Abstract
The nervous system plays an important but poorly understood role in modulating longevity. GABA, a prominent inhibitory neurotransmitter, is best known to regulate nervous system function and behaviour in diverse organisms. Whether GABA signalling affects aging, however, has not been explored. Here we examined mutants lacking each of the major neurotransmitters in C. elegans, and find that deficiency in GABA signalling extends lifespan. This pro-longevity effect is mediated by the metabotropic GABAB receptor GBB-1, but not ionotropic GABAA receptors. GBB-1 regulates lifespan through G protein-PLCβ signalling, which transmits longevity signals to the transcription factor DAF-16/FOXO, a key regulator of lifespan. Mammalian GABAB receptors can functionally substitute for GBB-1 in lifespan control in C. elegans. Our results uncover a new role of GABA signalling in lifespan regulation in C. elegans, raising the possibility that a similar process may occur in other organisms.
Similar content being viewed by others
Introduction
Aging is a complex physiological process modulated by a multitude of genetic and environmental factors1,2. Recent work has revealed an important role of the nervous system in modulating aging1,3,4. For example, in insulin/IGF-1 signalling, one of the best characterized longevity-regulatory pathways, the DAF-2 insulin/IGF-1 receptor primarily acts in the nervous system to regulate lifespan in Caenorhabditis elegans5. Interestingly, work in mice revealed a similar mechanism6. Environmental factors also affect aging by impinging on the nervous system1,3,4,7,8. Apparently, neurons can act cell nonautonomously to modulate aging, and neuroendocrine signalling plays an important role in this process1,3,4,7,8. However, whether and how neurotransmitter signalling modulates aging is poorly understood.
GABA is the primary inhibitory neurotransmitter in the mammalian brain9,10. It is critical for the function and development of the nervous system and plays a key role in behavioural control11,12,13,14. GABA acts through both ionotropic and metabotropic receptors15,16. As ion channels, ionotropic GABAA receptors mediate the acute, fast actions of GABA17,18. By contrast, metabotropic GABAB receptors are G protein coupled receptors (GPCRs) that execute the slow, long-lasting effects of GABA19,20,21,22.
C. elegans is a popular model organism widely utilized to study the biology of aging, featuring a short generation time and lifespan, as well as conserved genetic mechanisms that regulate longevity1,2. C. elegans has also been widely used as a genetic model for neurobiology23. Although C. elegans possesses a relatively small nervous system, it produces all the major neurotransmitters (for example, ACh, glutamate, GABA and biogenic amines) and their cognate receptors, all of which are encoded by evolutionarily conserved gene families23. While the role of neurotransmitters in controlling behaviour and nervous system development and function has been extensively characterized, little is known about their role in aging.
Here we interrogated the role of neurotransmitters in lifespan control in C. elegans and found that GABA regulates lifespan through GABAB receptor signalling. We further identified a genetic pathway that cell nonautonomously transmits longevity signals from the GABAB receptor in motor neurons to the transcription factor FOXO/DAF-16 in the intestine. Our results identify a novel function of GABA beyond its conventional role in modulating animal behaviour and nervous system function and development.
Results
Deficiency in GABA signalling extends lifespan
The nervous system of C. elegans hermaphrodites is relatively simple23,24, making it convenient to characterize the role of neurotransmission in lifespan control. We first assayed the lifespan of unc-13 mutant worms that are defective in synaptic transmission25,26. These mutant animals are long lived (Fig. 1a and Supplementary Table 1), consistent with previous results27. The fact that unc-13 mutant animals are long-lived indicates that deficiency in neurotransmitter signalling extends lifespan. To ascertain which neurotransmitter(s) may contribute to this effect, we examined mutant animals defective in each of the major neurotransmitters, including unc-17 (ACh), eat-4 (glutamate), cat-2 (dopamine), tph-1 (serotonin), tdc-1 (tyramine), tbh-1 (octopamine) and unc-25 (GABA)28,29,30,31,32,33 (Fig. 1b–h). Among these mutants, unc-25 worms lived the longest life, showing the most pronounced phenotype (Fig. 1h), while others only had modest or no effect (Fig. 1b–g). While these data by no means exclude a role of other neurotransmitters, it reveals an important function of GABA signalling in lifespan control.
(a) unc-13 mutant worms are long-lived. UNC-13 is required for the release of neurotransmitters (log-rank test, P<0.001, n=74–82 for different genotypes). (b–g) Loss of octopamine (b), dopamine (c), glutamate (d), tyramine (e), serotonin (f) or acetylcholine signalling (g) modestly affect lifespan (log-rank test, P=0.755, P=0.811, P=0.371, P=0.046, P=0.069 and P=0.912, respectively. n=44–114 for different genotypes). (h) Loss of GABA signalling extends lifespan (log-rank test, P<0.001, n=57–85 for different genotypes). All lifespan assays were carried out at 20 °C and were repeated at least twice. 5-Fluoro-2′-deoxyuridine (FUDR) was included in assays involving unc-13 and unc-17 mutant worms, which are defective in egg-laying. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
Loss of the GABAB receptor gene gbb-1 extends lifespan
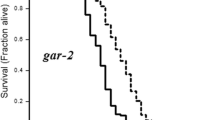
GABA acts through both ionotropic and metabotropic receptors. The C. elegans genome encodes two metabotropic GABAB receptor genes, gbb-1 and gbb-2, which are highly homologous to their mammalian counterparts34. We found that gbb-1 but not gbb-2 mutants were long-lived (Fig. 2a). gbb-1;gbb-2 double mutant was indistinguishable from gbb-1 single mutant (Fig. 2a), indicating a specific role of gbb-1 in regulating lifespan. By contrast, mutant worms lacking unc-49, which encodes the sole evolutionarily conserved GABAA receptor family member35, were normally lived (Fig. 2b). These results identify a role for the GABAB receptor gene gbb-1 in lifespan control.
