Abstract
Parkinson’s disease (PD) is a heterogeneous movement disorder with different motor subtypes including tremor dominant (TD), indeterminate and postural instability, and gait disturbance (PIGD) motor subtypes. Plasma glial fibrillary acidic protein (GFAP) was elevated in PD patients and may be regarded as a biomarker for motor and cognitive progression. Here we explore if there was an association between plasma GFAP and different motor subtypes and whether baseline plasma GFAP level can predict motor subtype conversion. Patients with PD classified as TD, PIGD or indeterminate subtypes underwent neurological evaluation at baseline and 2 years follow-up. Plasma GFAP in PD patients and controls were measured using an ultrasensitive single molecule array. The study enrolled 184 PD patients and 95 control subjects. Plasma GFAP levels were significantly higher in the PIGD group compared to the TD group at 2-year follow-up. Finally, 45% of TD patients at baseline had a subtype shift and 85% of PIGD patients at baseline remained as PIGD subtypes at 2 years follow-up. Baseline plasma GFAP levels were significantly higher in TD patients converted to PIGD than non-converters in the baseline TD group. Higher baseline plasma GFAP levels were significantly associated with the TD motor subtype conversion (OR = 1.283, P = 0.033) and lower baseline plasma GFAP levels in PIGD patients were likely to shift to TD and indeterminate subtype (OR = 0.551, P = 0.021) after adjusting for confounders. Plasma GFAP may serve as a clinical utility biomarker in differentiating motor subtypes and predicting baseline motor subtypes conversion in PD patients.
Similar content being viewed by others
Introduction
Astrocytes constitute a crucial component of the central nervous system. These cells play axon homeostasis and synaptic function1. Glial fibrillary acidic protein (GFAP) is considered as a marker of astroglia activation2. When the central nervous system is damaged, astrocytes release GFAP into the peripheral blood1. Research has demonstrated that astroglia activation contributes to the development of Parkinson’s disease (PD)3,4,5. Several studies indicate that plasma GFAP levels were higher in PD patients compared to healthy controls (HCs)6 and were associated with cognitive impairment in PD patients7,8. Our previous study has suggested that plasma GFAP could serve as a biomarker for monitoring and predicting motor symptoms progression and cognitive function deterioration in PD9. Higher baseline plasma GFAP predicted a more rapid progression to postural instability evaluated with H&Y stage9. However, the association between plasma GFAP and motor subtypes, and whether plasma GFAP could predict the conversion of motor subtypes in PD were not explored in previous study9.
PD is a clinically heterogeneous disease with patients exhibiting a diverse array of motor and non-motor symptoms10,11. Based on clinical symptoms and disease progression, various clinical classification subtypes exist. Among these, tremor dominant (TD), postural instability and gait disturbance (PIGD), and indeterminate subtypes are the most common subtypes in clinical settings and research12,13. PIGD subtypes usually had more severe cognitive impairment, faster disease progression, and poor quality of life14,15,16. TD subtypes respond effectively to dopaminergic drugs and exhibit slower disease progression17. Motor subtypes are unstable and can transform into one another as the disease progresses18,19. Research indicates that approximately 50% of PD patients experience motor subtype conversion within the first two years18, with most TD subtypes converting to PIGD and indeterminate subtypes, while PIGD subtypes show limited conversion to other subtypes19.
AD co-pathology is frequently found in postmortem examinations of individuals with PD, particularly those who develop dementia and dementia with Lewy bodies (DLB)20. A study indicated that the PIGD motor subtype had higher cortical Lewy bodies and a greater burden of cortical amyloid-β plaque compared to the TD motor subtype in PD21. PIGD motor subtype was associated with AD co-pathology in DLB22. Autopsy indicated that plasma GFAP may serve as a sensitive biomarker for concomitant AD pathology in Lewy body spectrum disorders23.
At present, the diagnosis of motor subtypes is based mainly on clinical evaluation, and reliable biomarkers to differentiate motor subtypes are lacking, especially the biomarker of predicting motor subtype conversion. Whether plasma GFAP can be used as a potential biomarker to distinguish motor subtypes and predict motor subtype conversion remains unknown. Thus, we measured plasma GFAP in a longitudinal Chinese PD patients cohort including TD, PIGD, and indeterminate motor subtypes and explored the difference between different subtypes at baseline and over time. We also investigated the relationship between baseline levels of GFAP and motor subtype conversion. Furthermore, we compared levels of plasma GFAP with other blood markers, that is, neurofilament light chain (NfL), p-tau181, Aβ42, and Aβ40.
