Abstract
The re-use of genes in new organs forms the base of many evolutionary novelties. A well-characterised case is the recruitment of the posterior spiracle gene network to the Drosophila male genitalia. Here we find that this network has also been co-opted to the testis mesoderm where is required for sperm liberation, providing an example of sequentially repeated developmental co-options. Associated to this co-option event, an evolutionary expression novelty appeared, the activation of the posterior segment determinant Engrailed to the anterior A8 segment controlled by common testis and spiracle regulatory elements. Enhancer deletion shows that A8 anterior Engrailed activation is not required for spiracle development but only necessary in the testis. Our study presents an example of pre-adaptive developmental novelty: the activation of the Engrailed transcription factor in the anterior compartment of the A8 segment where, despite having no specific function, opens the possibility of this developmental factor acquiring one. We propose that recently co-opted networks become interlocked, so that any change to the network because of its function in one organ, will be mirrored by other organs even if it provides no selective advantage to them.
Similar content being viewed by others
Introduction
It has been observed that genes playing particular roles during organ development can be recruited to perform novel functions in other organs. The re-use, or co-option, of developmental genes in new organs, is the base of many evolutionary novelties. An example of this is the highly transparent crystallin proteins that refract light in the eye lens. In all vertebrates, α-crystallin evolved from the co-option of a small heat shock protein to the eye; while in birds δ-crystallin evolved from the co-option of a different protein, the Arginosuccinase lyase involved in arginine biosynthesis1.
Although there is abundant research on single-gene co-option, few studies have considered the functional consequences of full-gene network co-option. One of the best-characterised co-option cases is the recruitment of the appendage-forming gene network to form the eye-spots that decorate butterfly wings in several species2. Similarly, in the Drosophila melanogaster subgroup, the larval respiratory posterior spiracles and the adult male genitalia share the expression of numerous genes due to the recent co-option into the male genital disc primordium of a pre-existing gene network controlling the formation of the external larval respiratory organs3. The co-option of this gene network to the male genitalia resulted in the formation of the posterior lobe, a structure present in D. simulans and D. mauritiana, closely related to Drosophila melanogaster, but not in the more distant D. biarmipes or D. ananassae species3.
The formation of the posterior spiracles and the posterior lobes have been well-studied in D. melanogaster. Posterior spiracle organogenesis is regulated by a gene network activated in the eighth abdominal larval segment (A8) by the Hox protein Abdominal-B (Abd-B) (Fig. 1a, b)4. The internal spiracular chamber is formed from A8 anterior compartment cells (A8a) when Abd-B activates in the dorsal ectoderm the transcription of the JAK/STAT signalling pathway ligand Unpaired (Upd) as well as the Empty spiracles (Ems) and the Cut (Ct) transcription factors. The external protruding stigmatophore is formed from both anterior and posterior compartment A8 cells when Abd-B activates the Spalt (Sal) transcription factor (Fig. 1b), which in turn activates engrailed (en) transcription in a unique A8 pattern5,6. These primary factors activate the RhoGAP Cv-c and RhoGEF64C cytoskeletal regulators, the cell polarity gene crumbs (crb) and various Cadherins6. Abd-B also modulates the expression of wingless and the EGF regulator rhomboid genes generating A8 specific segmental information distinct from that in more anterior segments5.
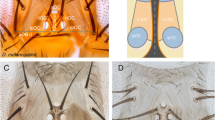
a Close-up of the larval posterior spiracles showing the internal filters (fz) formed by the spiracular chamber cells, lodged in the protruding stigmatophores (st) and connecting to the tracheae (tr). b Scheme of the st11 early posterior spiracle organogenetic gene network activated in A8 by Abd-B. Light blue represents the spiracular chamber primordium that eventually will invaginate to form at st16 the fz (brown, inset). Olive green represents the cells of the stigmatophore primordium. The spatial expression domain of the Abd-B m (orange line) and Abd-Br (mauve line) isoforms and the location of the antero-posterior A8 compartment boundary are indicated. All spiracular chamber cells form in the Abd-Bm expressing anterior compartment, while the stigmatophore primordium comprises both anterior and posterior dorsal A8 cells. c, d hh-lacZ embryos double stained with anti-En (green) and anti-ßGal (magenta) at progressively later stages: (c-c”) st11, (d-d”) st14. Magenta arrowheads point to anterior A8 cells gaining En expression at st14, white arrowheads point at the A8 posterior cells losing En expression. The panel to the right shows both channels. To facilitate segment comparison between the extended germ band embryo (c) and the retracted germ band embryo (d), the close-up of the squared region shown in panel c, has been rotated 180°. The location of the posterior abdominal (A6–A9) compartments is labelled. e–g Wild-type embryos double stained with anti-Sal (magenta) and anti-En (green) in Drosophila melanogaster (e-e”), Drosophila virilis (f-f”) and Episyrphus balteatus (g-g”). White arrowheads in (e’-g’) point at the stigmatophore Sal expression and asterisks mark the spiracle opening. Note that the cross-reactive Sal antibody staining has some background (g’) due to unspecific binding to the vitelline membrane that cannot be completely removed. h–j Cuticles of recently hatched Drosophila melanogaster (h), Drosophila virilis (i) and Episyrphus balteatus larvae (j) with arrows pointing to the protruding stigmatophore. Scale bars: 50 µm in c–g and 100 µm in h–j.
The posterior lobe is a hook-shaped structure used by the male to grasp the female during mating, which may act as a prezygotic reproductive isolation barrier facilitating speciation7. In D. melanogaster, ten genes of the spiracle gene network are required for the formation of the posterior lobe, with their activation in at least seven cases being regulated in both structures by the same cis-regulatory elements (CRE). The study of two of these enhancers, revealed that the same DNA-binding sites activate the CRE’s expression in both organs, making this one of the best-characterised cases of whole gene network co-option3.
The recruitment of a gene network to a new organ exposes it to different selective pressures that may accelerate the appearance of novelties8. One such novelty is the expression of the posterior segment determinant engrailed in the anterior compartment cells of the A8 segment5. Engrailed is crucial during segmentation, and its expression in the anterior compartment is surprising given that En has been localised to the posterior cells all along arthropod evolution9,10,11,12.
Drosophila segmentation results from the activation of the segment-polarity genes engrailed (en), hedgehog (hh) and wingless (wg) in periodic stripes of cells along the antero-posterior axis of the embryo13,14,15. Once activated, the segment-polarity genes engage in cross-regulatory interactions that maintain their expression. As a result, En and its direct target hh become expressed throughout development in the posterior compartment of every segment, where they regulate cell tension and adhesive characteristics that prevent posterior cells from mixing with anterior compartment cells, generating stable signalling boundaries16. Mutant embryos for either en or hh result in an almost complete fusion of segments. As the posterior spiracle is one of the few circumferential organs in the embryo, en activity in anterior A8 cells could be required for establishing the circumferential information pattern necessary for spiracle organogenesis that contributed to the evolution of the protruding posterior spiracles characteristic of dipteran larvae.
Here, we investigate when en was recruited to the anterior A8 cells (A8a) and the cis and trans regulatory elements responsible for it. We find that A8a expression appeared in Diptera before the evolution of the posterior lobe and present evidence that this is associated with a previous posterior spiracle gene network co-option event to the testis mesoderm. Working with Drosophila melanogaster, we show that Engrailed expression in the A8a compartment is not required for the spiracle’s development and that the engrailed CRE controlling spiracle expression is required in the testis cyst cells for spermiation. Our work presents an example of repeated sequential gene network co-option events involving tissues of different germ layers, and shows how this resulted in the generation of a bona fide pre-adaptive developmental expression novelty: the activation of the En transcription factor in the anterior compartment of the embryonic A8 segment where, despite having no specific function, it opens the possibility of this important developmental factor acquiring one in the future. We show that the expression of en in the anterior compartment of the A8 segment, was likely caused by the regulatory interlocking of the co-opted networks. We propose gene network interlocking occurs as the result of the use of the same gene network in several organs, so that any change to the network because of its functionality in one organ, will be mirrored in all organs even if it has no selective advantage in some of them.