(a) gbb-1 mutant worms are long-lived, but gbb-2 mutant worms are not (log-rank test, P<0.001, gbb-1 mutant worms compared with wild type; P=0.518, gbb-2 mutant worms compared with wild type; P<0.001, gbb-1;gbb-2 mutant worms compared with wild type. n=44–67 for different genotypes). (b) unc-49 mutant worms show normal lifespan (log-rank test, P=0.353, n=67–76 for different genotypes). All lifespan assays were carried out at 20 °C and were repeated at least three times. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
gbb-1 is enriched in the nervous system but is also found to be expressed in other tissues such as muscle and intestine34. We therefore asked where gbb-1 acts to regulate lifespan. Using tissue-specific promoters, we found that expression of gbb-1 cDNA in neurons, but not in muscle or intestine, rescued gbb-1 lifespan phenotype (Fig. 3a). Thus, gbb-1 appears to act in neurons to regulate lifespan.
(a) Transgenic expression of gbb-1 in neurons (log-rank test, P=0.198), but not in the intestine (log-rank test, P<0.001) or muscles (log-rank test, P<0.001), suppresses the long-lived phenotype of gbb-1 mutant worms (log-rank test, P<0.001). The rgef-1, ges-1 and myo-3 promoters were used to drive gbb-1 cDNA expression in neurons, intestine and muscles, respectively61,62,63 (n=47–63 for different genotypes). (b) Overexpression of gbb-1 cDNA in neurons shortens lifespan (log-rank test, P=0.001, n=53–72 for different genotypes). Each plasmid DNA listed above was injected at a concentration of 50 ng μl−1. All lifespan assays were carried out at 20 °C and were repeated at least three times. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
We crossed the gbb-1 neuronal transgene into wild-type background and found that it rendered worms short-lived (Fig. 3b). Thus, overexpression of gbb-1 shortens lifespan. This is consistent with our mutant data that loss of gbb-1 extends lifespan, providing further evidence supporting that gbb-1 regulates longevity.
GBB-1-dependent lifespan regulation requires DAF-16/FOXO
We wondered how gbb-1 controls lifespan. Various genetic and environmental factors regulate lifespan by converging on a subset of transcription factors1. We therefore examined the major transcription factors known to regulate lifespan, and found that RNAi of daf-16 completely suppressed the lifespan-extending phenotype of gbb-1 mutant worms (Fig. 4a), while RNAi of other transcription factors such as hsf-1, skn-1 and pha-4 did not (Fig. 4b–d). Thus, daf-16 is required for the function of gbb-1, suggesting that gbb-1 signals to daf-16 to regulate lifespan.
(a) daf-16 RNAi fully suppresses the long-live phenotype of gbb-1 mutant worms (log-rank test, P=0.502, n=78–91 for different genotypes). (b–d) RNAi of pha-4 (b, log-rank test, P<0.001), skn-1 (c, log-rank test, P<0.001) or hsf-1(d, log-rank test, P<0.001) fail to suppress the long-lived phenotype of gbb-1 mutant worms (n=79–94 for different genotypes). For RNAi experiments, NGM plates included carbenicillin (25 μg ml−1) and IPTG (1 mM). HT115 bacteria-carrying vector or RNAi plasmid were seeded on RNAi plates 2 days before the experiment. Worms were fed RNAi bacteria from the egg stage. All RNAi lifespan assays were carried out at 20 °C, and were repeated at least three times. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
Gi/o-PLCβ transmits longevity signals from GBB-1 to DAF-16
DAF-16 is best known to act downstream of insulin/IGF-1 signalling to regulate lifespan. Does GBB-1 genetically interact with insulin/IGF-1 signalling? To test this, we assayed the lifespan of gbb-1;daf-2 double mutant. daf-2 encodes the sole worm insulin/IGF-1 receptor homologue36. The double mutant exhibited a lifespan significantly longer than both gbb-1 and daf-2 single mutants (Supplementary Fig. 1), suggesting that gbb-1 and daf-2 may act in different pathways. However, as a reduction-of-function allele of daf-2 was used here (daf-2 nulls are lethal), we did not further test this model. Thus, we do not rule out the possibility that gbb-1 and daf-2 may act in the same or overlapping pathways.
Then how does GBB-1 signal to DAF-16? As a GPCR, GBB-1 is unlikely to communicate with DAF-16 directly. We then sought to identify genes that transduce longevity signals from GBB-1 to DAF-16. As the GABAB receptor is known to be coupled to Gi/o-PLCβ signalling10,37, we examined this pathway. The worm genome encodes one Go orthologue, goa-1 (refs 38, 39), and at least half a dozen Gi-related genes40. However, unlike gbb-1, goa-1 mutant worms are not long-lived (Supplementary Fig. 2), suggesting the presence of functional redundancy from other Gi/o genes. To overcome this difficulty, we attempted to block Gi/o signalling using PTX (Pertussis toxin) by expressing it as a transgene in worm neurons. We found that inactivation of Gi/o by PTX extended lifespan (Fig. 5a). This PTX-dependent lifespan extension also required daf-16, as it can be blocked by daf-16 RNAi (Fig. 5a), suggesting that Gi/o may act upstream of DAF-16 to regulate lifespan. Further, the PTX transgene also suppressed the short-lived phenotype caused by overexpression of gbb-1 (Fig. 5b), suggesting that Gi/o acts downstream of gbb-1. These data support our model that Gi/o acts downstream of GBB-1 but upstream of DAF-16.
