Abstract
Multiple endocrine neoplasia type 1 (MEN1) is a hereditary cancer syndrome caused by germline mutations of the MEN1 gene located in chromosome 11q13. In patients with MEN1, multicentric tumors develop in the involved organs; however, precise evaluation of genetic changes in these multicentric tumors has not been performed. In the present study, using whole-exome sequencing, we analyzed germline and somatic genetic changes in blood cells, two pancreatic endocrine tumors and one duodenal tumor obtained from a patient with MEN1 gastrinoma. We found that this patient possessed a novel germline mutation of the MEN1 gene [NM_137099.2:c.1505dupA (p.Lys502Lysfs); the localization was Chr11:64572134 on Assembly GRCh37], in which an adenine insertion in codon 502 of the MEN1 gene resulted in a frame shift and a premature stop codon. In terms of heterozygosity, the mutated allele was heterozygous in blood cells, hemizygous in the two pancreatic tumors and homozygous in the duodenal tumor. Immunohistochemical staining confirmed that only truncated menin protein accumulated in the nucleus of the tumor tissues. Further evaluation of tumor-specific somatic mutations in two pancreatic tumors did not detect single-nucleotide variations (SNVs) in 609 cancer-associated genes designated by the COSMIC cancer gene census, suggesting that the germline MEN1 mutation and resultant loss of heterozygosity played a major role in tumorigenesis. In the duodenal tumor, in addition to the germline MEN1 mutation, single-nucleotide variations in two cancer-associated genes were found. Further studies are required to clarify the role of these somatic single-nucleotide variations in the progression of MEN1 tumors.
Similar content being viewed by others
Introduction
Multiple endocrine neoplasia type 1 (MEN1, OMIM 131100) is a hereditary cancer syndrome inherited as an autosomal dominant trait, which is caused by germline mutations of the MEN1 gene located in chromosome 11q13.1 Patients with MEN1 develop signs and symptoms of primary hyperparathyroidism, pituitary adenoma, entero-pancreatic neuroendocrine tumors (NET), adrenocortical tumors, thymic tumors, bronchial carcinoids and various types of non-NET. In patients with MEN1, multicentric tumors develop in the involved organs, as is the case for hereditary cancer syndromes.2 However, precise evaluation of genetic changes in these multicentric tumors has not been performed. The crystal structure3 and biological study of the menin protein have suggested a wide range of roles for menin as a scaffold protein.4 In this study, we performed whole-exome sequencing (WES) using blood cells and tumor tissues to assess germline and somatic mutations in a patient with MEN1, who was diagnosed with a rare case of gastrinoma with dysphagia resulting from esophageal stricture.
Materials and Methods
Patient
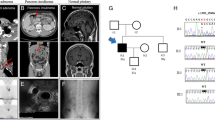
A 43-year-old male patient presenting with dysphagia caused by esophageal stricture was referred to the Shizuoka Cancer Center from a local hospital on a suspicion of MEN1. Further evaluation revealed elevated levels of serum gastrin (⩾3,000 pg ml−1 (normal: ⩽200 pg ml−1)), glucagon (253 pg ml−1 (normal: 70–174 pg ml−1)), calcium (10.9 mg dl−1 (normal: 8.5–10.2 mg dl−1)) and intact parathyroid hormone (104 pg ml−1 (normal: 10–65 pg ml−1)). Enhanced computed tomography showed multiple highly vascular lesions within the pancreas and duodenum (Figure 1a–d). These findings indicated that the patient is suffering from gastrinoma. Total pancreatectomy was performed for the multiple masses within the pancreas and the duodenum. Pathological studies revealed that the patient had more than 10 tumors in the pancreas and the duodenum. In the pancreas, one tumor (7 mm in diameter) was detected in the head, two (5, 3 mm) in the body and another two (19, 5 mm) in the tail. In the duodenum, there was one (5 mm) in the bulb, another one (10 mm) and some smaller ones (about 1 mm) in the descending limb. Histology of multiple tumors in the pancreas and duodenum indicated NET with histological grade 2.5
Clinical and pathological manifestations in this MEN1 patient. CT image and pathology specimens. The patient had more than 10 tumors in the pancreas and the duodenum. The pancreas head had one tumor (7 mm in diameter), the body had two tumors (5, 3 mm) and the tail had two tumors (19, 5 mm). In the duodenum, the bulb had one tumor (5 mm), and the descending limb had one tumor (10 mm) and smaller ones (about 1 mm). (a) Duodenum. (b) Pancreatic head. (c) Pancreatic body. (d) Pancreatic tail. Yellow arrows indicate tumor. (e) Pedigree of the patient. P indicates the proband and E+ indicates germline mutation of the MEN1 gene (c.1505dupA). CT, computed tomography; MEN1, multiple endocrine neoplasia type 1.