Results
Engrailed expression in the posterior part of the segment has been conserved in arthropods for over 500 million years9,10,11,12. Despite such wide conservation, some exceptions have been described in D. melanogaster where en becomes activated in anterior cells of the wing primordium and in the posterior spiracles5,17. We have found that although hh and en are initially expressed in a coincident stripe of A8 posterior cells (Fig. 1c-c”), after stage 11, En expression in A8 diverges from that of hh, being turned off from the ventral posterior cells and activated in dorsal anterior cells (Fig. 1d). This pattern re-specification generates a circumference of En expressing cells around the spiracle opening that could be important for stigmatophore morphogenesis.
To infer when this expression novelty appeared we stained different Diptera species using the cross-reactive antibodies anti-Sal, which serves as a marker for the stigmatophore, and anti-En. For this purpose, we analysed three cyclorrhaphan species with similar larval body plans (Supplementary Fig. 1). En and Sal expression patterns are almost indistinguishable between Drosophila melanogaster and Drosophila virilis, which diverged about 40 million years ago (Sophophora Drosophila) (Fig. 1e, f). In contrast, while Sal is expressed in the stigmatophore of Episyrphus balteatus, which diverged from Drosophila about 100 million years ago18, En does not form a ring around the spiracle opening, being expressed as a stripe (Fig. 1g). This stripe is very similar to that formed in the A9 segment suggesting that En expression in Episyrphus is restricted to the posterior compartment and that expression of En in A8a has been recently acquired in the brachiceran diptera. Comparison of Episyrphus and Drosophila larvae shows that the stigmatophore of E. balteatus is noticeably less protrusive than that of D. melanogaster or D. virilis (Fig. 1h–j) suggesting that En recruitment to the A8a compartment may be responsible for this organ shape difference.
Regulation of engrailed anterior compartment expression
To understand the genetic and molecular mechanisms driving activation of en in the anterior compartment, we searched in D. melanogaster for the specific CRE activating its expression in A8a cells and the transcriptional regulators interacting with it. Our search was facilitated by a previous analysis that identified the CREs present in the invected-engrailed locus19. Analysis of the enH-lacZ, enM-lacZ and enP-lacZ reporters driving stripe expression at different stages, reveals they are exclusively active in the posterior cells of the A8 segment (Supplementary Fig. 2a–d). Two reporters are specifically expressed in A8: the enX-lacZ reporter is active in small groups of isolated cells that probably represent spiracle sensory elements, while the enD-lacZ reporter shows expression in a ring of cells surrounding the spiracle’s opening (Supplementary Fig. 2e–f).
Dissection of enD localised the posterior spiracle-specific enhancer to a 439 bp region (enD0.4, Supplementary Fig. 3). The activation of reporter genes containing this element appears first in a dorsal stripe in the anterior compartment of A8 (Supplementary Fig. 3c–e) that expands at later stages to surround the spiracle opening coinciding with En protein expression in the stigmatophore (Fig. 2a). As all reporters containing these regulatory elements drive similar expression (Supplementary Fig. 3f–g), in what follows we use them interchangeably to study en regulation in the posterior spiracle using either enD-lacZ, enD-ds-GFP, or enD-0.4-mCherry as specified in each particular figure.
Expression of enD enhancer in wild type (a–c, e, g) or posterior spiracle mutant embryos (d, f, h). a enD-lacZ embryo double stained with anti-En (a’ green) and anti-ßGal (a” magenta). b, c e enD-lacZ embryos double stained with anti-ßGal (green) and either anti-Ct (b), anti-AbdB (c), or anti-Sal (e) in magenta. g enD-GFP upd0.43-lacZ embryo double stained with anti-GFP (g’ green) and anti-ßGal (g” magenta). d, f, h enD-lacZ embryos homozygous mutant for either Abd-BM1 (d), Df(2)5 sal-salr (f) or the upd locus deficiency Df(1)os1A (h), double stained with anti-ßGal (green) and anti-En (magenta). Panels b and g also show orthogonal sections to demonstrate Ct and upd0.43 are expressed in the internal spiracular chamber cells and enD-lacZ in the external stigmatophore cells. All embryos are st14. Scale bar: 20 µm.
To identify the upstream regulators controlling engrailed expression in the anterior compartment we analysed the reporter’s activation in mutants for the segment polarity and for the posterior spiracle gene networks. Expression of enD-lacZ in mutants for either enE, hhAC or wgCX4 homozygous embryos is maintained in the posterior spiracles despite their global abnormal morphology (Supplementary Fig. 4a–c), suggesting the enhancer is not under the direct regulation of the segment-polarity gene network.
We then determined the spatial relationship of spiracle cells activating the enD enhancer with respect to various posterior spiracle gene network regulators6,20,21 and found they belong to the protruding stigmatophore (Fig. 2b, c, e, g).
Analysis of posterior spiracle gene network mutants shows the enD reporters are not expressed in Abd-BM1 nor in sal null mutants (Fig. 2d, f) or in embryos carrying the Df(1)os1A deletion that lack the Upd, Upd2 and Upd3 ligands activating the JAK/STAT signalling pathway (Fig. 2h). Mutations in other posterior spiracle genes tested do not affect enD expression despite having abnormal spiracle development (Supplementary Fig. 5a, b).
As upd is not transcribed in the stigmatophore, these results suggest that Upd diffusion from the spiracular chamber primordium activates the JAK/STAT receptor in the adjacent Sal expressing stigmatophore cells. We tested this model by expressing ectopically in all ectodermal cells either UAS-upd or UAS-sal with the pan-ectodermal driver 69B-Gal4 line. Ectopic Upd expression results in the expansion of enD-reporter expression to all dorsal A8 and A9a Sal-expressing cells (Supplementary Fig. 5c). In contrast, ectopic expression of Sal in the whole ectoderm does not modify enD activation in A8 (Supplementary Fig. 5d). To test if simultaneous Sal and Upd activation of JAK/STAT would suffice to activate enD expression in the dorsal ectoderm, we simultaneously expressed UAS-sal and UAS-upd. Although the co-expression of both proteins results in additional ectoderm expression, this only appears in the posterior A7 segment (Supplementary Fig. 5e) suggesting that Abd-B or another posterior abdominal factor is also required for enD activation.
Regulation of enD expression by STAT and Abd-B
To find out if the enD CRE is directly regulated by Abd-B, Sal or STAT, we searched the region bioinformatically to identify putative DNA-binding sites (Supplementary Fig. 6a).
Using electro mobility shift assays (EMSA), we observed that activated STAT binds to an oligo containing the TTC(4n)GAA STAT-binding site22. Mutation of this sequence prevents activated STAT binding to the oligo (Supplementary Fig. 6b). Similarly, we observed full-length Abd-B binding to oligos containing its putative DNA-binding sites (Supplementary Fig. 6c, lane 6). Binding specificity was confirmed by the super-shift generated when adding anti-Abd-B antibody (lane 7) and by the lack of binding of an Abd-B protein with a mutation affecting Asn51 amino acid in the DNA-binding homeodomain (lanes 8 and 9). We failed to confirm direct Sal protein binding in EMSA experiments using the Sal zinc-finger domain.
We tested the in vivo relevance of the biochemically identified sites on the enD0.4 reporter element. Mutation of the 4n TTCCAGCGAA STAT-binding site in enD0.4 to TTCCAGCGtt in enD0.4StatMut, decreased reporter activation in the posterior spiracle at the early 11–12 stages (Supplementary Fig. 6d, e). However, enD0.4StatMut spiracle expression reappears at stage 14, indicating that this reporter element is also controlled by late spiracle gene network elements independently of STAT. Mutation of both Abd-B putative sites [site 260 CCCATAAAAAT to CCCAcggAAAT and site 359 GGATTTATGGC to GGATccgTGGC] in enD0.4AbdB260-359Mut completely abolished reporter expression (Supplementary Fig. 6f). These results confirm the direct regulation of en in the posterior spiracles by Abd-B and STAT.
