Abstract
Purpose
Craniometaphyseal dysplasia (CMD) is a rare skeletal disorder that has progressive thickening and increased density of the craniofacial bones along with abnormal metaphyses of the long bones. Other features include a wide nasal bridge, paranasal bossing, hypertelorism, and an increase in the zygomatic width. We present a series of cases that showcase the three-dimensional radiographic features of this rare condition and compare it with an age and gender-matched controlled group using cone beam computed tomography (CBCT).
Objective
To evaluate the three-dimensional radiographic features of craniometaphyseal dysplasia (CMD).
Materials and methods
Retrospective analysis of CBCT scans of 7 patients who were diagnosed with the rare condition craniometaphyseal dysplasia was evaluated. Radiographic features of the craniofacial bones, the paranasal sinuses prominently maxillary, and frontal and sphenoid sinuses were evaluated. Skull bones were also evaluated, and the size and shape of the sella turcica and external auditory meatus were measured. Retained primary teeth and impactions were evaluated using a panoramic reformatted image. The type of occlusion and buccolingual dimensions of jaw bones were also assessed. Age and gender-matched control samples were used to measure the same features for comparing CMD patients to healthy controls. The quality of normal and syndromic patients’ cortical and trabecular bone was determined by measuring pixel intensity values (PIV) generated by CBCT scans.
Results
Cone beam CT images of patients with CMD were analyzed to evaluate cranial bones, their density, and any abnormalities associated with the sinuses and foramina. Patients with CMD had bones increased in size. A significant increase in the amount of bone formed was found in the inner table of the frontal and occipital bones. All seven patients with CMD had smaller foramina due to the deposition of sclerosed bone in the foramina of the skull base. The paranasal sinuses, prominently maxillary and frontal and sphenoid sinuses, were smaller than the age and gender-matched controls. The buccolingual dimensions of jawbones were increased. The maxillary and mandibular arch relationship ranged from class II to class III. The mean nasal bridge measurement for the CMD patients was 26.77 mm, while in the controls, the mean nasal bridge measurement was 19.48 mm. The mean measurements of the right and left orbits of CMD patients were 30.6 mm and 31.07 mm respectively, and the mean measurements of the right and left orbits of controls were 32.45 mm and 32.04 mm. Pixel intensity values (PIVs) representing density ranged between 100 and 1000 PIVs for cortical bone and between − 60 and 258 for trabecular bone suggesting a densely sclerotic texture, while in the control group patients, the PIVs for cortical bone were > 1000 and 150–300 for trabecular bone.
Conclusion
CMD patients had significantly larger bone widths, a lower density of the bone, and smaller sinuses compared to the control group.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Craniometaphyseal dysplasia (CMD) is a rare skeletal disorder that has progressive thickening and increased density of the craniofacial bones along with abnormal metaphyses of the long bones [1]. Jackson et al. in 1954 first described this disease [2]. Patients with this condition have progressive hyperostosis of craniofacial bones clinically evident as a wide nasal bridge, paranasal bossing, and hypertelorism with an increase in bi-zygomatic width, along with a prominent mandible [3]. In this condition, there is an increased deposition of ossified bone, and a failure of its breakdown prevents the normal remodeling of the long bones, the skull base, and the facial bones causing progressive expansion of bones [4].
CMD is diagnosed during early childhood and monitoring of the features continues during the lifetime as there is an increase in the density of craniofacial bones and the metaphysis, most often starting at the skull base. Sclerosis and progressive thickening of the bones may cause narrowing and obliteration of the cranial foramina and can lead to abnormalities of the corresponding nerve that is getting compressed due to the obliteration of the foramen and can cause both visual and neurological impairments like facial palsy, blindness, and deafness.
Dysfunction of the osteoblast/osteoclast is attributed to the possible pathophysiology of this condition [5]. CMD can be classified into the autosomal dominant form (ANKH) and autosomal recessive form (GJA1). The ANKH gene is linked with the autosomal dominant form and expressivity is variable. The function of ANKH is to move a chemical called pyrophosphate in the body [6]. The recessive form is rare and has been mapped to chromosome 6q21–22. The function of GJA1 is to create a passage for small molecules between cells [7].