After the operation, the hypergastrinemia improved, and the patient remained symptom free for 2 years without recurrence.
The patient gave written informed consent for a detailed genetic examination, which was approved by the Institutional Review Board of the Shizuoka Cancer Center. When the patient was referred for genetic counseling, the family history revealed that several family members possessed past histories of hyperparathyroidism and NET (Figure 1e). Although the attending physician proposed genetic testing for the family members, agreement was not obtained from these family members.
Sample acquisition and isolation of DNA and RNA
Samples were flash-frozen in liquid nitrogen. Genomic DNA was extracted from surgically resected tissues and buffy coat cells of the peripheral blood, using the QIAamp DNA Blood Midi Kit (Qiagen, Hilden, Germany). DNA concentration and purity were assessed using Qubit and Nanodrop (Thermo Fisher Scientific, Waltham, MA, USA). For RNA extraction, fresh tissues were submerged in QIAzol Lysis Reagent (Qiagen) and disrupted using a TissueLyser (Qiagen). Total RNA was isolated using the miRNeasy mini kit (Qiagen) and quantified using a Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA).6
Whole-exome sequencing analysis
The Ion AmpliSeq Exome RDY Kit (Thermo Fisher Scientific) was used to construct an Ion Torrent adapter-ligated library according to the manufacturer’s instructions. The Ion Proton Sequencing 200 Kit (Thermo Fisher Scientific) was used for nucleotide sequencing according to the manufacturer’s protocol. Germline and somatic mutations were detected using the torrent variant caller analysis and Ion Reporter software (Thermo Fisher Scientific), respectively. Detected mutations were validated using the Integrative Genomics Viewer (IGV),7,8 followed by Sanger and pyrosequencing. We searched COSMIC (http://cancer.sanger.ac.uk/cosmic/) and the Human Gene Mutation Database (HGMD (http://www.hgmd.cf.ac.uk/ac/index.php)) Professional v.2016.3 (BIOBASE, Beverly, MA, USA) to determine whether detected mutations had been previously reported as being associated with diseases. All of the genomic linear positions were based on human genome reference version UCSC Genome Assembly GRCh37.9
Comprehensive cancer panel analysis
To confirm the presence of tumor-specific somatic mutations in pancreatic and duodenum tumors, a comprehensive cancer panel analysis for 409 cancer-associated genes10 was performed. A DNA library comprising these genes was prepared using the Ion AmpliSeq Comprehensive Cancer Panel Kit (Thermo Fisher Scientific). Amplified libraries were quantified using the StepOnePlus Real-Time PCR System (Thermo Fisher Scientific). Templates for sequencing were prepared using the Ion PI Template OT2 200 Kit (Thermo Fisher Scientific) with an Ion OneTouch 2 (Thermo Fisher Scientific) according to the manufacturer’s instructions. Data were processed, and variants were identified as described above for WES.
Sanger sequencing
Sanger sequencing of PCR products was performed by TAKARA BIO (Kusatsu, Japan). PCR reactions were performed using the HotStarTaq DNA Polymerase (Qiagen) and MEN1-specific primers (forward primer: 5′-TCATCTGCACTTGCGACTGT-3′; reverse primer: 5′-ACCTTGCTCTCACCTTGCTC-3′). For sequencing, the forward primer was used. PCR reactions were performed using the following conditions: initial PCR activation step at 95 °C for 15 min; 35 cycles of denaturation at 94 °C for 30 s, annealing at 65 °C for 30 s and extension at 72 °C for 30 s; and a final extension step at 72 °C for 10 min. Amplicons were purified using the Illustra ExoProStar system (GE Healthcare, Little Chalfont, UK). Mutated allele frequency was determined using the ab1 Peak Reporter (Thermo Fisher Scientific).
