Abstract
Many circRNAs are involved in the carcinogenesis of breast cancer (BCa) through the transcription of microRNAs (miRNAs) and mRNAs. This study investigated circRBM33 regulation of the miR-542-3p/hypoxia-inducible factor-1α (HIF-1α) axis in BCa. BCa clinical tissue samples were collected to test differential expressions of circRBM33, miR-542-3p, and HIF-1α. MCF-7 cells were subjected to normoxia or hypoxia and transfected with plasmids that regulated CircRBM33, miR-542-3p, and HIF-1α expression levels. Glycolysis was evaluated by measuring glucose consumption, lactic acid production, and protein expression of hexokinase 2, glucose transporter type 1 and lactic dehydrogenase A. Cell proliferation and apoptosis were also assessed, and the interactions between genes were explored. CircRBM33 and HIF-1α were upregulated, while miR-542-3p was downregulated in BCa tissue samples and cell lines. Hypoxia induced circRBM33 expression in BCa cells, which negatively regulated miR-542-3p expression. CircRBM33 knockdown or miR-542-3p rescue reduced glycolysis and proliferation and promoted apoptosis of BCa cells. MiR-542-3p inhibition rescued circRBM33 knockdown-mediated glycolysis, proliferation and apoptosis of BCa cells. MiR-542-3p targeted HIF-1α, and the overexpression of HIF-1α reversed the effect of miR-542-3p upregulation on glycolysis, proliferation, and apoptosis of BCa cells. Collectively, downregulating circRBM33 suppresses miR-542-3p-targeted HIF-1α expression, resulting in the inhibition of glycolysis and proliferation and the promotion of BCa cells’ apoptosis.
Similar content being viewed by others
Introduction
Breast cancer (BCa) is the fifth leading cause of death [1] and manifests with symptoms including redness or rash around the skin or nipples, changes in breast size or shape, lumps or thickening, nipple discharge, axilla swelling, and persistent pain [2]. The length of the menstrual life, especially the part that occurs before the first full-term pregnancy, is the main risk for BCa [3]. Primary prevention to eliminate pathogenic factors and to enhance immune system, such as improving diet and reducing alcohol intake, and secondary prevention to detect BCa, such as mammography and ultrasonography, are recommended to the risk of BCa occurrence and development [4]. Tumor cells mainly rely on glycolysis to produce energy, and aerobic glycolysis is a scientifically recognized marker of cancer cell metabolism [5]. Therefore, a targeted regulation of glycolysis may provide a therapeutic direction for cancer treatment.
Oncogenic or anti-oncogenic circRNAs suggest their potential use in the treatment of BCa [6]. For instance, circRNF20 enhances the proliferation and glycolysis of BCa cells [7], while the downregulation of neoplasia domain containing 4 C (circDENND4C) decreases glycolysis and invasiveness of BCa cells under hypoxia [8]. CircRBM33 participates in the progression of gastric cancer (GC), and its expression closely correlated with metastasis, differentiation, position, and size of tumors [9]. It was shown that CircRBM33 is a trigger for glycolysis, and contributes to the growth of cervical cancer (CC) cells [10]; however, research on the mechanism of CircRBM33 in BCa is very limited. MiR-542-3p is an effective target that suppresses BCa resistance to paclitaxel [11], and that has synergistic effects when combined with doxorubicin on fighting [12]. More importantly, miR-542-3p is an indicator of cancer stages and has an antiangiogenic effect on primary BCa [13]. Regarding BCa cellular growth, miR-542-3p suppresses the biological behaviors of BCa cells through regulating its target [14]. Hypoxia provides energy for cancer development, and hypoxia-inducible factor (HIF) regulates many genes related to cancer development [15]. HIF-1α was selected as a target of miR-542-3p via bioinformatics analysis and has been implied to partly orchestrate hypoxia-induced metabolic changes to glycolysis and autophagy of BCa cells [16]. Moreover, Jinmei Jin et al. showed that the repression of the HIF-1α-dependent metabolism blocks glycolysis and growth of BCa cells [17]. CircRNAs roles as mediators of microRNAs (miRNAs) transcriptional regulatory functions associated with the development of cancer are widely accepted. Based on this background, we hypothesized that the circRBM33/miR-542-3p/HIF-1α axis may mediate BCa growth and progression. This mechanism may involve a role of circRBM33 in enhancing the growth and glycolysis of BCa cells through upregulating miR-542-3p targeting of HIF-1α.