Results
Clinical and demographic profiles
In the study, 184 PD patients and 95 HCs were included. At baseline 184 patients were enrolled, and the enrolled 184 patients had clinical and plasma data at the 1-year follow-up. The enrolled 175 patients (one patient died and eight patients were lost to follow-up) had clinical data and the enrolled 125 patients had plasma data at the 2-year follow-up. The age of the PD patients was 57.78 ± 11.17 years old with an age of onset of 55.81 ± 11.55 years, a disease duration of 1.89 (1.75) years, and a H-Y stage of 2 (0). In the control group (n = 95), 51.6% were male, with a mean age of 55.07 ± 7.31 years. Among the total patients, 62 (33.69%) were classified into the TD subtype, 84 (45.65%) with the PIGD subtype, and 38 (20.65%) had an indeterminate subtype. As shown in Table 1, there were no significant differences in the age, sex, age of onset, disease duration, LEDD, UPDRS-I, UPDRS-II, UPDRS-III, MoCA scores, and PDQ39 scores among the different motor subtypes at baseline. H-Y stage was significantly higher in the PIGD group compared to the TD group (2 (0) vs. 2 (0), Z = 2.723, P = 0.006). MoCA scores were lower in the PIGD group than in the TD group, but no statistical difference at the 2-year follow-up (25.00 (5) vs. 26.00 (4), t = 1.596, P = 0.113). UPDRS-III scores were significantly higher in the PIGD group compared to the TD group at 2-year follow-up (32.83 ± 10.15 vs. 28.34 ± 8.54, t = 2.562, P = 0.011) (Supplementary Table 1).
Plasma GFAP between patients with different motor subtypes of PD
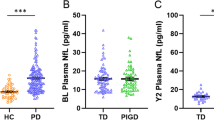
We found baseline plasma GFAP levels were significantly elevated in PD than that of HCs (69.80 ± 36.18 pg/mL vs. 57.89 ± 23.54 pg/mL, P = 0.016) (Fig. 1A). However, baseline plasma GFAP levels were not significantly different between TD and PIGD motor subgroups (68.14 (45.92) pg/mL vs. 69.83 ± 38.31 pg/mL, Z = 0.471, P = 0.638) (Fig. 1B). Interestingly, we found that plasma GFAP levels of the PIGD subgroup were significantly higher than that of the TD subgroup at the two-year follow-up time point (86.01 ± 47.10 pg/mL vs. 68.57 ± 27.69 pg/mL, t = −2.035, P = 0.045) (Fig. 1C). Similarly, baseline plasma NFL levels were no difference between TD and PIGD subtypes, but plasma NFL levels of the PIGD subgroup were significantly higher than that of the TD subgroup at a two-year follow-up time point (14.26 ± 10.00 pg/mL vs 10.37 ± 3.93 pg/mL, t = −2.223, P = 0.028). There were no significant differences in the plasma p-tau181, Aβ42, and Aβ40 between TD and PIGD subtypes at baseline and two-year follow-up time points (Supplementary Table 2).
Plasma GFAP in PD patients and HCs were compared at baseline (A). Plasma GFAP in TD and PIGD groups were compared at baseline (B) and two-year follow-up (C). T-test and Wilcoxon test were used for group comparison. Errors bars represent mean ± standard deviation (*P < 0.05). GFAP Plasma Glial fibrillary acidic protein, PD Parkinson’s disease, TD tremor dominant, PIGD postural instability and gait disturbance.
Correlations between plasma GFAP and clinical characteristics at baseline
In all PD patients, plasma GFAP was significantly associated with age (r = 0.628, P < 0.001, duration (r = −0.189, P = 0.010), and MoCA (r = −0.151, P = 0.040). There were no associations between plasma GFAP and UPDRS-III (r = 0.095, P = 0.199), H-Y (r = 0.083, P = 0.265), and LEDD (r = 0.002, P = 0.975). In the PIGD group, plasma GFAP was correlated with age (r = 0.630, P < 0.001) and duration (r = −0.249, P = 0.023). In the TD group, only age was associated with plasma GFAP (r = 0.640, P < 0.001). Plasma GFAP levels were all associated positively with plasma NFL, P-tau181, Aβ42, and Aβ40 at baseline and 2-year follow-up (Supplementary Table 3). Plasma GFAP and NfL levels were positively associated with PIGD scores at 2-year follow-up (Supplementary Table 4).
The conversion of different motor subtypes at two-year follow-up
Among 184 PD patients, 184 (100%) and 175 (95.1%) patients were assessed at 1-year follow-up and 2-year follow-up, respectively. Therefore, 175 patients (41 TD, 30 indeterminate, and 104 PIGD) with available follow-up data of 2 years were included in subsequent analysis to evaluate the prediction capacity of plasma GFAP levels. The shift of different motor subtypes is shown in Fig. 2. Longitudinally, 30% of baseline TD patients converted to PIGD subtypes, and 15% of them changed to indeterminate subtypes at 2 years follow-up. Finally, 85% of PIGD patients were still categorized as PIGD subtypes at 2 years follow-up, only 4% baseline PIGD patients converted to TD, with 11% shifted to indeterminate subtype. Moreover, 14% of indeterminate patients changed to TD, and 53% changed to PIGD at 2 years follow-up.
Different motor subtypes conversion at 1 year follow-up and 2 years follow-up. TD, indeterminate, and PIGD subtypes were represented by green, blue, and red, respectively. The number of patient subtype conversions was shown in the row. TD tremor dominant, PIGD postural instability and gait disturbance.