Pyrosequencing
PyroMark Gold Q96 and PyroMark Q24 systems (Qiagen) were used for pyrosequencing according to the manufacturer’s protocol. Primers used for the pyrosequencing were biotinylated primer (5′-CGCCAGGGTTTTCCCAGTCACGAC-3′), forward primer (5′-CGCCAGGGTTTTCCCAGTCACGACGGCTGTGCCAGCGACAGT-3′), reverse primer (5′-GCAGAAGGTGCGCATAGTG-3′) and sequencing primer (5′-AAGCCAGCACTGGAC-3′). PCR amplification was performed as follows: initial PCR activation step at 95 °C for 15 min; 45 cycles of denaturation at 94 °C for 30 s, annealing at 65 °C for 30 s and extension at 72 °C for 30 s; and a final extension step at 72 °C for 10 min. PCR amplification was performed twice. The forward primer and reverse primer were used for the first amplification, whereas the biotinylated primer was used for the second amplification.11
Copy number and gene expression analyses
Copy number variation analysis with WES data was performed using Ion Reporter. Real-time PCR was used for copy number and gene expression analyses with the StepOnePlus Real-Time PCR System (Thermo Fisher Scientific). The QuantStudio3D system (Thermo Fisher Scientific) was used for digital PCR analyses. TaqMan primers/probes Hs00270415_cn (Chr11:64577357 on build GRCh37) (Probe1) and Hs01778293_cn (Chr11:64571825 on build GRCh37) (Probe2) (Thermo Fisher Scientific) were used for the analyses. For complimentary DNA synthesis, 1 μg of total RNA was used. The Omniscript Kit (Qiagen) and oligo(dT) primer (Thermo Fisher Scientific) were used for reverse transcription PCR according to the manufacturer’s protocol.
The MEN1 gene expression in tumors obtained from the present case was compared with that in other types of pancreatic tumors, including adenocarcinoma, adenosquamous carcinoma and endocrine tumors of the pancreas, in non-MEN1 patients selected from 2,042 cases in our cancer genome project, Project HOPE.12
Immunohistochemistry
Immunohistochemical studies were performed for the cellular localization of menin-like immunoreactivity using normal and tumor tissues obtained from the present case. Sections of a pancreatic tumor (Pancreas-1) and a duodenal tumor were studied. Tissue sections with a thickness of 3 μm were incubated with two anti-menin antibodies; the N-terminal antibody B-9 (Santa Cruz Biotechnology, Dallas, TX, USA) recognizes amino-terminal region of menin and the C-terminal antibody ab103561 (Abcam, Cambridge, UK) recognizes the carboxyl terminal region of menin. Both antibodies were used at the dilution of 1:100 (Supplementary Figure 1). Following the peroxidase reaction, the existence of specific immunostaining was evaluated.
Results
Novel MEN1 germline mutation in a patient with MEN1
Tissue samples were collected from a patient with MEN1 during removal of pancreatic and duodenal tumors, and peripheral blood was collected during total pancreatectomy. When the three tumor samples and blood were evaluated by WES, a novel germline mutation in the MEN1 gene was identified upon validation of the WES results using Integrative Genomics Viewer. Because the MEN1 gene was the minus strand, an insertion of thymine in Chr11:64572133–64572134 was detected in all samples (Figure 2a). The thymine insertion was confirmed using Sanger sequencing (Figure 2b) and pyrosequencing (Figure 2c). The insertion of adenine in exon 10 codon 502 of the MEN1 gene, which caused a frame shift and a stop codon in codon 530, was also identified. This is a novel MEN1 germline mutation [NM_137099.2:c.1505dupA (p.Lys502Lysfs); the localization was Chr11:64572134 on Assembly GRCh37] in patients with MEN1.
