Abstract
The ependymal epithelium forms the cerebrospinal fluid barrier, separating the brain and spinal cord from the cerebrospinal fluid. However, in specific regions of the central nervous system, there are neurons that directly interface with the cerebrospinal fluid, including neuronal bodies, dendrites, or axons, This constitutes what is referred to as the "cerebrospinal fluid contacting neurons system (CSF-CNS)". The research team led by Professor Zhang has successfully utilized cholera toxin subunit B coupled horseradish peroxidase complex (CB-HRP) to selectively label the specialized neuron system that interfaces with cerebrospinal fluid, pioneeringly designating it as the "cerebrospinal fluid-contacting nucleus", commonly referred to as the "CSF-contacting nucleus". For the first time, the discovery of the CSF-contacting nucleus provides compelling morphological evidence for the existence of a distinct neural structure within the brain parenchyma that establishes a connection with the cerebrospinal fluid, thereby suggesting its potential significance in facilitating material and information exchange between the brain parenchyma and cerebrospinal fluid. After conducting a comprehensive series of studies on the morphological structure, material expression, gene analysis and functional aspects of the CSF-contacting nucleus in rodents and non-human primates, it has been revealed that there are fibrous connections between the CSF-contacting nucleus and the cerebral cortex and subcortical nuclei being involved in the regulatory mechanisms of pain, cognition, learning and memory, emotion, addiction, stress and anxiety responses, visceral activity, olfaction, vision processing and perception, auditory processing, perception, motor control and coordination, homeostasis regulation including maintenance of body energy and fluid balance, as well as the control of sleep–wake cycles and synchronization of biological rhythms. Current experiments have confirmed that the CSF-contacting nucleus is related to pain, morphine dependence and withdrawal, learning and memory, as well as stress. This present article offers a comprehensive review of the neurobiological characteristics and recent advancements in pain regulation of the CSF-contacting nucleus. The aim is to provide novel insights into the investigation of pain regulation within bidirectional regulatory pathway between the brain and cerebrospinal fluid, with a specific focus on elucidating the role of the CSF-contacting nucleus as a bridge structure. Additionally, the objective of this research is to propose innovative strategies for pain management and associated disorders in the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The cerebrospinal fluid barrier, formed by the ependymal epithelium, separates the brain and spinal cord from the cerebrospinal fluid; in specific regions of the central nervous system, there are neuronal bodies, dendrites, or axons that directly interface with the cerebrospinal fluid. The term coined by Vigh-Teichmann for this group of neurons is the "cerebrospinal fluid contacting neurons system" (CSF-CNS) [1]. According to the positional relationship between cerebrospinal fluid contacting neurons and the ependyma, they can be broadly classified into three categories [2]: (1) Ependymal-associated cerebrospinal fluid contacting neurons are situated within the ventricular wall and central canal of the spinal cord; (2) Supependymal cerebrospinal fluid contacting neurons are located adjacent to the ependyma; (3) Distally cerebrospinal fluid contacting neurons in brain parenchyma extend their processes into cerebrospinal fluid, thereby modulating non-synaptic information transmission through absorption or release of bioactive substances. The first two types of cerebrospinal fluid contacting, also known as proximal cerebrospinal fluid contacting neurons, are positioned adjacent to the ventricular wall or directly immersed in cerebrospinal fluid, and numerous scholars have conducted comprehensive investigations into their structural and functional properties [1,2,3,4,5,6,7,8,9]. However, it becomes challenging to distinguish the distal cerebrospinal fluid contacting neurons from other central neurons employing specialized methods, which results in limited research conducted on this particular neuron.
The cholera toxin subunit B coupled horseradish peroxidase complex (CB-HRP) is an originally employed tracer for peripheral nerves. Professor Licai Zhang's research team has innovatively injected CB-HRP into the ventricular system, revealing that it can specifically label dista cerebrospinal fluid contacting neurons in the brain parenchyma [10,11,12], and initially designated this cluster of neurons as the "cerebrospinal fluid-contacting nucleus," abbreviated as "CSF-contacting nucleus" [13]. Given that the bodies of neurons in the CSF-contacting nucleus are situated in the brain parenchyma and extend into the cerebrospinal fluid, they serve as a crucial link between these two compartments. Therefore, it is hypothesized that the CSF-contacting nucleus plays a pivotal role in facilitating material exchange, information transmission, and functional regulation between the brain parenchyma and cerebrospinal fluid.
A series of experimental studies were conducted on the morphological structure, material expression, gene analysis, and function of the CSF-contacting nucleus in the brains of rodents and non-human primates [14,15,16,17,18,19,20,21,22], which suggests the CSF-contacting nucleus are potentially involved in regulating various bodily functions such as pain perception, cognition and learning memory processes, emotional responses, addiction-related mechanisms, stress modulation, visceral activities control, olfaction processing as well as vision-, hearing-, movement-related functions, maintaining homeostasis regulation including energy balance management and fluid equilibrium maintenance,contributing to sleep–wake cycles synchronization along with other biological rhythms [23,24,25,26,27,28,29,30,31,32,33,34,35,36]. The present review provides a comprehensive of the biological characteristics and recent advancements in pain regulation of the CSF-contacting nucleus, aim to offer novel insights into the study of pain regulation through the brain-cerebrospinal fluid bidirectional regulatory pathway, with particular emphasis on the role of the CSF-contacting nucleus as a bridge structure. Furthermore, it proposes a promising avenue for treating pain and related disorders.
