Abstract
Objectives
Non-communicable diseases (NCDs) account for over 30% of disability-adjusted life years in South Africa. In this research, we offer an estimate of the potential reduction in NCD incidence that would arise from an improvement in diet, combined with a reduction in both tobacco and alcohol consumption.
Methods
We apply the PRIME model, which simulates the effect of risk reduction on NCD incidence. The model inputs baseline data related to the population, risky consumption behaviour and NCD incidence. The model allows for counterfactual scenarios altering the risky consumption behaviour to yield revised NCD incidence.
Results
We find that reducing salt, tobacco and alcohol, along with improved fruit, vegetable and fiber consumption would yield a 10% reduction in NCDs from the 2018 baseline. NCD incidence reductions arise primarily from ischemic heart disease (49%), cerebrovascular diseases (33%) and bronchus and lung disease (11%).
Conclusion
South Africa’s NCD incidence is high because of relatively poor behavioural choices, despite plans and policies aimed at changing this. South Africa should increase their efforts to reach NCD goals. If the government is able to reduce harmful behaviour, with respect to a number of the underlying consumption choices, NCD incidence is expected to fall precipitously.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
1.1 NCDs in South Africa
Non-communicable diseases (NCDs)—cancer, type 2 diabetes mellitus, respiratory illnesses (such as chronic obstructive pulmonary disease) and mental health disorders—are an increasing global burden [1]. An estimated 41 million people could die prematurely from NCDs, potentially costing USD47 trillion over the next few decades [2]. NCDs disproportionately affect low- and middle-income countries (LMICs), where over 75% of NCD-related deaths occur and NCDs are likely to become the biggest cause of death [2,3,4].
South Africa, has a quadruple burden of disease—communicable diseases, NCDs, maternal and child health, and injury-related disorders [5,6,7]. Furthermore, there is extensive interpersonal violence and there is evidence of increasing multimorbidity, which raises both the demand for and cost of healthcare, when health budgets are limited [8,9,10,11,12,13,14]. By 2010, NCDs were amongst the top causes of death, with a large share of deaths being premature (i.e. before the age of 60) [15]. Relatively recent estimates of NCD burdens suggest that they account for approximately 30% of disability-adjusted life years (DALYs) and nearly two-thirds of catastrophic health expenditure (CHE) [16].
NCDs are commonly associated with socio-environmental and behavioural factors, including tobacco and alcohol use, sedentary lifestyles and unhealthy diets, which have become more common in less developed countries [5, 6, 17]. The NCD burden remains high, partly due to associations with antiretroviral therapy, and therefore continues to deserve attention [18, 19]. In this study, we evaluate the potential health improvement that can be derived in South Africa from reductions in NCD behavioural risk factors, such as excessive consumption of salt, tobacco and alcohol, as well as insufficient intake of fiber, fruits and vegetables. We do so through the analysis of data from 2018 through the lens of the Preventable Risk Integrated Model (PRIME). We find an approximate 10% reduction in NCD incidence achievable, if the South African population is able to change their lifestyle consumption habits.
2 Background
2.1 NCD risks in South Africa
Inequality is rife in South Africa. Its Gini coefficient hovers around 0.69, while 83% of households without at least one employed member experience poverty, which contributes to NCD prevalence [20,21,22,23,24]. There are also inequities in education, malnutrition outcomes and access to food and energy, similar to those related to income and health [25,26,27,28,29,30,31,32].
Although access to food and nutrition is unequal, consumption of sugar-sweetened beverages (SSB), packaged, and fast foods, has increased overall, including amongst the poor [33]. 40% of South Africans consume enough energy, but not enough nutritional quality [34]. This increase in unhealthy consumption has been fuelled by marketing, product placement and the increased availability of high-energy products [35].
Along with an ageing population, there is evidence of high salt consumption, which correlates with hypertension, and ultimately, increased cardiovascular disease (CVD) [5, 6, 36]. Premature mortality and long-term disability, which affect government health expenditure, labour productivity and economic growth, are obvious problems associated with CVD [37, 38]. CVD is also likely to out-of-pocket (OOP) expenditures, which tend to be higher in lower-income households, rural and underserved communities [39, 40]. A modest salt reduction could have substantial health gains [41, 42].
The literature on tobacco consumption and health effects in South Africa captures tobacco-attributable deaths, as well as race-based differences in mortality rates and economic costs associated with tobacco-related diseases [43,44,45,46]. While the smoking prevalence has dropped in most countries, it increased from 19% in 2017 to 24% in 2021 in South Africa [104]. Tobacco consumption is mainly driven by male adults, with a smoking prevalence of 39% in 2021. The increase in smoking prevalence has potentially been fuelled by illicit cigarette trade. Between 2017 and 2021, illicit trade rose from 35 to 54% [47, 48]. The illicit cigarette market not only endangers individual health—due to potentially poor cigarette quality—but also constrains the government budget, due to tobacco tax losses. An illicit market share of 54% means that the South African government could double its tobacco tax revenues, if smokers could be convinced to switch to legal tobacco products. Tobacco is responsible for approximately 10% of deaths, while the costs of premature death, morbidity and healthcare are estimated at near 1% of GDP; larger cost estimates also exist [43, 46, 49].
Alcohol consumption also tends to be high, while binge and other risky drinking patterns are common. Of particular concern is annual per capita alcohol consumption, which is around 9.5 L [50, 51]. Furthermore, South Africa is one of only nine countries globally with the second-highest (4 out of 5) patterns-of-drinking score [52]. While illicit alcohol trade has not yet reached the levels of illicit cigarette trade, it is responsible for a substantial loss to South Africa’s budget. Illicit alcohol trade constituted 22% of the total market in 2020, producing a fiscal deficit of R11.3 billion [53].
2.2 Policy context
The United Nations High-Level NCD Meeting focused on the potential impact of an increasing NCD burden on LMIC health systems, while the World Health Organization (WHO) has emphasised ‘best buys’—cost effective, feasible and inexpensive interventions, that offer large improvements in public health [54, 55]. Given that public health spending has not generally met the 15% government budget share proposed in the Abuja Declaration in Africa, and that both health insurance and access to healthcare is incomplete, many costs are likely to be covered by OOP expenditures [56,57,58,59]. OOP costs are associated with poor health outcomes especially for the poor, elderly and those with chronic conditions [60,61,62]. Thus, there is a need to manage costs, if not at the national level, then at least at the individual level, via prevention.