Methods and materials
Clinical samples
BCa and normal tissue specimens were excised from 56 patients (>18 years old) in Second Hospital of Jiaxing City from September 2016 to March 2018. All cases were diagnosed as BCa and received no neoadjuvant radiotherapy or chemotherapy before surgery.
Culture and transfection of cells
The human normal breast epithelial cell line, MCF-10A, and the human BCa cell lines, MCF-7, MDA-MB-231, and SKBR3A (ATCC, VA, USA), were cultured in Roswell Park Memorial Institute-1640 medium with 10% fetal bovine serum (FBS). For hypoxic induction, the cells were exposed to 1% O2, 5% CO2, and 94% N2.
After reaching a 70–80% confluence, the cells were detached using 0.25% trypsin and passaged. The cells were divided into a normoxia group (without hypoxic induction) and an hypoxia group (hypoxic induction for 24 h) and transfected with si-negative control (NC), si-CircRBM33, miR-NC, miR-542-3p/miR-149/miR-758-3p inhibitor, miR-542-3p/miR-149/miR-758-3p mimic, overexpressed (oe)-NC, and oe-HIF-1α, respectively, using Li-pofectamine 2000.
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
Total RNA in tissues and cells was extracted by Trizol method (Takara, Dalian, China) and analyzed to determine concentration and purity. The reverse transcription of RNA into cDNA was conducted using the PrimeScript RT kit (Takara). The fluorescent quantitative PCR was processed on the ABI PRISM® 7300 system (Applied Biosystems, MA, USA). Taking Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control, and gene expression was measured using the 2−△△Ct method [18]. The primer sequences (Table 1) were designed by Sangon (Shanghai, China).
Western blot assay
Protein samples were obtained from cells after treatment with radio-immunoprecipitation assay lysis solution (Beyotime, Shanghai, China) and then quantified by the bicinchoninic acid kit (Beyotime). The proteins were mixed with the sample buffer (Beyotime), denatured, and electrophoresed at 80 V. After transferring onto a membrane, this latter was blocked in the blocking solution, incubated with the primary antibodies, HIF-1α (1:6000, ab205718), hexokinase 2 (HK2, 1:1000, ab209847), LDHA (1:5000, ab52488), GLUT1 (1:100000, ab115730, (Abcam, MA, USA), and then washed in PBS and incubated with secondary horseradish peroxidase-labeled goat anti-rabbit immunoglobulin G (IgG) antibody (1:5000, CWBIO, Beijing, China). The membrane was developed, and protein bands were visualized using a chemiluminescence imaging system (Bio-rad, CA, USA).
Detection of glucose consumption and lactate production
The supernatant of each cell culture medium was collected to determine glucose and lactic acid concentrations with the glucose colorimetric determination kit and lactic acid determination kit (both from Biovision, Wehrheim, Germany). The relative concentration was normalized to total proteins [19].
Cell counting kit (CCK)−8 assay
The cells were trypsinized, stained with trypan blue staining, supplemented with Dulbecco’s Modified Eagle Medium and FBS, and added to a 96-well plate with 2000 cells/well for 24, 48, 72 h, respectively. After culture, the cells were incubated with CCK-8 solution (10 μL/well) for 3 h, and optical density at 450 nm was measured on a microplate reader.
Flow cytometry
Annexin V-fluorescein isothiocyanate (FITC) Apoptosis Detection Kit (Thermo Fisher Scientific, MA, USA) was used to measure cell apoptosis. Cell suspension (1 × 106 cells/mL, 200 µL) was mixed with binding buffer (300 µL), supplemented with Annexin V-FITC/propidium iodide (5 µL) and detected on a flow cytometer.
Dual luciferase reporter gene assay
The binding sites of circRBM33 and miR-542-3p and that of miR-542-3p and HIF-1α were predicted online using http://starbase.sysu.edu.cn/. Wild-types circRBM33 (WT-CircRBM33) and HIF-1α (WT-HIF-1α) reporters were constructed by cloning the sequence of circRBM33 or HIF-1α containing miR-542-3p complementary sequence into the pmirGLO vector (Promega, WI, USA). The site-directed mutant reporters MUT-CircRBM33 and MUT-HIF-1α were produced by site-directed mutagenesis kit (Yeasen, Shanghai, China). The reporter, along with miR-542-3p mimic or miR-NC, were co-transfected into cells to detect luciferase activity on the dual luciferase reporter gene detection system (Promega).
