Abstract
In a prospective study (NCT02866149), we assessed the efficacy of fulvestrant and everolimus in CDK4/6i pre-treated mBC patients and circulating tumor DNA (ctDNA) changes throughout therapy. Patients treated with fulvestrant and everolimus had their ctDNA assessed at baseline, after 3–5 weeks and at disease progression. Somatic mutations were identified in archived tumor tissues by targeted NGS and tracked in cell-free DNA by droplet digital PCR. ctDNA detection was then associated with clinicopathological characteristics and patients’ progression-free survival (PFS), overall survival (OS) and best overall response (BOR). In the 57 included patients, median PFS and OS were 6.8 (95%CI [5.03–11.5]) and 38.2 (95%CI [30.0-not reached]) months, respectively. In 47 response-evaluable patients, BOR was a partial response or stable disease in 15 (31.9%) and 11 (23.4%) patients, respectively. Among patients with trackable somatic mutation and available plasma sample, N = 33/47 (70.2%) and N = 19/36 (52.8%) had ctDNA detected at baseline and at 3 weeks, respectively. ctDNA detection at baseline and PIK3CA mutation had an adverse prognostic impact on PFS and OS in multivariate analysis. This prospective cohort study documents the efficacy of fulvestrant and everolimus in CDK4/6i-pretreated ER + /HER2- mBC and highlights the clinical validity of early ctDNA changes as pharmacodynamic biomarker.
Similar content being viewed by others
Introduction
Breast cancer is the most common cancer among women globally, with an estimated 2.3 million new cases and 685 000 deaths in 2020 in the world [1]. Recently, the prognosis of estrogen receptor-positive/HER2-negative (ER + /HER2-) metastatic breast cancer (mBC) has been improved following the development of cyclin-dependent kinase 4/6 inhibitors (CDK 4/6i). Three CDK4/6i (palbociclib, ribociclib and abemaciclib) [2,3,4] in combination with aromatase inhibitors [5] or fulvestrant (a selective estrogen receptor degrader) [6] have extended progression-free survival (PFS) and overall survival (OS) [7]. These drugs became standard of care as first line treatment for ER + /HER2- mBC patients, in the absence of visceral crisis.
Upon progression on first line CDK4/6i and endocrine therapy (ET), the combination of fulvestrant with everolimus (targeting the PI3K/AKT/mTOR pathway) is recognized as a valid second line treatment option by National Comprehensive Cancer Network (NCCN) and European Society for Medical Oncology (ESMO) [8] guidelines for ER + /HER2- mBC. The underlying evidence mostly consists in two randomized phase 2 trials, PrE0102 [9] and MANTA [10], both conducted in ER + /HER2- mBC patients whose mBC has become resistant to aromatase inhibitors. In these studies, patients treated with fulvestrant and everolimus showed a median PFS of 10.3 and 12.3 months, compared to 5.1 and 5.4 months with fulvestrant alone, respectively. However, these efficacy data were obtained without prior CDK4/6i therapy and no biomarker associated with fulvestrant and everolimus efficacy has been validated so far.
Circulating tumor DNA (ctDNA), a fraction of total cell-free circulating DNA (cfcDNA), has emerged in the last decade as a promising biomarker for diagnosis, prognosis, and monitoring treatment efficacy in different cancer types [11,12,13]. In ER + /HER2- mBC treated by ET and CDK4/6i, our group and others have previously shown that early ctDNA decrease was a prognostic factor for PFS [14,15,16] and the efficiency of palbociclib and fulvestrant can be monitored by serial analyses of ctDNA [16].
This study was set up to prospectively document the efficacy of the everolimus plus fulvestrant combination after progression on CDK4/6i and, with the use of personalized droplet digital PCR (ddPCR) assays, to investigate the clinical validity of ctDNA early changes as pharmacodynamic marker in ER + /HER2- mBC patients.
Results
Patient characteristics
From February 2018 to October 2021, 57 patients with ER + /HER2- mBC after progression on a CDK4/6i were enrolled in this study. The main characteristics of the 57 patients are as follows: median age was 57 years (range [36–79]), 30 (52.6%) patients had ≥3 metastatic sites and 33 (57.9%) had visceral metastases. Most patients (N = 48, 84.2%) had received only one prior line of systemic therapy, ET (aromatase inhibitor) and CDK4/6i. Only one patient has already received fulvestrant in the previous lines of metastatic disease. Patient characteristics are shown in Table 1.