The South African government has responded to the country’s NCD threat via its NCD plan, which includes multiple stakeholders and focuses on reducing prevalence, and, therefore, burden [63, 64]. Amongst the government’s goals and targets are [32, 63]:
-
1.
25% reduction in relative NCD premature mortality (< 60 years) by 2020
-
2.
20% reduction in alcohol and tobacco consumption by 2020
-
3.
Salt intake reduction to < 5 g per day by 2020
-
4.
10% reduction in the rate of obesity and/or overweight by 2020
-
5.
20% reduction in the prevalence of high blood pressure by 2020
-
6.
10% increase in physical activity prevalence
-
7.
Cervical cancer screening for every woman: three screens per lifetime or every five years for those with an STD, or according to policy for HIV-positive women
-
8.
30% increase in the share of those able to control their hypertension, diabetes and asthma by 2020
-
9.
30% increase in screening for mental disorders by 2030
NCD policies are meant to drive behavioural change and eventually reduce NCDs; such policies were generally based on community and public participation, although this does not appear to have affected implementation or NCD prevalence [65, 66]. In support of the NCD plan, the government has enacted reforms meant to reduce: tobacco consumption, advertising for unhealthy foods, and the consumption of certain ingredients or components in unhealthy foods, such as fatty acids, salt and sugar [66]. By 2021, 8 policies were designed to affect smoking, a further 7 for alcohol, 8 for unhealthy diets and 5 on inactivity [67]. Thus, NCD policies are in place, but only tobacco control has been at least partially effective, likely contributing to the small reduction reported in NCD deaths [7, 18, 66]. South Africa is not unique in this regard—NCDs are increasing almost everywhere, especially in LMICs [53, 68,69,70]. Some of the problems are related to limited physical activity, as a lack of green or safe spaces, especially in urban informal settlements, deters outdoor activities [71].
The country’s endeavours were supported by international developments, such as the UN endorsement of the declaration for controlling and preventing NCDs along with international NCD reduction targets [55, 70]. Even though international agreements could be used to support NCD policy, many regulations were not easily implemented due to industry pushback [72,73,74,75,76,77,78,79,80,81,82,83,84,]. By 2021, only 13 (6 unhealthy diets, 3 tobacco control and 4 physical activity) of the identified programs had been implemented [64,65,66,67,67, 71, 73,74,75,76,77,78,79,80,81,82,83,84,85]. The government has also been moving, in fits and starts, towards universal health coverage via a national health insurance scheme [86, 87].
In summary, the current approach appears to be rather ineffective in curbing behavioural risks and thus NCDs. If the South African government aims to reach its NCD goals and improve the healthcare system, it needs to follow a different harm-based approach. Additional measures do not necessarily require costly interventions but could be built upon consumer education and awareness-raising. If consumers were more conscious about their lifestyles, they might make less harmful choices, i.e. healthier diets, lower alcohol consumption and reduced-risk tobacco products instead of smoking.
3 Methods and data
We evaluate the potential health improvement in South Africa that can be derived from reductions in NCD behavioural risk factors, such as excessive consumption of salt, tobacco and alcohol, as well as, insufficient intake of fiber, fruits and vegetables. To do so, we analyze data from 2018 through the lens of the Preventable Risk Integrated Model (PRIME) [88]. The Preventable Risk Integrated Model (PRIME), an Oxford-developed tool is a publicly accessible scenario modeling tool designed to assess the impact of changes in non-communicable disease (NCD) risk factors on morbidity and mortality.
3.1 Brief overview
The tool compares real-world baseline data with hypothetical scenarios derived from extensive meta-analyses (counterfactual scenario) or policy prescriptions. From our baseline, we know the incidence of NCDs, as well as the consumption of numerous goods that are known to be NCD risk factors. The comparison arises from, first, simulating the number of incidences that would arise from a change in unhealthy consumption choices, such that, second, the averted/prevented incidences can be calculated as the difference between the baseline and the simulation.
As implied by the above, the simulations are underscored by relative risks derived from the existing literature for each disease, age group, and gender. Using the relative risks, population attributable fractions (PAFs) are calculated in the model, determining the percentage of disease cases potentially preventable under alternative risk factor scenarios. The model compares actual data with an improved hypothetical scenario to estimate the number of avertable incidences. By categorizing complex data into distinct groups and applying PAFs to the incidence count in each, PRIME estimates the impact of modifying risk factors on total morbidity [88].
PRIME needs three sets of input data for a given year: age and gender distribution of the population, NCD incidence rates for each disease (ICD-10) by age and gender, and risk factor behaviour by age and gender. Below, we describe how the data is developed for the model.
3.2 Unhealthy consumption goods
To estimate the average consumption of salt and fiber (and standard deviations to capture the distribution) per gender and age group, data from the Global Dietary Database (GDD) 2018 is used [105]. However, salt intake is underreported in the data, because the metadata only includes added salt used during cooking, whereas the salt added to industrialized products is not considered [89, 90]. Therefore, we calculate the total salt consumption based on the assumption that on average, 55% of salt intake in South Africa is from processed food [91].
Vegetables—not including starches such as potatoes—and fruit intake are calculated from the South African Demographic and Health Survey (SADHS) from 2016 [106]. Because the GDD data is aggregated, some information, such as the percentage consuming < 1 fruit/vegetable portions daily, cannot be calculated [91]. The SADHS dataset provides only the number of fruit and vegetable types consumed daily, without specifying the amounts. To address this gap, we utilize the GDD to calculate the average quantity consumed, the standard deviation, and the percentage of individuals consuming less than one portion daily. With the insights from the GDD data, we can determine the average daily consumption of fruits and vegetables in grams. This involves dividing the total consumption by the number of types to estimate the average intake per fruit or vegetable type. We then use these estimates to calculate the daily fruit and vegetable intake for each respondent. This variable enables us to generate the necessary inputs for the PRIME.
The share of current, former, and never smokers, as well as daily alcohol consumption, is also obtained from the SADHS. Regarding current smoking behaviour, we account for both daily and weekly smokers. For alcohol consumption, we consider the number of drinks per day, with the assumption that a standard drink contains 12 g of alcohol [92]. These values represent the baseline scenario in the PRIME model.
Although the counterfactual scenarios can be modified in PRIME, we use the policy prescriptions from WHO recommendations for consumption of these goods [91]. Thus, it is assumed that mean South African population consumption behaviour is shifted, but the spread of consumption remains unchanged, such that standard deviations in the counterfactual match the baseline scenario [93].