RNA immunoprecipitation (RIP) assay
RIP analysis was performed with Magna RIP kit (Millipore, MA, USA). Magnetic beads coated with human anti-Ago2 antibody were reacted with RIP buffer and cell lysates. Normal IgG was used as a negative control (NC). After treatment with proteinase K, RNA was used for RT-qPCR.
Statistical analysis
The statistical analysis was performed using GraphPad prism7, and all data were expressed as mean ± standard deviation. The comparison between two groups was performed by t test, multiple groups by one-way analysis of variance (ANOVA), while pairwise comparison was performed by the Tukey’s method. The correlation was assessed using Pearson correlation analysis. P < 0.05 was indicative of a statistically significant difference.
Results
CircRBM33 is upregulated in BCa tissues and hypoxia-induced BCa cells
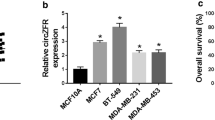
To explore the potential role of circRBM33 in BCa, we measured its expression in BCa tissues and cell lines using RT-qPCR and found that circRBM33 is highly expressed in BCa tissue samples compared with that of normal tissue samples (Fig. 1A). CircRBM33 expression was also higher in human BCa cell lines (MCF-7, MDA-MB-231, and SKBR3A) compared with that in the MCF-10A normal breast epithelial cells (Fig. 1B). Since the highest level of circRBM33 was seen in MCF-7 cells, MCF-7 cells were selected for subsequent experiments. After hypoxia induction, circRBM33 expression increased in MCF-7 cells in a time-dependent manner (Fig. 1C). These results demonstrate that circRBM33 is highly expressed in BCa, and that its expression is induced under hypoxic conditions.
A CircRBM33 was upregulated in BCa tissues; B CircRBM33 was upregulated in BCa cells (Compared with MCF-10A cells, circRBM33 expression was 1.7 times higher in MCF-7 cells); C CircRBM33 was upregulated in hypoxia-induced MCF-7 cells; **P < 0.01; *P < 0.05 compared with the 0 h group. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison between two groups was performed by t test, that among multiple groups was by one-way ANOVA, and pairwise comparison was by Tukey’s method.
Hypoxia increases glycolysis and inhibits apoptosis of BCa cells
Several studies reported that hypoxia promotes glycolysis and proliferation and inhibits apoptosis of cancer cells [20,21,22,23,24]. Therefore, glycolysis and apoptosis of BCa cells were tested under normoxia and hypoxia. The results showed an increased cell glucose consumption and lactate production under hypoxic conditions compared with those normoxic conditions (Fig. 2A, B). The protein expression levels of the glycolysis-related proteins, HK2, LDHA, and GLUT1, were detected by Western blot, and the results showed that HK2, LDHA, GLUT1, and GLUT1 protein expressions increased under hypoxic conditions (Fig. 2C). Cell proliferation and apoptosis were detected by CCK-8 and flow cytometry, respectively. Under hypoxic conditions, cell proliferation was promoted, and cell apoptosis was inhibited (Fig. 2D, E) (all P < 0.05). In summary, glycolysis, and cell proliferation of BCa cells can be induced under hypoxic conditions, while apoptosis is inhibited.
A Glucose consumption of MCF-7 cells under hypoxic conditions was 2.5 times that under normoxia; B Lactate production of MCF-7 cells under hypoxic conditions was 2.8 times that under normoxia; C Western blot detected HK2, LDHA and GLUT1 expression under hypoxia (HK2 expression of MCF-7 cells under hypoxic conditions was 2.8 times that under normoxia, LDHA expression of MCF-7 cells under hypoxic conditions was 1.4 times that under normoxia, GLUT1 expression of MCF-7 cells under hypoxic conditions was 1.9 times that under normoxia); D CCK-8 detected MCF-7 cell proliferation under hypoxia; E Flow cytometry detected MCF-7 cell apoptosis under hypoxia (cell apoptosis rate decreased from 8.86% to 3.12%); *P < 0.05. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison between two groups should be performed by t test.
Knockout of circRBM33 reduces glycolysis and promotes apoptosis of BCa cells
It was previously reported that circRBM33 knockdown inhibits cervical cancer glycolysis [25]. CircRBM33 is overexpressed in BCa and can be induced under hypoxic conditions. To further study the mechanism by which hypoxia induces glycolysis and cell proliferation and inhibits apoptosis of BCa cells, we designed si-CircRBM33 to knock down CircRBM33 expression in BCa cells (Fig. 3A). Transfection of si-CircRBM33 reduced glucose consumption and lactate production of BCa cells in normoxia and hypoxia groups (Fig. 3B, C). Western blot analysis showed that circRBM33 downregulation decreases HK2, LDHA, GLUT1 and GLUT1 protein expressions in both normoxic and hypoxic BCa cells (Fig. 3D). The results of CCK-8 and flow cytometry demonstrated that si-CircRBM33 reduces proliferation and promotes apoptosis of BCa cells in the normoxia and hypoxia groups (Fig. 3E, F). Collectively, these results demonstrate that of circRBM33 knockout reduces glycolysis and proliferation and promotes apoptosis of BCa cells.