2 Morphology and cellular structure of the CSF-contacting nucleus
2.1 Morphology characteristics of the CSF-contacting nucleus
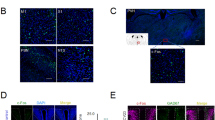
Since Zhang et al. successfully introduced CB-HRP to label and track the CSF-contacting nucleus for the first time [10, 37, 38], the research group has further developed cholera toxin B subunit labeled with fluorescein isothiocyanate (CB-FITC), CB, and Green fluorescent RetroBeads (Lumafluor, Inc.) [39, 40] to directly label it, thereby providing a favorable experimental methodology and technical means for morphological studies of the CSF-contacting nucleus, and the concurrent application of immunoelectron microscopy enhances the effectiveness of investigating its ultrastructure [13, 37, 41]. The morphological characteristics of the CSF-contacting nucleus can be clearly visualized using the CB-HRP, CB-FITC CB, and Green fluorescent RetroBeads [10, 37,38,39,40]: The coronal section of brain displays two distinct A and B clusters with a distribution pattern of "Y", primarily composed of small neuronal cells. Cluster A is located ventrally along the midline of the midbrain aqueduct, while cluster B consists of two symmetrical parts distributed in the upper end of the pontine's basal gray matter within the fourth ventricle (Fig. 1). Licai Zhang et al. utilized immunofluorescence microscopy imaging technology, laser confocal Z-axis sequential scanning overlay reconstruction, Imaris 3D/4D/multidimensional image rendering, and interactive analysis methods to perform three-dimensional reconstruction of the CSF-contacting nucleus in rat (Fig. 2). The findings indicates that the nucleus group is located within the ventral gray matter of the midbrain aqueduct at the junction of midbrain and pons, as well as in the basal gray matter of the upper region of fourth ventricle, it gradually tapers towards its tail and eventually disappeared within the basal gray matter of fourth ventricle, and it is adjacent to dorsal raphe nucleus, yet longer than. The CSF-contacting nucleus shares a common location with 12 pairs of cranial nerves, exhibiting bilateral symmetry. Its overall morphology resembles a "rivet", featuring a wide rostral side and measuring approximately 2000 μm in length. By utilizing fluorescence characteristic tracers and whole brain transparency technology, the computer high-resolution image processing accurately reproduced the 3D images of both cell bodies and fibers of this specific group of neurons. Furthermore, it provided a detailed display of the size, spatial location, adjacency, and projection connections among different nuclei. Additionally, it defined the three-dimensional coordinates for the CSF-contacting nucleus in the brain [17,18,19,20].
The location of the CSF-contacting nucleus in the brain and tracing methods by different labeling markers for the CSF-contacting nucleus in rats. CSF cerebrospinal fluid, CB Cholera toxin B subunit, CB-FITC Cholera toxin B subunit labeled with fluorescein isothiocyanate, CB-HRP Cholera toxin subunit B coupled horseradish peroxidase complex
The studies have confirmed that the neurons within CSF-contacting nucleus possess the necessary structural foundation to traverse both the brain-cerebrospinal fluid barrier and blood–brain barrier, enabling them to recruit substances from plasma/cerebrospinal fluid (such as hormones, neurotransmitters, etc.). Additionally, some of their neuronal processes extend into the cerebrospinal fluid and vasculature system in order to facilitate intercellular fluid/cerebrospinal fluid mediated chemical messengers through volume diffusion function [3, 41, 42]. Moreover, recent studies have demonstrated that the elongated projections of the CSF-contacting nucleus extend towards the superficial dorsal horn of the spinal cord, indicating their structural basis for neurotransmission through synaptic connections [30, 35, 36]. From a morphological perspective, this finding substantiates that the CSF-contacting nucleus is indeed a distinct type group of neurons that establish connectivity between brain parenchyma and cerebrospinal fluid, forming a neural-humoral regulatory circuit via synaptic connections with other regions, and it may play a pivotal role in facilitating substance and information exchange between brain parenchyma and cerebrospinal fluid.
2.2 Cellular structure and synaptic connectivity of the CSF-contacting nucleus
Although the ultrastructural characteristics of the proximal cerebrospinal fluid contacting neurons have been confirmed in previous studies [1, 3,4,5,6,7,8,9], no relevant research on the CSF-contacting nucleus has been reported. The investigation of this problem will contribute to elucidating the mechanisms and nature of material and information exchange between brain parenchyma and cerebrospinal fluid, thereby providing valuable guidance for studying the functionality of the CSF-contacting nucleus.
2.2.1 Cellular structural characteristics of the CSF-contacting nucleus under light microscope
The morphology of the neurons within the CSF-contacting nucleus varies, and a majority of them exhibit an oval shape, with size 16 μm × 10 μm and the large nucleus located in the center of the cell body [41, 42]. Recent study have found that μ opioid receptor (MOR) is expressed in the membrane of neurons within the the CSF-contacting nucleus, and 5-hydroxytryptamine (5-HT) is contained in the cytoplasm, and 5-HT immunoreactive positive fibers are projected to the dorsal horn of the spinal cord, which suggests that the CSF-contacting nucleus may play an important role in the regulation of descending pain [35].
2.2.2 Cellular ultrastructural characteristics of the CSF-contacting nucleus under electron microscope
The cellular ultrastructure of neurons within the CSF-contacting nucleus has the characteristics of the general subcellular structure of neurons, including mitochondria, trachyplasmic reticulum, free ribosome, Golgi complex, lysosome and other organelles [41, 42], but the synaptic end-like structures containing a large number of synaptic vesicles can be seen deep into the compartment [19]. The ultrastructure of neurons within the CSF-contacting nucleus suggests that it has the function of general neurons, and may have similar functional characteristics to peripheral neurons, which can directly feel the external information (cerebrospinal fluid) or transmit the information and substances in the brain to other parts through the cerebrospinal fluid.