3.3 Tobacco products risk reduction
For risk reduction in smoking, we follow a different approach. While quitting smoking always remains the best solution for health, convincing all smokers to quit immediately is deemed unrealistic. Additionally, the PRIME model does not allow for a reduction in consumption levels for smoking, unlike for other risk factors, which limits the potential counterfactual scenarios to a reduction in the number of current smokers. To overcome both limitations, we follow a more pragmatic approach, wherein we allow a certain share of smokers to switch to less harmful alternatives or quit smoking. Due to robust toxicological evidence and the increasing attractiveness of non-combustible alternatives to consumers, we include them in our model.
Non-combustible alternatives include nicotine-containing products that heat instead of burn nicotine, such as e-cigarettes and heated tobacco products. These products still contain nicotine and liquids or aerosols, which can have harmful health effects. Moreover, it is currently unclear what long-term health consequences may result from the continuous use of e-cigarettes. Given their recent market introduction, epidemiologic research is still needed to determine whether these alternatives can reduce the disease risk associated with smoking and, if so, to what extent. Unlike e-cigarettes, heated tobacco products are not tobacco-free. [94] Their harm-reducing effect arises from the lower temperatures at which the tobacco vaporizes, but dangerous chemicals can still be produced, albeit often in lower concentrations than in combustible cigarettes. [95, 96] Based on toxicological studies and expert opinions, commissioned by governmental authorities or research institutes, various methodologies are employed, with most researchers estimating cancer potencies or biomarkers to assess health risks. The range of risk reduction reported in these studies is between 65% and 97%. [95,96,97,98,99,100,101,102,103] However, there remain significant uncertainties regarding the long-term health effects of these alternatives.
For our model, we assume a risk-reduction of 50%, which is below the reported lower risk of 65–97%. We use this conservative estimate to account for the uncertainty on long-term health effects. In our counterfactual scenario, this means that if half of all smokers switch to these alternatives and they pose half the health risk of traditional smoking products, we assume that 25% of smokers will have the same relative risks as ex-smokers, while 25% will remain at their current smoking status. By taking this conservative approach, we aim to provide a balanced and realistic assessment of the potential health benefits of these alternatives, acknowledging both the promising evidence and the existing uncertainties.
3.4 Population and incidences
Population data by age and gender is obtained from the World Bank for 2018 in South Africa [107]. However, since the World Bank data aggregates from the age of 80, we have taken the distribution of 80–84-year-olds among 80 + year-olds from the 2011 census, applying it to the World Bank data, assuming the distribution has not changed between 2011 and 2018 [108].
The incidences per disease by age group and gender are obtained from the Global Burden of Disease for 2018 [109]. Due to the difference in longevity of cancer diseases (ICD codes C00–C14: Lip, oral cavity and pharynx cancer, C16: Stomach cancer, C25: Pancreas cancer, C18–20: Colorectum cancer, C50: Breast cancer, C64: Kidney cancer, C22: Liver cancer, C53: Cervix cancer, C34: Bronchus and lung), the incidence is calculated using the following formula:
, with.
Total Icancer,n=10 represents the incidence per each cancer for year 10.
Total Icancer,n-1 represents the incidence per each cancer from the previous year.
Icancer represents the number of new incidences per year for each cancer.
SRcancer denotes the yearly survival rate, derived from the survival rate per cancer after 10 years.
n is the year for which the incidence is being calculated [89].
This formula calculates the 10-year incidence of diseases, based on acute and chronic incidences [110]. Chronic diseases, such as cancer, are typically evaluated over a 10-year period. This is because only considering new cases within a given year could potentially underestimate the incidence of these diseases due to their longevity.[101] The formula assumes a consistent decline in the survival rate over those 10 years, presuming that after 10 years, individuals are no longer affected by cancer.
3.5 PRIME benefits and limitations
As with any modelling exercise, PRIME is underscored by numerous assumptions. One of the main features, and, thus, benefits, of the model is that its structure limits the potential for double-counting, which might arise if risk factors were not mediated [88]. However, the parameters in the model are underscored by the epidemiological literature, which, for the most part, focuses more on developed than developing countries. Thus, it is possible that the parameters that we apply are not appropriate for the context—South Africa—wherein it is applied. Hopefully, in future, additional local research will be undertaken and will be able to verify or improve the parameters used in PRIME. Even though the model limits double-counting, the parameters are dependent upon what is known at this moment; certainly, the parameters may not entirely capture all forms of mediation, such that some double counting is likely to remain. Finally, due to the modelling approach, it is not possible to estimate the total health impact for each risk factor—we can only attribute incidence to each factor.
PRIME, in full, allows for the inclusion of diet, physical activity, smoking and alcohol consumption. For our analysis, we do not have access to information on physical activity. Given the rates of obesity in the country, the body mass index for 41% of women and 11% of men exceeds 30 kg/m2, it is likely that physical activity is too low in the country, in addition to concerns over diet [106]. Unfortunately, we are not able to directly pinpoint the potential benefits of improved exercise plans on NCD incidence. Also, as been highlighted, the available dietary data underreports salt consumption, requiring us to derive the amount of salt in the South African diet from an assumption on processed food consumption. If we are assuming too much salt consumption in the diet, our estimated improvements are too high, and vice versa. Clearly, if the baseline data is incorrect, the results will also not be entirely accurate.
4 Results
4.1 Modelling data
As noted earlier, PRIME requires inputs by age and gender for a variety of (potentially unhealthy) consumption goods, which we captured from the GDD, SADHS and Census [105, 106, 108]. Table 1 reports simple averages for the baseline for males and females, which were averaged across all age groups (not weighted by population), as well as the counterfactual values, which are based on WHO recommendations and our tobacco risk reduction methods described above. The data suggests that too many South Africans do not eat any fruit or vegetables, although average consumption of fruit and vegetables is in line with recommendations. Furthermore, South Africans consume too much salt and alcohol, but not enough fiber, while too many South Africans smoke. Thus, the differences between baseline and counterfactual consumption are large in many cases.
When it comes to ‘bad’ risky consumption, our baseline data reveals that men consume more salt, alcohol and tobacco. On the other hand, when it comes to ‘good’ risky consumption, we see that men consume a larger amount of vegetables, and are more likely to consume vegetables than women. However, women consume more fruit and are more likely to consume fruit than men. Furthermore, men consume more fiber than women. Although, smoking prevalence and drinking are high, as discussed earlier, the baseline data suggests that fruit and vegetable consumption is inordinately low—and, as our results suggest—improvements in their consumption could be greater benefit—and to more people—than reducing alcohol and tobacco consumption.
