Abstract
Understanding the genomic underpinnings of antipredatory behaviors is a hot topic in eco-evolutionary research. Yellow-bellied toad of the genus Bombina are textbook examples of the deimatic display, a time-structured behavior aimed at startling predators. Here, we generated the first de novo brain transcriptome of the Apennine yellow-bellied toad Bombina pachypus, a species showing inter-individual variation in the deimatic display. Through Rna-Seq experiments on a set of individuals showing distinct behavioral phenotypes, we generated 316,329,573 reads, which were assembled and annotated. The high-quality assembly was confirmed by assembly validators and by aligning the contigs against the de novo transcriptome with a mapping percentage higher than 91.0%. The homology annotation with DIAMOND (blastx) led to 77,391 contigs annotated on Nr, Swiss Prot and TrEMBL, whereas the domain and site protein prediction made with InterProScan led to 4747 GO-annotated and 1025 KEGG-annotated contigs. The B. pachypus transcriptome described here will be a valuable resource for further studies on the genomic underpinnings of behavioral variation in amphibians.
Measurement(s) | Transcriptome |
Technology Type(s) | RNA-seq of total RNA |
Factor Type(s) | Behavior |
Sample Characteristic - Organism | Bombina pachypus |
Sample Characteristic - Environment | natural environment |
Sample Characteristic - Location | Italy |
Similar content being viewed by others
Background & Summary
Inter-individual variation in antipredatory behavior has long attracted scientific curiosity and has been investigated in a wide range of animal species, from mammals to fishes, insects and even to marine invertebrates1. However, we still know little about the specific molecular mechanisms underlying the origin of this variation. In fact, while some behavioral traits have been linked to epigenetic mechanisms2, the observation that behavior can be heritable supports a role for modulation of standing genetic variation within populations3,4. The recent advances in omic-sciences opens for investigating the genetic basis of behavioral trait variation5, and thus for understanding the genomic underpinnings of the inter-individual variation in antipredatory strategies.
Deimatism is a common anti-predatory strategy. It consists in suddenly unleashing unexpected defenses to frighten predators and to stop their attack, and it combines cryptism and aposematism in a complex and time structured antipredatory strategy6. Deimatic displays typically involve either chromatic and behavioral components. Inter-individual variation in warning signals have traditionally been considered maladaptive. Yet, individual variation in morphological and chromatic components have been widely reported in many organisms7,8,9,10,11. Conversely, the occurrence of polymorphism in the behavioral component of warning signals is still almost unexplored.
With this study, we aim at providing genomic resources to investigate the genetic underpinnings of inter-individual behavioral differences in warning signals. We generated the first de novo brain transcriptome of a species showing polymorphism in behavioral traits associated with deimatic displays, the Apennine yellow-bellied toad Bombina pachypus12. The yellow-bellied toads of the genus Bombina are textbook examples of unken-reflex, a deimatic behavior which consists in toads arching their body and exposing their aposematically colored ventral side. Experimental evidence has shown within-population variation in the way B. pachypus toads reacted to predation stimuli: about half of the toads quickly reacted with a long and intense body arching and aposematic display (i.e. the unken-reflex), while the other half of the individuals analysed did not show deimatic behavior, but rather moved away12. These two antipredatory strategies have been proposed to reflect the way individuals cope with environmental challenges, i.e. reactive vs proactive coping style, and arguably linked to differential sympathetic-parasympathetic activities13. We focused on brain transcriptome, as the brain tissues have shown differential gene expression profiles linked to distinct behavioral states in response to environmental stimuli14,15,16, also in closely related Bombina species17,18. Thus, the transcriptome analysis of the brain can reveal the ways in which distinct molecular pathways can modulate anti-predatory behaviour19.
The data presented in this study consist of assembled transcriptome sequences of the brain of B. pachypus at the adult stage. The de novo transcriptome has been annotated to provide a transcriptome reference for further analysis of differential gene expression profiles. Since the B. pachypus genome has not been sequenced so far, the transcriptome presented here will be a valuable resource for further eco-evolutionary studies on the behavioral repertoire of amphibians.
Methods
Sample collection and RNA preparation
We analyzed 6 adult yellow-bellied toad individuals representative of distinct behavioral profiles, i.e. prolonged unken-reflex display vs no unken-reflex display (thereafter referred as “ + ” and “-“, respectively). Behavioral profiles were scored as in Chiocchio et al.12: 3 toads showed prolonged unken-reflex (+), whereas the other 3 did not show unken-reflex (−), as reported in Table 1. Sampling procedures were approved by the Italian Ministry of Ecological Transition and the Italian National Institute for Environmental Protection and Research (ISPRA; permit number: 20824, 18-03-2020). After dissection, brain tissue was immediately stored in RNAprotect Tissue Reagent (Quiagen) until RNA extraction. RNA extractions were performed using the RNeasy Plus Kit (Quiagen), according to the manufacturer’ instructions. RNA quality and concentration were assessed by means of both a spectrophotometer and a Bioanalyzer (Agilent Cary60 UV-vis and Agilent 2100, respectively - Agilent Technologies, Santa Clara, USA).