Progressive facial weakness, frontonasal hyperostosis, and sclerosis of the forehead are the typical clinical manifestations of this disease. Excessive bone formation is one of the striking features of this condition; other facial features include prognathism, a flattened nasal bridge, and frontonasal bossing [2]. Increased density of craniofacial bones is a characteristic feature and is initially noted at the base of the skull. CMD is diagnosed during early childhood, and it has been observed that the features become more prominent throughout life, often resulting in the narrowing of the cranial foramina, including the foramen magnum. If untreated, bone deposition leads to foraminal obliteration that can compress cranial nerves and can lead to disabling conditions such as facial palsy, blindness, or deafness [3]. Leontiasis ossea (lion face) was used to describe this severe facial abnormality in early literature; the majority of patients with ANKH mutations present with severe craniosclerosis and less severe facial abnormalities. Metaphyses of long bones are widened in CMD patients; they were described as Erlenmeyer flask deformities. These changes at the metaphyses of long bones in children mimic the symptoms of rickets and must be considered during diagnosis and treatment planning [3,4,5,6,7].
Patients with CMD may have a delay in dentition development, and teeth may fail to erupt because of hyperostosis and sclerosis of the alveolar bone. These patients require various dental procedures like multiple extractions, tooth exposures, and orthodontic treatment. The orthodontic tooth movement involves bone remodeling by osteoclastic-initiated resorption which happens on the pressure side, while osteoblast-initiated bone formation occurs on the tension side. This imbalance causes a delay in orthodontic tooth movement [8].
Currently, the treatment for CMD is dependent on the location of excessive bone deposition and the associated symptoms and includes multiple surgeries like relieving foraminal stenosis and bone augmentation procedures that may have to be repeated several times throughout life. Also, these patients require multiple progressive radiographic evaluations exposing them to large doses of ionizing radiation that is harmful to the patient. While there is uncertainty about the risks following lower doses from CT scans (5–50 mGy per organ), in a study conducted by Mathews et al., in 680,000 Australians exposed to a CT scan when aged 0–19 years, cancer incidence increased by 24% compared with the incidence in over 10 million unexposed people. The proportional increase in risk was evident at short intervals after exposure and was greater for persons exposed at younger ages [8]. This is an important consideration for clinicians to use lower dose options when available and deemed appropriate for the diagnostic task.
Cone-beam computed tomography (CBCT) is increasingly used to acquire three-dimensional information about the maxillofacial skeleton as it exposes patients to a lower radiation dose compared the multi-detector medical CT. It is mainly used for dental and craniofacial diagnoses because it provides high-resolution 3D images, and the cross-sectional images rendered by CBCT can be used to study structures of dentition, craniofacial bones, and sinuses. Evaluation of bone density in CBCT scans can be done by measuring pixel intensity values (PIVs).
In CMD patients, CBCT helps in evaluating the dimensions of the bones and obliteration of any foramina, and, in this particular case, we noted several impacted teeth and CBCT imaging provides an opportunity to study these teeth and their relationships to adjacent structures and for orthodontic treatment planning purposes. Compared to other advanced modalities, CBCT provides all the information about the gnathic structures and particularly the osseous components in high resolution and low dose. Because of this, it is possible to retake scans at a later time point to monitor the progress of the condition or any treatment rendered.
Three-dimensional radiographic features of this rare genetic disorder have not been sufficiently investigated. The entire healthcare team needs to get adequate information about the status of the involved areas to provide a comprehensive treatment plan. The evolution of low-cost, low-dose CBCT scanners has made it possible to image and document the radiographic features of this rare disease to understand it better.
The objective of this study was to evaluate and compare the three-dimensional radiographic features of craniometaphyseal dysplasia (CMD) with a normal population using cone-beam computed tomography imaging.
Materials and methods
The present study is an evaluation of CBCT scans of 7 patients who were diagnosed with CMD by genetic testing and were found to have known autosomal dominant ANK mutations. These CMD patients were referred to the UConn School of Dental Medicine for diagnosis and treatment planning for dental-related problems. Age and gender-matched control samples were used to evaluate the same features for comparing patients with this syndrome to normal age- and gender-matched controls. In this study, a large field-of-view CBCT scan that covers the entire maxilla, mandible, and craniofacial bones including the sinuses and sinonasal complex was used.