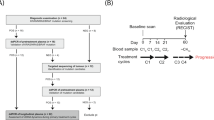
Of these 57 patients, 31 patients had archived tumor tissue available; 29 of them displayed at least one somatic mutation in tumor DNA by targeted NGS. Further targeted NGS, performed on baseline cfcDNA in those without available tissue DNA, identified another 19 patients with detectable mutations (Fig. 1). Finally, 47 patients with available ddPCR assays were tested for ctDNA levels before and after treatment targeting the identified mutation (PIK3CAmut: N = 25; ESR1mut: N = 7; TP53mut: N = 4; AKTmut: N = 2; GATA3mut: N = 2; CUX1mut: N = 1; PTENmut: N = 1; APCmut: N = 1; ATMmut: N = 1; RETmut: N = 1; TRAPPmut: N = 1; KRASmut: N = 1; Supplementary Table 1). Of the 25 patients identified with a PIK3CA mutation (including the one with a PIK3CA variant of unknown significance), 20 patients had tumor tissue DNA available, and 5 patients were tested with baseline cfcDNA.
Patients’ outcome
With a median follow-up of 29.7 months, median PFS and OS with everolimus plus fulvestrant post CDK4/6i (palbociclib) were 6.8 (95%CI [5.03–11.5]) months and 38.2 (95%CI [30.0-not reached (NR)]) months, respectively (Fig. 2A, B). Among the 47 patients with measurable disease at baseline, 15 (31.9%) experienced a partial response (PR). Eleven (23.4%) patients had SD and 21 (44.7%) had PD as BOR (Supplementary Table 2).
We assessed whether the duration of treatment with palbociclib (using median PFS as threshold, 13.6 months) affected survival in patients treated with everolimus and fulvestrant. Interestingly, there was no effect on survival with everolimus plus fulvestrant (for PFS: median PFS of 6.8 (95%CI [3.75–15.3]) months for patients with longer exposure versus 5.3 (95%CI [3.82–11.5]) months, p = 0.49 and for OS: median OS of 30.0 (95%CI [24.0-NR]) months for patients with longer exposure versus 38.2 (95%CI [14.1-NR]) months, p = 0.68 (Fig. 2C, D)). Patients with a long duration of response to ET + palbociclib treatment in the previous line (PFS > 24 months) had a numerically longer PFS than those with a PFS ≤ 24 months (median PFS: 13.0 months versus 5.3 months, p = 0.16). Patients with a rapid progression to ET + palbociclib treatment (PFS < 6 months) had a shorter PFS, but it was not significantly associated (4.5 months versus 6.8 months for patients with a PFS ≥ 6 months, p = 0.41) (Supplementary Figure 1). We also evaluated survival in the 25 patients with PIK3CA mutation. A shorter median PFS and OS was observed (for PFS: 3.4 (95%CI [2.83-8.91]) months versus 9.9 (95%CI [5.30-15.33]) months, p = 0.008 and for OS 22.9 (95%CI [10.8-NR]) months versus 40.0 (95%CI [30.0-NR]) months, p = 0.06, respectively) (Supplementary Figure 2A, B). The prognostic impact of other (i.e. non-PIK3CA) mutations was also investigated. No significant difference was observed on PFS with ESR1 mutation (N = 7/47, p = 0.15) (Supplementary Fig. 3). For TP53 (N = 4) or AKT1 (N = 2) mutation, due to the limited number of patients included, the analysis was lack of statistical power and data was not shown.
Safety
The most common adverse events of any grade occurring in at least 25% of patients were mucositis (N = 39/57, 68.4%), asthenia (N = 27/57, 47.4%), decreased weight (N = 18/57, 31.6%), rash (N = 18/57, 31.6%), hypercholesterolemia (N = 16/57, 28.1%), hypertriglyceridemia (N = 15/57, 26.3%), alanine aminotransferase increased (N = 15/57, 26.3%) and dyspnea (N = 15/57, 26.3%). Grade 3 or 4 adverse events were mucositis (N = 6/57, 10.5%), hypertriglyceridemia (N = 2/57, 3.5%), neutropenia (N = 1/57, 1.8%), decreased body weight (N = 1/57, 1.8%) and pneumonitis (N = 1/57, 1.8%) (Table 2). Ten patients (17.5%) discontinued everolimus due to toxicity and 17 (29.8%) patients required a dose reduction.
ctDNA detection and prognostic value
Among the 47 patients with a trackable mutation, N = 33/47 patients (70.2%) showed detectable ctDNA at baseline with a median allelic frequency of 1.62% (range (0.0-52.6)) (Fig. 3A and Supplementary Table 3). Three weeks after treatment, matched plasma samples were available for 36 patients, of which N = 19/36 patients (52.8%) were ctDNA-positive with a median allelic frequency of 0.2% (range [0.0-55.1]). Nine patients had no ctDNA detected at both baseline and 3–5 W. Seventeen patients (47.2%) showed a ctDNA decrease at 3–5 W, including 8 patients with ctDNA clearance (22.2%), while 10 patients showed an increase of ctDNA (50.0%). At the time of tumor progression, N = 12/18 (66.7%) patients had ctDNA detected with a median allelic frequency of 1.9% (range [0.0-42.5]) (Fig. 3A, B and Supplementary Table 3). No significant association was found for ctDNA levels between the different time points (Fig. 3A). The timing of clinical relapse of the different patients is shown in Fig. 3B.