With regard to somatic mutations in the two pancreatic tumors, we did not detect genetic changes in 609 cancer-associated genes belonging to the COSMIC cancer gene census (http://cancer.sanger.ac.uk/census). In one pancreatic tumor (Pancreas-1), a somatic mutation of RPS16 was detected, and in the other pancreatic tumor (Pancreas-2), that of C1orf100 was detected; both genes were not included in the list of cancer-associated genes. With regard to the duodenal tumor, somatic mutations were found in SF3B1 and CLTC, which were categorized as cancer-associated genes in the COSMIC cancer gene census. In addition, somatic mutations were detected in eight genes considered to be other genes (Table 1).
In the Comprehensive Cancer Panel (CCP) analyses for 409 cancer-associated genes, none of the somatic mutations were detected in the two pancreatic tumors and the duodenal tumor; two cancer-associated genes detected by the WES study, SF3B1 and CLTC, were not included in the CCP.
Genotype of the MEN1 gene mutation
The frequency of the mutant allele identified by the Sanger sequencing and that identified by pyrosequencing was comparable. The frequency of the mutant allele was ~50% in the blood, 80% in the pancreatic tumors and 90% in the duodenal tumor. The presence of the wild-type allele in tumor tissue samples was considered to represent contamination by normal tissue.
A difference in MEN1 gene copy number between samples was suggested by Ion Reporter in the WES analysis. Therefore, real-time and digital PCR were performed to assess the gene copy number. Comparable results were obtained using TaqMan probes targeting either the 5′ or 3′ end of the MEN1 gene (Figure 3a): the blood and duodenal tumors had two copies each, whereas the two pancreatic tumors had one copy (Figure 3b). These results were in agreement with those inferred from the depth of WES. Table 1 summarizes the genotype of each sample obtained from the patient: the blood was heterozygous, the pancreatic tumors were hemizygous and the duodenal tumor was homozygous for MEN1.
MEN1 gene expression
Using RNA extracted from normal and tumor tissues of the pancreas, MEN1 gene expression was analyzed by real-time PCR. In this study, tissues obtained from the patients with MEN1 were compared with the samples obtained from the patients with pancreatic tumors including adenocarcinoma, adenosquamous carcinoma and sporadic endocrine tumor. The tumor tissues obtained from patients with adenocarcinoma and adenosquamous carcinoma had cancer-associated gene mutations (TP53, KRAS and CDKN2A) and the tumor tissue from a patient with a sporadic endocrine tumor had neither cancer-associated gene mutation nor abnormal hormone secretion. MEN1 gene expression in normal tissue of the MEN1 patient was approximately fivefold higher than that observed in normal tissue of patients with non-endocrine carcinomas, suggesting overexpression of the wild-type MEN1 gene; the high MEN1 expression in the tumor of the MEN1 patient may be attributed to contamination with normal tissue (Supplementary Figure 2).
Immunohistochemistry
In the pancreatic tumor, normal pancreatic cells as well as tumor cells were positively stained by the N-terminal antibody (Figure 4a). In contrast, the C-terminal antibody positively stained normal cells, but tumor cells were negative (Figure 4b). In the duodenal tumor, tumor cells were positively stained by the N-terminal antibody (Figure 4c), but they were not stained by the C-terminal antibody (Figure 4d). In the cells determined to be positive by the N-terminal antibody, cell nuclei were stained in both normal and tumor cells, and in tumor cells the staining was relatively stronger than that in normal cells, suggesting the accumulation of truncated protein in the nuclei of tumor cells.
Immunohistochemical staining of menin for normal and tumor tissues of a patient with MEN1. (a) Menin staining in the pancreas using an N-terminal menin antibody. (b) Menin staining in the pancreas using a C-terminal menin antibody. Tumor can be seen on the right side of the micrograph, while normal tissue can be seen on the left side. (c) Menin staining in the duodenum using an N-terminal menin antibody. (d) Menin staining in the duodenum using a C-terminal antibody (both micrographs show tumor tissues). MEN1, multiple endocrine neoplasia type 1.
Discussion
MEN1 is an autosomal dominant hereditary disease, and to date, more than 600 germline mutations in the MEN1 gene have been identified.13,14 In the present study, we analyzed blood cells and surgically resected tissues obtained from a patient with MEN1, and identified a novel germline mutation in the MEN1 gene. The validation studies by Sanger sequencing and pyrosequencing revealed that the mutation was an insertion of adenine in exon 10, codon 502 of the MEN1 gene, which caused a frame shift and introduced a stop codon in codon 530.