CBCT analysis
All the images were obtained by using the Hitachi MercuRay CBCT machine (Hitachi, Tokyo, Japan). Exposure parameters were 100 kVp and 10 mA with a 10-s scan time. CB Works version 3.0, a CBCT reconstruction software program (Cyber Med, Seoul, Korea), was used to evaluate the scans in the multiplanar reformation (MPR) mode. The MPR mode displayed the area of interest in axial, sagittal, and coronal planes. A coronal plane that best depicts both cranial and jaw bones was used for the measurement of pixel intensity values (PIVs), which reflect bone density.
Panoramic, cephalometric, and cross-sectional views were generated for further analysis of dental structures. The radiographic features like patency and size of the paranasal sinuses and size of temporal, frontal, parietal, and occipital bones were evaluated. Foramina in the skull base, external auditory meatus, and sella turcica were evaluated. Along with these, dental abnormalities, impactions, and type of occlusion were assessed. The bone quality of normal and syndromic patients was evaluated by measuring pixel intensity (PIV) values generated by the CBCT reconstruction program.
Statistical analysis
To analyze the data, descriptive analysis and unpaired t-test (p-values) were used. Among the raters, intra-rater and inter-rater agreement for the image analysis and measurements were done using kappa analyses.
Results
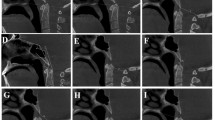
Analysis of the CBCT scans showed that the bone density in seven patients with CMD appeared to be altered in both cortical and trabecular bones but reduced in the trabecular compartment of cranial and maxillofacial bones. Figures 1 and 2 show the differences in the buccolingual width (1a, 2a), sizes of maxillary sinuses (1b, 2b), the shape of the sella turcica, (1c, 2c), and the opacification of mastoid air cells in CMD patients (Fig. 2d).
CBCT imaging of dentofacial examination of patients with craniometaphyseal dysplasia. a Axial view showing the buccolingual width. b Sagittal section showing the alteration in the shape of the sella turcica (circle). c Coronal section showing the reduction in size of maxillary sinus. d Axial section of CBCT showing the hypo pneumatization of the mastoid air cells (arrows)
All CMD patients showed hyperostotic cranial bones. The frontal and occipital bones had a significant deposition of bone on the inner table. All seven patients with CMD had smaller foramina due to increased bone deposition in foramina of the skull base. The paranasal sinuses, especially the maxillary, frontal and sphenoid sinuses, were smaller than the gender-age-matched controls. The buccolingual dimensions of jawbones were markedly increased. The relationship between the maxilla and mandible in the anterior–posterior axis was highly variable, from class II (4 CMD patients) to class III (3 CMD patients). The mean nasal bridge measurement for the CMD patients was 26.77 mm, while in the controls, the mean nasal bridge measurement was 19.48 mm. The mean measurements of the right and left orbits of CMD patients were 30.6 mm and 31.07 mm respectively, and the mean measurements of the right and left orbits of the controls were 32.45 mm and 32.04 mm.
Pixel intensity values (PIVs) derived from CBCT volumes to evaluate the quality of bone was between the range of 100 and 1000 for cortical bone and between − 60 and 258 for trabecular bone in these patients suggesting densely sclerotic texture, while in the normal or control group patients, the PIVs of cortical bone was > 1000 and 150–300 for trabecular bone. Table 1 and Fig. 3 shows the CBCT measurements of parameters among CMD patients and the normal control group.
Some of the CMD patients had retained deciduous teeth, and the permanent teeth were impacted due to the delay in the eruptive mechanism. Figure 4 shows a panoramic view of a CMD patient with multiple impactions in the maxilla and mandible with retained deciduous teeth. This finding was insignificant among the healthy control patients.
Discussion
The dentofacial anomalies in patients with CMD occur at different ages of their life. The dentist needs to recognize the symptoms and be familiar with treatment options and potential complications. Awareness of the possible symptoms and challenges will help the dentist assemble a craniofacial team to provide the appropriate care for patients to achieve a better quality of dental health and overall health. Studying the clinical and radiographic features will help in developing treatment guidelines and recommendations for management. CBCT is a very low-dose option to study the maxillofacial skeleton in three dimensions and to detect any changes in dental development and bone deposition. CBCT provides high resolution, has a 3D capability, and exposes patients to less radiation than multi-detector medical CTs [7,8,9,10,11,12,13]. Imaging patients with CBCT is a valuable way to capture the maxillofacial region in three dimensions to better evaluate the condition and help in providing the appropriate care.