A CircRBM33 expression in MCF-7 cells after knocking down circRBM33; B Glucose consumption in MCF-7 cells after knocking down circRBM33; C Lactate production in MCF-7 cells after knocking down circRBM33; D Western blot detected HK2, LDHA and GLUT1 expression in MCF-7 cells after knocking down circRBM33; E CCK-8 detected MCF-7 cell proliferation after knocking down circRBM33; F Flow cytometry detected MCF-7 cell apoptosis after knocking down circRBM33 (Under normoxia, the apoptotic rate increased from 8.15% to 17.25% after knocking down circRBM33; Under hypoxia, the apoptotic rate increased from 2.58% to 8.95% after knocking down circRBM33); *P < 0.05. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison between two groups should be performed by t test.
CircRBM33 negatively regulates miR-542-3p expression
The StarBase database showed that circRBM33 and miR-542-3p may bind to each other (Fig. 4A). The interaction between the two was confirmed by dual luciferase reporter experiment, as shown by the reduction of the luciferase activity of the cells that were co-transfected with miR-542-3p mimic and WT-CircRBM33 (Fig. 4B). To verify the endogenous association between circRBM33 and miR-542-3p, RIP experiment was conducted in MCF-7 cells overexpressing miR-542-3p using an anti-Ago2 antibody. The results demonstrated that miR-542-3p overexpression enhances circRBM33 enrichment (Fig. 4C).
A StarBase database showed binding sites between circRBM33 and miR-542-3p; B Dual luciferase reporter experiment confirmed the interaction between circRBM33 and miR-542-3p; C RIP experiment confirmed the interaction between circRBM33 and miR-542-3p; D MiR-542-3p expression after knocking down circRBM33; E miR-542-3p was downregulated in BCa tissues; F MiR-542-3p was downregulated in hypoxia-induced BCa cells; G. Pearson test analyzed the correlation between circRBM33 and miR-542-3p levels; **P < 0.01; *P < 0.05. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison between two groups should be performed by t test, that among multiple groups was by one-way ANOVA, while pairwise comparison was by Tukey’s method. The correlation was analyzed using Pearson correlation analysis.
Furthermore, miR-542-3p expression was measured by RT-qPCR, and the finding indicated that miR-542-3p was increased when circRBM33 was inhibited in the cells (Fig. 4D). Moreover, the reduction of miR-542-3p expression was tested in BCa and normal tissue samples (Fig. 4E). We found that miR-542-3p expression is reduced by hypoxia in BCa cells in a time-dependent manner (Fig. 4F). Besides, Pearson test confirmed the negative correlation between circRBM33 and miR-542-3p (Fig. 4G), further indicating that circRBM33 interacts with miR-542-3p in BCa. The above results indicate that circRBM33 negatively regulates miR-542-3p expression through of its interaction with miR-542-3p.
Inhibition of miR-542-3p rescues circRBM33 knockout-mediated glycolysis and apoptosis for BCa cells
Subsequently, using a miR-542-3p inhibitor, we successfully inhibited miR-542-3p expression in si-CircRBM33-transfected BCa cells (Fig. 5A). The results showed that the co-transfection of si-CircRBM33 and miR-542-3p inhibitor promotes glucose consumption and lactate production in BCa cells in the normoxia and hypoxia groups (Fig. 5B, C), and increases HK2, LDHA, and GLUT1 protein expressions (Fig. 5D). Cell proliferation and apoptosis analysis revealed that the downregulation of circRBM33 and miR-542-3p accelerates proliferation, while also suppressing apoptosis of BCa cells in the normoxia and hypoxia groups (Fig. 5E, F). In summary, miR-542-3p downregulation can reverse the effect of circRBM33 knockdown on glycolysis, proliferation, and apoptosis of BCa cells.