The copy number analyses showed that the mutant allele was heterozygous in blood cells, hemizygous in the two pancreatic tumors and homozygous in the duodenal tumor. It is possible to postulate that tumorigenesis in this case can be explained by zygosity of the mutated MEN1 gene, as is the example of the Knudson’s two-hit hypothesis:15 the first hit is the germline MEN1 mutation, and the second hit is the loss of the wild-type allele. In the present case, the second hit in two pancreatic tumors with hemizygosity might be induced by subchromosomal deletion or unbalanced translocation.16 In the duodenal tumor with homozygosity, loss of heterozygosity could be explained by duplication of the chromosome carrying the mutated MEN1 gene or by mitotic recombination.16
Because hereditary cancer syndromes are rare morbidities, there are few opportunities to analyze somatic mutations in multicentric tumors obtained from one patient. Our WES analysis of the two pancreatic tumor tissues revealed that, except for a germline MEN1 mutation, none of these tumors possessed tumor-specific somatic mutations in the 609 cancer-associated genes designated by the COSMIC cancer gene census (http://cancer.sanger.ac.uk/census). The CCP evaluation confirmed these results for 409 cancer-associated genes. Furthermore, changes of genes such as PHLDA3 reported to be important for the formation of pancreatic NET17 were not detected. Moreover, no somatic fusion genes with the activity of cancer driver genes were detected in these tumors.6 With regard to the remaining genes (~18700), one tumor-specific mutation in each of the pancreatic tumors was detected. Because these changes are likely to be passenger mutations and are not associated with tumor development, MEN1 gene mutation played a major role in tumorigenesis in the two pancreatic tumors. In the duodenal tumor, tumor-specific single-nucleotide variations of two cancer-associated genes and eight other genes were found. Further studies are required to determine whether somatic mutations of cancer-associated genes contribute to tumorigenesis. It is worth noting that the tumors in these patients possess the lowest number of tumor-specific mutations, i.e., 1–10 per tumor (nonsynonymous), when compared to more than 2,000 tumors evaluated in the Project HOPE cohort (mean; 128.7 non-synonymous mutations, unpublished data). This observation may reflect the fact that endocrine tumors developed in patients with MEN1 are low-grade malignancies in many cases.
To analyze the roles of menin, we investigated the molecular dynamics of menin with immunohistochemistry in the present case. The MEN1 genetics of the present case provide us a unique opportunity to assess menin dynamics for several reasons; (i) The truncated menin in this patient is supposed to be menin (1–529), which loses the C-terminal portion of intact menin. The position of the truncation was located between two nuclear localization signals; (ii) The mutated MEN1 gene is heterozygous in normal tissues, but hemizygous or homozygous in tumor tissues. This finding indicates that normal tissues contain both intact and truncated menin, and that tumor tissues contain only truncated protein; (iii) Two antibodies recognizing the N-terminal and C-terminal portions of menin are available in the present study. The immunohistochemical studies demonstrated that cell nuclei of normal tissues were stained by both antibodies, indicating that both intact and truncated menin are present in normal cells. In contrast, cell nuclei of tumor tissues were stained by the N-terminal antibody, but they were not stained by the C-terminal antibody. Since the staining in tumor cell nuclei is strongly positive, it is reasonable to speculate that the truncated menin accumulates in the nuclei of the tumor cells. It is worth noting that menin possesses two nuclear localization signals in the C-terminus, and that the mutation and stop codon in the present case were located between the two nuclear localization signals. Guru et al.18 reported that the presence of one nuclear localization signals could explain the accumulation of truncated menin in the nuclei.
Many studies have elucidated the function of menin in cell biology. Matkar et al.4 recently proposed that menin is a scaffold or adaptor protein that regulates gene expression both positively and negatively, and interacts with many proteins with diverse functions. Because our genetic analyses suggest that truncated menin plays a major role in the tumorigenesis of multicentric pancreatic and endocrine tumors, further genetic and biochemical analyses on these tumors including the gene expression profile may provide further information on the pathogenic role of menin identified here.