Prediction of motor subtype conversion using baseline plasma GFAP
Baseline plasma GFAP levels were significantly higher in TD patients converted to PIGD than non-converters in the baseline TD group (91.67 ± 47.39 pg/mL vs. 65.40 ± 25.98 pg/mL, t = 2.615, P = 0.012) (Fig. 3). ROC analysis indicated that plasma GFAP predicted the motor subtype transformation with an AUC of 0.6656, 95% CI: 0.4985–0.8328, sensitivity = 52.63%, specificity = 85.29%). Similarly, we also found that baseline plasma GFAP levels were significantly elevated in indeterminate patients converted to PIGD compared with non-converters in the baseline indeterminate group (78.57(56.67) pg/mL vs. 51.99 ± 16.14 pg/mL, Z = 1.784, P = 0.074). Baseline plasma GFAP levels were lower in PIGD patients converted to TD and indeterminate subtypes compared to that of non-converters in the baseline PIGD group (48.07 ± 14.11 pg/mL vs. 74.07 ± 40.49 pg/mL, t = 2.191, P = 0.032). ROC analysis indicated that plasma GFAP predicted the PIGD subtype transformation with an AUC of 0.7376, 95% CI: 0.6092–0.8659, sensitivity=71.64%, specificity=66.67%). Plasma GFAP levels were significantly higher in TD convert to PIGD group than TD stable group at 2-year follow-up (109.42 ± 59.29 pg/mL vs.70.48 ± 28.72 pg/mL, t = 2.811 P = 0.008). There were no differences in plasma GFAP levels between non-converters and converters in the Ind group at 2-year follow-up (51.49 ± 12.56 vs 70.44 ± 25.66, t = 1.846 P = 0.078). Plasma GFAP levels were higher in PIGD stable group than PIGD convert to TD group at 2-year follow-up (71.27 (56.84) vs 45.47 ± 11.97, Z = 2.846 P = 0.004) (Supplementary Fig. 1). No significant differences were observed at the baseline plasma NFL, p-tau181, Aβ42, and Aβ40 levels among different motor subtype conversions (Supplementary Table 5).
Baseline plasma GFAP was compared in non-converters and converters at baseline TD group (A), Ind group (B), and PIGD group (C). T-test and Wilcoxon test were used for group comparison. Errors bars represent mean ± standard deviation (*P < 0.05). GFAP Plasma Glial fibrillary acidic protein, TD tremor dominant, Ind indeterminate, PIGD postural instability and gait disturbance.
The logistic regression analyses between baseline plasma GFAP levels and motor subtype conversion are summarized in Table 2. Univariate logistic regression analysis with TD motor subtype conversion as the dependent variable showed a significant association with baseline plasma GFAP (OR = 1.251, P = 0.027). PIGD motor subtype conversion was correlated significantly with baseline plasma GFAP (OR = 0.614, P = 0.017). To better explore the independent association between baseline plasma GFAP and motor subtype conversion, a multivariate logistic regression model was applied with non-converters in baseline TD and PIGD groups as the reference. Overall, a higher baseline GFAP was significantly associated with the TD motor subtype conversion (OR = 1.283, P = 0.033) after adjusting for age and UPDRS-III. With every 10 units increasing baseline plasma GFAP levels, TD patients were 28.3% more likely to convert to the PIGD subtype (Fig. 4A). Similarly, a lower baseline GFAP was related to the PIGD motor subtype shift (OR = 0.551, P = 0.021) after adjusting for age, H-Y, and UPDRS-III. Every 10 units decreasing baseline plasma GFAP levels, PIGD patients were 45.9% more likely to convert to the TD and indeterminate subtypes. (Fig. 4B).
Relationship between baseline plasma GFAP and the probability of TD (A) and PIGD (B) motor subtypes after correction for confounders. Predicted probabilities for stable subtypes were represented with a red line. Predicted probabilities of motor subtype converters were shown in the blue line. GFAP Plasma Glial fibrillary acidic protein, TD tremor dominant, PIGD postural instability and gait disturbance.
Plasma GFAP, cognitive and motor data in PD patients over different time points
Longitudinal data of plasma GFAP, cognitive, and motor scores are summarized in Table 3. In the total PD patients, plasma GFAP did not elevate significantly over time (P = 0.560). Similarly, no significant difference in plasma GFAP was observed in the TD group (P = 0.901) and indeterminate group (P = 0.057) over time. However, plasma GFAP levels were significantly increased over time in the PIGD group (P = 0.038).
Discussion
In the study, we showed that plasma GFAP levels were higher in PIGD subtypes compared to TD subtypes over two years. Additionally, we demonstrated that plasma GFAP levels were significantly elevated over time in the PIGD subtypes, but not in TD subtypes. Moreover, our study suggested that higher baseline plasma GFAP levels in the TD group can predict the conversion of TD motor subtypes to PIGD subtypes, and lower baseline plasma GFAP levels in the PIGD group can predict the conversion to TD and indeterminate subtypes within a relatively short follow-up period of two years.