4.2 Baseline incidence
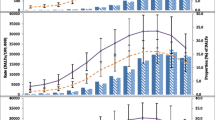
We begin by describing the baseline population disease incidence; see Fig. 1. It shows higher incidence of disease for women, which is primarily due to them making up a larger share of the relatively older population due to lower life expectancy for men, as well as representing all or nearly all cases of cervical and breast diseases (which represents approximately 15% of incidence). Total incidence in the data is 840 908, just over 355 000 for men and 485 000 for women, with the most common incidences being diabetes (20%), Ischaemic heart diseases (12%), chronic obstructive pulmonary disease (8%), breast disease (8%), cervical disease (7%), bronchus and lung disease (5%) and colorectum disease (5%). As we describe below, the counterfactual incidence suggests that male health benefits relatively more from the healthier consumption and behaviour choices underpinning the counterfactual scenario in the PRIME model.
4.3 Avertable incidence
As noted above, there are extensive differences between current South African consumption of salt, fruit, vegetables, fiber, alcohol and tobacco, relative to the recommendations. Inappropriate consumption of those goods has been linked to disease; thus, a revision in consumption towards recommendations could reduce disease. The difference in incidence between the baseline and the counterfactual is presented in Fig. 2. We refer to this as avertible incidence, as it arises from reductions in risky consumption behaviour. We further dissect Fig. 2, presenting avertable incidence by disease category in Fig. 3.
The total reduction in incidence arising from consumption modifications is close to 80 000; with baseline incidence near 840 900, that represents a decrease near 9.5% from baseline. The reduced incidence falls primarily within ischemic heart disease (49%), cerebrovascular diseases (33%) and bronchus and lung disease (11%).
Under our counterfactual scenario, given the relatively poor consumption of fruits and vegetables in the baseline data, the model finds that 49% of the reduction can be attributed to improved fruit and vegetable intake across a wider swathe of the population, while 21% can be attributed to a reduction in salt intake, 14% to reduction in smoking and smoking risk and 14% from improvements in fiber consumption.
4.4 Avertible incidence by gender
Given that women disproportionately suffered from disease, while, arguably, men were the worst consumption culprits, we briefly consider the improvements by gender; see Fig. 4.
Our baseline data suggested that approximately 90% of men and women were not eating at least one fruit per day, while between 50% (men) and 65% (women) were not eating at least one vegetable per day. Thus, reaching the counterfactual fruit and vegetable consumption level of one of each per day affects nearly everyone. On other hand, only about 38% of men and 7% of women were current smokers in the baseline data.
Such differences in the baseline explain why the reduction in incidence is dominated by the difference in behaviours by gender; thus, overall, the reduction in incidence is larger for men. Overall, there are 44 500 fewer cases for men and approximately 34 700 fewer cases for women, in the counterfactual. The reduction in incidence due to the reduction in smoking risk is 2.7 times larger for men than women; women were at least 5 times more likely to be current smokers, while we assumed an approximate 50% reduction in risk for those that remained smokers. For alcohol, the reduction is 12 times larger for men, while for salt, it is 1.3 times; the reduction to get to the counterfactual alcohol consumption was approximately 12 times larger for men than women, while the ratio of salt reduction required (3.75 for men and 2.99 for women) was approximately 1.3. These results and this discussion are suggestive of the likely impact of applying further sensitivity analysis by, for example, reducing the sale intake further or further reducing the risk associated with tobacco consumption. In what follows, we consider the latter.
4.5 Tobacco sensitivity
Finally, we offer insight into the sensitivity of our findings to assumptions related to tobacco consumption. Our initial assumption is based on a 50% risk reduction factor and 50% of smokers switching to less harmful alternatives; see Fig. 5, which leads to 10 881 fewer incidences. Doubling the risk reduction factor to 100%—indirectly implying half of all smokers would quit all nicotine products—leads to double the incidence reduction: 21 762 fewer incidences. Relative to the counterfactual avertible incidences, close to 80 000, the overall reduction in incidences would rise to 11% of the baseline, compared to the initially calculated 9.5%. Thus, further progress with regards to smoking cessation would pay substantial dividends.
5 Conclusion
5.1 Summary
South Africa is known for its quadruple burden of disease, which includes issues related to communicable diseases, NCDs, maternal and child health, as well as injury-related problems [5,6,7]. NCDs were amongst the top causes of death in South Africa in 2010, accounting for 12.9% of all deaths in 2008 rising to 17.6% in 2018; estimates suggest that they account for up to 30% of the DALYs [2, 15, 16, 102]. The government’s NCD plan is a response to the threat that NCDs pose on the health of the population, and, by extension, the healthcare system [32, 63, 64]. Similarly, the government has a variety of policies in place to regulate sugar, salt, tobacco and alcohol, although not all such policies had been fully implemented by 2021 [66,67,, 67, 73].
Despite those plans and policies, the initial baseline for our model points to a population consuming too much alcohol and salt, smoking too much, and not consuming enough fruit, vegetables or fiber. The model we have applied—the PRIME model—is used to examine the likely impact of reduced NCD risk consumption activities, such as a reduction in tobacco, alcohol and salt or an increase in fruits, vegetables and fiber [88]. The reductions that we assume are in line with WHO recommendations, and match the government’s NCD plans.
Our baseline data suggests that NCD incidences are higher for women than for men, primarily due to cervical and breast diseases, despite the fact that NCD behaviours are riskier for men than for women. After applying our counterfactual scenario, we find an approximate 10% reduction in NCD incidence. However, due to the fact that NCD risky behaviour was initially higher for men, they would need to reduce the risks associated with their behaviour by more than women would; thus, we find that NCD incidence falls by more for men than women.
5.2 Policy implications
Political action is required to realise the potential gains from a significant reduction in NCD incidence from less harmful consumption. An appropriate policy approach that incentivizes South Africans to reduce unhealthy consumption is crucial to improving public health. This approach does not necessarily require costly interventions. For example, restricting alcohol consumption and smoking are relatively easy objectives to reach. Tobacco taxes have been successful in reducing smoking in the country [103]. However, there is also evidence of extensive illicit cigarette sales [47, 48]. Policymakers could and should do more to control the illicit market. Simultaneously, they should promote less harmful alternatives, such as e-cigarettes, heat-not-burn products, and low-alcohol beverages. Incentivizing less harmful alternatives implies lower taxation compared to their more harmful counterparts. By doing so, consumers would be incentivized to consume those less harmful alternatives, rather than purchasing illicit products of questionable quality. Individual health would improve as a result, reducing NCDs. Additionally, the state treasury could see a much-needed improvement thanks to the partial recovery of South Africa’s tax losses from illicit trade.