References
Chandrasekharappa SC, Guru SC, Manickam P, Olufemi SE, Collins FS, Emmert-Buck MR et al. Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 1997; 276: 404–407.
Schussheim DH, Skarulis MC, Agarwal SK, Simonds WF, Burns AL, Spiegel AM et al. Multiple endocrine neoplasia type 1: new clinical and basic findings. Trends Endocrinol Metab 2001; 12: 173–178.
Huang J, Gurung B, Wan B, Matkar S, Veniaminova NA, Wan K et al. The same pocket in menin binds both MLL and JUND but has opposite effects on transcription. Nature 2012; 482: 542–546.
Matkar S, Thiel A, Hua X . Menin: a scaffold protein that controls gene expression and cell signaling. Trends Biochem Sci 2013; 38: 394–402.
Fred T, Bosman FC, Ralph HH, Neil DT . WHO Classification of Tumours of the Digestive System. Vol. 3. IARC Press: Lyon, France, 2010.
Urakami K, Shimoda Y, Ohshima K, Nagashima T, Serizawa M, Tanabe T et al. Next generation sequencing approach for detecting 491 fusion genes from human cancer. Biomed Res 2016; 37: 51–62.
Robinson JT, Thorvaldsdottir H, Winckler W, Guttman M, Lander ES, Getz G et al. Integrative genomics viewer. Nat Biotechnol 2011; 29: 24–26.
Thorvaldsdottir H, Robinson JT, Mesirov JP . Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief Bioinform 2013; 14: 178–192.
Nagashima T, Shimoda Y, Tanabe T, Naruoka A, Saito J, Serizawa M et al. Optimizing an ion semiconductor sequencing data analysis method to identify somatic mutations in the genomes of cancer cells in clinical tissue samples. Biomed Res 2016; 37: 359–366.
Shimoda Y, Nagashima T, Urakami K, Tanabe T, Saito J, Naruoka A et al. Integrated next-generation sequencing analysis of whole exome and 409 cancer-related genes. Biomed Res 2016; 37: 367–379.
Royo JL, Hidalgo M, Ruiz A . Pyrosequencing protocol using a universal biotinylated primer for mutation detection and SNP genotyping. Nat Protoc 2007; 2: 1734–1739.
Yamaguchi K, Urakami K, Ohshima K, Mochizuki T, Akiyama Y, Uesaka K et al. Implementation of individualized medicine for cancer patients by multiomics-based analyses-the Project HOPE. Biomed Res 2014; 35: 407–412.
Concolino P, Costella A, Capoluongo E . Multiple endocrine neoplasia type 1 (MEN1): an update of 208 new germline variants reported in the last nine years. Cancer Genet 2016; 209: 36–41.
Guru SC, Manickam P, Crabtree JS, Olufemi SE, Agarwal SK, Debelenko LV . Identification and characterization of the multiple endocrine neoplasia type 1 (MEN1) gene. J Intern Med 1998; 243: 433–439.
Knudson AG Jr . Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA 1971; 68: 820–823.
Devilee P, Cleton-Jansen AM, Cornelisse CJ . Ever since Knudson. Trends Genet 2001; 17: 569–573.
Ohki R, Saito K, Chen Y, Kawase T, Hiraoka N, Saigawa R et al. PHLDA3 is a novel tumor suppressor of pancreatic neuroendocrine tumors. Proc Natl Acad Sci USA 2014; 111: E2404–E2413.
Guru SC, Goldsmith PK, Burns AL, Marx SJ, Spiegel AM, Collins FS et al. Menin, the product of the MEN1 gene, is a nuclear protein. Proc Natl Acad Sci USA 1998; 95: 1630–1634.
Acknowledgements
The authors thank the staff at the Shizuoka Cancer Center Hospital for the clinical support and sample preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information for this article can be found on the Human Genome Variation website
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Naruoka, A., Ohnami, S., Nagashima, T. et al. Germline and somatic genetic changes in multicentric tumors obtained from a patient with multiple endocrine neoplasia type 1. Hum Genome Var 4, 17013 (2017). https://doi.org/10.1038/hgv.2017.13
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/hgv.2017.13
- Springer Nature Limited