(a) Inhibition G protein function by transgenic expression of PTX in neurons extends lifespan (log-rank test, P<0.001), which can be fully suppressed by daf-16 RNAi (log-rank test, P=0.046). The H20 promoter drives expression of PTX in neurons64 (n=68–96 for different genotypes). (b) Transgenic expression of PTX fully suppresses the short-lived phenotype caused by gbb-1 transgene (log-rank test, P<0.001, xuEx2117 compared with the wild type; P=0.56, xuEx1964;xuEx2117 compared with xuEx1964. n=65–88 for different genotypes). (c) Loss of egl-8 extends lifespan, which can by fully suppressed by daf-16(mgDf47) mutation (log-rank test, P<0.001, egl-8 mutant worms compared with the wild type; P=0.781, egl-8;daf-16 mutant worms compared with daf-16 mutant worms. n=57–115 for different genotypes). (d) Loss of egl-8 fully suppresses the short-lived phenotype caused by gbb-1 overexpression (log-rank test, P<0.001, xuEx1967 compared with the wild type; P=0.382, egl-8;xuEx1967 compared with egl-8 mutant worms. n=72–102 for different genotypes). All lifespan assays were carried out at 20 °C and were repeated at least twice. 5-Fluoro-2′-deoxyuridine (FUDR) was included in assays involving egl-8 mutant worms, which show a defect in egg laying. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
The GABAB receptor is known to pass signals from Gi/o heterotrimeric G proteins to PLCβ (refs 10, 37). The worm genome encodes one PLCβ homologue, EGL-8 (refs 41, 42). Similar to gbb-1 mutant and PTX-transgenic worms, egl-8 mutant worms were long-lived (Fig. 5c), consistent with the model that egl-8 acts in the pathway. Importantly, the long-lived phenotype associated with egl-8 mutant worms was suppressed by daf-16 (Fig. 5c), suggesting that egl-8 acts upstream of daf-16. Furthermore, loss of egl-8 suppressed the short-lived phenotype caused by overexpression of gbb-1 (Fig. 5d), suggesting that egl-8 acts downstream of gbb-1. A prior study also showed that EGL-8 regulates lifespan in a DAF-16-dependent manner43. These results together support the model that G protein-PLCβ signalling transduces longevity signals from GBB-1 to DAF-16.
DKF-2/PKD acts upstream of DAF-16 to regulate lifespan
DAF-16 is best known to be regulated by kinases1. We next set out to identify kinases that act downstream of PLCβ but upstream of DAF-16. Protein kinase C (PKC), a group of kinases activated by DAG and/or Ca2+, is a primary downstream target of PLCβ. The worm genome encodes four PKC homologues: tpa-1, pkc-1, pkc-2 and pkc-3. However, none of these genes, when mutated or knocked down, gave rise to a long-lived phenotype as did egl-8 and gbb-1 (Supplementary Fig. 3a–d). These data suggest that PKC is unlikely to mediate the effect of EGL-8/PLCβ in longevity signalling.
In addition to PKC, DAG can also activate another type of kinases, protein kinase D (PKD), in both a PKC-dependent and -independent manner44. Two PKD homologues are found in C. elegans: dkf-1 and dkf-2 (ref. 45). We thus considered a potential role of these two PKD genes. dkf-1 mutant worms were short-lived (Supplementary Fig. 3e–f), suggesting that it is probably not involved. By contrast, worms lacking dkf-2 were long-lived (Fig. 6a), a phenotype similar to gbb-1 and egl-8 mutants. A previous study also showed that loss of dkf-2 extends lifespan and does so by regulating DAF-16 (ref. 45). Indeed, we found that the long-lived phenotype of dkf-2 worms was completely suppressed by loss of daf-16 (Fig. 6b), consistent with the notion that dkf-2 acts upstream of daf-16 to regulate lifespan.
(a) dkf-2 acts in the gbb-1 pathway to regulate lifespan. dkf-2 mutant worms were long-lived (log-rank test, P<0.001). dkf-2; gbb-1 double mutant showed a similar lifespan to single mutants (log-rank test, P=0.919), suggesting that they are in the same pathway (n=61–72 for different genotypes). (b) Loss of daf-16 fully suppresses the long-lived phenotype of dkf-2 mutant worms (log-rank test, P=0.114, n=72–82 for different genotypes). (c) qPCR analysis of DAF-16 target genes. qPCR reactions were run in triplicates for each genotype. Each experiment was repeated three times. Error bars, s.e.m. *P<0.05 (analysis of variance (ANOVA) with Bonferroni test). (d) Quantification of SOD-3::GFP fluorescence intensity. SOD-3::GFP is encoded by the transgene muIs84 (ref. 46). The left panels show representative images (selected from 10 similar images). Image quantification was performed as previously described56. n≥15. Error bars, s.e.m. **P<0.001 (t-test). Scale bar, 100 μm. (e) Overexpression of dkf-2 in neurons fully suppresses the long-lived phenotype of dkf-2 mutant worms. Prgef-1 is a neuron-specific promoter62 (log-rank test, P=0.143, n=77–98 for different genotypes). (f) Overexpression of dkf-2 modestly shortens lifespan (log-rank test, P=0.005, n=58–69 for different genotypes). (g) Overexpression of dkf-2 fully suppresses the long-lived phenotype of gbb-1 mutant worms (log-rank test, P=0.462, n=44–72 for different genotypes). (h) Overexpression of dkf-2 fully suppresses the long-lived phenotype of egl-8 mutant worms (log-rank test, P=0.967, n=50–98 for different genotypes). All lifespan assays were carried out at 20 °C and were repeated at least twice. FUDR was included in assays involving egl-8 mutant worms, which show a defect in egg laying. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
To obtain further evidence that dkf-2 regulates daf-16, we examined whether loss of dkf-2 can promote daf-16 function. We found that the mRNA levels of DAF-16 target genes, such as mtl-1, sod-3 and dod-3, were upregulated in dkf-2 mutant background (Fig. 6c). We also assayed the protein level of SOD-3::GFP fusion, a commonly used reporter for DAF-16 activity, and found that it was upregulated in dkf-2 worms (Fig. 6d). Thus, dkf-2 appears to regulate daf-16 function, providing additional evidence supporting that DKF-2 acts upstream of DAF-16 to control lifespan.