A miR-542-3p expression in MCF-7 cells after inhibiting circRBM33 and miR-542-3p; B Glucose consumption in MCF-7 cells after inhibiting circRBM33 and miR-542-3p; C Lactate production in MCF-7 cells after inhibiting circRBM33 and miR-542-3p; D Western blot detected HK2, LDHA and GLUT1 expression in MCF-7 cells after inhibiting circRBM33 and miR-542-3p; E CCK-8 detected MCF-7 cell proliferation after inhibiting circRBM33 and miR-542-3p; F Flow cytometry detected MCF-7 cell apoptosis after inhibiting circRBM33 and miR-542-3p (Under normoxia, the apoptotic rate dropped from 16.82% to 10.52% after inhibiting circRBM33 and miR-542-3p; Under hypoxia, the apoptotic rate decreased from 9.80% to 3.25% after inhibiting circRBM33 and miR-542-3p); *P < 0.05. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison between two groups should be performed by t test.
MiR-542-3p targets HIF-1α
Using the StarBase data, we searched for the binding sites between HIF-1α and miR-542-3p (Fig. 6A). The outcome from the dual luciferase reporter experiment showed that the transfection of miR-542-3p mimic weakens the luciferase activity of WT-HIF-1α (Fig. 6B). Meanwhile, RIP detection demonstrated that HIF-1α and miR-542-3p are enriched in Ago2 immunoprecipitation (Fig. 6C).
A The binding site between miR-542-3p and HIF-1α; B Dual luciferase reporter experiment confirmed the interaction between miR-542-3p and HIF-1α; C RIP experiment confirmed the interaction between miR-542-3p and HIF-1α; D, E HIF-1α expression after downregulating or upregulating miR-542-3p; F HIF-1α was upregulated in BCa tissues; G HIF-1α was upregulated in hypoxia-induced BCa cells; H Pearson test analyzed the correlation between miR-542-3p and HIF-1α; **P < 0.01; *P < 0.05. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison between two groups should be performed by t test, that among multiple groups was by one-way ANOVA, while pairwise comparison was by Tukey’s method. The correlation was analyzed using Pearson correlation analysis.
Subsequently, we applied RT-qPCR and Western blot methods to measure HIF-1α levels in cells and found that HIF-1α expression decreases in the cells transfected with miR-542-3p mimic, while this expression was increased in the cells transfected with the miR-542-3p inhibitor (Fig. 6D, E).
In addition, higher HIF-1α levels were observed in BCa tissue samples compared to those in normal tissue samples, and in hypoxic BCa cells compared to those in normoxic BCa cells (Fig. 6F, G). Pearson analysis confirmed a negative correlation between HIF-1α and miR-542-3p expressions (Fig. 6H). In summary, HIF-1α is a target of miR-542-3p.
Overexpression of HIF-1α reverses the inhibitory effect of upregulated miR-542-3p on glycolysis and apoptosis of BCa cells
To further investigate this mechanism, we overexpressed miR-542-3p and HIF-1α in BCa cells (Fig. 7A-B). We found that miR-542-3p mimic inhibits glycolysis and proliferation of BCa cells in the normoxia and hypoxia groups, while the co-transfection of HIF-1α reverses the effects of miR-542-3p mimic on glycolysis and proliferation (Fig. 7C–F). According to the flow cytometry results, we found that the overexpression of miR-542-3p promotes the apoptosis of BCa cells in the normoxia and hypoxia groups, and that such effects can be mitigated when HIF-1α was overexpressed (Fig. 7G). Together, these results demonstrate that miR-542-3p inhibits glycolysis and proliferation and promotes cell apoptosis of BCa cells through HIF-1α.
A, B HIF-1α expression in MCF-7 cells after upregulating miR-542-3p and HIF-1α; C Glucose consumption in MCF-7 cells after upregulating miR-542-3p and HIF-1α; D Lactate production in MCF-7 cells after upregulating miR-542-3p and HIF-1α; E Western blot detected HK2, LDHA and GLUT1 expression in MCF-7 cells after upregulating miR-542-3p and HIF-1α; F CCK-8 detected MCF-7 cell proliferation after upregulating miR-542-3p and HIF-1α; G Flow cytometry detected MCF-7 cell apoptosis after upregulating miR-542-3p and HIF-1α (Under normoxia, after upregulating miR-542-3p, the apoptotic rate increased from 7.28% to 16.88%, and after upregulating miR-542-3p and HIF-1α, the apoptotic rate decreased from 17.25% to 9.14%; Under hypoxia, after upregulating miR-542-3p, the apoptotic rate increased from 3.20% to 7.88%, and after upregulating miR-542-3p and HIF-1α, the apoptotic rate decreased from 8.50% to 3.340%); *P < 0.05. Data were expressed in the form of mean ± standard deviation (Repetitions = 3). The comparison among multiple groups should be performed by one-way ANOVA, while pairwise comparison was by Tukey’s method.