A ctDNA levels at different time points quantified by ddPCR. Each dot represents the level of ctDNA of a patient measured. Mann–Whitney test was used to compare ctDNA levels at different time points. B Swimmer plot showing the dynamic changes of ctDNA levels, best overall response and timing of clinical relapse. Each bar represents one patient. Red circles indicate ctDNA detection and blue circles indicate no ctDNA detection. The black triangle indicates disease progression (PD). *SD ≥ 6 months. Bsl: baseline; 3–5 W: 3–5 weeks, PD progressive disease, PR partial response, SD stable disease, NA not available.
The prognostic value of ctDNA detection at baseline and 3–5 W was then investigated. Both ctDNA detection at baseline and 3–5 W were associated with shorter median PFS (5.1 months versus 12.4 months, HR = 2.04, 95%CI [1.20–3.45], p = 0.005 and 4.3 months versus 12.7 months, HR = 2.56, 95%CI [1.41–4.76], p = 0.002, respectively). OS was also shorter in patients with both ctDNA detection at baseline and 3–5 W (24 months versus NR months, HR = 3.85, 95%CI [1.39–10.99], p = 0.002 and 13.2 months versus 40.0 months, HR = 3.85, 95%CI [1.56–10.00], p = 0.001, respectively) (Fig. 4A–D). Among patients with ctDNA detected at baseline, ctDNA levels was not significantly associated with PFS (ctDNA > vs < median copy number, HR = 1.40, 95%CI [0.69-3.00], p = 0.33) (Supplementary Table 4). Among the patients with PIK3CA mutation, ctDNA detection at baseline showed no prognostic value (Supplementary Figure 4).
Early ctDNA dynamics as pharmacodynamic marker
Among 27 patients with positive ctDNA detection at baseline and/or 3–5 W, 25 patients were available for analysis of correlation with the first tumor assessment (one patient was not followed up and the other patient had no measurable disease according to RECIST criteria). The majority of patients with an increase in ctDNA between baseline and 3–5 W had disease progression (N = 11/13, 84.6%) at the first tumor assessment (2 patients had a PR). Among patients with a decrease in ctDNA, 3 patients (N = 3/12, 25%) had PD at the first tumor assessment (6 patients had a PR and 3 patients had SD) (Fig. 5A). Overall, using a ‘ctDNA decreased at 3–5 W’ vs ‘ctDNA increased at 3–5 W’ threshold, ctDNA changes accuracy to predict disease progression at first tumor assessment was 80.0%, with N = 20 correct predictions out of 25 assessable patients.
Tumor response according to ctDNA change from baseline (A). *SD ≥ 6 months. Progression-free survival and overall survival according to early ctDNA variation (B, C) and ctDNA clearance (D, E). PD progressive disease, PR partial response, SD stable disease, PFS progression-free survival; OS: overall survival.
In terms of survival, patients with a ctDNA decrease at 3–5 W (regardless of threshold) had a significantly longer median PFS (7.5 months vs 2.2 months; HR = 0.15, 95%CI [0.06-0.41], p < 0.0001) and median OS (40.0 months vs 12.2 months; HR = 0.27, 95%CI [0.09-0.79], p = 0.011) (Fig. 5B, C). The same trend was observed for ctDNA clearance at 3–5 W: longer median PFS (7.5 months vs 4.2 months; HR = 0.36, 95%CI [0.136-0.99], p = 0.04) and median OS (40.0 months vs 13.2 months; HR = 0.31, 95%CI [0.09-1.10], p = 0.065) were found in patients with ctDNA clearance (Fig. 5D, E). We explored two other thresholds of ctDNA changes, -30% and -50% in copy number at 3–5 W: both were associated with PFS (HR = 0.15, 95%CI [0.06-0.41], p < 0.0001 and HR = 0.34, 95%CI [0.15-0.81], p = 0.011, respectively) and OS (HR = 0.27, 95%CI [0.09-0.79], p = 0.011 and HR = 0.27, 95%CI [0.07-0.84], p = 0.017, respectively) (data not shown).