Regulation of enD expression by Sal
Sal is crucial for enD expression and previous work showed sal is the earliest gene expressed in a circumferential stigmatophore-specific pattern4 with spatio-temporal dynamics of expression preceding enD reporter expression. Thus, to understand how the gene network controlling stigmatophore organogenesis has been established, it is relevant to know how sal spiracle transcription is controlled.
The sal locus spans over 110kbp between the salr and the sal genes23. To identify a sal CRE potentially active in the posterior spiracles, we searched bioinformatically for regions containing putative binding sites for spiracle cascade regulators. We identified a 1,8kbp fragment (sal2.1) driving reporter expression in the stigmatophore overlapping with Abd-B, Sal and anterior En expression (Fig. 3a–c). Interestingly, a comparison with a hh-lacZP30 enhancer trap line24 shows sal2.1 is expressed exclusively in the anterior A8 compartment (Fig. 3e).
a–c Expression of sal2.1-lacZ in wild type st13 embryos double stained with anti-ßGal (green, arrowheads) and anti-AbdB (a), anti-Sal (b), or anti-En (c) in magenta. d Expression of sal2.1-lacZ in Abd-BM5 mutant embryos lacking the m isoform. e Expression of sal2.1-PH-GFP in hh-lacZ embryos stained with anti-GFP (green) and anti-ßGal (magenta). f Expression of sal2.1-PH-GFP (green) in wild type embryos stained with anti-En (magenta). g, h Ectopic activation with the ectoderm-specific line 69B-Gal4 of the UAS-Abd-Bm (g) or UAS-Abd-Br isoform (h) in sal2.1-PH-GFP embryos stained with anti-GFP (green) and anti-En (magenta). Arrowheads in a–e point at anterior A8 cells, asterisk in panel d indicates hindgut expression. Scale bar: 50 µm.
To identify sal2.1 regulators we analysed its expression in posterior spiracle mutants. The Abd-B gene gives rise to two isoforms with different spatial patterns of expression (Fig. 1b). The Abd-Br isoform is expressed from A8p to A10, while Abd-Bm is expressed from A5-A1025. Thus Abd-Bm is the only isoform expressed in A8a. We compared the regulatory capability of each isoform by ectopically expressing them in the ectoderm with the 69B-Gal4 line. Abd-Bm induces ectopic sal2.1 activation in anterior trunk segments while Abd-Br does not result in strong sal2.1 ectopic activation (Fig. 3f–h) suggesting an isoform-specific regulation. Next, we analysed Abd-BM5 mutant embryos, which lack exclusively the Abd-Bm isoform function25,26, and found sal2.1 expression disappears from anterior A8 (Fig. 3b, d). Mutations in other posterior spiracle genes tested do not affect sal2.1 expression despite having abnormal spiracle development (Supplementary Fig. 5f–i).
Taken together, our results suggest that the novel A8a segment-specific activation of En required the function of at least two Abd-B regulated enhancers: sal2.1 inducing Sal expression in A8a at stage 11 and enD integrating the Abd-B, Sal and JAK/STAT signalling pathway inputs.
Analysis of enD enhancer function
To find out what is enD required for during development, we generated enDΔ, a deletion in the endogenous gene encompassing the enD0.4 element that eliminates En expression from the A8a compartment (Supplementary Fig. 3a, h, i and Methods). Unexpectedly, enDΔ homozygous mutants develop normal posterior spiracles but are male sterile.
Despite enD reporter being active in third instar larval and pupal genital discs (Supplementary Fig. 7a, b), the enDΔ adult male genitalia appear normal (Supplementary Fig. 7c–f). Quantification shows that the posterior lobe area size in enDΔ mutants is intermediate to that of the wild type and the enDΔ/+ males, both of which are fertile, indicating that male sterility is not due to genital defects (Supplementary Fig. 8). To test if the sterility could be due to abnormal spermatogenesis we labelled the sperm tails with the Dj::GFP transgene27. In control males, Dj::GFP labelling is first detected in the elongated sperm tails extending along most of the testis as they individualise (Fig. 4a), and is maintained when the sperms exit into the seminal vesicle (Fig. 4b, c). In enDΔ3; Dj::GFP mutant testis, we only observe GFP labelling at the sperm individualisation stage, with no GFP detectable in the seminal vesicle nor at the end of the testis terminal region which only contains cyst cell remnants (Fig. 4d). The same is observed when labelling the sperm axoneme with anti-Axo49 antibody (Fig. 4e, f). These results indicate that enD may be required when the mature sperms abandon the testis.
Five-day-old adult testis with the sperm tails labelled with Dj::GFP (green, or grey) in control heterozygous (a, c) or homozygous enDΔ males (d). b Schematic drawing of the Drosophila melanogaster wild-type testis terminal region indicating (1) the Head Cyst Cells (HCC, green) tightly holding the sperm heads (yellow); (2) the coiled sperms and (3) the sperms after their release to the seminal vesicle. c Close-up of the squared region in a. In heterozygous males (a, c) spermiation leads to sperm accumulation in the seminal vesicle (asterisk). In homozygous enDΔ males (d) sperms cannot be detected in the seminal vesicle (asterisk) nor in the distal part of the terminal testis region (right of the discontinuous yellow line). e, f Testes with the sperm tails labelled with Axo49 (magenta) in the testis terminal region and seminal vesicle (asterisk) of control heterozygous (e) or enDΔ homozygous (f) males. All testes are counter-stained with DAPI (yellow). In (a, c, d) filamentous Actin is labelled with Rhodamine phalloidin (magenta). Scale bar: 100 µm.
Posterior spiracle gene network expression in the testis
In Drosophila melanogaster, the germ cell niche is located at the testis apex in a structure known as the hub (Fig. 4a). When the germ stem cell divides, it gives rise to a sperm progenitor cell (gonialblast) that separates from the hub and becomes encapsulated by two highly specialised mesodermal cells (the cyst cells). The cyst cells do not proliferate but will protect and signal to the gonialblast as it divides28,29. Inside each cyst, the gonialblast experiences four mitoses and a meiosis generating 64 clonal spermatids that elongate (Fig. 4a). When the spermatids individualise to give rise to the spermatozoa, the two cyst cells differentiate to form a head cyst cell (HCC) that forms an Actin basket holding tightly the 64 sperm heads, and a tail cyst cell (TCC) that elongates to surround the growing sperm tails (Fig. 4b). After the 64 spermatozoa individualise, they coil at the testis terminal region where, during the phase known as spermiation, generate forces that lead to their liberation from the cyst and exit to the seminal vesicle (Fig. 4c)30,31.