The connection of neurons within the CSF-contacting nucleus is close, which is in line with the morphological and structural characteristics of electrical synapses [41, 42], similar to the connection between cardiomyocytes, making the CSF-contacting nucleus a "syncytial". This structure is conducive to the completion of rapid, bidirectional and synchronous functional activities to ensure the consistency of activities of neurons in the CSF-contacting nucleus.
The pre-synaptic component is formed by the structure of the non-CSF-contacting neurons, and the postsynaptic component is formed by the structure of the CSF-contacting neurons, and the synaptic connection is formed by axo-dendritic synapses, dendro-dendritic synapses, axo-axo-dendritic synapses, and the dendritic central synaptic spheroid structure formed by multiple dendrites of non-CSF-contacting neuron. The predominant synapse types observed in the CSF-contacting neurons were analyzed, encompassing both excitatory and inhibitory synapses. The CSF-contacting nucleus is postulated to receive innervation from neurons in other brain regions and transmit information towards the cerebrospinal fluid, based on the synaptic connectivity characteristics [13, 41]. The use of whole-brain projection circuit tracer technology has confirmed the origin of nerve fibers with synaptic connections in the CSF-contacting nucleus [16,17,18,19]. However, further investigation is required to elucidate the regulatory mechanisms and neurotransmitters involved in the biological functions of the CSF-contacting nucleus, which comprises 235 functional nuclei across 24 functional areas including the whole-brain cortex, subcortical and limbic system, diencephalon, brainstem, and spinal cord.
3 The CSF-contacting nucleus and its role in pain regulation
The transmission of peripheral nociceptive information to the brain is facilitated through the spinal cord and modulated by descending pathways originating from relevant nuclei in the brain, with a significant involvement of the midbrain in pain modulation [43,44,45,46,47]. Group A of the CSF-contacting nucleus is located on the ventral side of the midbrain aquaduct and distributes longitudinally along the midline. Group B of the CSF-contacting nucleus consists of two parts and distributes symmetrically in the basal gray matter of the fourth ventricle at the upper end of the pontine. Studies have found that some substances that are closely related to pain in the CSF-contacting nucleus are expressed under pain stimuli, such as Nitric oxide (NO), Fos protein, Substance P (SP), Ca2+ /calmodulin-dependent protein kinase-IIα (CaMK-IIα), Nicotinamide adenine dinucleotide phosphate-d, Purinergic P2X4 receptor (P2X4R), Purinergic P2X3 receptor, Mitogen-activated protein kinase1 (MAPK1), Transient receptor potential cation channel subfamily V member 1 (TRPV1), gamma-aminobutyric acid-B1 (GABA-B1), Hyperpolarization-activated cyclic nucleotide-gated channel 1, Mas-related G-protein coupled receptor member A, Acid-sensing ion channel 3 (ASIC3), etc. [22, 24,25,26, 29, 33, 34, 48, 49] (Table 1). In view of the special anatomical location, morphological characteristics and the expression changes of some substances during the pain state, it is speculated that the CSF-contacting nucleus may play an important role in the process of pain modulation.
3.1 The CSF-contacting nucleus and its role in inflammatory pain
The pain response to spinal injury caused by pathological processes such as tissue injury and inflammation can be augmented by SP, and the upregulation of SP expression in the cerebrospinal fluid has been observed in patients with diseases such as fibromyalgia and irritable bowel syndrome [50]. The formalin-induced pain model showed that the expression of SP increased in the the CSF-contacting nucleus [51,52,53], and intrathecal administration of local anesthetic bupivacaine can lead to down-regulate the expression of SP in the CSF-contacting nucleus. It is speculated that the CSF-contacting nucleus plays a crucial in pain modulation between brain and cerebrospinal fluid through the expression of SP in both synaptic and volumetric connection [53].
The activation of ASIC3 can elicit non-selective cation influx and enhance neuronal excitability. Wang et al. [54, 55] found that ASIC3 is expressed and distributed on the neuronal membrane within the CSF-contacting nucleus. Under the condition of inflammatory pain induced by complete Freund's adjuvant, the expression of ASIC3 in the CSF-contacting nucleus was upregulated, and administration of specific antagonist APETx2 (a peptide toxin effector of ASIC3) into the lateral ventricle could significantly alleviate the behavioral response to inflammatory pain. However, it has not been clarified whether the CSF-contacting nucleus cause its own excitatory changes by detecting cerebrospinal fluid or PH changes in the brain through ASIC3, or if it regulates the functions of other nuclei to participate in the process of persistent inflammatory pain.
3.2 The CSF-contacting nucleus and its role in neuropathic pain
The International Association for the Study of Pain defines neuropathic pain as pain caused by direct damage to the peripheral or central nervous system. In recent years, there has been extensive research conducted on the cellular and molecular mechanisms underlying neuropathic pain caused by peripheral or central nerve injury, focusing on primary sensory neurons at the peripheral level and spinal cord, leading to a substantial accumulation of data. However, there are few studies on the supraspinal mechanisms of neuropathic pain, mainly focusing on the two aspects of the reduced inhibitory effect of descending pain and the enhanced facilitation of descending pain. Early studies found changes in the expression of some substances in the CSF-contacting nucleus under pain conditions, suggesting that it is closely related to pain. Subsequent studies found changes in the expression of ion channels, receptors and substances that affect the excitability and synaptic plasticity of the CSF-contacting nucleus [22, 24,25,26, 29, 33, 34, 48, 49].