DKF-2 acts downstream of EGL-8 to regulate lifespan
We then asked whether dkf-2 functions in the gbb-1 pathway to regulate lifespan. dkf-2;gbb-1 double mutant exhibited a lifespan similar to single mutants (Fig. 6a), suggesting that they act in the same pathway. To gather additional evidence, we first rescued dkf-2 long-lived phenotype by expressing wild-type dkf-2 cDNA as a transgene in neurons in dkf-2 mutant worms (Fig. 6e). In addition, this dkf-2 transgene modestly shortened lifespan in wild-type background (Fig. 6f and Supplementary Table 1). Importantly, this transgene suppressed the long-lived phenotype of gbb-1, as well as egl-8 mutant worms (Fig. 6g–h), providing additional evidence supporting that DKF-2 acts downstream of GBB-1 and EGL-8/PLCβ to regulate lifespan.
Neuron-to-intestine signalling transmits longevity signals
Apparently, the aforementioned genetic pathway, which transmits longevity signals from GBB-1 to DAF-16, does not inform where the pathway operates. Our data showed that GBB-1 acts in neurons to regulate lifespan (Fig. 3a), yet DAF-16 is best known to function in the intestine46. This raises the question whether GBB-1 signals DAF-16 through neuron-to-intestine signalling. However, DAF-16 also functions in neurons46, which confounds our data interpretation. To clarify this issue, we interrogated the site of action of DAF-16 in the GBB-1 pathway. We found that transgenic expression of daf-16 cDNA in the intestine but not in neurons rescued the lifespan phenotype of gbb-1;daf-16 double mutant (Fig. 7a), indicating that DAF-16 acts in the intestine in the GBB-1 pathway.
(a) Transgenic expression of daf-16 in the intestine (log-rank test, P=0.892 compared with gbb-1 mutant worms), but not in neurons (log-rank test, P<0.001 compared with gbb-1 mutant worms), suppresses the short-lived phenotype of gbb-1;daf-16 double mutant worms. The rgef-1 and ges-1 promoters were used to drive daf-16 cDNA expression in neurons and the intestine, respectively (n=55–93 for different genotypes). (b) Transgenic expression of gbb-1 in subsets of interneurons fails to suppress the long-lived phenotype of gbb-1 mutant worms. The glr-1 promoter was used to drive gbb-1 cDNA expression in interneurons (log-rank test, P<0.001, n=37–60 for different genotypes). (c) Transgenic expression of gbb-1 in sensory neurons fails to suppress the long-lived phenotype of gbb-1 mutant worms. The osm-6 promoter was used to drive gbb-1 cDNA expression in sensory neurons (log-rank test, P<0.001, n=41–103 for different genotypes). (d) Transgenic expression of gbb-1 in ventral cord motor neurons fully suppresses the long-lived phenotype of gbb-1 mutant worms (log-rank test, P=0.799, n=35–70 for different genotypes). The acr-2 promoter was used to drive gbb-1 cDNA expression in ventral cord motor neurons. All lifespan assays were carried out at 20 °C and were repeated at least twice. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
GBB-1 is broadly expressed in the nervous system34. We further asked in which groups of neurons GBB-1 regulates lifespan. Transgenic expression of gbb-1 cDNA in sensory neurons under the osm-6 promoter did not rescue gbb-1 mutant phenotype (Fig. 7c), nor did expression of gbb-1 in subsets of interneurons using the glr-1 promoter (Fig. 7b). By contrast, restoring gbb-1 in ventral cord motor neurons through the acr-2 promoter fully rescued the gbb-1 longevity phenotype (Fig. 7d). Thus, GBB-1 can act in ventral cord motor neurons to regulate lifespan. This suggests a motor neuron-to-intestine signalling axis that transmits longevity signals from GBB-1 to DAF-16.
Rat GABAB receptor can functionally substitute for GBB-1
We expressed the rat GABAB receptor, GB1/GB2 (refs 47, 48, 49), as a transgene in gbb-1 mutant background using a neuron-specific promoter, and found that the transgene rescued the long-lived phenotype of gbb-1 mutant worms (Fig. 8a). This suggests that mammalian GABAB receptor can functionally substitute for worm GBB-1 in regulating lifespan.
(a) Transgenic expression of rat GABAB receptors (rGB1 and rGB2) fully suppresses the long-lived phenotype of gbb-1 mutant worms (log-rank test, P=0.11, n=49–66 for different genotypes). (b,c) Mammalian GABAB receptor antagonist CGP36216 (b) and SCH50911 (c) do not have a notable effect on lifespan in wild-type worms (log-rank test, P=0.389 and P=0.527, respectively. n=57–127 for the different treatments). Drug concentration: 1 μM. (d,e) Mammalian GABAB receptor antagonist CGP36216 (d) and SCH50911 (e) extend the lifespan of transgenic worms expressing rat GABAB receptor (log-rank test, P<0.001 in the each case, n=44–87 for the different case). Drug concentration: 1 μM antagonist. (f) Schematic model illustrating a genetic pathway through which GABA regulates longevity. The putative site-of-action of each gene (neuron versus intestine) is denoted. All lifespan assays were carried out at 20 °C and were repeated at least twice with the exception of b,c. Please see Supplementary Table 1 for detailed statistical analysis of lifespan data.
Unlike worm GBB-1, the mammalian GABAB receptor has been extensively characterized pharmacologically15,48,50,51. We thus took this advantage by testing some known antagonists of the GABAB receptor such as CGP36216 (ref. 52) and SCH50911 (ref. 53). These chemicals did not have a notable effect on lifespan in wild-type animals (Fig. 8b,c). Remarkably, when these chemicals were applied to transgenic worms expressing the rat GABAB receptor GB1/GB2, they extended lifespan and did so as efficiently as gbb-1 mutation (Fig. 8d,e). These data present further evidence supporting that the mammalian GABAB receptor can functionally substitute for worm GBB-1 in lifespan regulation, raising the possibility that it may play a similar role in mammals.