Because of the observed late spermatogenesis defects, we analysed enD activity in the adult testis. enD expression shows it is active at the testis terminal region in the head cyst cells (HCC), which also express low levels of En (Fig. 5a–c). We next studied if the same gene network regulates enD in the testis and in the spiracles. Abd-B expression has been previously observed in the embryo’s testis mesodermal somatic cells32. However, in the larval and adult testis, Abd-B is no longer expressed in the mesoderm cells although it is detectable in the germ cells33. We confirmed the lack of Abd-B expression in mesodermal testis cells, which accords with a report showing that ectopic Abd-B induction interferes with testicular development34. In contrast, we found expression of Sal in the HCC nuclei at the testis terminal region (Fig. 5d–f). We also found that the upd-Gal4 enhancer trap line driving the UAS-H2AmChery-P2A-eGFP-PH reporter labels the HCC nuclei and membranes (Supplementary Fig. 9b). Furthermore, the 10xSTAT-GFP reporter, which is expressed in cells activating the JAK/STAT signalling pathway35,36, is also active in the HCC where we can also detect nuclear STAT-GFP expressed from a BAC element (Supplementary Fig. 9c, d). To find out if the enhancers driving the expression of sal and upd in the HCC are the same as those responsible for their activation in the posterior spiracles, we tested the spiracle-specific upd0.43-lacZ reporter and sal2.1-GFP. In both cases, we observed activation in the HCC (Fig. 5d, e). Other spiracle network genes like cv-c and crb are also activated in the HCC and in the case of crb using the same enhancer driving spiracle expression (Supplementary Fig. 9e, f)37 indicating that part of the posterior spiracle gene network has been co-opted to the HCC mesodermal cells.
a–e Expression of enD, sal2.1 and upd0.43 enhancers in the HCC at the testis terminal region. (a) enDds-GFP (green) is expressed in the HCCs holding the 64 sperm head bundles. Sperm tails labelled with Axo49 are shown in magenta, and sperm heads are in yellow with DAPI. (a’) shows only GFP and DAPI stainings and a close-up of an HCC holding the sperm heads. b The HCC labelled in green with enDds-GFP can be distinguished by the formation of an actin basket [labelled with phalloidin (magenta or white)] holding the 64 sperm head bundles (yellow). c enDds-GFP is active in HCC cells which also express low levels of endogenous Engrailed (magenta). d sal2.1-mCherry is active in cells expressing low levels of endogenous Sal protein. e upd0.43-lacZ (magenta) is active in a subset of HCC expressing Sal (green). Wild type enD0.4-mCherry reporter expression in HCCs (f). Reporter expression is maintained in enD0.4 when the putative Abd-B DNA-binding sites are mutated (g), or when the putative STAT sites are mutated (h). In (a, b, d–h) DAPI staining is yellow. Scale bar: 20 µm.
To test the extent to which the enD enhancer regulation is conserved, we analysed the expression of the Abd-B and STAT mutated binding site reporters. As could be expected from the absence of Abd-B expression in the HCC, mutating the Abd-B binding sites does not affect the testis enhancer expression (Fig. 5g). Mutation of the STAT-binding sites does not affect the construct’s expression either (Fig. 5h) , this may be because it still has a secondary input from the gene network as happens in the posterior spiracles (see above).
Finally, we analysed if Sal and En are expressed in the testis of D. virilis and E. balteatus (Fig. 6). We detected equivalent expression of both Sal and En in the testis terminal region of D. virilis (Fig. 6c, d) but not in E. balteatus (Fig. 6e, f), suggesting these genes were co-opted to the adult testes in the Drosophilids after they diverged from Episyrphus ancestors. In fact, Drosophila and Episyrphus testis morphology is very different and even the sperm of Episyrphus balteatus have shorter tails as shown by staining the axoneme with Axo49 antibody (Fig. 6b, d, f), suggesting that, among other factors, the posterior spiracle gene network co-option may have contributed to their morphological divergence.
Testis terminal region of D. melanogaster (a, b), D. virilis (c, d) and whole testis of E. balteatus (e, f) stained with anti-Engrailed and anti-Spalt (a, c, e) or anti-Spalt, and the axoneme marker anti-Axo49 (b, d, f). D. virilis testes are larger but similar in shape to those of D. melanogaster and have a similar expression of En and Sal in the terminal region (a, c) where the sperms coil and individualise (b, d). The testis morphology of E. balteatus is completely different and expresses neither En nor Sal (e). Axoneme staining with anti-Axo49 and DAPI reveals the formation of shorter sperms in E. balteatus that do not coil (f). DAPI DNA staining in yellow. Scale bar: 100 µm.
Discussion
Genetic co-option is an evolutionary source of novelty1,8,38,39,40,41. Here we studied how the expression of the posterior compartment determinant Engrailed in anterior cells, was acquired during the organogenesis of the posterior spiracles. Unexpectedly, we found this novelty is required for spermiation, the process by which the sperms are released from the somatic cyst cells encapsulating them, but is functionally irrelevant for the development of the larval posterior spiracles.
Previous work showed that the ectodermally expressed posterior spiracle gene network had been co-opted to the ectodermal male genitalia in the Drosophila melanogaster clade3. Here, we show the same gene network has also been co-opted to the mesodermal head cyst cells in Drosophila. We found that part of the posterior spiracle gene network becomes activated in the HCCs using the same enhancers driving expression in the posterior spiracles.
Although Abd-B is expressed in the somatic testis cells during embryogenesis, it is not expressed in the adult HCCs. We suggest two alternative explanations. The first possibility is that one of the spiracle primary targets, many of which encode transcription factors, becomes expressed in the testis HCC independently of Abd-B regulation, and this results in the activation of the other spiracle genes due to cross-regulatory network interactions. Alternatively, the embryonic Abd-B expression could be speculated to epigenetically modify the spiracle gene network leaving it in a poised state that later could become activated in the absence of Abd-B.
The finding that the posterior spiracle gene network has been co-opted twice to different organs, suggests that the coordinated activation of several transcription factors and signalling molecules of the network does not necessarily cause detrimental effects. The reported effects of experimentally inducing the ectopic activation of the eye gene regulatory network in D. melanogaster, a situation akin to what may happen during co-option, may help in understanding this. eyeless activation in the imaginal primordia results in the formation of ectopic eyes and causes morphological alterations leading to lethality42. However, it has been noted that ectopic Eyeless expression in the wing only induces eye development in proximal cells where Dpp is also expressed, and similar results were observed with Hh43,44 showing that the activation of a gene network inducer does not cause developmental transformations in all cells where it is expressed. Thus, when the co-option of a gene network causes developmental transformations reducing the animal’s fitness, it will be lost. However, if the co-option had no influence on local developmental processes, it could be tolerated giving the opportunity to the gene network elements to interact with allelic variants present in the population whose interaction could result in selective pressures fixing the trait. Such a series of steps may have led to the co-option of other developmental gene networks such as the activation of the appendage gene network in the butterfly wings that resulted in the formation of novel wing spot patterns2,45.
Expression of Engrailed in posterior metameric stripes is characteristic of arthropods, but is also observed in Onycophorans and in certain worms indicating an ancient origin12,46,47. In flies, anterior En activation has rarely been reported48, and in D. melanogaster, anterior expression is the exception. En activation in A8a associated to the posterior spiracle is present in D. virilis, but not in E. balteatus, suggesting it originated in the higher Diptera (Brachicera). This is supported by analyses showing that in other Diptera like Bactrocera dorsalis (Tephritidae) and Lucilia sericata (Calliphoridae), that present well-developed larval posterior spiracles, engrailed expression is restricted to the posterior A8 segment49,50. We have found that A8a engrailed spiracle expression does not depend on the segmentation gene network, but is regulated by the posterior spiracle network through Abd-B, Sal and the JAK/STAT signalling pathway. Despite its complex regulation, this en enhancer has no function in Drosophila melanogaster’s spiracle organogenesis. Taken together, Engrailed expression in the anterior compartment of A8 was acquired after functional posterior spiracles already existed in dipteran larvae and its origin had to do with testis evolution. Future experiments should establish if this occurred due to the de novo appearance of the enD CRE, or to a silent enD cryptic enhancer becoming activated due to the recruitment of a new transregulatory factor in the spiracles/testis gene network or to changes in chromatin accessibility.