Patients with CMD had a short anterior cranial base, short upper facial height, short maxillary length, and approximately 2-year delay in the eruption of permanent teeth. There were several changes noted in CMD patients mainly in the sella turcica, size of the sphenoid sinus, and diploe. The shape and size of sella turcica usually help the clinician in approximating the size of the pituitary gland. This information can help in distinguishing normal developmental patterns and pathology [14]. The findings of this study showed that there was a significant change in the shape and size of the sella with a mean difference of 2.17 mm when compared to normal individuals. The diploe is a homogenous spongy bone tissue that tightly fills in the distance between external and internal lamina in cranial bones [15]. In our study, diploe measurements among the CMD patients were 26.3 mm, whereas with normal individuals, it was 4.94 mm, which was a significant difference.
Hearing loss and vision impairment were noted in 2 patients and correlated with the radiographic findings; others did not have any cranial involvement in optic and otic structures but had multiple impacted teeth and increased dimensions of the facial bones with a lower density number measured by PIVs indicating the presence of abnormal bone. The progressive craniofacial bone thickening and narrowing of cranial foramina can lead to severe visual and neurological impairment, such as facial palsy and deafness. Careful monitoring of these structures in CMD patients is required.
Hereditary dysplasias like CMD produces changes in the skull bones and facial bones, by forming dense osteosclerotic bone, which narrows and partially obliterates the maxillary and other paranasal sinuses [15]. There was partial obliteration of paranasal sinuses compared with the healthy control group (Table 1). Mastoid air cells were opacified in 6 cases while there were well-pneumatized mastoid air cells. These findings confirmed the Chen et al. study [12].
Our CBCT data showed that the bone density in seven patients with CMD appeared to be altered in both cortical and trabecular compartments of cranial and jawbones. Interestingly, there were no changes in the external auditory meatus and the osseous structures of the TMJ in these patients compared to the normal control group. Most patients with CMD need orthodontic treatment due to either delayed eruption or impaction. One possible explanation for this is that there is an increased dimension of the bone along with poorer or less dense bone as measured by the PIVs. Orthodontic treatment should consider these unique features in the treatment plan and some increase in the overall required treatment time should be expected.
Limitations of the study
With this disease being a very rare condition, the sample size is small, and therefore it is challenging to make very definitive comments on the radiographic features across the entire spectrum of the disease. However, this is one of the few studies in the literature that compares and quantifies patients with this rare disease to healthy age and gender-matched controls.
Conclusion
Patients with craniometaphyseal dysplasia have a poorer bone quality, and larger buccolingual/palatal bone dimensions leading to an increased width, increased nasal bridge dimensions, and significantly smaller sinuses compared to the control group of age and gender-matched individuals.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Wu B, Jiang Y, Wang O, Li M, Xing XP, Xia WB. Craniometaphyseal dysplasia with obvious biochemical abnormality and rickets-like features. Clin Chim Acta. 2016;456:122–7. https://doi.org/10.1016/j.cca.2016.01.021. Epub 2016 Jan 25. PMID: 26820766.
Martin K, Nathwani S, Bunyan R. Craniometaphyseal dysplasia: a review and novel oral manifestation. J Oral Biol Craniofacial Res. 2017;7(2):134–6. https://doi.org/10.1016/j.jobcr.2017.04.007.
Esteves SS, Silva AP, Coutinho MB, Abrunhosa J, Sousa CAE. Craniometaphyseal dysplasia and otolaryngology findings. Int J Pediatr Otorhinolaryngol Extra. 2015;10(1):11–3. https://doi.org/10.1016/j.pedex.2014.12.002.
Haverkamp F, Emons D, Straehler-Pohl HJ, Zerres K. Craniometaphyseal dysplasia as a rare cause of a severe neonatal nasal obstruction. Int J Pediatr Otorhinolaryngol. 1996;34:159–64.