As noted in the study, South Africans do not consume fruits and vegetables often enough. Thus, working to improve diet diversity—with a focus on fruits, vegetables and other sources of fiber—in the country is an important policy goal, as well. While a change in dietary lifestyle primarily depends on income levels and prices, which are already subsidized for the poor through social grants and zero-rated value-added taxes. Additional research suggests that such subsidies are successful [103]. However, grants and subsidies are costly, especially when one considers the consumption gap. Thus, further research is needed to find less expensive ways to promote the consumption of better foods.
Data availability
The data used for this model is publicly available, as is the PRIME model. However, in order to use the data, which is "housed" by others, the potential user must create an account and register.
References
World Health Organization. Noncommunicable diseases country profiles 2018. Geneva: WHO; 2018.
Allen LN, Feigl AB. What’s in a name? A call to reframe non-communicable diseases. Lancet Glob Health. 2017;5:129–30. https://doi.org/10.1016/S2214-109X(17)30001-3.
World Health Organization. Non‐Communicable disease, country profiles: South Africa. 2018. https://www.who.int/nmh/countries/2018/zaf_en.pdf?ua=1.
Alwan A. Global status report on non-communicable diseases 2010. Geneva: World Health Organization; 2011.
Mayosi BM, Flisher AJ, Lalloo UG, et al. The burden of non-communicable diseases in South Africa. Lancet. 2009;374:934–47. https://doi.org/10.1016/S0140-6736(09)61087-4.
Maimela E, Alberts M, Modjadji SEP, et al. The Prevalence and determinants of chronic non-communicable disease risk factors amongst adults in the Dikgale health demographic and surveillance system (HDSS) site, Limpopo Province of South Africa. PLoS ONE. 2016;11(2):e0147926. https://doi.org/10.1371/journal.pone.0147926.
Pillay-van Wyk V, Msemburi W, Laubscher R, et al. Mortality trends and differentials in South Africa from 1997 to 2012: second national burden of disease study. Lancet Glob Health. 2016;4(9):e642–53. https://doi.org/10.1016/S2214-109X(16)30113-9.
Nojilana B, Bradshaw D, Pillay-van Wyk V, et al. Emerging trends in non-communicable disease mortality in South Africa, 1997–2010. S Afr Med J. 2016;106(5):477–84. https://doi.org/10.7196/samj.2016.v106i5.10674.
Berkowitz N, Okorie A, Goliath R, et al. The prevalence and determinants of active tuberculosis among diabetes patients in Cape Town, South Africa, a high HIV/TB burden setting. Diabetes Res Clin Pract. 2018;138:16–25. https://doi.org/10.1016/j.diabres.2018.01.018.
Folb N, Timmerman V, Levitt NS, et al. Multimorbidity, control and treatment of noncommunicable diseases among primary healthcare attenders in the Western Cape, South Africa. South Afr Med J. 2015;105(8):642–7. https://doi.org/10.7196/SAMJNEW.7882.
Murphy A, Palafox B, Walli-Attaei M, et al. The household economic burden of non-communicable diseases in 18 countries. BMJ Glob Health. 2020;5:e002040. https://doi.org/10.1136/bmjgh-2019-002040.
Isaacs AA, Manga N, Le Grange C, et al. A snapshot of noncommunicable disease profiles and their prescription costs at ten primary healthcare facilities in the in the western half of the Cape Town Metropole. South Afr Family Pract. 2014;56(1):43–9. https://doi.org/10.1080/20786204.2014.10844582.
Lee JT, Hamid F, Pati S, et al. Impact of noncommunicable disease multimorbidity on healthcare utilisation and out-of-pocket expenditures in middle-income countries: cross sectional analysis. PLoS ONE. 2015;10(7):e0127199. https://doi.org/10.1371/journal.pone.0127199.
Roomaney RA, van Wyk B, Cois A, et al. Inequity in the distribution of non-communicable disease multimorbidity in adults in South Africa: an analysis of prevalence and patterns. Int J Public Health. 2022;67:1605072. https://doi.org/10.3389/ijph.2022.1605072.
Norman R, Schneider M, Bradshaw D, et al. Interpersonal violence: an important risk factor for disease and injury in South Africa. Popul Health Metrics. 2010;8:32. https://doi.org/10.1186/1478-7954-8-32.
Haakenstad A, Coates M, Bukhman G, et al. Comparative health systems analysis of differences in the catastrophic health expenditure associated with non-communicable vs communicable diseases among adults in six countries. Health Policy Plan. 2022;37(9):1107–15. https://doi.org/10.1093/heapol/czac053.
Silvaggi F, Leonardi M, Guastafierro E, et al. Chronic diseases & employment: an overview of existing training tools for employers. Int J Environ Res Public Health. 2019;16:718. https://doi.org/10.3390/ijerph16050718.
Pillay-van Wyk V, Msemburi W, Laubscher R, et al. Mortality trends and differentials in South Africa from 1997 to 2012: second national burden of disease study. Lancet Global Health. 2016;4(9):e642-653. Erratum in: Lancet Global Health. 2017;5(3):e275. https://doi.org/10.1016/S2214-109X(16)30113-9.
Mashinya F, Alberts M, Van Geertruyden JP, et al. Assessment of cardiovascular risk factors in people with HIV infection treated with ART in rural South Africa: a cross sectional study. AIDS Res Ther. 2015;12:42. https://doi.org/10.1186/s12981-015-0083-6.
Mosomi JW. The labor market in South Africa 2000–2017. IZA World of Labor. 2020. https://doi.org/10.15185/izawol.475.
Finn A. A national minimum wage in the context of the South African labour market. National Minimum Wage Research Initiative Working Paper Series No. 1. University of the Witwatersrand. 2015.
Stringhini S, Carmeli C, Jokela M, et al. Socioeconomic status and the 25 × 25 risk factors as determinants of premature mortality: a multicohort study and meta-analysis of 17 million men and women. Lancet. 2017;389:1229–37. https://doi.org/10.1016/S0140-6736(16)32380-7.
Sulla V, Zikhali P. Overcoming poverty and inequality in South Africa: an assessment of drivers, constraints and opportunities. Washington, DC: The World Bank; 2018. https://doi.org/10.1596/29614.