Library preparation and sequencing
Library preparation and RNA sequencing were performed by NOVOGENE (UK) COMPANY LIMITED using Illumina NovaSeq platform. Library construction was carried out using the NEBNext® Ultra ™ RNA Library Prep Kit for Illumina®, following manufacturer instructions. Briefly, after the quality control check, the mRNA sample was isolated from the total RNA by using magnetic beads made of oligos d(T)25 (i.e. polyA-tail mRNA enrichment). Subsequently, mRNA was randomly fragmented, and a cDNA synthesis step proceeded using random hexamers and the reverse transcriptase enzyme. Once the synthesis of the first chain has finished, the second chain was synthesized with the addition of the Illumina buffer, dNTPs, RNase H and polymerase I of E.coli, by means of the Nick translation method. Then, the resulting products went through purification, repair, A-tailing and adapter ligation. Fragments of the appropriate size were enriched by PCR, the indexed P5 and P7 primers were introduced, and the final products were purified. Finally, the Illumina Novaseq 6000 sequencing system was used to sequence the libraries, through a paired-end 150 bp (PE150) strategy. We obtained on average 52.7 million reads for each library. The sequencing data are available at the NCBI Sequence Read Archive (project ID PRJNA76401320).
Pre-assembly processing stage
A total of 316,329,573 pairs of reads was generated by Illumina sequencing. All of them went to a cleaning analytic step. The quality of the raw reads was assessed with the FastQC 0.11.5 tool (http://www.bioinformatics.bbsrc.ac.uk/projects/fastqc), in order to estimate the RNAseq quality profiles. The quality estimators were generated for both the raw and trimmed data. The quality assessment metrics for trimmed data were aggregated across all samples into a single report for a summary visualization with MultiQC software tool21 v.1.9 (see Fig. 1). To remove low quality bases and adapter sequences, raw reads were also analyzed through a quality trimming step with Trimmomatic22, v.0.39 (setting the option SLIDINGWINDOW: 4: 15, MINLEN: 36, and HEADCROP: 13). All the unpaired reads were discarded. After the cleaning step and removal of low-quality reads, 297,354,405 clean reads (i.e. 94% of raw reads) were maintained for building the de novo transcriptome assembly (see Table 1).
De novo transcriptome assembly and quality assessment
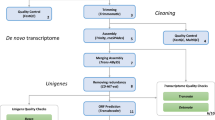
As there is no reference genome for B. pachypus, we performed a de novo transcriptome assembly procedure. The workflow of the bioinformatic pipelines is shown in Fig. 2. All the described bioinformatics analyses were performed on the high-performance computing systems provided by ELIXIR-IT HPC@CINECA23.
To construct an optimized de novo transcriptome, avoiding chimeric transcripts, we employed rnaSPAdes24, a tool for de novo transcriptome assembly from RNA-Seq data implemented in the SPAdes v.3.14.1 package. rnaSPAdes automatically detected two k-mer sizes, approximately one third and half of the maximal read length (the two detected k-mer sizes were 45 and 67 nucleotides, respectively). At this stage, a total of 1,118,671 assembled transcripts were generated by rnaSPAdes runs, with an average length of 689.41 bp and an N50 of 1474 bp (Table 2).
Results from the assembly procedures were validated through three independent validator algorithms implemented in BUSCO25 v.4.1.4, DETONATE26 v.1.11 and TransRate27 v.1.0.3. These tools generate several metrics used as a guide to evaluate error sources in the assembly process and provide evidence about the quality of the assembled transcriptome. Busco provides a quantitative measure of transcriptome quality and completeness, based on evolutionarily-informed expectations of gene content from the near-universal, ultra-conserved eukaryotic proteins (eukaryota_odb9) database. Detonate (DE novo TranscriptOme rNa-seq Assembly with or without the Truth Evaluation) is a reference-free evaluation method based on a novel probabilistic model that depends only on the assembly and the RNA-Seq reads used to construct it. Transrate generates standard metrics and remapping statistics. No reference protein sequences were used for the assessment with Transrate. The main metrics resulted from the assembly validators are shown in Table 2 (“Before CD-HIT-est” column). After this triple assessment validation step, the result of the assembly procedure become the input for the CD-HIT-est v.4.8.128 program, a hierarchical clustering tool used to avoid redundant transcripts and fragmented assemblies common in the process of de novo assembly, providing unique genes. CD-HIT-est was run using the default parameters, corresponding to a similarity of 95%. Subsequently, a second validation step was launched on the CD-HIT-est output file. To refine the final transcriptome dataset, a further hierarchical clustering step was performed by running CORSET v1.0629. Then, the output of CORSET was validated by BUSCO, and quality assessment was performed with HISAT230,31 by mapping the trimmed reads to the reference transcriptome (unigenes). Results from all validation steps are shown in Table 2 and discussed in the “Technical Validation” paragraph.