Discussion
BCa is a heterogeneous disease that leads to a high mortality rate in female [26]. Our study investigated the mechanism and effects of circRBM33 in BCa. Our results showed that circRBM33 downregulation increases miR-542-3p expression, which reduces HIF-1α expression leading to the suppression of glycolysis and proliferation of BCa cells.
In this study, we first showed that circRBM33 expression is high in BCa tissues, and that its expression is induced by hypoxia. Subsequently, we functionally observed that circRBM33 silencing in BCa cells, under normoxia or hypoxia, reduces glycolysis and proliferation, and enhanced apoptosis of BCa cells. To our best knowledge, other studies reported a similar effect of circRBM33 in other cancer types. It has been shown that circRBM33 silencing inhibits proliferation, migration, and invasion, and promotes apoptosis of gastric cancer cells through targeting the miR-149/IL-6 axis [9]. Furthermore, circRBM33 knockdown have been shown to regulate the miR-758-3p/PUM2 axis to inhibit tumor growth in vivo, suppress cervical cancer cell proliferation, migration, invasion, and glycolysis, and promote cervical cancer cell apoptosis in vitro [25]. However, we found that miR-149 and miR-758-3p do not affect the expression of HIF1α (Supplementary Fig. 1). As indicated, circRBM33 gene expression is upregulated in GC, and the reduction of CircRBM33 expression enhances apoptotic but blocks proliferative, invasive, and migratory phenotypes of GC cells via a targeted increase of miR-149 expression [9]. Another report also reported an increase of circRBM33 expression in CC, and showed that of circRBM33 depletion reduces proliferation and glycolysis, and enhances apoptosis of malignant cells [10]. These reports suggest that circRBM33 is a promoter of cancer progression.
Next, we found that miR-542-3p interacts with circRBM33 and regulates BCa proliferation and progression. For this, we investigated the potential functions of upregulated miR-542-3p in BCa and observed that its overexpression exhibits similar effects to those of circRBM33 downregulation in BCa cells. H-X Wu et al. have reported that miR-542-3p is expressed at low levels in BCa, and that the introduction of ectopic miR-542-3p into BCa cells reduces cell growth [14]. Moreover, miR-542-3p mimic-induced upregulation of miR-542-3p in BCa cells strengthens the efficacy of paclitaxel in chemoresistant BCa [11]. On the other hand, a study confirmed that the combination of miR-542-3p expression and, doxorubicin can promote the apoptosis of triple negative BCa cells, indicating a potential use of this combination as a promising therapy for BCa [12]. Similarly, the reduction of miR-542-3p level was tested in colorectal cancer, while its overexpression provides resistance against the aggressive behaviors of cancer cells by negatively modulating the expression of miR-542-3p’s targets [27]. Additionally, another publication reported that restoring miR-542-3p is capable of restraining proliferation, whilst miR-542-3p knockdown exerts the opposite effect on epithelial ovarian cancer cells [28]. In general, all these studies have confirmed our finding that miR-542-3p increased expression depresses tumorigenesis.
Finally, the target of miR-542-3p, HIF-1α, has captured our attention. Through rescue experiment, we noticed that HIF-1α is a miR-542-3p downstream factor in mediating BCa, as shown by the results that demonstrated that HIF-1α overexpression mitigates the role of upregulated miR-542-3p in BCa cells. Recently, it was reported that HIF-1 stabilization has anti-tumor effects on BCa [29]. A former study has showed that Parkin-mediated ubiquitination and degradation of HIF-1α is partly involved in reducing metastasis of BCa cells [30]. HIF-1 activation is an active driver of BCa glycolysis [31], and a marker of enhanced aerobic glycolysis associated with drug resistance [32]. A recent study reported that in cardamonin-treated BCa, HIF-1α inhibition is partly involved in reducing glucose uptake and lactic acid production [17].
Conclusion
Overall, this study demonstrates that the inhibition of circRBM33 prevents BCa progression by reducing glycolysis and proliferation and by promoting apoptosis of BCa cells. It also shows that this mechanism involves miR-542-3p-mediated downregulation of HIF-1α. Our experimental discovery may provide new insights into the molecular mechanism of BCa. In the future, more studies are required to further validate and develop the results of our research.