Samodien E, Abrahams Y, Muller C, et al. Non-communicable diseases-a catastrophe for South Africa. South Afr J Sci. 2021. https://doi.org/10.17159/sajs.2021/8638.
Ataguba JE, Akazili J, McIntyre D. Socioeconomic-related health inequality in South Africa: evidence from general household surveys. Int J Equity Health. 2011. https://doi.org/10.1186/1475-9276-10-48.
Spaull N. Poverty & privilege: primary school inequality in South Africa. Int J Educ Dev. 2013;33(5):436–47. https://doi.org/10.1016/j.ijedudev.2012.09.009.
May J, Timæus IM. Inequities in under-five child nutritional status in South Africa: what progress has been made? Dev South Afr. 2014;31(6):761–74. https://doi.org/10.1080/0376835X.2014.952896.
Jonah CM, May JD. Evidence of the existence of socioeconomic-related inequality South African diets: a quantitative analysis of the 2017 General Household Survey. World Nutr. 2019;10(4):27–42. https://doi.org/10.26596/wn.201910427-42.
Koch SF. Equivalence scales in a developing country with extensive inequality. South Afr J Econ. 2022;90(4):486–512. https://doi.org/10.1111/saje.12326.
Koch SF. Basic needs (in)security and subjective equivalence scales. Soc Indic Res. 2023;169(3):723–57. https://doi.org/10.1007/s11205-023-03178-7.
Ye Y, Koch SF. Towards accessibility or affordability? Multidimensional energy poverty across the South African urban–rural divide. Energy Res Soc Sci. 2023. https://doi.org/10.1016/j.erss.2023.103002.
Day C, Groenewald P, Laubscher R, et al. Monitoring of non-communicable diseases such as hypertension in South Africa: challenges for the post-2015 global development agenda. S Afr Med J. 2014;104(10):680–7. https://doi.org/10.7196/SAMJ.7868.
Igumbor EU, Sanders D, Puoane TR, et al. “Big food”, the consumer food environment, health, and the policy response in South Africa. PLoS Med. 2012;9:e1001253. https://doi.org/10.1371/journal.pmed.1001253.
Shisana O, Labadarios D, Rehle T, et al. South African National Health and Nutrition Examination Survey (SANHANES-1). Cape Town: HSRC Press; 2013.
Freudenberg N. Lethal but Legal: corporations, consumption, an protecting public health. Oxford: Oxford University Press; 2014.
Charlton KE, Steyn K, Levitt NS, et al. Diet and blood pressure in South Africa: intake of foods containing sodium, potassium, calcium, and magnesium in three ethnic groups. Nutrition. 2005;21:39–50. https://doi.org/10.1016/j.nut.2004.09.007.
Marquez PV, Farrington JL. The challenge of non-communicable diseases and road traffic injuries in Sub-Saharan Africa: an overview. Washington, DC: The World Bank; 2013.
McIntyre D. The public-private health sector mix in South Africa. In: health care financing in South Africa. Cape Town: Health Economics Unit University of Cape Town; 2009.
Goudge J, Gilson L, Russell S, et al. The household costs of health care in rural South Africa with free public primary care and hospital exemptions for the poor. Tropical Med Int Health. 2009;14:458–67. https://doi.org/10.1111/j.1365-3156.2009.02256.x.
Harris B, Goudge J, Ataguba JE, et al. Inequities in access to health care in South Africa. J Public Health Policy. 2011;32:S102-123. https://doi.org/10.1057/jphp.2011.35.
Bertram MY, Tollman S, Hofman KJ, et al. Reducing the sodium content of high-salt foods: effect on cardiovascular disease in South Africa. S Afr Med J. 2012;102:743–5. https://doi.org/10.7196/SAMJ.5832.
Watkins DA, Olson ZD, Verguet S, et al. Cardiovascular disease and impoverishment averted due to a salt reduction policy in South Africa: an extended cost-effectiveness analysis. Health Policy Plan. 2016;31(1):75–82. https://doi.org/10.1093/heapol/czv023.
Groenewald P, Vos T, Norman R, et al. Estimating the burden of disease attributable to smoking in South Africa in 2000. S Afr Med J. 2007;97(8 Pt 2):674–81. https://doi.org/10.7196/SAMJ.661.
Sitas F, Urban M, Bradshaw D, et al. Tobacco attributable deaths in South Africa. Tob Control. 2004;13:396–9. https://doi.org/10.1136/tc.2004.007682.
Sitas F, Egger S, Bradshaw D, et al. Differences among the coloured, white, black, and other South African populations in smoking-attributed mortality at ages 35–74 years: a case-control study of 481 640 deaths. The Lancet. 2013;382(9893):685–93. https://doi.org/10.1136/tc.2004.007682.
Boachie MK, Rossouw L, Ross H. The economic cost of smoking in South Africa, 2016. Nicotine Tob Res. 2021;23(2):286–93. https://doi.org/10.1093/ntr/ntaa162.
Vellios N, van Walbeek C, Ross H. Illicit cigarette trade in South Africa: 2002–2017. Tob Control. 2020;29:s234-242. https://doi.org/10.1136/tobaccocontrol-2018-054798.
Vellios N. How big is the illicit cigarette market in South Africa?. Econ3x3. 2022. https://www.econ3x3.org/article/how-big-illicit-cigarette-market-south-africa#.
Pearce A, Sharp L, Hanly P, et al. Productivity losses due to premature mortality from cancer in Brazil, Russia, India, China, and South Africa (BRICS): a population-based comparison. Cancer Epidemiol. 2018;53:27–34. https://doi.org/10.1016/j.canep.2017.12.013.
World Health Organization. Global status report on alcohol and health 2018. Geneva: World Health Organization; 2019.
Vellios NG, Van Walbeek CP. Self-reported alcohol use and binge drinking in South Africa: evidence from the national income dynamics study, 2014–2015. South Afr Med J. 2018. https://doi.org/10.7196/SAMJ.2018.v108i1.12615.108(1):33-39.
World Health Organization. Global status report on alcohol and health. Geneva: World Health Organization; 2014.
Witt D, Nagy J. Understanding the drivers of illicit alcohol: an analysis of selected country case studies. World Customs J. 2022;16(2):81–98.
Press Release UN. Non-communicable diseases deemed development challenge of “epidemic proportions” in political declaration adopted during landmark General Assembly summit. 2011. Available from: https://press.un.org/en/2011/ga11138.doc.htm#:~:text=Proclaiming%20the%20spread%20of%20non,to%20devise%20voluntary%20policies%20that.