Discussion
As the primary inhibitory neurotransmitter, GABA is best known to regulate the function and development of the brain and it plays a key role in controlling animal behaviour11,12,13,14. In the current study, we identified a new role of GABA signalling in aging. Interestingly, the effect of GABA on longevity is mediated by GABAB rather than GABAA receptor. As a GPCR, GABAB receptor generally meditates the slow, long-lasting actions of GABA, which is consistent with its role in regulating longevity. A previous study reported that knocking down Drosophila GABAB receptor in insulin-producing cells shortens lifespan54, an effect opposite to that observed in C. elegans. However, unlike extended lifespan, it is difficult to interpret a short-lived phenotype. As such, it remains unclear whether GABA and GABAB receptor modulate aging in this organism. On the other hand, mammalian GABAB receptor can functionally substitute for its worm homologue in lifespan control; furthermore, antagonists of mammalian GABAB receptor can extend the lifespan of transgenic worms expressing this receptor, raising the possibility that GABA and GABAB receptor may play a role in aging in other organisms. It will be interesting to test this hypothesis in future studies.
Recent studies have uncovered an increasingly important role of the nervous system in longevity1,3,4,7,8. Neurons regulate lifespan in a cell-nonautonomous manner presumably through neurotransmission1,3,4,7,8. Aside from neuroendocrine signalling, neurotransmitters are also believed to play a key role in this process. For example, endoplasmic reticulum (ER) stress in neurons affects lifespan through neurotransmitter rather than neuroendocrine signalling55. The identity of this neurotransmitter(s), however, remains elusive. By characterizing mutant strains defective in each of the major neurotransmitters in C. elegans, we found that GABA signalling plays an important role in regulating lifespan. To the best of our knowledge, this represents the first comprehensive analysis examining the role of all neurotransmitters in lifespan control in any organism.
Our results uncovered a genetic pathway that transmits longevity signals from GABA via G protein-PLCβ signalling to the FOXO transcription factor DAF-16, which is a key regulator of lifespan (Fig. 8f). Specifically, this genetic pathway consists of GBB-1, G protein, EGL-8/PLCβ, DKF-2/PKD and DAF-16/FOXO (Fig. 8f). It is interesting that PKD rather than PKC transmits signals from PLCβ in our case, although PKC plays an important role in temperature-dependent lifespan regulation in C. elegans56. One remaining question concerns how DKF-2 regulates DAF-16. The simplest model would be that DKF-2 phosphorylates DAF-16. However, we did not detect such phosphorylation in in vitro kinase assays (J.G., X.Z.S.X. and J.L., unpublished data). This suggests that DKF-2 likely regulates DAF-16 indirectly. Consistent with this model, we found that GBB-1 acts in motor neurons while DAF-16 functions in the intestine. Expression of DKF-2 in neurons is also sufficient to rescue dkf-2 lifespan phenotype. Other components in the GBB-1 genetic pathway are also best known to act in the nervous system41,42. This reveals a motor neuron-to-intestine signalling axis that transmits longevity signals from GBB-1 to DAF-16. Recent studies have pointed to a critical role of such neuron-to-intestine signalling in lifespan control55,57. However, exactly how longevity signals are transmitted between neurons and intestinal cells remains a difficult question to address. Future efforts are needed to unravel the underlying mechanisms.
Despite the observation that loss of GABA signalling seems to elicit the most pronounced effect on lifespan among all the major neurotransmitters, this does not necessarily indicate that other neurotransmitters do not have a role in lifespan control. Previous studies have demonstrated that worms experience a decline in dopamine and serotonin levels with age58. Octopamine has also been reported to play a key role in mediating CREB-regulated transcription coactivator (CRTC)-dependent lifespan regulation in neurons57. These neurotransmitters all bind to multiple types of receptors, including both GPCRs and ion channels. It is possible that their receptors have opposing effects on longevity, with some promoting lifespan and others inhibiting it, which may account for the modest effect resulting from a complete loss of these neurotransmitters. It is also possible that these neurotransmitters may play a more prominent role in controlling lifespan under certain specific physiological conditions. Our studies will encourage others to investigate how the nervous system regulates lifespan through neurotransmitter signalling, an interesting but poorly understood question in the biology of aging.
Methods
Genetics and molecular biology
Wild-type: N2. TQ2181:unc-13(e51). TQ4927: tbh-1(n3247) × 8 outcrossed. TQ4935: cat-2(e1112) × 8 outcrossed. TQ4933: eat-4(ky5) × 8 outcrossed. TQ4929: tdc-1(n3419) × 8 outcrossed. TQ4931: tph-1(mg280) × 8 outcrossed. unc-17(e245) × 4 outcrossed. TQ6057: unc-25(e156) × 4 outcrossed. TQ4912: gbb-1(tm1406) × 8 outcrossed. TQ4911: gbb-2(tm1165) × 8 outcrossed. TQ6054: unc-49(e407) × 4 outcrossed. TQ5117: xuEx1611[Pges-1::gbb-1::SL2::mCherry]. TQ5119: xuEx1613[Pmyo-3::gbb-1::SL2::mCherry]. TQ5123: xuEx1617[Prgef-1::gbb-1::SL2::mCherry]. TQ5842: xuEx1964[PH20::ptx+Prgef-1::DsRed]. xuEx2117[Prgef-1::gbb-1::SL2::YFP]. TQ4914: egl-8(n488) × 4 outcrossed. TQ4077: daf-16(mgDf47) × 4 outcrossed. TQ5450: dkf-2(pr3) × 6 outcrossed. xuEx2106[Prgef-1::dkf-2::SL2::mCherry], xuEx2109[Prgef-1::dkf-2::SL2::YFP], TQ5940: xuEx1976[Prgef-1::rGB1::SL2::mCherry+Prgef-1::rGB2::SL2::YFP]. TQ5492: goa-1(sa734) × 2 outcrossed. TQ4915: tpa-1(k530) × 4 outcrossed. TQ5576: pkc-1(nj3) × 4 outcrossed. TQ5008: dkf-1(ok2695) × 4 outcrossed. TQ5447: dkf-1(pr2) × 6 outcrossed. The pkc-3 RNA interference (RNAi) clone was generated in the laboratory, while other RNAi clones were from the Ahringer library and were confirmed by sequencing. All mutants in the GBB-1 pathway are null alleles, including gbb-1, egl-8, dkf-2 and daf-16. These mutants have been outcrossed to our N2 strain for four to eight times before lifespan assay.