Finally, the CORSET output was run on TransDecoder32,33, the current standard tool that identifies long open read frames (ORFs) in assembled transcripts, using default parameters. TransDecoder by default performs ORF prediction on both strands of assembled transcripts regardless of the sequenced library. It also ranks ORFs based on their completeness, and determines if the 5 ‘end is incomplete by looking for any length of AA codons upstream of a start codon (M) without a stop codon. We adopted the “Longest ORF” rule and selected the highest 5 AUG (relative to the inframe stop codon) as the translation start site.
Transcriptome annotation
We employed different kinds of annotations for the de novo assembly. We introduced DIAMOND34, an open-source algorithm based on double indexing that is 20,000 times faster than BLASTX on short reads and has a similar degree of sensitivity. Like BLASTX, DIAMOND attempts to determine exhaustively all significant alignments for a given query. Most sequence comparison programs, including BLASTX, follow the seed-and-extend paradigm. In this two-phase approach, users search first for matches of seeds (short stretches of the query sequence) in the reference database, and this is followed by an ‘extend’ phase that aims to compute a full alignment. The following parameter settings were applied: DIAMOND-fast DIAMOND BLASTX-t 48 -k 250 -min-score 40; DIAMOND-sensitive: DIAMOND BLASTX -t 48 -k 250 -sensitive -min-score 40.
Contigs were aligned with DIAMOND on Nr, SwissProt and TrEMBL to retrieve the corresponding best annotations. An annotation matrix was then generated by selecting the best hit for each database. Following the analysis of BLASTX against Nr, SwissProt and TremBL, we obtained respectively: 123,086 (64.57%), 77,736 (40.78%), 122,907 (64.48%) contigs. The results obtained following the analysis with BLASTP against Nr, SwissProt and TrEMBL were 96,321 (50.53%), 57,877 (30.36%) and 97,256 (51.02%) contigs respectively. All the information on the resulting datasets is resumed in Table 3.
The output obtained by the BLASTX annotation consisted in a total of 77391 sequences simultaneously mapped on the three queried databases (i.e., Nr, SwissProt and TrEMBL). The output obtained following the BLASTP annotation consisted in a total of 57704 sequences simultaneously mapped on the three databases. Venn diagrams are presented in Fig. 3, showing the redundancy of the annotations in the different databases for both DIAMOND BLASTX (Fig. 3a) and DIAMOND BLASTP (Fig. 3b). Furthermore, the ten most represented species and the ten hits of the gene product obtained respectively with BLASTX and BLASTP by mapping the transcripts against the reference database Nr are shown in Figs. 4 and 5. Since BLASTX translated nucleotide sequence searches against protein sequences the BLASTX results are more exhaustive than BLASTP results. Contigs were also processed with InterProScan35 to detect InterProScan signatures. The InterPro database (http://www.ebi.ac.uk/interpro/) integrates together predictive models or ‘signatures’ representing protein domains, families and functional sites from multiple, diverse source databases: Gene3D, PANTHER, Pfam, PIRSF, PRINTS, ProDom, PROSITE, SMART, SUPERFAMILY and TIGRFAMs. The obtained InterProScan results for all the unigenes are available on Figshare in the form of Tab Separated Values (tsv) file format, which includes the GO and KEGG annotated contigs, respectively.
Comparison with Bombina orientalis brain transcriptome
We compared the brain de novo transcriptome of B. pachypus with the brain de novo transcriptome of B. orientalis, recently produced in the frame of a prey-catching conditioning experiment17,18. The B. orientalis transcriptome resource was downloaded from GEO archive of NCBI (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE171766). To make the datasets comparable, we first performed ORF prediction on B. orientalis trascriptome using Transdecoder, using default settings. Results from the B. orientalis trascriptome ORF prediction are available in Figshare at the following link https://doi.org/10.6084/m9.figshare.20319633/). We also applied the makedb function implemented in DIAMOND to create the protein database index. Then, we aligned the B. pachypus predicted coding sequences and proteins (query files) against the B. orientalis protein database (reference) using DIAMOND BLASTX and BLASTP, respectively. We obtained 167041 matches from BLASTX and 156248 matches for BLASTP. Results from the BLASTX and BLASTP comparisons, and the most matched proteins, are available on Figshare36 (link available in next paragraph).