Organization of African Unity. Abuja declaration on HIV/AIDS, Tuberculosis and other related infectious diseases. 2001. Available from: http://www.un.org/ga/aids/pdf/abuja_declara. https://au.int/sites/default/files/pages/32894-file-2001-abuja-declaration.pdf.
World Health Organization. First global ministerial conference on healthy lifestyles and noncommunicable disease control. Moscow: World Health Organization; 2011. p. 28–9.
Xu K, Evans DB, Kawabata K, et al. Household catastrophic health expenditure: a multicountry analysis. Lancet. 2003;362(9378):111–7. https://doi.org/10.1016/S0140-6736(03)13861-5.
Abegunde DO, Mathers CD, Adam T, et al. The burden and costs of chronic diseases in low-income and middle-income countries. Lancet. 2007;370(9603):1929–38. https://doi.org/10.1016/S0140-6736(07)61696-1.
Ebrahim S, Pearce N, Smeeth L, et al. Tackling non-communicable diseases in low-and middle-income countries: Is the evidence from high-income countries all we need? PLoS Med. 2013;10(1):e1001377. https://doi.org/10.1371/journal.pmed.1001377.
Chandra A, Gruber J, McKnight R. Patient cost-sharing and hospitalization offsets in the elderly. Am Econ Rev. 2010;100(1):193–213. https://doi.org/10.1257/aer.100.1.193.
Trivedi AN, Moloo H, Mor V. Increased ambulatory care co-payments and hospitalizations among the elderly. N Engl J Med. 2010;362(4):320–8. https://doi.org/10.1056/NEJMsa0904533.
Choudhry NK, Avorn J, Glynn RJ, et al. Full coverage for preventive medications after myocardial infarction. N Engl J Med. 2011;365(22):2088–97. https://doi.org/10.1056/NEJMsa1107913.
National Department of Health. Strategic Plan for the Prevention and Control of Non-Communicable Diseases 2013-17. Pretoria: NDoH, 2013.
Cecchini M, Sassi F, Lauer JA, et al. Tackling of unhealthy diets, physical inactivity, and obesity: health effects and cost-effectiveness. Lancet. 2010;376(9754):1775–84. https://doi.org/10.1016/S0140-6736(10)61514-0.
Ndinda C, Ndhlovu TP, Juma P, et al. The evolution of non-communicable diseases policies in post-apartheid South Africa. BMC Public Health. 2018;18(Supplement 1):956. https://doi.org/10.1186/s12889-018-5832-8.
Uwimana-Nicol J, Hendricks L, Young T. Population-level interventions targeting risk factors of diabetes and hypertension in South Africa: a document review. BMC Public Health. 2021;21(1):2283. https://doi.org/10.1186/s12889-021-11910-6.
Reddy PJ, Sewpaul R, Sifunda S, et al. A decade of tobacco control: the South African case of politics, health policy, health promotion and behaviour change. S Afr Med J. 2013;103:835–40. https://doi.org/10.7196/samj.6910.
World Health Organization (WHO). Non-communicable disease prevention and control, risk factor reduction, mental health and injury and violence. Geneva: World Health Organization; 2016.
World Health Organization (WHO). WHO Country Sooperation strategy 2016–2020 South Africa. Geneva: World Health Organization; 2016.
World Health Organization (WHO). Noncommunicable diseases (NCD) country profiles: South Africa. Geneva: World Health Organization; 2014.
Ndinda C, Hongoro C, Labadarios D, et al. Baseline assessment for future impact evaluation of informal settlements targeted for upgrading. Pretoria: Human Science Research Council; 2016.
Boseley, S. Threats, bullying, lawsuits: tobacco industry’s dirty war for the African market. 2017. Available from: https://www.theguardian.com/world/2017/jul/12/big-tobacco-dirty-war-africa-market.
Ndinda C, Hongoro C. Analysis of non-communicable diseases prevention policies in Africa-A case study of South Africa. Pretoria: Human Science Research Council; 2017.
Yusuf S, Hawken S, Ounpuu S, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet. 2004;364(9438):937–52. https://doi.org/10.1016/S0140-6736(04)17018-9.
Afroz A, Alramadan MJ, Hossain MN, et al. Cost-of-illness of type 2 diabetes mellitus in low and lower-middle income countries: a systematic review. BMC Health Serv Res. 2018;18:1. https://doi.org/10.1186/s12913-018-3772-8.
Brouwer ED, Watkins D, Olson Z, et al. Provider costs for prevention and treatment of cardiovascular and related conditions in low-and middle-income countries: a systematic review. BMC Public Health. 2015;15:1183. https://doi.org/10.1186/s12889-015-2538-z.
Aminde LN, Takah NF, Zapata-Diomedi B, et al. Primary and secondary prevention interventions for cardiovascular disease in low-income and middle-income countries: a systematic review of economic evaluations. Cost Eff Resour Allocat. 2018. https://doi.org/10.1186/s12962-018-0108-9.
Gheorghe A, Griffiths U, Murphy A, et al. The economic burden of cardiovascular disease and hypertension in low-and middle-income countries: a systematic review. BMC Public Health. 2018;18(1):975. https://doi.org/10.1186/s12889-018-5806-x.
Husereau D, Drummond M, Petrou S, et al. Consolidated health economic evaluation reporting standards (CHEERS) statement. Value Health. 2013;16(2):e1-5. https://doi.org/10.3111/13696998.2013.784591.
International Monetary Fund. World economic outlook‐ inflation rate, average consumer prices. 2021. https://www.imf.org/external/datamapper/PCPIPCH@WEO/ZAF?zoom=ZAF&highlight=ZAF.
Day K, Booyens S. The cost-effectiveness of managed care regarding chronic medicine prescriptions in a selected medical scheme. Curationis. 1998;21(4):65–70. https://doi.org/10.4102/curationis.v21i4.684.
Suhrcke M, Boluarte TA, Niessen L. A systematic review of economic evaluations of interventions to tackle cardiovascular disease in low-and middle-income countries. BMC Public Health. 2012;12(1):2. https://doi.org/10.1186/1471-2458-12-2.
Edwards PR, Lunt DW, Fehrsen GS, et al. Improving cost-effectiveness of hypertension management at a community health centre. S Afr Med J. 1998;88(5):549–53.