Microinjections were performed using standard protocols. Each plasmid DNA listed above in the transgenic line was injected at a concentration of 50 ng μl−1. For those experiments involving transgenes, three independent transgenic lines were tested for lifespan to confirm the results. For simplicity and clarity, only the data from one transgenic line were shown.
Lifespan assay
Lifespan studies were performed on 60-mm nematode growth medium (NGM) plates at 20 °C as previously described56,59,60. For each lifespan assay, 70–110 worms were included and transferred every other day to fresh NGM plates with 14 worms per plate. The first day of adulthood was considered day 1. Survival was scored every 1–2 days, and worms were censored if they crawled off the plate, hatched inside or lost the vulva integrity during reproduction. 5-Fluoro-2′-deoxyuridine was included in assays involving unc-13, unc-17 and egl-8 mutant worms, which show egg-laying defects. For RNAi experiments, NGM plates included carbenicillin (25 μg ml−l) and isopropyl-β-D-thiogalactoside (1 mM). HT115 bacteria with vector or RNAi plasmid were seeded on RNAi plates 2 days before experiment. Worms were fed RNAi bacteria, beginning at the egg stage.
For GABAB receptor antagonist experiments, 1 μM CGP36216 or SCH50911 (Tocris) was first spread out on NGM plates to let it diffuse for 1 day. L4 hermaphrodites were then transferred to these plates to assay lifespan. Worms were transferred to fresh drug plates every 2–3 days until day 10, after which animals remained on the same drug plates until death.
Graphpad Prism 5 (GraphPad Software Inc.) and IBM SPSS Statistics 19 (IBM Inc.) were used to analyse lifespan data. P values were calculated with the log-rank (Kaplan–Meier) method.
Overexpression of rat GABAB receptor in C. elegans
cDNA encoding the rat GABAB receptor subunits, GB1 or GB2, was driven by the neuron-specific promoter rgef-1. The two plasmids were co-injected (50 ng μl−1 each) into gbb-1 mutant worms to generate the transgenic animals overexpressing rat GABAB receptor.
qRT–PCR and microscopy
Total RNA was isolated from ∼200 Day-3 adult worms using TRI Reagent (Life Technologies). Quantitative PCR (qPCR) experiments were performed with CYBR Green (Life Technologies) according to the protocol provided by the manufacturer to analyse the amount of mRNA of daf-16 target genes. qPCR data were analysed with the ΔΔCt method using act-1 (actin) as an internal reference for normalization. Primer sequences used for qPCR are (all 5′–3′) listed as below. act-1: 5′-CCAGGAATTGCTGATCGTATGCAGAA-3′, 5′-TGGAGAGGGAAGCGAGGATAGA-3′. mtl-1: 5′-TGCAGTCTCCCTTACATCCA-3′, 5′-TGCAGTGGAGACAAGTGTTG-3′. sod-3: 5′-TATTAAGCGCGACTTCGGTTCCCT-3′, 5′-CGTGCTCCCAAACGTCAATTCCAA-3′. dod-3: 5′-AAAAAGCCATGTTCCCGAAT-3′, 5′-GCTGCGAAAAGCAAGAAAAT-3′.
Quantification of SOD-3::GFP fluorescence intensity was performed on an Olympus BX51 upright microscope as described previously56. Images were acquired with a Roper CoolSnap charge couple device camera controlled with MetaMorph (Molecular Devices Inc.) and analysed with ImageJ (NIH). SOD-3::GFP is encoded by the transgene muIs84 (ref. 46).
Additional information
How to cite this article: Chun, L. et al. Metabotropic GABA signalling modulates longevity in C. elegans. Nat. Commun. 6:8828 doi: 10.1038/ncomms9828 (2015).
References
Kenyon, C. J. The genetics of ageing. Nature 464, 504–512 (2010).
Fontana, L., Partridge, L. & Longo, V. D. Extending healthy life span--from yeast to humans. Science 328, 321–326 (2010).
Allen, E. N., Ren, J., Zhang, Y. & Alcedo, J. Sensory systems: their impact on C. elegans survival. Neuroscience 296, 15–25 (2014).
Jeong, D. E., Artan, M., Seo, K. & Lee, S. J. Regulation of lifespan by chemosensory and thermosensory systems: findings in invertebrates and their implications in mammalian aging. Front. Genet. 3, 218 (2012).
Wolkow, C. A., Kimura, K. D., Lee, M. S. & Ruvkun, G. Regulation of C. elegans life-span by insulinlike signaling in the nervous system. Science 290, 147–150 (2000).
Taguchi, A., Wartschow, L. M. & White, M. F. Brain IRS2 signaling coordinates life span and nutrient homeostasis. Science 317, 369–372 (2007).
Apfeld, J. & Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 402, 804–809 (1999).
Alcedo, J. & Kenyon, C. Regulation of C. elegans longevity by specific gustatory and olfactory neurons. Neuron 41, 45–55 (2004).
Bettler, B., Kaupmann, K. & Bowery, N. GABAB receptors: drugs meet clones. Curr. Opin. Neurobiol. 8, 345–350 (1998).
Bettler, B., Kaupmann, K., Mosbacher, J. & Gassmann, M. Molecular structure and physiological functions of GABA(B) receptors. Physiol. Rev. 84, 835–867 (2004).
D’Souza, M. S. & Markou, A. The "stop" and "go" of nicotine dependence: role of GABA and glutamate. Cold Spring Harb. Perspect. Med. 3, doi:10.1101/cshperspect.a012146 (2013).
Vithlani, M., Terunuma, M. & Moss, S. J. The dynamic modulation of GABA(A) receptor trafficking and its role in regulating the plasticity of inhibitory synapses. Physiol. Rev. 91, 1009–1022 (2011).