Univariate and multivariate analysis
Finally, univariate and multivariate proportional hazards models were performed to evaluate the prognostic value of prospectively collected clinicopathological factors combined with ctDNA detection at different time points. As shown in Supplementary Table 4, PIK3CA mutation detection (in a subgroup of N = 25 (43.8%) patients with PIK3CA mutations), ctDNA detection at baseline and 3–5 W, ctDNA decrease between baseline and 3–5 W, and ctDNA clearance at 3–5 W were significantly associated with PFS and OS in univariate analysis (except for the presence of PIK3CA mutation and ctDNA clearance at 3–5 W, where there was no significant difference in OS), while age and performance status were only associated with OS (HR = 1.00, 95%CI [1.00–1.10], p = 0.03 and HR = 2.90, 95%CI [1.20-6.90], p = 0.02).
In multivariate analysis, baseline ctDNA positivity and PIK3CA mutation were found as independent prognostic factors for PFS and OS (for PFS, HR = 2.36, 95%CI [1.38-4.04], p = 0.002; HR = 2.67, 95%CI [1.39-5.10], p = 0.003; for OS, HR = 6.33, 95%CI [2.07-19.36], p = 0.001; HR = 4.58, 95%CI [1.57-13.35], p = 0.005, respectively) (Table 3).
Discussion
To our knowledge, this is the first prospective study to evaluate the efficacy of everolimus in combination with fulvestrant after progression on CDK4/6i in ER + /HER2- mBC. We document a median PFS of 6.8 months with everolimus and fulvestrant. This median PFS is apparently shorter than that reported in the PrE0102 and MANTA trials, which included patients not pre-treated with CDK4/6i [9, 10]. A similar shortening of second line PFS in CDK4/6i pretreated patients was previously observed with the fulvestrant-alpelisib combination, with median PFS of 11.0 months when given to CDK4/6i-naïve (SOLAR-1 trial [17]) versus 7.3 (BYLieve trial [18]) and 5.3 (French early access program [19]) months in CDK4/6i-pretreated mBC patients. In the post CDK4/6i setting, few retrospective studies have reported short-lived efficacy of everolimus-based combination (with exemestane or fulvestrant) : median PFS was 4.9 months in N = 12 patients [20], 4.2 months in N = 41 patients [21] and 3.8 months in N = 79 patients [22]. These PFS were obtained in patients treated in 2nd to 5th lines and whose prior exposure to CDK4/6 inhibitors ranged from 4 to 21 months. Measurable disease according to RECIST was not a prerequisite for inclusion in these trials. Of note, in these retrospective reports, patients received everolimus as second or later line of therapy, which may explain the apparently shorter PFS compared to our prospective study. Finally, the fulvestrant and everolimus combination observed in our study appears somehow similar to the efficacy reported with fulvestrant and capivasertib combination: in the phase III CAPItello-291 trial (Patients were required to have RECIST measurable disease (version 1.1) or at least one lytic or mixed lytic–blastic bone lesion with identifiable soft-tissue components), the AKT inhibitor given in combination with fulvestrant led to a median PFS of 7.2 months and 5.5 months in patients with prior CDK4/6i exposure [23]. An appealing characteristic of everolimus is that its label allows using it in all patients, independently of their tumor mutational landscape. PIK3CA mutations have previously been reported to be associated with worse outcome in patients with ER + /HER2- mBC [24], while the efficacy of everolimus in clinical trial data is uncertain. In the neoadjuvant setting, Baselga et al. [25] found that the presence of PIK3CA exon 9 mutations conferred an improved response to the combination of everolimus with letrozole [25]. In various metastatic cancers, patients treated with PI3K/AKT/mTOR pathway inhibitors in combination with other treatments (ET, anti-HER2 therapy, or chemotherapy) had a longer time to progression compared to patients without PIK3CA mutations [26]. However, in the BOLERO-2 trial, the authors found no association between the presence of a PIK3CA mutation (either by next generation sequencing on tumor tissue or in cell-free DNA by ddPCR) and response to everolimus [27, 28]. In our study, we found that patients with PIK3CA mutation had a limited benefit, with a median PFS of 3.4 months and a median OS of 22.9 months. This median PFS appears to be shorter than reported with alpelisib in the BYLieve study [18] (7.3 months). The small sample size of this subpopulation in our study makes it difficult to draw definitive conclusions, but the impact of PIK3CA mutation on everolimus and fulvestrant efficacy after CDK4/6i needs to be further explored.