3.2.1 Excitatory changes of neurons within the CSF-contacting nucleus and its role in pain regulation
Ionic receptors expressed on the cell membrane play a crucial role in the initiation and sustenance of neuronal action potentials. Transient receptor potential cation channel subfamily M member 8 (TRPM8) [34, 56], TRPV1 [24, 57, 58] and Transient receptor potential cation channel subfamily C member 6 (TRPC6) [59, 60] are characterized by their non-selective permeability to cations. When activated, these channels facilitate the influx of non-selective cation and subsequently enhance neuronal excitability. The expression of these receptors in the neurons of the CSF-contacting nucleus provides morphological basis for the perception of the changes in cerebrospinal fluid. Further investigations revealed that the expression levels of TRPV1, TRPC6 and ASIC3 in the CSF-contacting nucleus were upregulated in the presence of neuropathic pain. Moreover, administration of their respective specific antagonists via intracerebroventricular injection significantly ameliorated neuropathic pain behaviors.
The large-conductance Ca2+-activated K+ channel alpha subunit (BKCa-α) has the ability to attenuate intracellular calcium overload, affect membrane potential hyperpolarization, and suppress excessive neuronal firing. The involvement of this process in the repolarization and rapid hyperpolarization of action potentials contributes to the reduction of neuronal excitability following activation. thus playing a crucial role in the pain transduction [61]. It was found that under the condition of neuropathic pain, the BKCa-α expression of neurons in the CSF-contacting nucleus decreased [62]. The presence of BKCa-α in the CSF-contacting nucleus suggests its involvement in a range of pathological and physiological activities, triggered by BKCa-α, through intracellular calcium overload. This involvement affects membrane potential hyperpolarization, leads to neuron overfiring, and contributes to neuropathic pain.
The P2X4R is an extracellular ATP-gated, non-selective cation membrane channel with high permeability to Ca2+. P2X4R is involved in pain signal transduction, and the up-regulation of P2X4R expression in highly activated microglia is necessary for the generation and maintenance of allodynia after nerve injury. There have been numerous studies investigating the involvement of spinal cord microglia in neuropathic pain through P2X4R expression,, however, no relevant reports regarding the CSF-contacting nucleus have been published. Song et al. [63] confirmed that P2X4R is expressed on the membrane of neurons within the CSF-contacting nucleus, and their expression increases under neuropathic pain conditions, which provides an important morphological basis for considering the potential impact of microglial activity on the CSF-contacting nucleus.
3.2.2 Changes in synaptic plasticity of neurons within the CSF-contacting nucleus and its role in pain regulation
Synaptic plasticity serves as a crucial pathophysiological basis for the initiation and sustenance of pain. Drebrin, as the primary binding protein of dendritic spine cytoskeleton, is intimately linked to synaptic plasticity and intracellular signal transduction [64]. Geng et al. [65] found that the expression of drebrin increased in the CSF-contacting nucleus of chronic constriction injury (CCI) model rats, suggesting that neuropathic pain induces the translocation of drebrin from dendritic spines to cell bodies and its involvement in synaptic plasticity changes associated with neuropathic pain.
3.2.3 The expression changes of receptors and substances iwithin the CSF-contacting nucleus and its role in pain regulation
The periaqueductal gray of the midbrain exhibits a high distribution of 5-HT and its receptors, and the CSF-contacting nucleus also locates in the midbrain. It was found that the expression of 5-HT1A receptor was constant in the CSF-contacting nucleus and decreased with the development of neuropathic pain [66]. The intrathecal administration of tramadol or intramuscular injection of 8-hydroxy-2-(di-n-propylamino) tetralin, a specific agonist for the 5-HT1A receptor, effectively alleviated neuropathic pain and upregulated 5-HT1A receptor [67, 68], suggesting that the expression of 5-HT1A receptor on neurons of the CSF-contacting nucleus in the midbrain was associated with neuropathic pain. The neuropathic pain may be regulated by the 5-HT1A receptor of the CSF-contacting nucleus, establishing the information link between the brain parenchyma and the cerebrospinal fluid, or by the descending nociceptive regulatory system. The subsequent studies [35, 36] revealed that the targeted destruction of the CSF-contacting nucleus and reduction in the basic pain threshold were achieved by introducing CB-saporin (SAP) into the lateral ventricle, thereby facilitating the pain behavior response in the pain model. The double immunofluorescence labeling shows that the CSF-contacting nucleus is composed of a population of MOR-expressing neurons, which contain 5-HT in the cytoplasm. The CSF-contacting nucleus contains 5-HT neurons that project descending 5-HT fibers to the spinal cord. This pathway regulates the release of neuroactive substance 5-HT, and MOR may exert pain inhibition regulation by regulating the release of 5-HT transmitter in descending projection neurons of the CSF-contacting nucleus.
Receptor activation necessitates the involvement of a cascade of signaling pathways, including intracellular kinases, among which the role of the MAPK family in pain has been unequivocally elucidated. The expression levels of Extracellular signal-regulated kinase1/2 (ERK1/2), and its downstream phosphorylated cAMP-responsive element binding protein were found to be increased in the CSF-contacting nucleus of a rat pain model induced by CCI [69,70,71]. These signal molecules may be implicated in the neuropathic pain process by regulating downstream gene expression. Furthermore, it has been observed that the expression of matrix metalloproteinase-2/9 was significantly upregulated in rats subjected to neuropathic pain stimulation, which aligns with the manifestation of increased pain intensity [72]. The findings of this study further validate the involvement of signaling molecules in the pathogenesis of neuropathic pain by regulating the expression of downstream gene proteins.