Most insects have a pair of non-protruding spiracle openings in each trunk segment (Fig. 7a branch A). In the hemimetabolous bug Oncopeltus fasciatus nymph or the holometabolous beetle Tribolium castaneum larva these are formed by an internal spiracular chamber that expresses the Ct protein51,52, however, in Oncopeltus, Ct expression is not regulated by Hox proteins as it is in Drosophila, but depends on the tracheal protein Trachealess. Although in Tribolium, Spalt is expressed in the lateral ectoderm, it is not associated with the spiracles. Similarly, Engrailed is restricted to a posterior stripe of cells in the segment that does not surround the spiracular opening53. Our analysis of E. balteatus embryos suggests Sal was recruited in Diptera to the protruding stigmatophore before engrailed was expressed in A8a (Fig. 7a branch B). Analysis of Sal and En expression does not reveal any activation in E. balteatus testis, indicating the spiracle network is not expressed in the gonads of all Diptera. This situation changes in Drosophila species where both Sal and En are expressed in A8a associated with the spiracles as well as in the testes (Fig. 7a branch C). The co-option of the posterior spiracle cascade to the testis and the recruitment of Engrailed expression may have been the result of the major sperm size and gonad morphology changes occurring at some point after the divergence of Episyrphus and Drosophila that could have required the selection of new genetic variants to maintain fertility (Fig. 7b purple band). As the enD enhancer has no apparent function in the posterior spiracles, but is required for male spermiation in D. melanogaster, we propose that originally this CRE was selected for its testicular function and that expression in the posterior spiracles was a side effect caused by the interlocked regulation of the co-opted gene networks in both organs which was not eliminated because it had no deleterious effects (Fig. 7b blue band). Finally, after the divergence from the virilis group, a second co-option event of the spiracle gene network in the male genital disc of D. melanogaster and closely related species led to the evolution of the posterior lobe (Fig. 7a branch D). Given the extreme variability of the posterior lobe sizes between various D. melanogaster genotypes it is unclear whether the enD enhancer has a direct function on lobe development or if its expression there is aphenotypic, and only the result of its interlocking with other functional elements of the co-opted gene network.
a Simplified evolutionary tree representing the consecutive co-option events that sequentially recruited the posterior spiracle gene network to the testis and the male genital posterior lobe in Drosophila melanogaster. Temporal lines not to scale. b Schematic representation of the co-opted posterior spiracle gene network elements (green) to the testis HCC and the posterior recruitment of Engrailed function (red) to the testis that resulted in its simultaneous aphenotypic recruitment to the posterior spiracles caused by the networks’ interlocking. Genes highlighted in grey are not expressed in HCC.
Our results suggest that the selection of a new trait in a co-opted gene network in one organ, results in its activation in all organs where the network is expressed. This implies a slower acquisition of new traits in co-opted gene networks as novelties would become discarded if they had detrimental effects in any of the interlocked organs. On the other hand, if the new trait (for example the activation of a novel transcription factor) was selected for its function in one organ, this would lead to the fixation of non-functional pre-adaptive traits with functional potential in all interlocked organs. Thus, we propose that co-option can lead to sensu stricto pre-adaptation cases, as opposed to the associated term of exaptation. While in exaptation the co-opted character had a previous selective function that has been recruited to perform a novel function (i.e., heat shock proteins recruited to form the eye crystallin, or feathers that could have served for heat regulation or sexual display before being co-opted for flight) cases like the expression of Engrailed in the posterior spiracle provide no selective advantage but could conceivably acquire it in the future, as has happened in the anterior wing of several Diptera where En has acquired a new role in wing pigmentation48.
Methods
Fly stocks
Wild type flies: Drosophila melanogaster Oregon-R and Drosophila virilis were reared using standard Drosophila melanogaster fly food. An Episyrphus balteatus colony was maintained at 22 ± 2 °C in the laboratory with a 12:12 h light:dark photoperiod. Larvae were fed with aphids maintained in infested broad bean plants, while adults were fed with pollen and a 30% honey solution. Eggs were collected every 24 h and dechorionated and fixed using Drosophila melanogaster standard protocols.
The following D. melanogaster mutant stocks were used: Abd-BM1 null mutant for both Abd-B isoforms and Abd-BM5 mutant for the Abd-Bm isoform26, Df(1)os1A deficient for the three Unpaired ligands (upd1, upd2 and upd3)54, Df (2L)32FP-5 deficient for spalt and sal-r genes23 (Barrio et al., 1999), hhAC (BL-1749), wgCX4 (BL-2980) null, Df(2R) enE (BL-2216) deficient for engrailed and invected genes55, Df(3L)H99 (BL-1576) deficient for reaper, grim, and hid56,57,58, Df(2L)drmP2 deficiency for drm, sob and odd59, ctdb760, ems9H8361, grn7j86 and grn7L12 null mutants62.
Reporter lines: hhP30-lacZ (BL-5530)63, enD e702–lacZ, enH sk11a-lacZ, enM slk5-lacZ, enX sn2-lacZ19; Dj::GFP27. 10xSTAT-GFP reporter35, STAT92E-GFP BAC[BAC CH321-73F24 P[acman]64, crb518-lacZ37, cv-c::GFP65.
Gal4 and UAS lines: 69B-Gal466, UAS-upd67; UAS-AbdBm 1,168; UAS-AbdBr22, UAS-AbdBAsn5169, UAS-H2AmChery-P2A-eGFP-PH70.
EMSA assays
We searched bioinformatically for Abd-B, Sal and STAT putative binding sites in enD0.4 using JASPAR (https://jaspar.genereg.net/). To test the physical binding capacity of Abd-B, Sal or STAT to the enD0.4 DNA fragment, the enD0.4 DNA fragment was subdivided into six overlapping fragments. Each fragment consisted of two radioactively labelled antiparallel annealed oligos. Oligo sequences are listed in Supplementary Table 1. Labelling, protein expression and binding reaction as well as gel running and imaging were performed as described37. Once the binding sites in the larger fragments were identified, we confirmed binding using smaller fragments of oligos 2, 4 and 6. Mutations of the putative STAT or Abd-B binding sites in these smaller fragments were used to test direct binding in EMSA. Oligos are listed in Supplementary Table 1. Protein extracts for binding reaction were obtained as described at Pinto et al.37. Briefly, we transiently transfected S2R+ cells (DGRC Stock 150; https://dgrc.bio.indiana.edu/stock/150; RRID:CVCL_Z831) with the following plasmids using Effectene Transfection Reagent (Qiagen # 301425): For STAT expression, pAC-GAL4, pUASt-STAT-GFP, pAC-HopTML; for Abd-B expression, pAC-GAL4 and pUASt-HA-Abd-B; for Abd-B mutant on Asn51, pAC-GAL4 and pUASt-Abd-B mut Asn5169. All cDNAs used encode full-length protein sequence. Transfected cells were incubated at 20 °C, harvested 72 h later and lysed using lysis buffer (10 mM HEPES pH7.0; 10 mM KCl; 5 mM MgCl2; 1% Triton X-100; 50% Glycerol; 1 mM DTT; 10uM Na-O-Ortovanadate; 1 mM PMSF). Crude extract from these cells was added directly to EMSA assays.
CRISPR-Cas9 deletions
Three independent enDΔ deletions removing a 2kb-region [located at 2 R:11516500–11518500 (Flybase release: r6.45)] were induced by Genetivision. The deletions were induced using the sgRNAs enD gRNA1 and enD gRNA2 flanking enD (Supplementary Table 1) after inserting a 3xP3-GFP cassette. Sequencing showed all three deletions remove an identical region, but enDΔ1 had a lethal off-target mutation and was discarded. Both enDΔ2 and enDΔ3 are viable and male sterile either when homozygous or when hemizygous over Df(2)enE which removes engrailed and invected.
Constructs
enD19 was subdivided into shorter fragments called enD-ds, enD-E OL1.2 and enD-0.4 using the primer pairs enD1for and enD3rev; enD2for and enD4rev; enDEOLfor and enDEOLrev1.2 and enDEOLfor and enD4rev respectively (Supplementary Table 1). These cis-regulatory elements were used to create enD-ds-GFP, enD-E OL-GFP, enD0.4-mCherry (Supplementary Fig. 3 and Supplementary Table 3).