GFAP serves as a biomarker of reactive astrogliosis, exhibiting elevated levels in plasma and cerebrospinal fluid (CSF) in neurodegenerative diseases such as Alzheimer’s disease (AD)24,25, dementia with Lewy bodies26, frontotemporal lobar degeneration27, and PD28. Plasma GFAP levels not only increased in AD but also raised in preclinical AD compared to cognitively unimpaired older adults24. A recent study suggests that the magnitude change of plasma GFAP levels was higher than that in CSF in AD patients29. These results indicate that plasma GFAP could serve as a convenient, sensitive biomarker for detecting reactive astrogliosis. Therefore, dynamic changes in plasma GFAP of PD patients were measured during a two-year follow-up period. Our results showed that plasma GFAP levels of PD patients were higher than those of controls, which was supported by some previous studies7,8.
No significant differences in the plasma GFAP levels were observed between PIGD and TD subtypes at baseline, which may be due to both PD subtype patients being in the early disease stage with a median disease duration of 1.89 (1.75) years. The lack of difference in plasma GFPA levels between TD and PIGD at baseline may depend on the fact that each subgroup also includes future converters. Reactive astrogliosis has been observed in the substantia nigra pars compacta of both PD patients and PD animal models, leading to dysfunction in dopaminergic neurons30,31. Studies suggested that the akinetic-rigid dominant subtype or PIGD motor subtype experienced more impairment of DA neurons than the TD motor subtype13,21. Our study reveals significantly elevated plasma GFAP levels in the PIGD subgroup compared to the TD group at the two-year follow-up, indicating a more intense pathophysiology in PIGD patients.
Previous studies have shown that the PIGD subtype not only exhibited greater cognitive progression, worse hallucinations, and poorer fatigue14,15,32 but also increased risk of falls and gait freezing compared to the TD subtype32,33. With a poorer prognosis and more rapid disease progression14, the PIGD motor subtype could be a reliable predictor of disease progression. Therefore, reliable biomarkers for diagnosing motor subtypes are essential. Our study highlights the importance of plasma GFAP in the PD motor subtype.
In addition to plasma GFAP, other plasma biomarkers were investigated to differentiate motor phenotype in our study. The results suggest that patients with the PIGD subtype exhibited higher plasma NFL levels compared to that of patients with the TD subtype at the two-year follow-up time point, however, no difference at baseline, which was consistent with a previous study34. Our results showed that plasma GFAP levels were significantly associated with plasma NFL levels. Plasma NfL is a reliable biomarker for neuronal injury and axonal degeneration and it was associated with the disease severity and progression of PD35 and other neurodegenerative disorders36. Plasma GFAP showed a significant correlation with plasma NfL in AD29 and FTD37. Our results indicated that axonal damage and astroglia activation were coexistent in PD, with even greater intensity in the PIGD motor subtype.
In the current study, we found plasma p-tau181, Aβ42, and Aβ40 did not differ between TD and PIGD subgroups at baseline and the two-year follow-up time point. AD co-pathology is frequently found in postmortem examinations of individuals with PD38, particularly those who develop dementia and DLB20. An autopsy indicated that plasma GFAP may serve as a sensitive biomarker for AD co-pathology in Lewy body spectrum disorders, especially the accumulation of β-amyloid plaques23. Our results showed a correlation between plasma GFAP levels and plasma P-tau181, Aβ42, and Aβ40 in PD, indicating an association between plasma GFAP with AD co-pathology in PD.
Research showed that the PIGD motor subtype had higher mean pathological grading of cortical Lewy bodies and more cortical amyloid-β plaque load than the TD motor subtype in PD21. A study showed that PIGD scores were related to the CSF t-tau/Aβ1-42 ratio, indicating AD co-pathology may contribute to PIGD motor signs in LBD22. Plasma GFAP levels were positively associated with PIGD scores at 2-year follow-up in our study. Plasma GFAP is a marker of astrogliosis, elevation tends to be highest in AD Simone Baiardi39 and may serve as a sensitive biomarker for concomitant AD pathology in LBD23. Therefore, we speculated that AD co-pathology may contribute to PIGD motor symptoms in PD. Besides, AD co-pathology may also contribute to worse cognition and faster disease progression in the PIGD motor subtype in PD. Reliable biomarkers for diagnosing motor subtypes are essential. Our study highlights the importance of plasma GFAP and AD co-pathology in the PD motor subtype.
In addition to plasma Aβ42 and Aβ40, the Aβ42/Aβ40 ratio was typically used in AD, and the ratio was associated with neocortical amyloid burden40. A study showed that APOE-ε4 status was associated with the plasma Aβ42/Aβ40 ratio and cognitive decline in PD41. However, there was no difference in the Aβ42/Aβ40 ratio between the TD group and the PIGD group at baseline and 2-year follow-up. Astrocyte reactivity is a crucial initial event linking Aβ and tau pathology in preclinical AD42. We speculated that astrocyte reactivity may occur before Aβ and tau pathology in early PD, leading to no difference in the Aβ42/Aβ40 ratio between the TD group and the PIGD group.