3.3 The CSF-contacting nucleus and its role in visceral pain
The expression of Fos protein expression is closely associated with the central response to various nociceptive stimuli. It was found that Fos protein expression was observed to be increased in the CSF-contacting nucleus of a rat model with sigmoid pain [73,74,75], and this reaction could be inhibited by intrathecal injection of ketamine, which may be related to GABA, although the precise mechanism of action requires further investigation. In the rat model with sigmoid pain, the number and distribution range of Fos protein in the CSF-contacting nucleus increased much more than that of nitric oxide synthase (NOS) immunoreactive neurons, indicating that NO may enhance the transmission of visceral injurious information by activating the CSF-contacting nucleus. The inhibition of visceral pain may be achieved by administering the NOS inhibitor L-IV-nitroarginine methyl ester, which can effectively reduce the production of NO [76, 77]. However, the existing research on the perception and regulation of visceral pain is constrained in terms of its scope, thereby demanding additional exploration into the underlying mechanisms.
4 Conclusion and prospect
The CSF-contacting nucleus is located within the brain parenchyma at the junction of the midbrain and pons, with its cell body located in the brain parenchyma, extensive projectional connections with the functional nuclei in the brain, and the protrusions extend into the cerebrospinal fluid,. The CSF-contacting nucleus really acts as a bridge between the functional structure of deep brain parenchyma and cerebrospinal fluid. The CSF-contacting nucleus not only plays a crucial role in the regulation of inflammatory pain, neuropathic pain and visceral pain, but also may encompasse a wide range of cerebrospinal fluid-related physiological activities and diseases due to its axonal termination within the cerebrospinal fluid. Therefore, a novel comprehension and emphasis on the CSF-contacting nucleus may provide a fresh perspective in the field of life sciences and disease research. Shifting from the traditional focus on neuron-neuron dialogue to neuron-humoral dialogue, particularly brain-cerebrospinal fluid communication, may be imperative for advancing our understanding in this field.
Availability of data and materials
Not applicable.
Abbreviations
- 5-HT:
-
5-Hydroxytryptamine
- ASIC3:
-
Acid-sensing ion channel 3
- BKCa-α:
-
Large-conductance Ca2+ -activated K + channel alpha subunit
- CaMK-IIα:
-
Ca2+ /calmodulin-dependent protein kinase-IIα
- CB-FITC:
-
Cholera toxin B subunit labeled with fluorescein isothiocyanate
- CB-HRP:
-
Cholera toxin subunit B coupled horseradish peroxidase complex
- CCI:
-
Chronic constriction injury
- CSF-CNS:
-
Cerebrospinal fluid contacting neurons system
- CSF-contacting nucleus:
-
Cerebrospinal fluid-contacting nucleus
- ERK1/2:
-
Extracellular signal-regulated kinase1/2
- GABA-B1:
-
Gamma-aminobutyric acid-B1
- MAPK:
-
Mitogen-activated protein kinase
- MOR:
-
μ Opioid receptor
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- P2X4R:
-
P2X4 receptor
- SAP:
-
Saporin
- SD:
-
Sprague-Dawley
- TRPC6:
-
Transient receptor potential cation channel subfamily C member 6
- TRPM8:
-
Transient receptor potential cation channel subfamily M member 8
- TRPV1:
-
Transient receptor potential cation channel subfamily V member 1
References
Vigh-Teichmann I, Vigh B, Aros B. [Enzyme histochemical studies on the nervous system. IV. Acetylcholinesterase activity in the liquor contacting neuronal system of various vertebrates]. Histochemie. 1970;21(4):322–37. German.
Barreiro-Iglesias A, Villar-Cervino V, Anadon R, Rodicio MC. Descending brain-spinal cord projections in a primitive vertebrate, the lamprey: cerebrospinal fluid-contacting and dopaminergic neurons. J Comp Neurol. 2008;511(6):711–23.
Vigh B, e Silva Manzano MJ, Frank CL, Vincze C, Czirok SJ, Szabo A, et al. The system of cerebrospinal fluid-contacting neurons. Its supposed role in the nonsynaptic signal transmission of the brain. Histology Histopathol. 2004;19(2):607–28.
Calle M, Claassen IE, Veening JG, Kozicz T, Roubos EW, Barendregt HP. Opioid peptides, CRF, and urocortin in cerebrospinal fluid-contacting neurons in Xenopus laevis. Ann N Y Acad Sci. 2005;1040:249–52.
Carrera I, Molist P, Anadon R, Rodriguez-Moldes I. Development of the serotoninergic system in the central nervous system of a shark, the lesser spotted dogfish Scyliorhinus canicula. J Comp Neurol. 2008;511(6):804–31.
Fernandez-Lopez B, Villar-Cervino V, Valle-Maroto SM, Barreiro-Iglesias A, Anadon R, Rodicio MC. The glutamatergic neurons in the spinal cord of the sea lamprey: an in situ hybridization and immunohistochemical study. PLoS ONE. 2012;7(10):e47898.
Orts-Del’immagine A, Wanaverbecq N, Tardivel C, Tillement V, Dallaporta M, Trouslard J. Properties of subependymal cerebrospinal fluid contacting neurones in the dorsal vagal complex of the mouse brainstem. J Physiol. 2012;590(16):3719–41.
Veening JG, Barendregt HP. The regulation of brain states by neuroactive substances distributed via the cerebrospinal fluid; a review. Cerebrospinal Fluid Res. 2010;7:1.
Vigh B, Vigh-Teichmann I. Actual problems of the cerebrospinal fluid-contacting neurons. Microsc Res Tech. 1998;41(1):57–83.
Wang MS, Zhang LC. [A comparison of HRP vs CB-HRP for labelling CSF contacting neurons]. Acta Acad Med Xuzhou. 1992;12(4):286–7. Chinese.