Regulatory sal sequences were bioinformatically analysed to isolate regions with increased putative STAT-binding sites. Only sal2.1, amplified by PCR using primers Sal2.1for and Sal2.1rev (Supplementary Table 1), drove posterior spiracle expression when used as reporters (sal2.1-lacZ and sal2.1-GFP).
A 0.43 kb cis-regulatory element of upd, obtained amplifying with the primers upd F2 M for and upd F2 AT rev (Supplementary Table 1) was used to create upd0.43-lacZ which drives expression in the posterior spiracle and testis. All the above fragments were cloned into pGEMt-easy. After sequencing confirmation, fragments were excised with EcoRI and subcloned into pCASPER-NLS-LacZ, pCASPER-PH-eGFP or pCASPER-mCherry. Transgenic flies were obtained by the CBM-SO Drosophila transgenesis service (Spain).
In vitro mutagenesis: Putative binding sites were identified with JASPAR. enD0.4STATmut was made mutating the TTCCAGCGAA putative STAT-binding site in enD0.4 to TTCCAGCGtt known to abolish STAT binding as described at22,37. The mutation was generated using QuikChange protocol (Stratagene) starting from pGEMt-easy:enD0.4 as a template and using the enDdwn Stat-mut for and enDdwn Stat-mut rev primers (Supplementary Table 1).
enD0.4Abd-Bmut was achieved using Megaprimer PCR-based mutagenesis. As template pGEMt-easy:enD0.4 was used in combination with mutagenic primers (Supplementary Table 1). Fragments containing enD0.4WT, enD0.4STATmut or enD0.4Abd-Bmut were checked by sequencing and excised from pGEMt-easy with EcoRI and cloned into pCASPER-PH-eGFP and/or pCASPER-mCherry plasmids to generate transgenic flies. AbdB binding site substitutions were made as described in refs. 22,37 as follows: Site 260 CCCATAAAAAT and site 359 GGATTTATGGC were substituted by mut260 CCCAcggAAAT and mut359 GGATccgTGGC respectively.
In situ hybridisation
Probes were made by PCR amplification using a specific oligo followed by a second PCR reaction that introduced a T7 promoter. The QIAquick purified fragment was labelled using Digoxigenin RNA Labeling Kit (Roche). Embryos were fixed in 4% formaldehyde for 20’−30’ and hybridised at 55 °C overnight with the probe. We used anti-DIG-AP (Roche) to detect the DIG labelled probe following standard RNA in situ procedures.
Immunohistochemistry
Embryos: Drosophila eggs were collected overnight at 25 °C on apple juice agar plates supplemented with fresh yeast paste. Episyrphus eggs were collected from the broad bean plants. All eggs were treated similarly: dechorionated 2 min in commercial bleach diluted in water (1:1), washed thoroughly and fixed for 20 min in a PBS-formaldehyde 4%/heptane mix. After removing the fixative, methanol was added and shaken vigorously a few seconds to remove the vitelline membrane. After allowing both phases to separate, sinking embryos were recovered, washed in clean methanol, rehydrated in PBS-Tween 0.1% (PBT) and preadsorbed for an hour in PBT-1%BSA at room temperature (RT). Primary antibodies were used at the described concentration, diluted in PBT-1%BSA and incubated overnight at 4 °C. Primary antibodies were washed twice for 5 min in PBT and preadsorbed 1 h in PBT-1%BSA at RT. Embryos were incubated with the secondary antibody diluted at 1:400 in PBT-1%BSA at RT for 4 h. After incubation, embryos were washed four times 15 min in PBT at RT followed by to 2 rinses in PBS before mounting in Vectashield.
Testis: Five-day-old males were dissected in PBS and fixed 30 min at RT in 4% paraformaldehyde+0.3% Triton X-100, washed three times with PBS-Triton 0.3% and preadsorbed in PBS-Triton 0.3% + 1%BSA during 1 h. Primary antibodies were incubated overnight at 4 °C, and washed four times in PBS-Triton 0.3%. Testes were preabsorbed in PBS-Triton 0.3% + 1%BSA for 1 h at RT. Secondary antibodies diluted at 1:400 in PBS-Triton 0.3% + 1%BSA were incubated at RT for 4 h. After incubation testis were washed 4 × 15 min in PBS-Triton 0.3% at RT, rinsed twice in PBS and mounted in Vectashield.
Pupal and third larval instar genital discs were dissected in PBS. Fixation and staining protocol were performed as in embryos but with 0.1% Triton X-100.
The following primary antibodies were used (Supplementary Table 2): mouse α-Ct 2B10 1:20 (DSHB), mouse α-Abd-B 1A2E9 1:25 (DSHB), mouse anti-En 4D9 1:50 (DSHB), rabbit α-Sal (our laboratory), rat anti-RFP 1:500 (Chromotek, 5F8), mouse anti-Axo49 1:500 (Sigma-Aldrich, MABS276), mouse anti-βgal 1:1.000 (Promega, Z378A), chicken anti-βgal 1:500 (Abcam ab9361), rabbit anti-βgal 1:500 (MP Biomedicals, 8559762) rabbit anti-GFP 1:300 (Invitrogen, A11122), chicken anti-GFP 1:500 (Abcam, ab13970).
Secondary antibodies were coupled to Alexa488, Alexa555 or Alexa647 (Supplementary Table 2). Filamentous Actin was stained with Rhodamine phalloidin (Molecular Probes, R415). For RNA in situ hybridisation α-DIG-alcaline phosphatase conjugated was used (1:2000) (Roche, 11093274910).
Images were taken on an SPE or a Stellaris Leica confocal microscope making use of the company’s provided software and processed using FIJI ImageJ2 (version 2.9.0/1.54f), Imaris x64 (version 8.0.2) and Adobe Photoshop CS5 (version 12.0 ×64) programs.
Embryo cuticle preparation
Recently hatched first instar larvae were collected and mounted in Hoyer’s medium. The cuticles were kept on a 75 °C plate for two days until clear.
Posterior lobe measurements
The external genitalia of 18–28 young males (1–2 days old) were dissected and mounted in Hoyers medium. Images were taken in a Zeiss Axioplan 2 microscope at 40x magnification. Data were analysed using Microsoft Excel (version 16.16.27) and GraphPad Prism8 (version 8.4.3 (471)) (Supplementary Fig. 8, Supplementary Tables 4 and 5 and source data).
Statistics and reproducibility
Statistical analysis in Supplementary Fig. 8. was performed as described in ref. 7. The outline of each posterior lobe was traced manually using Fiji ImageJ (ImageJ2, version 2.9.0/1.54f) and enclosed with an artificial baseline drawn in line with the lateral plate. The posterior lobe areas of different genotypes measured in pixels were compared using a one-way analysis of variance (ANOVA) with Type II Sums of Squares as recommended for unbalanced groups. Data complied with previous assumptions of normality and homoscedasticity according to Shapiro-Wilks and Levene´s tests. Subsequently, as the ANOVA results indicated significant differences between groups (ANOVA; F3, 82 = 18.335; p-value = 3.407*10−9) a pairwise post hoc Tukey-Kramer test was performed to assess differences between genotypes. Statistical analyses were performed using the R free software environment (R Development Core Team, 2023). In all statistical analyses, significance was set at a 5% risk error (Supplementary Fig. 8 and Supplementary Table 5).
All experiments performed for this manuscript were repeated at least three times. For embryo stainings, we analysed more than 50 specimens of the relevant stages (5 to 16 h of development). For cuticle preparations, more than 50 first-instar larvae were analysed. For third instar imaginal discs imaginal disc and adult testis analysis a minimum of 20 specimens were studied.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data generated in this study are available within the Article and Supplementary Files. There are no data restrictions. Fly stocks can be freely obtained upon request. Source data are provided in this paper.