Finally, we investigated the impact of ctDNA dynamics as a predictive biomarker for the efficacy of everolimus plus fulvestrant. We found a prognostic impact of the baseline and 3–5 W ctDNA positivity on PFS and OS, and also a prognostic impact of the variation of ctDNA level between 3–5 W and baseline. However, among the patients with decreased ctDNA, three patients had a PD at the first radiological evaluation and two patients had a PR in spite of an increased ctDNA load. One hypothesis is that the mutations used for ctDNA monitoring in these patients were from a tumor subclone, which eventually couldn’t predict and represent the whole tumor evolution under treatment. Our results are in line with a recent study [29] monitoring ctDNA in fulvestrant-exemestane combination, where PFS was particularly short in patients with detectable ctDNA after 14 days of treatment: 2.1 months versus 5.0 months in those without detectable ctDNA (p = 0.012). A statistically significant longer PFS was observed for patients with ctDNA ratio (D14/baseline) below or equal to the median value (p ≤ 0.061): 4.4 months versus 1.9 months (HR = 2.5, 95%CI [1.0–6.2], p = 0.043). In the second line setting where multiple treatments are available and endocrine-resistance therapies are frequent, assessment of ctDNA dynamics during treatment could rapidly predict patients that will experience a tumor progression or response, but further studies are needed to assess its clinical utility. A phase 2 trial (“MONDRIAN”, NCT04720729) is underway to evaluate the use of early ctDNA level variation to monitor response to chemotherapy in HER2- mBC.
In conclusion, we report here the first prospective assessment of fulvestrant-everolimus in ER + /HER2- mBC patients who progressed on prior CDK4/6 inhibitor. We document that fulvestrant-everolimus is an active regimen in this population, with limited toxicity, whereas ctDNA detection demonstrated a satisfactory clinical validity for monitoring response to therapy.
Methods
Patients and blood sampling
All patients signed a written informed consent to this ethically approved study (Comité de Protection des Personnes “Ile de France VI”), registered with clinicaltrials.gov (NCT02866149, cohort #12). Eligibility criteria were: patients aged ≥18 years with ER + /HER2- mBC (immunoreactive for ER in ≥10% of tumor-cell nuclei by immunohistochemistry) treated at Institut Curie Hospitals (Paris and Saint Cloud), who progressed under prior CDK4/6i therapy (with no requirement for prior CDK4/6i duration). The presence of measurable disease (per RECIST criteria) was not a prerequisite for patient enrollment. All included patients were then treated with everolimus (10 mg/day, unless decided otherwise by physicians) and fulvestrant, per standard of care. Mucositis prophylaxis was left at the discretion of the physician. Tumor response to therapy was assessed at least every 3 months by CT-scan. The objective response rate (ORR) was calculated only with the patients with RECIST measurable disease, including those with unconfirmed responses. Any patient who did not achieve a complete/partial response or progressive disease (PD) within 6 months was considered as having a stable disease (SD). Adverse events were coded and graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (version 5.0). Patient characteristics and survival data were prospectively registered. Patients who discontinued everolimus treatment for any reason other than progression were required to follow the same schedule of assessments until progression. For each patient, longitudinal blood samples were collected: at baseline, after 3–5 weeks (3–5 W) of therapy and at time of disease progression. Variation in ctDNA was calculated using copies/ml.
Plasma sample preparation, storage, DNA extraction and quantification
Blood samples were collected in EDTA tubes and plasma was isolated within 2 h by a two-step centrifugation: 820 × g for 10 min, then 16000 × g for 10 min at 4 °C. DNA was extracted using the QIAmp Circulating Nucleic Acid Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions and quantified by Qubit 2.0 fluorometer (Invitrogen, Carlsbad, CA, USA) using the dsDNA HS Assay (Invitrogen, Carlsbad, CA, USA). Extracted DNA was stored at –20 °C before use.
Identification of trackable somatic mutations
Archived tumor samples, either from the primary tumor or a metastatic deposit, were retrieved from the Pathology department. Tumor DNA was extracted from macro-dissected tumor tissue (Macherey-Nagel nucleospin tissue kit) and subjected to a large amplicon-based targeted NGS panel developed at Institut Curie, covering 483 genes (Qiagen Qiaseq). For patients with no tumor tissue available or failure of tissue DNA sequencing, targeted-NGS was performed on cfcDNA collected at baseline.