Zhang LC, Wang MS, Zhang FZ, Zhu ZY. [A tracing study on neuron contacting cerebrospinal fluid in rat]. Chin J Anat. 1994;17(1):37–40. Chinese.
Zhang LC. [Discovery and scientific significance of a unique nucleus–cerebrospinal fluid-contacting nucleus]. Int J Anesth Resus. 2022;43(2):113–7. Chinese.
Zhang LC, Zeng YM, Ting J, Cao JP, Wang MS. The distributions and signaling directions of the cerebrospinal fluid contacting neurons in the parenchyma of a rat brain. Brain Res. 2003;989(1):1–8.
Song SY, Yuan YM, Xu LL, Jiang J, Li Y, Yan Y, et al. Genetic Architecture and Functional Implications of the CSF-Contacting Nucleus. Neurosci Bull. 2023;39(11):1638–54.
Song SY, Zhai XM, Shan CJ, Lu LL, Hong J, Cao JL, et al. A Special Cranial Nucleus (CSF-Contacting Nucleus) in Primates. Front Neuroanat. 2020;14:53.
Song SY, Zhai XM, Dai JH, Lu LL, Shan CJ, Hong J, et al. Novel projections to the cerebrospinal fluid-contacting nucleus from the subcortex and limbic system in rat. Front Neuroanat. 2020;14:57.
Song SY, Zhai XM, Dai JH, Lu LL, Shan CJ, Hong J, et al. The CSF-contacting nucleus receives anatomical inputs from the cerebral cortex: A combination of retrograde tracing and 3D reconstruction study in rat. Front Neuroanat. 2020;14:600555.
Song SY, Li Y, Zhai XM, Li YH, Bao CY, Shan CJ, et al. Monosynaptic input mapping of diencephalic projections to the cerebrospinal fluid-contacting nucleus in the rat. Front Neuroanat. 2020;14:7.
Song SY, Li Y, Zhai XM, Li YH, Bao CY, Shan CJ, et al. Connection input mapping and 3D reconstruction of the brainstem and spinal cord projections to the CSF-contacting nucleus. Front Neural Circuits. 2020;14:11.
Song SY, Li YH, Bao CY, Li Y, Yin PC, Hong J, Li WL, Shi Y, Zhang LC. Stereotaxic coordinates and morphological characterization of a unique nucleus (CSF-contacting nucleus) in rat. Front Neuroanat. 2019;13:47.
Song SY, Zhang LC. The establishment of a CSF-contacting nucleus “Knockout” model animal. Front Neuroanat. 2018;12:22.
Wu T, Cao J, Zhang LC. [Neuropathic pain enhances expression of HCN2 channel in rat cerebrospinal fluid-contacting nucleus]. Sheng Li Xue Bao. 2014;66(3):323–31. Chinese.
Zhai XM, Yuan YM, Xu LL, Jiang J, Li Y, Yan Y, et al. Cerebrospinal fluid contacting nucleus and its 5-HT: A new insight into the regulation mechanism of general intravenous anesthesia. Brain Res. 2023;1798:148168.
Xu LL, Yan Y, Yuan YM, Li Y, Jiang J, Zhang LC. TRPV1 and GABAB1 in the Cerebrospinal Fluid-Contacting Nucleus are Jointly Involved in Chronic Inflammatory Pain in Rats. J Pain Res. 2022;15:3931–9.
Song W, Yong Y, Zhou YL, Lu LY, Yu GJ, Tang W, et al. Activation of P2X4 receptors in midbrain cerebrospinal fluid-contacting nucleus leads to mechanical hyperalgesia in chronic constriction injury rats. Purinergic Signal. 2023;19(3):481–7.
Chen YF, Tian EQ, Wang GP, Zhou F, Zhang LC. [The distribution of Mas-related G protein-coupled receptor A in cerebrospinal fluid-contacting nucleus of normal rats and its up-regulation in neuropathic pain]. Sheng Li Xue Bao. 2022;74(3):353–8. Chinese.
Li Q, Liu H, Zhang LC, Zhang HX, Zhou F. The CSF-contacting nucleus regulates learning and memory through synaptic connections with the hippocampus. Neurosci Lett. 2020;728:134972.
Zhou QQ, Chen SS, Zhang QQ, Liu PF, Fang HZ, Yang Y, et al. Cerebrospinal fluid-contacting nucleus mediates nociception via release of fractalkine. Braz J Med Biol Res. 2017;50(9):e6275.
Liu PF, Fang HZ, Yang Y, Zhang QQ, Zhou QQ, Chen SS, et al. Activation of P2X3 receptors in the cerebrospinal fluid-contacting nucleus neurons reduces formalin-induced pain behavior via PAG in a rat model. Neuroscience. 2017;358:93–102.
Fei Y, Wang X, Chen SS, Zhou QQ, Zhang C, Li Y, et al. Role of the RVM in descending pain regulation originating from the cerebrospinal fluid-contacting nucleus. Neurochem Res. 2016;41(7):1651–61.
Xing D, Wu YH, Li GL, Song SY, Liu YP, Liu H, et al. Role of cerebrospinal fluid-contacting nucleus in sodium sensing and sodium appetite. Physiol Behav. 2015;147:291–9.
Wu YH, Song SY, Liu H, Wang X, Fei Y, Li GL, et al. Role of adrenomedullin in the cerebrospinal fluid-contacting nucleus in the modulation of immobilization stress. Neuropeptides. 2015;51:43–54.