One previous study revealed lower plasma Aβ42 levels in the PIGD group than that in the TD group43. The inconsistent results may be due to the disease duration being approximately 4 years in the previous study43. A study from the Parkinson’s Progression Markers Initiative (PPMI) cohort found that α-synuclein, Aβ1-42 and p-tau181 levels in CSF were lower in patients with PIGD subtypes than in patients with TD subtypes44. The BioFIND study on patients with moderate-advanced PD found that CSF α-synuclein levels were lower in patients with the PIGD subtype than in patients with other motor subtypes45. Although these CSF proteins could serve as biomarkers to distinguish different motor subtypes, lumbar puncture is an invasive examination and clinical application is limited. Therefore, reliable and non-invasive tests may be crucial for diagnosing and predicting motor subtypes.
Motor subtypes of PD can be unstable and may shift as the disease progresses. One previous study showed that one-third of the TD subtype at baseline remained the TD subtype during six years of follow-up and most of the patients with TD subtype at baseline converted into other motor subtypes including 15% indeterminate and 50% PIGD subtypes19. Our results showed that about one-half of PD patients had a motor subtype conversion at 2 years follow-up, which was also consistent with a previous study46. Several studies explored the factors influencing the conversion of baseline TD subtypes18,47,48. Research from the PPMI cohort revealed that patients who transitioned from the TD subtype to the PIGD subtype during four years of follow-up exhibited higher UPDRS-II scores, more severe autonomic dysfunction, and a higher proportion of levodopa-induced dyskinesia than non-convertors48. A study conducted by Garcıa et al. suggested that motor subtype converters had worse global non-motor symptoms measured by the NMSS, poorer cognitive function, and lower serum levels of methylmalonic acid at baseline than those with stable-TD motor phenotypes during disease progression49. However, predictive factors for motor subtype converters are mainly focused on clinical features.
Motor subtype converters experienced faster disease progression and worse clinical prognoses50. Lack of studies on biomarkers besides clinical factors that predict the conversion of TD to PIGD subtypes. In the present study, it was demonstrated that higher plasma GFAP in patients with TD subtypes at baseline could shift to the PIGD subtypes at a two-year follow-up. Conversely, lower plasma GFAP levels in patients with PIGD subtypes at baseline could convert to the TD and indeterminate subtypes during follow-up. Similar results were not observed for plasma NFL, p-tau 181, Aβ42, and Aβ40. Therefore, our findings suggest that baseline plasma GFAP levels could serve as a valid biomarker for predicting motor subtype conversion and disease progression in PD.
We acknowledge the limitations of our study. The current study was a single-center design and the sample size was still relatively small. Therefore, future larger and multicenter studies including more patients with the indeterminate subtype are encouraged. The follow-up period was only 2 years, which makes it difficult to conclude the long-term progression of different motor subtypes in PD. Additionally, medication effects on PD motor subtype classification at different follow times should be considered.
In conclusion, PD patients with the PIGD subtype have evidence of higher plasma GFAP compared to the TD subtype. Plasma GFAP may represent a potential biomarker for identifying motor subtypes and predicting the conversion of different motor subtypes in PD.
Methods
Participants
Patients with PD were from an ongoing perspective longitudinal cohort study supported at the Department of Neurology of West China Hospital of Sichuan University. All subjects agreed to participate in the study and written informed consent was obtained. The study was approved by the Ethics Committee of West China Hospital of Sichuan University.
One hundred and eighty-four early-stage PD patients (disease duration <3 and Hoehn & Yahr [H&Y] stage ≤3) and 95 healthy controls (HCs) were included in the study. The enrolled 150 PD patients come from our previous study9. All subjects were recruited between February 2015 and November 2020 and followed for up to 2 years. PD patients were diagnosed clinically according to the United Kingdom Brain Bank diagnostic criteria51. Clinically established PD patients were recruited after follow-up evaluation. Atypical parkinsonism such as progressive supranuclear palsy, multiple system atrophy, dementia with Lewy bodies, and secondary parkinsonism (vascular, drug-induced, inflammatory, immune-mediated, infectious, traumatic, etc.) was excluded. HCs were recruited from volunteers or spouses of PD patients who had no history of neurological diseases and other severe disorders.