References
True, J. R. & Carroll, S. B. Gene co-option in physiological and morphological evolution. Annu. Rev. Cell Dev. Biol. 18, 53–80 (2002).
Murugesan, S. N. et al. Butterfly eyespots evolved via cooption of an ancestral gene-regulatory network that also patterns antennae, legs, and wings. Proc. Natl Acad. Sci. USA 119, e2108661119 https://doi.org/10.1073/pnas.2108661119 (2022).
Glassford, W. J. et al. Co-option of an ancestral hox-regulated network underlies a recently evolved morphological novelty. Dev. Cell 34, 520–531 (2015).
Hu, N. & Castelli-Gair, J. Study of the posterior spiracles of Drosophila as a model to understand the genetic and cellular mechanisms controlling morphogenesis. Dev. Biol. 214, 197–210 (1999).
Merabet, S., Hombria, J. C., Hu, N., Pradel, J. & Graba, Y. Hox-controlled reorganisation of intrasegmental patterning cues underlies Drosophila posterior spiracle organogenesis. Development 132, 3093–3102 (2005).
Lovegrove, B. et al. Coordinated control of cell adhesion, polarity, and cytoskeleton underlies Hox-induced organogenesis in Drosophila. Curr. Biol. 16, 2206–2216 (2006).
Frazee, S. R. & Masly, J. P. Multiple sexual selection pressures drive the rapid evolution of complex morphology in a male secondary genital structure. Ecol. Evol. 5, 4437–4450 (2015).
McQueen, E. & Rebeiz, M. On the specificity of gene regulatory networks: How does network co-option affect subsequent evolution? Curr. Top. Dev. Biol. 139, 375–405 (2020).
DiNardo, S., Kuner, J. M., Theis, J. & O’Farrell, P. H. Development of embryonic pattern in D. melanogaster as revealed by accumulation of the nuclear engrailed protein. Cell 43, 59–69 (1985).
Patel, N. H., Kornberg, T. B. & Goodman, C. S. Expression of engrailed during segmentation in grasshopper and crayfish. Development 107, 201–212 (1989).
Damen, W. G. Parasegmental organization of the spider embryo implies that the parasegment is an evolutionary conserved entity in arthropod embryogenesis. Development 129, 1239–1250 (2002).
Prud’homme, B. et al. Arthropod-like expression patterns of engrailed and wingless in the annelid Platynereis dumerilii suggest a role in segment formation. Curr. Biol. 13, 1876–1881 (2003).
Akam, M. The molecular basis for metameric pattern in the Drosophila embryo. Development 101, 1–22 (1987).
Ingham, P. W. Segment polarity genes and cell patterning within the Drosophila body segment. Curr. Opin. Genet. Dev. 1, 261–267 (1991).
Sanson, B. Generating patterns from fields of cells. Examples from Drosophila segmentation. EMBO Rep. 2, 1083–1088 (2001).
Monier, B., Pelissier-Monier, A., Brand, A. H. & Sanson, B. An actomyosin-based barrier inhibits cell mixing at compartmental boundaries in Drosophila embryos. Nat. Cell Biol. 12, 60–69 (2010).
Blair, S. S. Engrailed expression in the anterior lineage compartment of the developing wing blade of Drosophila. Development 115, 21–33 (1992).
Wiegmann, B. M. et al. Episodic radiations in the fly tree of life. Proc. Natl Acad. Sci. USA 108, 5690–5695 (2011).
Cheng, Y. et al. Co-regulation of invected and engrailed by a complex array of regulatory sequences in Drosophila. Dev. Biol. 395, 131–143 (2014).
Hombria, J. C.-G., Sanchez-Higueras, C. & Sanchez-Herrero, E. In Organogenetic Gene Networks (eds. Castelli-Gair Hombría, J. & Bovolenta, P.) Ch. 12, 319–373 (2016).
Castelli Gair Hombria, J., Rivas, M. L. & Sotillos, S. Genetic control of morphogenesis—Hox induced organogenesis of the posterior spiracles. Int. J. Dev. Biol. 53, 1349–1358 (2009).
Rivas, M. L., Cobreros, L., Zeidler, M. P. & Hombria, J. C. Plasticity of Drosophila Stat DNA binding shows an evolutionary basis for Stat transcription factor preferences. EMBO Rep. 9, 1114–1120 (2008).
Barrio, R., de Celis, J. F., Bolshakov, S. & Kafatos, F. C. Identification of regulatory regions driving the expression of the Drosophila spalt complex at different developmental stages. Dev. Biol. 215, 33–47 (1999).
Lee, J. J., von Kessler, D. P., Parks, S. & Beachy, P. A. Secretion and localized transcription suggest a role in positional signaling for products of the segmentation gene hedgehog. Cell 71, 33–50 (1992).
Delorenzi, M. & Bienz, M. Expression of Abdominal-B homeoproteins in Drosophila embryos. Development 108, 323–329 (1990).
Casanova, J., Sanchez-Herrero, E. & Morata, G. Identification and characterization of a parasegment specific regulatory element of the abdominal-B gene of Drosophila. Cell 47, 627–636 (1986).
Santel, A., Winhauer, T., Blumer, N. & Renkawitz-Pohl, R. The Drosophila don juan (dj) gene encodes a novel sperm specific protein component characterized by an unusual domain of a repetitive amino acid motif. Mech. Dev. 64, 19–30 (1997).
Fuller, M. T. Spermatogenesis. In The Development of Drosophila melanogaster. (eds. Bate & Martinez Arias) 1, 71–147 (Cold Spring Harbour Laboratory Press, 1993).
Leatherman, J. L. & Dinardo, S. Germline self-renewal requires cyst stem cells and stat regulates niche adhesion in Drosophila testes. Nat. Cell Biol. 12, 806–811 (2010).
Dubey, P., Shirolikar, S. & Ray, K. Localized, reactive F-actin dynamics prevents abnormal somatic cell penetration by mature spermatids. Dev. Cell 38, 507–521 (2016).
Desai, B. S., Shirolikar, S. & Ray, K. F-actin-based extensions of the head cyst cell adhere to the maturing spermatids to maintain them in a tight bundle and prevent their premature release in Drosophila testis. BMC Biol. 7, 19 (2009).
DeFalco, T., Le Bras, S. & Van Doren, M. Abdominal-B is essential for proper sexually dimorphic development of the Drosophila gonad. Mech. Dev. 121, 1323–1333 (2004).
Papagiannouli, F., Schardt, L., Grajcarek, J., Ha, N. & Lohmann, I. The Hox gene Abd-B controls stem cell niche function in the Drosophila testis. Dev. Cell 28, 189–202 (2014).
Zhang, S. et al. Repression of Abd-B by Polycomb is critical for cell identity maintenance in adult Drosophila testis. Sci. Rep. 7, 5101 (2017).
Bach, E. A. et al. GFP reporters detect the activation of the Drosophila JAK/STAT pathway in vivo. Gene Expr. Patterns 7, 323–331 (2007).
Hombria, J. C. & Sotillos, S. JAK-STAT pathway in Drosophila morphogenesis: From organ selector to cell behavior regulator. JAKSTAT 2, e26089 (2013).
Pinto, P. B., Espinosa-Vazquez, J. M., Rivas, M. L. & Hombria, J. C. JAK/STAT and hox dynamic interactions in an organogenetic gene cascade. PLoS Genet. 11, e1005412 (2015).
Ganfornina, M. D. & Sanchez, D. Generation of evolutionary novelty by functional shift. Bioessays 21, 432–439 (1999).
Hu, Y. & Moczek, A. P. Wing serial homologues and the diversification of insect outgrowths: insights from the pupae of scarab beetles. Proc. Biol. Sci. 288, 20202828 (2021).