Analysis of ctDNA by ddPCR
The identified somatic mutations were used for ctDNA monitoring in plasma by ddPCR. Tumor or synthetic DNA carrying the target mutation was used as positive control. Genomic DNA from healthy donors purchased from Promega (Madison, WI, USA) was used as negative control. Primers and probes for detecting PIK3CA: c.3140 A > G mutation are described in Supplementary Table 1. Assays for detecting TP53: c.637 C > T mutation was provided by SAGA Diagnostics (Lund, Sweden). ddPCR assays for detecting other mutations were purchased from Biorad laboratories (Hercules, CA, USA) (Supplementary Table 1). ddPCR assays were performed using the BioRad QX100 system (Biorad laboratories, Hercules, CA, USA) with 900 nM of each primer, 250 nM of each probe with one probe targeting the wide-type sequence and the other one targeting the mutant sequence being labeled with different fluorophores. The amplification was under the following conditions: 95 °C for 10 min (1 cycle); 40 cycles of 94 °C for 30 sec and hybridization at a specific temperature for each mutation for 60 s, and 98 °C for 1 min (1 cycle). The hybridization temperatures used for each assay were in Supplementary Table 1. Cluster thresholding and quantification were performed with QuantaSoft v.1.7.4 software. Droplets were manually assigned as wild-type (WT) or mutant (MUT) based on their fluorescence amplitude: WT: VIC+ or HEX + ; MUT: FAM + . The mutant allelic frequencies (MAFs) were calculated as follows: copy numbers of MUT / (copy numbers of WT + copy numbers of MUT). The total copy number of cfcDNA in each patient was calculated as the sum of the copy numbers of WT and MUT sequences. All experiments met the minimum requirements for digital PCR data [30, 31].
ddPCR data analysis
The false-positive rate of each assay was estimated as previously reported [32, 33] using ≥ 15 replicates of WT DNA. The limit of blank (LOB) of different assays was defined as the upper 95% confidence limit of the mean false-positive measurements. Samples were considered as positive when MAF was higher than LOB with more than 1-3 positive droplets detected per analysis (Supplementary Table 1). When less than 300 amplifiable genomes were detected by ddPCR, the sample was not considered for further ctDNA analysis. The laboratory performing ddPCR experiments was blinded to patient outcomes.
Statistical analysis
Categorical variables were compared using Fisher’s exact test. Survival analysis was performed using Kaplan–Meier plots with significance tested using the log-rank test. Progression-free survival (PFS), defined as the time from initiation of treatment to disease progression or death from any cause, was collected prospectively. Patients with no disease progression were censored at the last follow-up visit. OS was defined as the time from initiation of treatment to the date of death from any cause. Best overall response was defined as the best response recorded from the start of therapy until its discontinuation.
Cox proportional hazards modeling was used for univariate and multivariate analysis to evaluate the prognostic significance of different predictors. Baseline clinical and pathological characteristics with a p value > 0.1 in univariate analysis were excluded from multivariate analysis. Statistical analyses were performed with GraphPad Prism (version 8.0) or R software (version 4.1.1). P-values ≤ 0.05 were considered statistically significant.
Data availability
The human sequence data underlying this article cannot be shared due to patient privacy. Other data generated in this study are available within the article and its supplementary data files.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Finn RS, Martin M, Rugo HS, Jones S, Im S-A, Gelmon K, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375:1925–36.
Hortobagyi GN, Stemmer SM, Burris HA, Yap Y-S, Sonke GS, Paluch-Shimon S, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375:1738–48.
Johnston S, Martin M, Di Leo A, Im S-A, Awada A, Forrester T, et al. MONARCH 3 final PFS: a randomized study of abemaciclib as initial therapy for advanced breast cancer. NPJ Breast Cancer. 2019;5:5.
Mouridsen H, Gershanovich M, Sun Y, Perez-Carrion R, Boni C, Monnier A, et al. Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the International Letrozole Breast Cancer Group. J Clin Oncol Off J Am Soc Clin Oncol. 2003;21:2101–9.
Di Leo A, Jerusalem G, Petruzelka L, Torres R, Bondarenko IN, Khasanov R, et al. Final overall survival: fulvestrant 500 mg vs 250 mg in the randomized CONFIRM trial. J Natl Cancer Inst. 2014;106:djt337.
Hortobagyi GN, Stemmer SM, Burris HA, Yap Y-S, Sonke GS, Hart L, et al. Overall survival with ribociclib plus letrozole in advanced breast cancer. N Engl J Med. 2022;386:942–50.
Gennari A, André F, Barrios CH, Cortés J, de Azambuja E, DeMichele A, et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann Oncol Off J Eur Soc Med Oncol. 2021;32:1475–95.
Kornblum N, Zhao F, Manola J, Klein P, Ramaswamy B, Brufsky A, et al. Randomized phase II trial of fulvestrant plus everolimus or placebo in postmenopausal women with hormone receptor-positive, human epidermal growth factor receptor 2-negative metastatic breast cancer resistant to aromatase inhibitor therapy: results of PrE0102. J Clin Oncol Off J Am Soc Clin Oncol. 2018;36:1556–63.