References
Fahad Ullah M. Breast cancer: current perspectives on the disease status. Adv Exp Med Biol. 2019;1152:51–64.
Pearce L. Breast cancer. Nurs Stand. 2016;30:15.
Libson S, Lippman M. A review of clinical aspects of breast cancer. Int Rev Psychiatry. 2014;26:4–15.
Kolak A, Kaminska M, Sygit K, Budny A, Surdyka D, Kukielka-Budny B, et al. Primary and secondary prevention of breast cancer. Ann Agric Environ Med. 2017;24:549–53.
Wu Z, Wu J, Zhao Q, Fu S, Jin J. Emerging roles of aerobic glycolysis in breast cancer. Clin Transl Oncol. 2020;22:631–46.
Li Z, Chen Z, Hu G, Jiang Y. Roles of circular RNA in breast cancer: present and future. Am J Transl Res. 2019;11:3945–54.
Cao L, Wang M, Dong Y, Xu B, Chen J, Ding Y, et al. Circular RNA circRNF20 promotes breast cancer tumorigenesis and Warburg effect through miR-487a/HIF-1alpha/HK2. Cell Death Dis. 2020;11:145.
Ren S, Liu J, Feng Y, Li Z, He L, Li L, et al. Knockdown of circDENND4C inhibits glycolysis, migration and invasion by up-regulating miR-200b/c in breast cancer under hypoxia. J Exp Clin Cancer Res. 2019;38:388.
Wang N, Lu K, Qu H, Wang H, Chen Y, Shan T, et al. CircRBM33 regulates IL-6 to promote gastric cancer progression through targeting miR-149. Biomed Pharmacother. 2020;125:109876.
Ding Y, Yuan X, and Gu W. Circular RNA RBM33 contributes to cervical cancer progression via modulation of the miR-758-3p/PUM2 axis. J Mol Histol. 2021;52:173–85.
Lyu H, Wang S, Huang J, Wang B, He Z, Liu B. Survivin-targeting miR-542-3p overcomes HER3 signaling-induced chemoresistance and enhances the antitumor activity of paclitaxel against HER2-overexpressing breast cancer. Cancer Lett. 2018;420:97–108.
Wang S, Zhang J, Wang Y, Chen M. Hyaluronic acid-coated PEI-PLGA nanoparticles mediated co-delivery of doxorubicin and miR-542-3p for triple negative breast cancer therapy. Nanomedicine. 2016;12:411–20.
He T, Qi F, Jia L, Wang S, Song N, Guo L, et al. MicroRNA-542-3p inhibits tumour angiogenesis by targeting angiopoietin-2. J Pathol. 2014;232:499–508.
Wu HX, Wang GM, Lu X, Zhang L. miR-542-3p targets sphingosine-1-phosphate receptor 1 and regulates cell proliferation and invasion of breast cancer cells. Eur Rev Med Pharm Sci. 2017;21:108–14.
Tirpe AA, Gulei D, Ciortea SM, Crivii C, and Berindan-Neagoe I. Hypoxia: Overview on Hypoxia-Mediated Mec hanisms with a Focus on the Role of HIF Genes. Int J Mol Sci. 2019;20:6140.
Lee S, Hallis SP, Jung KA, Ryu D, Kwak MK. Impairment of HIF-1alpha-mediated metabolic adaption by NRF2-silencing in breast cancer cells. Redox Biol. 2019;24:101210.
Jin J, Qiu S, Wang P, Liang X, Huang F, Wu H, et al. Cardamonin inhibits breast cancer growth by repressing HIF-1alpha-dependent metabolic reprogramming. J Exp Clin Cancer Res. 2019;38:377.
Tuo YL, Li XM, Luo J. Long noncoding RNA UCA1 modulates breast cancer cell growth and apoptosis through decreasing tumor suppressive miR-143. Eur Rev Med Pharm Sci. 2015;19:3403–11.
Shi J, Wang H, Feng W, Huang S, An J, Qiu Y, et al. Long non-coding RNA HOTTIP promotes hypoxia-induced glycolysis through targeting miR-615-3p/HMGB3 axis in non-small cell lung cancer cells. Eur J Pharm. 2019;862:172615.
Li L, Ma Y, Maerkeya K, Reyanguly D, Han L. LncRNA OIP5-AS1 regulates the warburg effect through miR-124-5p/IDH2/HIF-1alpha pathway in cervical cancer. Front Cell Dev Biol. 2021;9:655018.