Clinical evaluation
The detailed data collection method was described in our previous study11. Age, sex and education level were collected in all subjects at baseline. Age of onset and disease duration of PD patients were collected. The motor and non-motor symptoms of PD patients were carefully evaluated by experienced movement disorder specialists at baseline and every 12 months during the follow-up period. The assessment of motor was performed in the “OFF” state and the evaluation of non-motor symptoms including cognitive function was performed in the “ON” state in PD patients. PD symptoms were assessed with part I, II, and III sub-scales of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) and H-Y staging was undertaken for all PD patients. Montreal Cognitive Assessment (MoCA, Beijing Version) was used to assess cognitive function. Quality of life was evaluated with the 39-item Parkinson’s disease questionnaire (PDQ-39) with a higher score indicating a poorer quality of life. Levodopa equivalent daily dose (LEDD) was assessed according to the levodopa conversion formula52. Briefly, 100 mg levodopa = 133 mg entacapone = 1 mg pramipexole = 5 mg ropinirole = 10 mg selegiline = 1 mg rasagiline = 100 mg amantadine.
Motor subtypes
The motor subtype classification of PD patients was evaluated according to Stebbins et al.53. Briefly, the ratio of the mean MDS-UPDRS tremor scores (11 items) to the mean MDS-UPDRS PIGD scores (5 items) was calculated for each patient. If the ratio was ≤0.90, patients were classified as a PIGD subtype, if the ratio was ≥1.15 as a TD subtype, and if the ratio was between 0.90 and 1.15 as an indeterminate subtype. Motor subtypes were evaluated repeatedly at the 1-year and 2-year follow-up time points. Converters were grouped during follow-up according to TD/PIGD groupings at baseline.
Measurement of plasma GFAP, NFL, Aβ40, Aβ42, and p-Tau181
Fasting venous blood of 4 ml whole blood (EDTA anticoagulant tube) was collected from all PD patients and healthy controls, centrifuged at 4 °C within 2 h (2000 rpm, 10 min), the upper plasma was collected and stored in the refrigerator at −80 °C to avoid repeated freezing and thawing. Plasma GFAP, NfL, Aβ40, Aβ42, and p-tau181 were measured for all participants at baseline, 1-year, and 2-years follow-up using an ultrasensitive single-molecule array (SimoaTM; Quanterix, Billerica, MA, USA) on the automated SimoaTM HD-X platform (GBIO, Hangzhou, China). The Neurology 4-Plex E Assay Kit (Cat No:103670) and p-tau181 Advantage V2 Assay Kit (Cat No:103714) were used (Quanterix). The plasma sample was randomized, blinded, and measured using a batch of reagents from the same lot.
Statistical analysis
The Shapiro–Wilk test was applied to assess the normality of data. For normal variables, numbers are expressed as mean ± standard deviation (SD). Analysis of variance with Bonferroni as post hoc test and t-tests were used for group comparison. For non-normal or non-homoscedasticity variables, numbers are expressed as median (interquartile range). Multiple comparisons were performed by the non-parametric Kruskal–Wallis test. Categorical variables were compared by Chi-square tests and Fisher’s exact tests.
Spearman correlation analysis was performed to assess the association between baseline plasma GFAP levels and clinical characteristics in different groups.
After adjusting for age, factors that affect motor subtype transformation were investigated by logistic regression analysis. Variables for inclusion were carefully selected, considering the number of events available and the univariate relationship. The receiver operating characteristic (ROC) curve was used to analyze the capability of baseline plasma GFAP levels for predicting the shift of the baseline TD subtype.
All analyses were carried out using SPSS version 22.0 (IBM Corporation, Armonk, NY, USA). Scatter plots and ROC curves were generated using GraphPad Prism version 8.0 (GraphPad Software, Inc, San Diego, CA, USA). P < 0.05 was considered statistically significant.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Liddelow, S. A. & Barres, B. A. Reactive Astrocytes: Production, Function, and Therapeutic Potential. Immunity 46, 957–967 (2017).
Abdelhak, A. et al. Blood GFAP as an emerging biomarker in brain and spinal cord disorders. Nat. Rev. Neurol. 18, 158–172 (2022).
Croisier, E. & Graeber, M. B. Glial degeneration and reactive gliosis in alpha synucleinopathies: the emerging concept of primary gliodegeneration. Acta Neuropathol. 112, 517–530 (2006).
Rizor, A., Pajarillo, E., Johnson, J., Aschner, M. & Lee, E. Astrocytic Oxidative/Nitrosative Stress Contributes to Parkinson’s Disease Pathogenesis: The Dual Role of Reactive Astrocytes. Antioxidants 8, 265 (2019).
Huang, J., Li, C. & Shang, H. Astrocytes in Neurodegeneration: Inspiration From Genetics. Front. Neurosci. 16, 882316 (2022).
Su, W., Chen, H. B., Li, S. H. & Wu, D. Y. Correlational study of the serum levels of the glial fibrillary acidic protein and neurofilament proteins in Parkinson’s disease patients. Clin. Neurol. Neurosurg. 114, 372–375 (2012).
Tang, Y. et al. Plasma GFAP in Parkinson’s disease with cognitive impairment and its potential to predict conversion to dementia. NPJ Parkinsons Dis. 9, 23 (2023).
Mao, S. et al. Association of serum neurofilament light chain and glial fibrillary acidic protein levels with cognitive decline in Parkinson’s disease. Brain Res. 1805, 148271 (2023).