Schmid P, Zaiss M, Harper-Wynne C, Ferreira M, Dubey S, Chan S, et al. Fulvestrant plus vistusertib vs fulvestrant plus everolimus vs fulvestrant alone for women with hormone receptor-positive metastatic breast cancer: the MANTA phase 2 randomized clinical trial. JAMA Oncol. 2019;5:1556–64.
Gerratana L, Davis AA, Shah AN, Lin C, Corvaja C, Cristofanilli M. Emerging role of genomics and cell-free DNA in breast cancer. Curr Treat Options Oncol. 2019;20:68.
Cabel L, Proudhon C, Mariani P, Tzanis D, Beinse G, Bieche I, et al. Circulating tumor cells and circulating tumor DNA: what surgical oncologists need to know? Eur J Surg Oncol J Eur Soc Surg Oncol Br Assoc Surg Oncol. 2017;43:949–62.
Cabel L, Proudhon C, Romano E, Girard N, Lantz O, Stern M-H, et al. Clinical potential of circulating tumour DNA in patients receiving anticancer immunotherapy. Nat Rev Clin Oncol. 2018;15:639–50.
O’Leary B, Hrebien S, Morden JP, Beaney M, Fribbens C, Huang X, et al. Early circulating tumor DNA dynamics and clonal selection with palbociclib and fulvestrant for breast cancer. Nat Commun. 2018;9:896.
Jacot W, Dalenc F, Lopez-Crapez E, Chaltiel L, Durigova A, Gros N, et al. PIK3CA mutations early persistence in cell-free tumor DNA as a negative prognostic factor in metastatic breast cancer patients treated with hormonal therapy. Breast Cancer Res Treat. 2019;177:659–67.
Darrigues L, Pierga J-Y, Bernard-Tessier A, Bièche I, Silveira AB, Michel M, et al. Circulating tumor DNA as a dynamic biomarker of response to palbociclib and fulvestrant in metastatic breast cancer patients. Breast Cancer Res. 2021;23:31.
André F, Ciruelos E, Rubovszky G, Campone M, Loibl S, Rugo HS, et al. Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. N Engl J Med. 2019;380:1929–40.
Rugo HS, Lerebours F, Ciruelos E, Drullinsky P, Ruiz-Borrego M, Neven P, et al. Alpelisib plus fulvestrant in PIK3CA-mutated, hormone receptor-positive advanced breast cancer after a CDK4/6 inhibitor (BYLieve): one cohort of a phase 2, multicentre, open-label, non-comparative study. Lancet Oncol. 2021;22:489–98.
Bello Roufai D, Gonçalves A, De La Motte Rouge T, Akla S, Blonz C, Grenier J, et al. Alpelisib and fulvestrant in PIK3CA-mutated hormone receptor-positive HER2-negative advanced breast cancer included in the French early access program. Oncogene. 2023;42:1951–6.
Xi J, Oza A, Thomas S, Ademuyiwa F, Weilbaecher K, Suresh R, et al. Retrospective analysis of treatment patterns and effectiveness of palbociclib and subsequent regimens in metastatic breast cancer. J Natl Compr Cancer Netw. 2019;17:141–7.
Dhakal A, Antony Thomas R, Levine EG, Brufsky A, Takabe K, Hanna MG, et al. Outcome of everolimus-based therapy in hormone-receptor-positive metastatic breast cancer patients after progression on palbociclib. Breast Cancer Basic Clin Res. 2020;14:1178223420944864.
Mo H, Renna CE, Moore HCF, Abraham J, Kruse ML, Montero AJ, et al. Real-world outcomes of everolimus and exemestane for the treatment of metastatic hormone receptor-positive breast cancer in patients previously treated with CDK4/6 inhibitors. Clin Breast Cancer. 2022;22:143–8.
Turner NC, Oliveira M, Howell SJ, Dalenc F, Cortes J, Gomez Moreno HL, et al. Capivasertib in hormone receptor-positive advanced breast cancer. N Engl J Med. 2023;388:2058–70.
Mosele F, Stefanovska B, Lusque A, Tran Dien A, Garberis I, Droin N, et al. Outcome and molecular landscape of patients with PIK3CA-mutated metastatic breast cancer. Ann Oncol Off J Eur Soc Med Oncol. 2020;31:377–86.
Baselga J, Semiglazov V, van Dam P, Manikhas A, Bellet M, Mayordomo J, et al. Phase II randomized study of neoadjuvant everolimus plus letrozole compared with placebo plus letrozole in patients with estrogen receptor-positive breast cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2009;27:2630–7.