Chen P, Lin QS, Zhang LC. [The distribution of MAP kinase phosphatase-1 in the cerebrospinal fluid-contacting nucleus and its functional contribution to depressive behaviors]. Sheng Li Xue Bao. 2015;67(1):90–6. Chinese.
Wang XY, Yan WW, Zhang XL, Liu H, Zhang LC. ASIC3 in the cerebrospinal fluid-contacting nucleus of brain parenchyma contributes to inflammatory pain in rats. Neurol Res. 2014;36(3):270–5.
Liu H, Yan WW, Lu XX, Zhang XL, Wei JQ, Wang XY, et al. Role of the cerebrospinal fluid-contacting nucleus in the descending inhibition of spinal pain transmission. Exp Neurol. 2014;261:475–85.
Cao J, Wu T, Zhang LC. [Targeted damage of the cerebrospinal fluid-contacting nucleus contributes to the pain behavior and the expression of 5-HT and c-Fos in spinal dorsal horn of rats]. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2014;30(3):218–22. Chinese.
Lu X, Geng X, Zhang L, Zeng Y. The methodology for labeling the distal cerebrospinal fluid-contacting neurons in rats. J Neurosci Methods. 2008;168(1):98–103.
Lu XF. The improved methods for labeling the distal cerebrospinal fluid-contacting neurons in rats, substance P in these neurons relating with the inflammatory pain [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2008.
Zhou F, Wang J, Zhang H, Liu H, Zhao G, Zu C, et al. Evaluation of three tracers for labeling distal cerebrospinal fluid-contacting neurons. Neurosci Bull. 2013;29(5):576–80.
Zhou F. Research on the mechanism of cerebrospinal fluid--contacting neurons involved in pain [dissertation]. Shenyang (Liaoning): China Medical University; 2013.
Liang D, Zhang LC, Qin CW, Zeng YM. [The ultrastructure of distal cerebrospinal fluid-contacting neurons in the dorsal raphe and the relationships with their surrounding tissues]. Acta Anat Sin. 2007;(4):34–7. Chinese.
Liang D. The ultrastrueture of the distal CSF-contacting neurons in the parenchyma of brain the relationships between the neurons and their surrounding tissues [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2007.
Zhang LC, Ding J, Wang MS, Li F, Cao JP, Zeng YM. [Ultrastructure relationship between distal neurons contacting cerebrospinal fluid in the dorsal raphe nucleus and brain parenchyma]. Chin J Anat. 1999;22(5):388–92. Chinese.
Heinricher MM, Tavares I, Leith JL, Lumb BM. Descending control of nociception: Specificity, recruitment and plasticity. Brain Res Rev. 2009;60(1):214–25.
Ossipov MH, Dussor GO, Porreca F. Central modulation of pain. J Clin Investig. 2010;120(11):3779–87.
Staud R. The important role of CNS facilitation and inhibition for chronic pain. Int J Clin Rheumatol. 2013;8(6):639–46.
Tavares I, Lima D. From neuroanatomy to gene therapy: searching for new ways to manipulate the supraspinal endogenous pain modulatory system. J Anat. 2007;211(2):261–8.
Wang S. The distribution of CaMK-IIα in fcerebrospinal-contacting neurons and phosphorylation induced by nociception [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2009.
Wang S, Yuan YP, Chu ZX, Zhang LC. [Increase of pCaMK-IIα in the distal cerebrospinal fluid-contacting neurons by intrathecal [Sar9, Met(O2)11]-substance P in rat]. Chin J Pain Med. 2009;(5):42–6. Chinese.
Bjersing JL, Dehlin M, Erlandsson M, Bokarewa MI, Mannerkorpi K. Changes in pain and insulin-like growth factor 1 in fibromyalgia during exercise: the involvement of cerebrospinal inflammatory factors and neuropeptides. Arthritis Res Ther. 2012;14(4):R162.
Lu XF, Geng XJ, Zhang LC, Zeng YM, Dong HY, Yu H. Substance P expression in the distal cerebrospinal fluid-contacting neurons and spinal trigeminal nucleus in formalin-induced the orofacial inflammatory pain in rats. Brain Res Bull. 2009;78(4–5):139–44.
Lu XF, Geng XJ, Zhang LC, Zeng YM. [Expression of substance P in the spinal trigeminal nucleus and cerebrospinal fluid-contacting neuron following injection of formalin in the oroface of rats]. Chin J Pain Med. 2009;(2):24–8. Chinese.
Lu XF, Geng XJ, Zhang LC, Zeng YM. [Intrathecal levobupivacaine downregulates substance P expression in the distal cerebrospinal fluid contacting neuron for formalin-induced inflammatory pain in rats]. Chin Pharmacol Bull. 2008;(2):191–6. Chinese.
Wang XY. ASIC3 in the cerebrospinal fluid-contacting nucleus of brain parenchyma contributes to inflammatory pain in rats [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2013.
Du J. Expression of TRPM8 in the distal cerebrospinal fluid-contacting neurons in the brain mesencephalon of rats [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2009.
Du J, Yang X, Zhang L, Zeng YM. Expression of TRPM8 in the distal cerebrospinal fluid-contacting neurons in the brain mesencephalon of rats. Cerebrospinal Fluid Res. 2009;6:3.
Xu C. Distribution of TRPV1 in CSF-contacting neuclear of rat brain parenchyma and it’s expression in neuropathic pain [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2011.
Xu C, Zhao ZJ, Wu TT, Zhang LC. Distribution of TRPV1 in CSF Contacting Nucleus of Rat Brain Parenchyma and its expression in Neuropathic Pain. J Neurol Neurophysiol. 2011;2(3):114.