McCarthy, M. M., Auger, A. P. & Perrot-Sinal, T. S. Getting excited about GABA and sex differences in the brain. Trends Neurosci. 25, 307–312 (2002).
Koella, W. P. GABA systems and behavior. Adv. Biochem. Psychopharmacol. 29, 11–21 (1981).
Gassmann, M. & Bettler, B. Regulation of neuronal GABA(B) receptor functions by subunit composition. Nat. Rev. Neurosci. 13, 380–394 (2012).
Xu, C., Zhang, W., Rondard, P., Pin, J. P. & Liu, J. Complex GABAB receptor complexes: how to generate multiple functionally distinct units from a single receptor. Front. Pharmacol. 5, 12 (2014).
Bormann, J. The 'ABC' of GABA receptors. Trends Pharmacol. Sci. 21, 16–19 (2000).
Hosie, A. M., Aronstein, K., Sattelle, D. B. & ffrench-Constant, R. H. Molecular biology of insect neuronal GABA receptors. Trends Neurosci. 20, 578–583 (1997).
Kaupmann, K. et al. Expression cloning of GABA(B) receptors uncovers similarity to metabotropic glutamate receptors. Nature 386, 239–246 (1997).
Jones, K. A. et al. GABA(B) receptors function as a heteromeric assembly of the subunits GABA(B)R1 and GABA(B)R2. Nature 396, 674–679 (1998).
Kaupmann, K. et al. GABA(B)-receptor subtypes assemble into functional heteromeric complexes. Nature 396, 683–687 (1998).
White, J. H. et al. Heterodimerization is required for the formation of a functional GABA(B) receptor. Nature 396, 679–682 (1998).
Bargmann, C. I. Neurobiology of the Caenorhabditis elegans genome. Science 282, 2028–2033 (1998).
de Bono, M. & Maricq, A. V. Neuronal substrates of complex behaviors in C. elegans. Annu. Rev. Neurosci. 28, 451–501 (2005).
Maruyama, I. N. & Brenner, S. A phorbol ester/diacylglycerol-binding protein encoded by the unc-13 gene of Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 88, 5729–5733 (1991).
Richmond, J. E., Davis, W. S. & Jorgensen, E. M. UNC-13 is required for synaptic vesicle fusion in C. elegans. Nat. Neurosci. 2, 959–964 (1999).
Munoz, M. J. & Riddle, D. L. Positive selection of Caenorhabditis elegans mutants with increased stress resistance and longevity. Genetics 163, 171–180 (2003).
Alfonso, A., Grundahl, K., Duerr, J. S., Han, H. P. & Rand, J. B. The Caenorhabditis elegans unc-17 gene: a putative vesicular acetylcholine transporter. Science 261, 617–619 (1993).
Sze, J. Y., Victor, M., Loer, C., Shi, Y. & Ruvkun, G. Food and metabolic signalling defects in a Caenorhabditis elegans serotonin-synthesis mutant. Nature 403, 560–564 (2000).
Lints, R. & Emmons, S. W. Patterning of dopaminergic neurotransmitter identity among Caenorhabditis elegans ray sensory neurons by a TGFbeta family signaling pathway and a Hox gene. Development 126, 5819–5831 (1999).
Alkema, M. J., Hunter-Ensor, M., Ringstad, N. & Horvitz, H. R. Tyramine functions independently of octopamine in the Caenorhabditis elegans nervous system. Neuron 46, 247–260 (2005).
Lee, R. Y., Sawin, E. R., Chalfie, M., Horvitz, H. R. & Avery, L. EAT-4, a homolog of a mammalian sodium-dependent inorganic phosphate cotransporter, is necessary for glutamatergic neurotransmission in Caenorhabditis elegans. J. Neurosci. 19, 159–167 (1999).
Jin, Y., Jorgensen, E., Hartwieg, E. & Horvitz, H. R. The Caenorhabditis elegans gene unc-25 encodes glutamic acid decarboxylase and is required for synaptic transmission but not synaptic development. J. Neurosci. 19, 539–548 (1999).
Dittman, J. S. & Kaplan, J. M. Behavioral impact of neurotransmitter-activated G-protein-coupled receptors: muscarinic and GABAB receptors regulate Caenorhabditis elegans locomotion. J. Neurosci 28, 7104–7112 (2008).
Bamber, B. A., Beg, A. A., Twyman, R. E. & Jorgensen, E. M. The Caenorhabditis elegans unc-49 locus encodes multiple subunits of a heteromultimeric GABA receptor. J. Neurosci. 19, 5348–5359 (1999).
Kimura, K. D., Tissenbaum, H. A., Liu, Y. & Ruvkun, G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science 277, 942–946 (1997).
Tu, H. et al. GABAB receptor activation protects neurons from apoptosis via IGF-1 receptor transactivation. J. Neurosci. 30, 749–759 (2010).
Mendel, J. E. et al. Participation of the protein Go in multiple aspects of behavior in C. elegans. Science 267, 1652–1655 (1995).
Segalat, L., Elkes, D. A. & Kaplan, J. M. Modulation of serotonin-controlled behaviors by Go in Caenorhabditis elegans. Science 267, 1648–1651 (1995).
Roayaie, K., Crump, J. G., Sagasti, A. & Bargmann, C. I. The G alpha protein ODR-3 mediates olfactory and nociceptive function and controls cilium morphogenesis in C. elegans olfactory neurons. Neuron 20, 55–67 (1998).
Lackner, M. R., Nurrish, S. J. & Kaplan, J. M. Facilitation of synaptic transmission by EGL-30 Gqalpha and EGL-8 PLCbeta: DAG binding to UNC-13 is required to stimulate acetylcholine release. Neuron 24, 335–346 (1999).
Miller, K. G., Emerson, M. D. & Rand, J. B. Goalpha and diacylglycerol kinase negatively regulate the Gqalpha pathway in C. elegans. Neuron 24, 323–333 (1999).