Janku F, Wheler JJ, Westin SN, Moulder SL, Naing A, Tsimberidou AM, et al. PI3K/AKT/mTOR inhibitors in patients with breast and gynecologic malignancies harboring PIK3CA mutations. J Clin Oncol Off J Am Soc Clin Oncol. 2012;30:777–82.
Hortobagyi GN, Chen D, Piccart M, Rugo HS, Burris HA, Pritchard KI, et al. Correlative analysis of genetic alterations and everolimus benefit in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: results from BOLERO-2. J Clin Oncol Off J Am Soc Clin Oncol. 2016;34:419–26.
Moynahan ME, Chen D, He W, Sung P, Samoila A, You D, et al. Correlation between PIK3CA mutations in cell-free DNA and everolimus efficacy in HR+, HER2- advanced breast cancer: results from BOLERO-2. Br J Cancer. 2017;116:726–30.
Gombos A, Venet D, Ameye L, Vuylsteke P, Neven P, Richard V, et al. FDG positron emission tomography imaging and ctDNA detection as an early dynamic biomarker of everolimus efficacy in advanced luminal breast cancer. NPJ Breast Cancer. 2021;7:125.
Huggett JF, Foy CA, Benes V, Emslie K, Garson JA, Haynes R, et al. The digital MIQE guidelines: minimum Information for Publication of Quantitative Digital PCR Experiments. Clin Chem. 2013;59:892–902.
dMIQE Group, Huggett JF. The digital MIQE guidelines update: minimum information for publication of quantitative digital PCR experiments for 2020. Clin Chem. 2020;66:1012–29.
Jeannot E, Darrigues L, Michel M, Stern M-H, Pierga J-Y, Rampanou A, et al. A single droplet digital PCR for ESR1 activating mutations detection in plasma. Oncogene. 2020;39:2987–95.
Zonta E, Garlan F, Pécuchet N, Perez-Toralla K, Caen O, Milbury C, et al. Multiplex detection of rare mutations by picoliter droplet based digital PCR: sensitivity and specificity considerations. PloS One. 2016;11:e0159094.
Acknowledgements
High-throughput sequencing was performed by the ICGex NGS platform of the Institut Curie supported by the grants ANR-10-EQPX-03 (Equipex) and ANR-10-INBS-09-08 (France Génomique Consortium) from the Agence Nationale de la Recherche (“Investissements d’Avenir” program), by the ITMO-Cancer Aviesan (Plan Cancer III) and by the SiRIC-Curie program (SiRIC Grant INCa-DGOS-465 and INCa-DGOSInserm_12554). Data management, quality control and primary analysis were performed by the Bioinformatics platform of the Institut Curie. Further NGS data analysis was performed by the Clinical Bioinformatics team of the Institut Curie.
Author information
Authors and Affiliations
Contributions
AV, SF wrote the manuscript; FCB developed the design of this study; SR, FCB, LC, FL, DL, EB, PC, MPS, JYP reviewed the manuscript; Data was collected by OTG, CC, CH, WT, BR, AV; Data analysis was carried out by LC and AV.
Corresponding authors
Ethics declarations
Competing interests
This work was supported by SIRIC 3 Curie (grant INCa-DGOS-INSERM-ITMO Cancer_18000). FC Bidard received research support and honoraria for advisory board from AstraZeneca and Novartis. F Lerebours reports consulting for AstraZeneca, Eisai, Genomic Health, Eli Lilly, Laboratories Pierre Fabre, and Roche; and travel support from AstraZeneca, Eisai, Novartis, Pfizer, Roche, and Laboratories Pierre Fabre; and lecture fees from AstraZeneca, Eisai, Eli Lilly, Novartis, and Laboratories Pierre Fabre. MP Sablin reports lecture fees from Servier. JY Pierga reports consulting for AstraZeneca, Eisai, Eli Lilly, MSD, Gilead, Daiichy Sankyo, Novartis, Pfizer and travel support from AstraZeneca, MSD, Novartis, Pfizer, Gilead, Seagen, and lecture fees from AstraZeneca, MSD, Eli Lilly, Novartis, Gilead, Veracyte. No other potential conflicts of interest were reported.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vasseur, A., Cabel, L., Hego, C. et al. Fulvestrant and everolimus efficacy after CDK4/6 inhibitor: a prospective study with circulating tumor DNA analysis. Oncogene 43, 1214–1222 (2024). https://doi.org/10.1038/s41388-024-02986-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41388-024-02986-6
- Springer Nature Limited
This article is cited by
-
Precision therapeutics and emerging strategies for HR-positive metastatic breast cancer
Nature Reviews Clinical Oncology (2024)