Anderson AN, Wessels F, Moodley I, et al. AT1 receptor blockers-cost-effectiveness within the South African context. S Afr Med J. 2000;90(5):494–8.
Ker JA, Oosthuizen H, Rheeder P. Decision-making using absolute cardiovascular risk reduction and incremental cost-effectiveness ratios: a case study. Cardiovasc J Afr. 2008;19(2):97–101.
Ataguba J. Health care financing in South Africa: moving towards universal coverage. Continuing Med Edu. 2010;28:74–8.
Matsoso MP, Fryatt R. National health insurance: the first 18 months. S Afr Med J. 2013;103:156–8.
Scarborough P, Harrington RA, Mizdrak A, et al. The preventable risk integrated ModEl and its use to estimate the health impact of public health policy scenarios. Scientifica. 2014. https://doi.org/10.1155/2014/748750.
Rose D, Bourne LA, Bradshaw D. Food and nutrient availability in South African households: Development of a nationally representative database. Technical Report: South African Medical Research Council, Health and Development Research Group and the Burden of Disease Research Unit. 2002. Available from: https://www.samrc.ac.za/sites/default/files/attachments/2022-08/foodnutrientavail.pdf.
Eksteen G, Mungal-Singh V. Salt intake in South Africa: a current perspective. J Endocrinol Metab Diabetes S Afr. 2015;20(1):9–14. https://doi.org/10.1080/16089677.2015.1030878.
World Health Organization. NCDprime: modelling the impact of national policies on noncommunicable disease (NCD) mortality using PRIME: a policy scenario modelling tool. No. WHO/EURO: 2019-3652-43411-60952. 2019.
Van Heerden IV, Parry CD. If you drink alcohol, drink sensibly. South Afr Food-Based Dietary Guidelines. 2001;14(3):71–7.
World Health Organization, Food and Agriculture Organization. Population nutrient intake goals for preventing diet-related chronic diseases. 2002. https://www.fao.org/3/AC911E/ac911e07.htm. Accessed Feb 2024.
Nutt D, Philips L, Balfour D, et al. Estimating the harms of nicotine-containing products using the MCDA approach. Eur Addict Res. 2014;20(5):e218-225. https://doi.org/10.1159/000360220.
Forster M, Fiebelkorn S, Yurteri C, et al. Assessment of novel tobacco heating product THP1. 0. Part 3: comprehensive chemical characterisation of harmful and potentially harmful aerosol emissions. Regul Toxicol Pharmacol. 2018;93:14–33. https://doi.org/10.1016/j.yrtph.2017.10.006.
Mallock N, Böss L, Burk R, et al. Levels of selected analytes in the emissions of “heat not burn” tobacco products that are relevant to assess human health risks. Arch Toxicol. 2018;92(6):2145–9. https://doi.org/10.1007/s00204-018-2215-y.
Slob W, Soeteman-Hernández LG, Bil W, et al. A method for comparing the impact on carcinogenicity of tobacco products: a case study on heated tobacco versus cigarettes. Risk Anal. 2020;40(7):1355–66. https://doi.org/10.1111/risa.13482.
Jaccard G, Tafin D, Moennikes O, et al. Comparative assessment of HPHC yields in the Tobacco Heating System THS2.2 and commercial cigarettes. Regul Toxicol Pharmacol. 2017;90:1–8. https://doi.org/10.1016/j.yrtph.2017.08.006.
Stephens W. Comparing the cancer potencies of emissions from vapourised nicotine products including e-cigarettes with those of tobacco smoke. Tob Control. 2018;27:10–7. https://doi.org/10.1136/tobaccocontrol-2017-053969.
Bekki K, Inaba Y, Uchiyama S, et al. Comparison of chemicals in mainstream smoke in heat-not-burn tobacco and combustion cigarettes. J UOEH. 2017;39(3):201–7. https://doi.org/10.7888/juoeh.39.201.
Pichon-Riviere A, Alcaraz A, Palacios A, et al. The health and economic burden of smoking in 12 Latin American countries and the potential effect of increasing tobacco taxes: an economic modelling study. Lancet Glob Health. 2020;8(10):e1282-1294. https://doi.org/10.1016/S2214-109X(20)30311-9.
Statistics South Africa. Non-communicable diseases in South Africa 2008–2018. Report No. 03-08-01. Pretoria: Statistics South Africa; 2023.
Chelwa G, van Walbeek C, Blecher E. Evaluating South Africa’s tobacco control policy using a synthetic control method. Tobacco Control. 2017;26:509–17. https://doi.org/10.1136/tobaccocontrol-2016-053011.
Data References
Global Adult Tobacco Survey Collaborative Group. Cigarette prevalence in South Africa. Global Adult Tobacco Survey (GATS). 2021. https://southafrica.tobaccocontroldata.org/en/home/prevalence/.
Global Dietary Database. https://globaldietarydatabase.org/. Accessed Feb 2024.
National Department of Health (NDOH), Statistics South Africa (Stats SA), South African Medical Research Council (SAMRC), and ICF. South Africa demographic and health survey 2016. NDOH Stats SA, SAMRC and ICF Pretoria, South Africa, and Rockville. 2019. https://dhsprogram.com/data/dataset/South-Africa_Standard-DHS_2016.cfm?flag=0.
The World bank data. Available from: https://data.worldbank.org/https://data.worldbank.org/indicator/SP.POP.TOTL?locations=ZA&skipRedirection=true.
Statistics South Africa. South African Census 2011: population dynamics in South Africa 2015. Pretoria, Republic of South Africa. Available from: http://www.statssa.gov.za/publications/Report-03-01-67/Report-03-01-672011.pdf.
Institute for Health Metrics and Evaluation. Global burden of disease data. 2018. https://www.healthdata.org/research-analysis/gbd. Accessed Feb 2024.
Robert Koch Institute. Survival Data. Available from: https://www.krebsdaten.de/Krebs/SiteGlobals/Forms/Datenbankabfrage/EN/datenbankabfrage_stufe2_form.html.
Acknowledgements
I would like to thank the reviewers for their suggestions. I believe the manuscript is better for them.
Author information
Authors and Affiliations
Contributions
Steven F. Koch undertook the research for the manuscript, prepared tables and figures, and reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This research is based on publicly available secondary data. Ethics clearance was not sought for the research.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koch, S.F. Examining potential health gains arising from reduced risk consumption in South Africa via the PRIME model. Discov Soc Sci Health 4, 33 (2024). https://doi.org/10.1007/s44155-024-00093-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44155-024-00093-1