DiFrisco, J., Wagner, G. P. & Love, A. C. Reframing research on evolutionary novelty and co-option: Character identity mechanisms versus deep homology. Semin. Cell Dev. Biol. 145:3-12 https://doi.org/10.1016/j.semcdb.2022.03.030 (2022).
Almudí, I. & Pascual-Anaya, J. in Old Questions and Young Approaches to Animal Evolution (eds. Martín-Durán JM & Vellutini BC) Ch. 6, 107–132 (Springer Nature, 2019).
Halder, G., Callaerts, P. & Gehring, W. J. Induction of ectopic eyes by targeted expression of the eyeless gene in Drosophila. Science 267, 1788–1792 (1995).
Chen, R., Halder, G., Zhang, Z. & Mardon, G. Signaling by the TGF-beta homolog decapentaplegic functions reiteratively within the network of genes controlling retinal cell fate determination in Drosophila. Development 126, 935–943 (1999).
Kango-Singh, M., Singh, A. & Henry Sun, Y. Eyeless collaborates with Hedgehog and Decapentaplegic signaling in Drosophila eye induction. Dev. Biol. 256, 49–60 (2003).
Brunetti, C. R. et al. The generation and diversification of butterfly eyespot color patterns. Curr. Biol. 11, 1578–1585 (2001).
Ahzhanov, A. & Kaufman, T. C. Evolution of distinct expression patterns for engrailed paralogues in higher crustaceans (Malacostraca). Dev. Genes Evol. 210, 493–506 (2000).
Franke, F. A. & Mayer, G. Controversies surrounding segments and parasegments in onychophora: insights from the expression patterns of four “segment polarity genes” in the peripatopsid Euperipatoides rowelli. PLoS ONE 9, e114383 (2014).
Dufour, H. D., Koshikawa, S. & Finet, C. Temporal flexibility of gene regulatory network underlies a novel wing pattern in flies. Proc. Natl Acad. Sci. USA 117, 11589–11596 (2020).
Mellenthin, K. et al. Wingless signaling in a large insect, the blowfly Lucilia sericata: a beautiful example of evolutionary developmental biology. Dev. Dyn. 235, 347–360 (2006).
Suksuwan, W., Cai, X., Ngernsiri, L. & Baumgartner, S. Segmentation gene expression patterns in Bactrocera dorsalis and related insects: regulation and shape of blastoderm and larval cuticle. Int. J. Dev. Biol. 61, 439–450 (2017).
Hanna, L. & Popadic, A. A hemipteran insect reveals new genetic mechanisms and evolutionary insights into tracheal system development. Proc. Natl Acad. Sci. USA 117, 4252–4261 (2020).
de Miguel, C., Linsler, F., Casanova, J. & Franch-Marro, X. Genetic basis for the evolution of organ morphogenesis: the case of spalt and cut in the development of insect trachea. Development 143, 3615–3622 (2016).
Lim, J. & Choe, C. P. Functional analysis of engrailed in Tribolium segmentation. Mech. Dev. 161, 103594 (2020).
Hombria, J. C., Brown, S., Hader, S. & Zeidler, M. P. Characterisation of Upd2, a Drosophila JAK/STAT pathway ligand. Dev. Biol. 288, 420–433 (2005).
Tabata, T., Schwartz, C., Gustavson, E., Ali, Z. & Kornberg, T. B. Creating a Drosophila wing de novo, the role of engrailed, and the compartment border hypothesis. Development 121, 3359–3369 (1995).
Chen, P., Lee, P., Otto, L. & Abrams, J. Apoptotic activity of REAPER is distinct from signaling by the tumor necrosis factor receptor 1 death domain. J. Biol. Chem. 271, 25735–25737 (1996).
Grether, M. E., Abrams, J. M., Agapite, J., White, K. & Steller, H. The head involution defective gene of Drosophila melanogaster functions in programmed cell death. Genes Dev. 9, 1694–1708 (1995).
White, K. et al. Genetic control of programmed cell death in Drosophila. Science 264, 677–683 (1994).
Green, R. B., Hatini, V., Johansen, K. A., Liu, X. J. & Lengyel, J. A. Drumstick is a zinc finger protein that antagonizes Lines to control patterning and morphogenesis of the Drosophila hindgut. Development 129, 3645–3656 (2002).
Blochlinger, K., Bodmer, R., Jack, J., Jan, L. Y. & Jan, Y. N. Primary structure and expression of a product from cut, a locus involved in specifying sensory organ identity in Drosophila. Nature 333, 629–635 (1988).
Dalton, D., Chadwick, R. & McGinnis, W. Expression and embryonic function of empty spiracles: a Drosophila homeo box gene with two patterning functions on the anterior-posterior axis of the embryo. Genes Dev. 3, 1940–1956 (1989).
Brown, S. & Castelli-Gair Hombria, J. Drosophila grain encodes a GATA transcription factor required for cell rearrangement during morphogenesis. Development 127, 4867–4876 (2000).
Tabata, T. & Kornberg, T. B. Hedgehog is a signaling protein with a key role in patterning Drosophila imaginal discs. Cell 76, 89–102 (1994).
Sotillos, S., Krahn, M., Espinosa-Vazquez, J. M. & Hombria, J. C. Src kinases mediate the interaction of the apical determinant Bazooka/PAR3 with STAT92E and increase signalling efficiency in Drosophila ectodermal cells. Development 140, 1507–1516 (2013).
Sotillos, S., Aguilar-Aragon, M. & Hombria, J. C. Functional analysis of the Drosophila RhoGAP Cv-c protein and its equivalence to the human DLC3 and DLC1 proteins. Sci. Rep. 8, 4601 (2018).
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993).
Harrison, D. A., McCoon, P. E., Binari, R., Gilman, M. & Perrimon, N. Drosophila unpaired encodes a secreted protein that activates the JAK signaling pathway. Genes Dev. 12, 3252–3263 (1998).
Castelli-Gair, J., Greig, S., Micklem, G. & Akam, M. Dissecting the temporal requirements for homeotic gene function. Development 120, 1983–1995 (1994).
Sambrani, N. et al. Distinct molecular strategies for Hox-mediated limb suppression in Drosophila: from cooperativity to dispensability/antagonism in TALE partnership. PLoS Genet. 9, e1003307 (2013).
Sanchez-Higueras, C. & Hombria, J. C. Precise long-range migration results from short-range stepwise migration during ring gland organogenesis. Dev. Biol. 414, 45–57 (2016).
Acknowledgements
We thank Dr. Steve Brown and Dr. Maria Luisa Rivas for helping with the isolation of the upd0.43 cis-regulatory element, Dr. Fernando Casares and his laboratory members for help with Episyrphus cultures and Dr. Judith Kassis for stocks. This work was supported by Ministerio de Ciencia e Innovación grants BFU2016-76528 and PID2019-104656GB-I00 co-funded by the European Regional Development Fund (FEDER) to J.C.-G.H. and by the Spanish Ministry of Science and Innovation (PID2020-116041GB-I00) and Leonardo Grants for Researchers and Cultural Creators 2021 from BBVA Foundation (IN[21]_CMA_BIO_0056) to I. A. The CABD is supported by a María de Maeztu Unit excellence grant CEX-2020-001088-M.
Author information
Authors and Affiliations
Contributions
S.M.-G. performed experimental studies in the embryo, S.S. performed experimental studies in the adult testis, J.M.E.-V. performed EMSA and in vitro mutagenesis, I.A. introduced the Episyrphus model. J.C.-G.H. coordinated, supervised the study and wrote the manuscript in collaboration with all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Leslie Pick and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Molina-Gil, S., Sotillos, S., Espinosa-Vázquez, J.M. et al. Interlocking of co-opted developmental gene networks in Drosophila and the evolution of pre-adaptive novelty. Nat Commun 14, 5730 (2023). https://doi.org/10.1038/s41467-023-41414-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-023-41414-3
- Springer Nature Limited