Lin, J. et al. Plasma glial fibrillary acidic protein as a biomarker of disease progression in Parkinson’s disease: a prospective cohort study. BMC Med. 21, 420 (2023).
Li, C., Liu, J., Lin, J. & Shang, H. COVID-19 and risk of neurodegenerative disorders: A Mendelian randomization study. Transl. Psychiatry 12, 283 (2022).
Ou, R. et al. Longitudinal evolution of non-motor symptoms in early Parkinson’s disease: a 3-year prospective cohort study. NPJ Parkinsons Dis. 7, 58 (2021).
Xiao, Y. et al. Stability of motor-nonmotor subtype in early-stage Parkinson’s disease. Front. Aging Neurosci. 14, 1040405 (2022).
Nutt, J. G. Motor subtype in Parkinson’s disease: Different disorders or different stages of disease? Mov. Disord. 31, 957–961 (2016).
van der Heeden, J. F. et al. Postural instability and gait are associated with severity and prognosis of Parkinson disease. Neurology 86, 2243–2250 (2016).
Aleksovski, D., Miljkovic, D., Bravi, D. & Antonini, A. Disease progression in Parkinson subtypes: the PPMI dataset. Neurol. Sci. 39, 1971–1976 (2018).
Reijnders, J. S., Ehrt, U., Lousberg, R., Aarsland, D. & Leentjens, A. F. The association between motor subtypes and psychopathology in Parkinson’s disease. Parkinsonism Relat. Disord. 15, 379–382 (2009).
Zhang, Y. H. et al. The relationship between the phenotype of Parkinson’s disease and levodopa-induced dyskinesia. Neurosci. Lett. 556, 109–112 (2013).
Urso, D. et al. Disentangling the PIGD classification for the prediction of cognitive impairment in de novo Parkinson’s disease. J. Neurol. 269, 1566–1573 (2022).
von Coelln, R. et al. The inconsistency and instability of Parkinson’s disease motor subtypes. Parkinsonism Relat. Disord. 88, 13–18 (2021).
Coughlin, D. G. & Irwin, D. J. Neuropathological substrates of cognition in Parkinson’s disease. Progress in Brain Research 269, 177–193 (2022).
Selikhova, M. et al. A clinico-pathological study of subtypes in Parkinson’s disease. Brain 132, 2947–2957 (2009).
Walker, I. M. et al. Non-tremor motor dysfunction in Lewy body dementias is associated with AD biomarkers. Parkinsonism Relat. Disord. 100, 33–36 (2022).
Cousins, K. A. Q. et al. Plasma GFAP associates with secondary Alzheimer’s pathology in Lewy body disease. Ann. Clin. Transl. Neurol. 10, 802–813 (2023).
Chatterjee, P. et al. Plasma glial fibrillary acidic protein is elevated in cognitively normal older adults at risk of Alzheimer’s disease. Transl. Psychiatry 11, 27 (2021).
Pereira, J. B. et al. Plasma GFAP is an early marker of amyloid-beta but not tau pathology in Alzheimer’s disease. Brain 144, 3505–3516 (2021).
Ishiki, A. et al. Glial fibrillar acidic protein in the cerebrospinal fluid of Alzheimer’s disease, dementia with Lewy bodies, and frontotemporal lobar degeneration. J. Neurochem 136, 258–261 (2016).
Benussi, A. et al. Serum Glial Fibrillary Acidic Protein (GFAP) Is a Marker of Disease Severity in Frontotemporal Lobar Degeneration. J. Alzheimers Dis. 77, 1129–1141 (2020).
Teng, X. et al. The relationship between serum neurofilament light chain and glial fibrillary acidic protein with the REM sleep behavior disorder subtype of Parkinson’s disease. J. Neurochem. 165, 268–276 (2023).
Benedet, A. L. et al. Differences Between Plasma and Cerebrospinal Fluid Glial Fibrillary Acidic Protein Levels Across the Alzheimer Disease Continuum. JAMA Neurol. 78, 1471–1483 (2021).
An, H. et al. Adenovirus-induced Reactive Astrogliosis Exacerbates the Pathology of Parkinson’s Disease. Exp. Neurobiol. 30, 222–231 (2021).
An, H., Heo, J. Y., Lee, C. J. & Nam, M.-H. The Pathological Role of Astrocytic MAOB in Parkinsonism Revealed by Genetic Ablation and Over-expression of MAOB. Exp. Neurobiol. 30, 113–119 (2021).
Kwon, K. Y., Lee, E. J., Lee, M., Ju, H. & Im, K. Impact of motor subtype on non motor symptoms and fall-related features in patients with early Parkinson’s disease. Geriatr. Gerontol. Int 21, 416–420 (2021).
Pelicioni, P. H. S., Menant, J. C., Latt, M. D. & Lord, S. R. Falls in Parkinson’s Disease Subtypes: Risk Factors, Locations and Circumstances. Int. J. Environ. Res. Public Health 16, 2216 (2019).