Mintz S, Velez I. Craniometaphyseal dysplasia associated with obstructive sleep apnea syndrome. Dentomaxillofac Radiol. 2004;33:4.
White JJ, Mazzeu JF, Hoischen A, et al. DVL3 alleles resulting in a-1 frameshift of the last exon mediate autosomal-dominant Robinow syndrome. Am J Hum Genet. 2016;98(3):553–61. https://doi.org/10.1016/j.ajhg.2016.01.005.
Reichenberger E, Tiziani V, Watanabe S, et al. Autosomal dominant craniometaphyseal dysplasia is caused by mutations in the transmembrane protein ANK. Am J Hum Genet. 2001;68(6):1321–6. https://doi.org/10.1086/320612.
Singh S, Qin C, Medarametla S, Hegde SV. Craniometaphyseal dysplasia in a 14-month-old: a case report and review of imaging differential diagnosis. Radiol Case Reports. 2016;11(3):260–5. https://doi.org/10.1016/j.radcr.2016.04.006.
Mathews JD, Forsythe AV, Brady Z, Butler MW, Goergen SK, Byrnes GB, Giles GG, Wallace AB, Anderson PR, Guiver TA, McGale P. Cancer risk in 680 000 people exposed to computed tomography scans in childhood or adolescence: data linkage study of 11 million Australians. BMJ. 2013;346:f2360.
Scarfe WC, Farman AG. What is cone-beam CT and how does it work? Dent Clin North Am. 2008;52(707–730):8.
Scarfe WC, Farman AG. What is cone-beam CT and how does it work? Dent Clin North Am. 2008;52(4):707–30. https://doi.org/10.1016/j.cden.2008.05.005. PMID: 18805225.
Chen IP, Tadinada A, Dutra EH, Utreja A, Uribe F, Reichenberger EJ. Dental anomalies associated with craniometaphyseal dysplasia. J Dent Res. 2014;93(6):553–8. https://doi.org/10.1177/0022034514529304.
Alkofide EA. The shape and size of the sella turcica in skeletal class I, class II, and class III Saudi subjects. Eur J Orthod. 2007;29(5):457–63. https://doi.org/10.1093/ejo/cjm049. Epub 2007 Aug 9. PMID: 17693429.
Skrzat J, Brzegowy P, Walocha J, Wojciechowski W. Age dependent changes of the diploe in the human skull. Folia Morphol (Warsz). 2004;63(1):67–70.
Lawson W, Patel ZM, Lin FY. The development and pathologic processes that influence maxillary sinus pneumatization. Anat Rec Adv Integr Anat Evol Biol. 2008;291(11):1554–63. https://doi.org/10.1002/ar.20774.
Acknowledgements
We thank the patients for participating in this study and the National Institutes of Health [NIH]/National Institute of Dental and Craniofacial Research [NIDCR] for funding the study.
Funding
This work was supported by institutional funds and R01-DE019458 (National Institutes of Health [NIH]/ National Institute of Dental and Craniofacial Research [NIDCR]) to EJR, T32-DE007302 (NIH/NIDCR) to EHD.
Author information
Authors and Affiliations
Contributions
KSM was involved in the data evaluation, prepared the manuscript, and is the corresponding author. IPC was involved in the ideation, grant submission, data collection, and data evaluation and preparation of the manuscript. EJR was the primary investigator and received the grant and was involved in ideation, grant writing, submission, patient data collection, evaluation, manuscript preparation, and overall management of the project. AT was involved with the ideation phase, patient imaging, data collection, data evaluation, and manuscript preparation. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the University of Connecticut Health (UCH) Institutional Review Board (IRB #03–008-1). Informed written consent to participate in the study was provided by all participants (or their parent or legal guardian in the case of children under 18). There are no age, gender, and patient pictures relating to individual participants in the present article. The radiographs are anonymized.
Consent for publication
All the authors have full consent for publication.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Muttanahally, K.S., Chen, IP., Reichenberger, E.J. et al. Three-dimensional radiographic features of craniometaphyseal dysplasia—a comparative CBCT study. J Rare Dis 2, 5 (2023). https://doi.org/10.1007/s44162-023-00009-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44162-023-00009-8