141–66.
Paździor K, Bilińska L, Ledakowicz S. A review of the existing and emerging technologies in the combination of AOPs and biological processes in industrial textile wastewater treatment. Chem Eng J. 2019;376:120597.
Pignatello JJ, Oliveros E, MacKay A. Advanced oxidation processes for organic contaminant destruction based on the Fenton reaction and related chemistry. Crit Rev Environ Sci Technol. 2006;36(1):1–84.
Pophali GR, Kaul SN, Mathur S. Influence of hydraulic shock loads and TDS on the performance of large-scale CETPs treating textile effluents in India. Water Res. 2003;37(2):353–61.
Raghu S, Basha CA. Chemical or electrochemical techniques, followed by ion exchange, for recycle of textile dye wastewater. J Hazard Mater. 2007;149(2):324–30.
Robinson T, McMullan G, Marchant R, Nigam P. Remediation of dyes in textile effluent: a critical review on current treatment technologies with a proposed alternative. Biores Technol. 2001;77(3):247–55.
Shah HS, Ruparelia JP. Studies on potential of water quality trading at industrial zone Palsana, Surat. Int J Appl Eng Res. 2017;12(15):5359–66.
Shahedi A, Darban AK, Taghipour F, Jamshidi-Zanjani A. A review on industrial wastewater treatment via electrocoagulation processes. Curr Opin Electrochem. 2020;22:154–69.
Sharma J, Sharma S, Soni V. Classification and impact of synthetic textile dyes on Aquatic Flora: a review. Reg Stud Mar Sci. 2021;45:101802.
Singh MM, Szafran Z, Ibanez JG. Laboratory experiments on the electrochemical remediation of environment. part 4: color removal of simulated wastewater by electrocoagulation-electroflotation. J Chem Educ. 1998;758:1040.
Syam Babu D, Anantha Singh TS, Nidheesh PV, Suresh Kumar M. Industrial wastewater treatment by electrocoagulation process. Sep Sci Technol. 2020;55(17):3195–227.
Tortajada C, Saklani U, Biswas AK. Water scarcity and regional security in India. In: David R, editor. Water, security and US foreign policy. New York: Routledge; 2017. p. 237–52.
Vandevivere PC, Bianchi R, Verstraete W. Treatment and reuse of wastewater from the textile wet‐processing industry: review of emerging technologies. J Chem Technol Biotechnol 1998;72.4:289–302.
Acknowledgements
The authors wish to acknowledge the help and cooperation of the authorities of Vatva Industrial Estate (VIE), Ahmedabad, for providing samples and data of industries and the Green Environment Services Co-op. Society Ltd. (GESCSL), Vatva, Ahmedabad, for providing details and data of CETP-Vatva.
Author information
Authors and Affiliations
Contributions
Both authors are equally responsible for data collection and manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shah, H., Ruparelia, J. Comparative studies for the treatment of industrial effluents employing advanced processes: towards enhancement of environmental performance. Discov Water 2, 4 (2022). https://doi.org/10.1007/s43832-022-00012-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43832-022-00012-y