Wu TT, Zhao ZJ, Xu C, Zhang LC. Distribution of TRPC6 in the cerebrospinal fluid-contacting nucleus of rat brain parenchyma and its expression in morphine dependence and withdrawal. Neurochem Res. 2011;36(12):2316–21.
Wu TT. Distribution of TRPC6 in the cerebrospinal fluid-contacting nucleus of rat brain parenchyma and its expression in the model of morphine dependence and withdrawal [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2011.
Fuchs A, Lirk P, Stucky C, Abram SE, Hogan QH. Painful nerve injury decreases resting cytosolic calcium concentrations in sensory neurons of rats. Anesthesiology. 2005;102(6):1217–25.
Ni LH, Zhang LC. [The decrease of BKCa-α expression in CSF-contacting nucleus in neuropathic pain rats]. Chin J Pain Med. 2013;(11):29–34. Chinese.
Song W. Distributions of P2X4 receptors in periaqueductal gray and cerebrospinal fluid-contacting nucleus: a possible contribution to neuropathic pain [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2011.
Merriam EB, Millette M, Lumbard DC, Saengsawang W, Fothergill T, Hu X, et al. Synaptic regulation of microtubule dynamics in dendritic spines by calcium, F-actin, and drebrin. J Neurosci. 2013;33(42):16471–82.
Geng XJ, Lu XF, Zhang LC, Zeng YM. [Expression of drebrin in the distal cerebrospinal fluid contacting neurons of rats with chronic constriction injury of sciatic nerve]. Sheng Li Xue Bao. 2008;60(4):469–74. Chinese.
Jiang WX, Zhang LC. [Distribution and effects of 5-HT1A receptors in distal cerebral spinal fluid-contacting neurons in rat brain parenchyma in neuropathic pain]. Sheng Li Xue Bao. 2008;60(2):243–8. Chinese.
Jiang WX, Yin N, Wang L, Zhang LC. [Effect of tramadol on expression of 5-HT1A receptor in distal cerebrospinal fluid contacting neurons in mid-brain in a rat model of neuropatlic pain]. Chin J Anesthesiol. 2010;(6):708–11. Chinese.
Zhang S, Zhu P, Guo JR, Zhang LC. [Role of 5-HT1A receptor in distal cerebrespinal fluid-contacting neurons in neuropathic pain in rats]. Chin J Anesthesiol. 2011;(5):569–72. Chinese.
Chen QP, Zhang L, Xiao C, Zhang LC, Zeng YM. [ERK1/2 is activated in the distal cerebrespinal fluid-contacting neurons following chronic constriction injury induced neuropathic pain]. Chin J Pain Med. 2008;(6):47–50. Chinese.
Chen QP, Zhang YH, Cao S. [Changes in expression of phosphor-p38MAPK in distal cerebrospinal fluid contacting neurons in rats with neuropathic pain]. Chin J Anesthesiol. 2011;(10):1189–91. Chinese.
Chen QP. The experiment study on the chronic constriction injury induced expression of p-ERKl/2, p-p38MAPK, pCREB, Fos in the distal cerebrospinal fluid contacting neurons in the rat brain [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2008.
Dong JJ. The Expressions of matrix metalloproteinase 2/9 in cerebrospinal fluid-contacting nucleus of rat brain with neuropathic pain [dissertation]. Soochow (Jiangsu): Xuzhou Medical University; 2010.
Wang YF, Zhang LC, Si XM. [Effect of intrathecal injection of ketamine on expression of Fos protein in the distal cerebrospinal fluid contacting neuron in rat with visceral pain]. Jiangsu Med J. 2013;(4):381–3. Chinese.
Wang YF, Zhang LC, Zeng YM. [The sigmoid pain stimulation induced expression of Fos in the distal cerebrospinal fluid contacting neurons]. Chin J Pain Med. 2007;(2):102–5. Chinese.
Zhang ZW, Zhang LC, Zeng YM, Jia XQ, Cao JP. [Peripheral pain stimulation induces Fos protein expression in fluid-contacting neurons in the dorsal raphe nucleus of rats]. Chin J Anesthesiol. 2002;(5):286. Chinese.
Yang B, Zhang LC, Zeng YM. [Microinjection of L-NAME into dorsal raphe nucleus inhibits nociceptive response in sigmoid pain model of rats]. Sheng Li Xue Bao. 2003;55(5):577–82. Chinese.
Zhang ZW, Zhang LC, Zeng YM, Zhang HQ, Cao JP. [NOS positive distal cerebrospinal fluid-contacting neurons in rat dorsal raphe nucleus]. Chin J Histochem Cytochem. 2001;(2):186–9. Chinese.
Acknowledgements
Not applicable.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This work was supported in part by grants from the National Natural Science Foundation of China (NSFC82171227 and NSFC81300957 to HL, 2022 Original Exploration Program recommended by experts NSFC82150007 to LCZ), and the Natural Science Foundation of Zhejiang Province (LY22H090019 to HL). The funding agencies had no influence on this study.
Author information
Authors and Affiliations
Contributions
JQW, HL and LCZ contributed to the conception of the study and writing, reviewing as well as editing of the manuscript. FZ, XFL and HXZ contributed to the material collection.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors of the manuscript have read and agreed to publish.
Competing interests
Dr. He Liu is one member of the Editorial Board in Anesthesiology and Perioperative Science and recuses himself from every editorial procedure of this submission including peer-review and academic decisions. All the other authors reported no biomedical financial interests or potential conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wei, J., Liu, H., Zhou, F. et al. The recent research progress in neurobiological characteristics and pain regulation of the cerebrospinal fluid-contacting nucleus. APS 2, 17 (2024). https://doi.org/10.1007/s44254-024-00051-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44254-024-00051-9