Ch'ng, Q., Sieburth, D. & Kaplan, J. M. Profiling synaptic proteins identifies regulators of insulin secretion and lifespan. PLoS Genet. 4, e1000283 (2008).
McKinsey, T. A. Derepression of pathological cardiac genes by members of the CaM kinase superfamily. Cardiovasc. Res. 73, 667–677 (2007).
Feng, H., Ren, M., Chen, L. & Rubin, C. S. Properties, regulation, and in vivo functions of a novel protein kinase D: Caenorhabditis elegans DKF-2 links diacylglycerol second messenger to the regulation of stress responses and life span. J. Biol. Chem. 282, 31273–31288 (2007).
Libina, N., Berman, J. R. & Kenyon, C. Tissue-specific activities of C. elegans DAF-16 in the regulation of lifespan. Cell 115, 489–502 (2003).
Rondard, P. et al. Functioning of the dimeric GABA(B) receptor extracellular domain revealed by glycan wedge scanning. EMBO J. 27, 1321–1332 (2008).
Galvez, T. et al. Allosteric interactions between GB1 and GB2 subunits are required for optimal GABA(B) receptor function. EMBO J. 20, 2152–2159 (2001).
Pin, J. P. et al. Activation mechanism of the heterodimeric GABA(B) receptor. Biochem. Pharmacol. 68, 1565–1572 (2004).
Couve, A., Moss, S. J. & Pangalos, M. N. GABAB receptors: a new paradigm in G protein signaling. Mol. Cell Neurosci. 16, 296–312 (2000).
Comps-Agrar, L. et al. The oligomeric state sets GABA(B) receptor signalling efficacy. EMBO J. 30, 2336–2349 (2011).
Ong, J. et al. CGP 36216 is a selective antagonist at GABA(B) presynaptic receptors in rat brain. Eur. J. Pharmacol. 415, 191–195 (2001).
Froestl, W. Chemistry and pharmacology of GABAB receptor ligands. Adv. Pharmacol. 58, 19–62 (2010).
Enell, L. E., Kapan, N., Soderberg, J. A., Kahsai, L. & Nassel, D. R. Insulin signaling, lifespan and stress resistance are modulated by metabotropic GABA receptors on insulin producing cells in the brain of Drosophila. PLoS ONE 5, e15780 (2010).
Taylor, R. C. & Dillin, A. XBP-1 is a cell-nonautonomous regulator of stress resistance and longevity. Cell 153, 1435–1447 (2013).
Xiao, R. et al. A genetic program promotes C. elegans longevity at cold temperatures via a thermosensitive TRP channel. Cell 152, 806–817 (2013).
Burkewitz, K. et al. Neuronal CRTC-1 governs systemic mitochondrial metabolism and lifespan via a catecholamine signal. Cell 160, 842–855 (2015).
Yin, J. A., Liu, X. J., Yuan, J., Jiang, J. & Cai, S. Q. Longevity manipulations differentially affect serotonin/dopamine level and behavioral deterioration in aging Caenorhabditis elegans. J. Neurosci. 34, 3947–3958 (2014).
Hsu, A. L., Feng, Z., Hsieh, M. Y. & Xu, X. Z. Identification by machine vision of the rate of motor activity decline as a lifespan predictor in C. elegans. Neurobiol. Aging 30, 1498–1503 (2009).
Liu, J. et al. Functional aging in the nervous system contributes to age-dependent motor activity decline in C. elegans. Cell Metab. 18, 392–402 (2013).
Aamodt, E. J., Chung, M. A. & McGhee, J. D. Spatial control of gut-specific gene expression during Caenorhabditis elegans development. Science 252, 579–582 (1991).
Altun-Gultekin, Z. et al. A regulatory cascade of three homeobox genes, ceh-10, ttx-3 and ceh-23, controls cell fate specification of a defined interneuron class in C. elegans. Development 128, 1951–1969 (2001).
Fire, A. & Waterston, R. H. Proper expression of myosin genes in transgenic nematodes. EMBO J. 8, 3419–3428 (1989).
Cao, P. et al. Light-sensitive coupling of rhodopsin and melanopsin to G(i/o) and G(q) signal transduction in Caenorhabditis elegans. FASEB J. 26, 480–491 (2012).
Acknowledgements
We thank Charles Rubin and Ao-Lin Hsu for providing strains, Zhaoyang Feng for the PTX plasmid and Lingxiu Xu for technical assistance. Some strains were obtained from the CGC and Knockout Consortiums in the USA and Japan. This work was supported by the NSFC (31130028, 31225011 and 31420103909 to J.L.), the Program of Introducing Talents of Discipline to the Universities from the Ministry of Education (B08029 to J.L.), the Ministry of Science and Technology of China (2012CB51800 to J.L.), Natural Science Foundation of HuBei Province (2014CFA010 to J.L.) and the NIA (X.Z.S.X.).
Author information
Authors and Affiliations
Contributions
L.C. performed the experiments and analysed the data. J.G. and B.Z. initiated the project. J.G., F.Y., H.L., T.Z. and T.Y. performed some experiments. X.Z.S.X. and J.L. supervised the project. C.L., X.Z.S.X. and J.L. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-3 and Supplementary Table 1. (PDF 799 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chun, L., Gong, J., Yuan, F. et al. Metabotropic GABA signalling modulates longevity in C. elegans. Nat Commun 6, 8828 (2015). https://doi.org/10.1038/ncomms9828
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms9828
- Springer Nature Limited
This article is cited by
-
Peripheral modulation of antidepressant targets MAO-B and GABAAR by harmol induces mitohormesis and delays aging in preclinical models
Nature Communications (2023)
-
Dopamine D1- and D2-like receptors oppositely regulate lifespan via a dietary restriction mechanism in Caenorhabditis elegans
BMC Biology (2022)
-
The flight response impairs cytoprotective mechanisms by activating the insulin pathway
Nature (2019)
-
Aminergic Signaling Controls Ovarian Dormancy in Drosophila
Scientific Reports (2018)