Ng, A. S. L. et al. Utility of plasma Neurofilament light as a diagnostic and prognostic biomarker of the postural instability gait disorder motor subtype in early Parkinson’s disease. Mol. Neurodegener. 15, 33 (2020).
Lin, C. H. et al. Blood NfL: A biomarker for disease severity and progression in Parkinson disease. Neurology 93, e1104–e1111 (2019).
Hansson, O. et al. Blood-based NfL: A biomarker for differential diagnosis of parkinsonian disorder. Neurology 88, 930–937 (2017).
Heller, C. et al. Plasma glial fibrillary acidic protein is raised in progranulin associated frontotemporal dementia. J. Neurol., Neurosurg. Psychiatry 91, 263–270 (2020).
Coughlin, D. G. & Irwin, D. J. Fluid and Biopsy Based Biomarkers in Parkinson’s Disease. Neurotherapeutics 20, 932–954 (2023).
Baiardi, S. et al. Diagnostic value of plasma p-tau181, NfL, and GFAP in a clinical setting cohort of prevalent neurodegenerative dementias. Alzheimers Res. Therapy 14, 153 (2022).
Doecke, J. D. et al. Total Aβ42/Aβ40 ratio in plasma predicts amyloid-PET status, independent of clinical AD diagnosis. Neurology 94, e1580–e1591 (2020).
Lin, J. et al. Evolution and Predictive Role of Plasma Alzheimer’s Disease-related Pathological Biomarkers in Parkinson’s Disease. J. Gerontology Ser. A 78, 2203–2213 (2023).
Bellaver, B. et al. Astrocyte reactivity influences amyloid-β effects on tau pathology in preclinical Alzheimer’s disease. Nat. Med. 29, 1775–1781 (2023).
Ding, J. et al. Relationship between the plasma levels of neurodegenerative proteins and motor subtypes of Parkinson’s disease. J. Neural Transm. 124, 353–360 (2017).
Kang, J. H. et al. CSF biomarkers associated with disease heterogeneity in early Parkinson’s disease: the Parkinson’s Progression Markers Initiative study. Acta Neuropathol. 131, 935–949 (2016).
Goldman, J. G. et al. Cerebrospinal fluid, plasma, and saliva in the BioFIND study: Relationships among biomarkers and Parkinson’s disease Features. Mov. Disord. 33, 282–288 (2018).
Erro, R. et al. Comparing postural instability and gait disorder and akinetic-rigid subtyping of Parkinson disease and their stability over time. Eur. J. Neurol. 26, 1212–1218 (2019).
Herman, T., Shema-Shiratzky, S., Arie, L., Giladi, N. & Hausdorff, J. M. Who will remain tremor dominant? The possible role of cognitive reserve in the time course of two common Parkinson’s disease motor subtypes. J. Neural Transm. 125, 1007–1011 (2018).
Lee, J. W., Song, Y. S., Kim, H., Ku, B. D. & Lee, W. W. Alteration of Tremor Dominant and Postural Instability Gait Difficulty Subtypes During the Progression of Parkinson’s Disease: Analysis of the PPMI Cohort. Front. Neurol. 10, 471 (2019).
Santos Garcia, D. et al. Parkinson’s Disease Motor Subtypes Change with the Progression of the Disease: Results from the COPPADIS Cohort at 2-Year Follow-Up. J. Parkinsons Dis. 12, 935–955 (2022).
Eisinger, R. S. et al. Parkinson’s disease motor subtype changes during 20 years of follow-up. Parkinsonism Relat. Disord. 76, 104–107 (2020).
Hughes, A. J., Daniel, S. E., Kilford, L. & Lees, A. J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 55, 181–184 (1992).
Tomlinson, C. L. et al. Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov. Disord. 25, 2649–2653 (2010).
Stebbins, G. T. et al. How to identify tremor dominant and postural instability/gait difficulty groups with the movement disorder society unified Parkinson’s disease rating scale: comparison with the unified Parkinson’s disease rating scale. Mov. Disord. 28, 668–670 (2013).
Acknowledgements
We thank the patients and their family for their participation in this study. This study was supported by the National Key Research and Development Program of China (grant no. 2021YFC2501203) and the Sichuan Science and Technology Program (Grant No. 2022ZDZX0023).
Author information
Authors and Affiliations
Contributions
N.N.C. contributed with conception, organization and execution, data collection, statistical analysis, and drafting the manuscript. R.W.O. contributed with execution and manuscript revision. C.Y.L., L.Y.Z., Q.Q.W., S.C.W., Q.R.J., T.M.Y., Y.X., J.Y.L., B.Z., X.P.C. contributed with data collection. H.F.S. contributed with conception and organization, manuscript review and critique, and responsible for overall content as the guarantor.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Che, N., Ou, R., Li, C. et al. Plasma GFAP as a prognostic biomarker of motor subtype in early Parkinson’s disease. npj Parkinsons Dis. 10, 48 (2024). https://doi.org/10.1038/s41531-024-00664-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-024-00664-8
- Springer Nature Limited