Zheng X, Fan H, Liu Y, et al. Hypoxia boosts aerobic glycolysis of carcinoma: a complex process for tumor development.[J]. Curr Mol Pharmacol, 2021. https://doi.org/10.2174/1874467214666210811145752.
Alharbi M, Lai A, Sharma S, Kalita-de Croft P, Godbole N, Campos A, et al. Extracellular vesicle transmission of chemoresistance to ovarian cancer cells is associated with hypoxia-induced expression of glycolytic pathway proteins, and prediction of epithelial ovarian cancer disease recurrence. Cancers. 2021;13:3388.
Xu Y, Kuai R, Chu YM, Zhou L, Zhang HQ, Li J. Hypoxia facilitates the proliferation of colorectal cancer cells by inducing cancer-associated fibroblast-derived IL6. Neoplasma. 2021;68:1015–1022.
Zhang H, Zhao X, Guo Y, Chen R, He J, Li L, et al. Hypoxia regulates overall mRNA homeostasis by inducing Met(1)-linked linear ubiquitination of AGO2 in cancer cells. Nat Commun. 2021;12:5416.
Ding Y, Yuan X, Gu W. Circular RNA RBM33 contributes to cervical cancer progression via modulation of the miR-758-3p/PUM2 axis. J Mol Histol. 2021;52:173–85.
Januskeviciene I, Petrikaite V. Heterogeneity of breast cancer: The importance of interaction between different tumor cell populations. Life Sci. 2019;239:117009.
Yuan L, Yuan P, Yuan H, Wang Z, Run Z, Chen G, et al. miR-542-3p inhibits colorectal cancer cell proliferation, migration and invasion by targeting OTUB1. Am J Cancer Res. 2017;7:159–72.
Li J, Shao W, Feng H. MiR-542-3p, a microRNA targeting CDK14, suppresses cell proliferation, invasiveness, and tumorigenesis of epithelial ovarian cancer. Biomed Pharmacother. 2019;110:850–6.
Kachamakova-Trojanowska N, Podkalicka P, Bogacz T, Barwacz S, Jozkowicz A, Dulak J, et al. HIF-1 stabilization exerts anticancer effects in breast cancer cells in vitro and in vivo. Biochem Pharm. 2020;175:113922.
Liu J, Zhang C, Zhao Y, Yue X, Wu H, Huang S, et al. Parkin targets HIF-1alpha for ubiquitination and degradation to inhibit breast tumor progression. Nat Commun. 2017;8:1823.
Lu Y, Wang L, Ding W, Wang D, Wang X, Luo Q, et al. Ammonia mediates mitochondrial uncoupling and promotes glycolysis via HIF-1 activation in human breast cancer MDA-MB-231cells. Biochem Biophys Res Commun. 2019;519:153–9.
Woo YM, Shin Y, Lee EJ, Lee S, Jeong SH, Kong HK, et al. Inhibition of aerobic glycolysis represses Akt/mTOR/HIF-1alpha axis and restores tamoxifen sensitivity in antiestrogen-resistant breast cancer cells. PLoS One. 2015;10:e0132285.
Acknowledgements
We would like to acknowledge the reviewers for their helpful comments on this paper. We would like to express their gratitude to EditSprings (https://www.editsprings.cn/) for the expert linguistic services provided. The study is funded by Jiaxing Public Welfare Research Program (2020AD30064), Jiaxing Science and technology project (The expression of SDC-1 in breast cancer epithelial cells and its correlation with clinical pathology)(number:2021AD30119).
Author information
Authors and Affiliations
Contributions
FL: finished study design; YJ, DY: finished experimental studies; MZ, GH, JW: finished data analysis; YJ, MZ: finished manuscript editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The use of all specimens has been approved by families of the patients. This study was approved by the ethics committee of Second Hospital of Jiaxing City.
Consent for publication
The use of all specimens has been approved by families of the patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jiang, Y., Zhang, M., Yu, D. et al. CircRBM33 downregulation inhibits hypoxia-induced glycolysis and promotes apoptosis of breast cancer cells via a microRNA-542-3p/HIF-1α axis. Cell Death Discov. 8, 126 (2022). https://doi.org/10.1038/s41420-022-00860-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41420-022-00860-6
- Springer Nature Limited
This article is cited by
-
NREP, transcriptionally upregulated by HIF-1α, aggravates breast cancer cell growth and metastasis by promoting glycolysis
Cell Death Discovery (2024)
-
Non-coding RNAs in breast cancer: with a focus on glucose metabolism reprogramming
Discover Oncology (2023)