Data Records
We generated six files corresponding to the RNA-seq samples of the brain tissue of the six B. pachypus individuals analyzed for this study. The six files were deposited in the NCBI Sequence Read Archive database, under project identification number PRJNA76401320; the NCBI accessions for each individual are listed in Table 1 (Run ID SRR15927729 - SRR15927734). The datasets containing the rnaSPAdes transcriptome assembly, post CD-Hit-Est assembly, post corset assembly (unigenes), predicted ORF and homology and functional annotation files were deposited in Figshare (Project description36: https://doi.org/10.6084/m9.figshare.c.5696179; Assembly37: https://doi.org/10.6084/m9.figshare.16945270; Annotation38: https://doi.org/10.6084/m9.figshare.16945264; comparison with Bombina orientalis transcriptome39: https://doi.org/10.6084/m9.figshare.20319633).
Technical Validation
Quality of the raw reads and assembly validation
To assess overall data quality, we performed quality checks using FastQC and MultiQC for all samples before and after adaptor/sequence trimming. The mean read counts per quality score were higher than 35 (Fig. 1a). The mean quality scores in each base position were higher than 35 (Fig. 1b). The mean sequence lengths were 126–130 bp (Fig. 1c). The mean per sequence GC content was 40% (Fig. 1d).
Transcriptome assembly validation was done using Busco, Detonate and Transrate. Results from the triple validation step are shown in Table 2, and contain the scores obtained from the execution of the three analysis tools, both before and after running CD-HIT-est.
Using HISAT231 (a fast and sensitive alignment program for mapping next-generation sequencing reads, DNA and RNA), we verified that more than 91% of the reads were mapped back to the assembled transcriptome of the B. pachypus thus indicating a proper quality sequence reconstruction. Figure 6 shows the number of raw reads, paired-reads after trimming, and trimmed paired-reads that are mapped against B. pachypus de novo transcriptome.
Transrate assessment showed increased values for the “Transrate Optimal Score” item following hierarchical clustering using CD-HIT-est, passing from 0.088 to 0.178, and for the “Transrate Assembly Score” item, passing from 0.056 to 0.128 (more than twice). The value of “good contigs” increased after CD-HIT-est (due to redundancy removal), but with a value of 0.92 for the final assembly. Transrate also reported a value of GC around 40% after each validation step. The transcriptome obtained after CD-HIT-est included a total of 896,992 transcripts with a mean transcript length of 616.32 bp and an N50 of 1082 bp, with a value above the 94% of completeness for Busco assessment. In terms of redundancy removal, the further step of CORSET clustering produced a real improvement. In fact, the final version of the assembled transcriptome included 267,959 transcripts with a mean transcript length of 799 bp, the N50 value equals to 2314 and a value above the 96% for Busco assessment, improving the previous results computed by the CD-HIT-est tool. As shown in Table 2, CORSET greatly improved the assembled transcriptome removing redundancy and reducing the number of transcripts, thus improving the quality scores of the final assembly.
Quality control of annotation
The transcriptome was functionally annotated by performing DIAMOND and InterProScan. By selecting the best hit for Nr, SwissProt and TrEMBL databases, the annotation matrix generated with DIAMOND has led to the results listed in Table 3. In particular 77,391 (BLASTX) and 57,704 (BLASTP) contigs were annotated in all the three databases, NR, Swissprot, Trembl.
InterProScan provided as result the corresponding InterPro accession numbers and, among other accession IDs, the GO and Kegg annotation. It produced a total of 32142 annotated contigs, being 4747 contigs GO-annotated and 1025 contigs KEGG-annotated.
Code availability
All the software programs used in this article (de novo transcriptome assembly, pre and post-assembly steps, and transcriptome annotation) are listed in the Methods paragraph. In case of no details on parameters, the programs were used with the default settings.
References
Carere, C. & Maestripieri, D. Animal Personalities: Behavior, Physiology, and Evolution. (Chicago: University of Chicago Press, 2013).
Jensen, P. Behaviour epigenetics–the connection between environment, stress and welfare. Appl. Anim. Behav. Sci. 157, 1–7 (2014).
Van Oers, K., de Jong, G., van Noordwijk, A. J., Kempenaers, B. & Drent, P. J. Contribution of genetics to the study of animal personalities: a review of case studies. Behaviour 142, 1185–120610 (2005).
Van Oers, K. & Sinn, D. L. The quantitative and molecular genetics of animal personality. In: Carere, C. & Maestripieri, D. editors. Animal Personalities: Behavior, Physiology, and Evolution. (Chicago: University of Chicago Press; p. 148–200, 2013).
Ellegren, H. Genome sequencing and population genomics in non-model organisms. Trends Ecol. Evol. 29, 51–63 (2014).
Umbers, K. D. L., Lehtonen, J. & Mappes, J. Deimatic displays. Curr. Biol. 25, R58eR59 (2015).
Joron, M. & Mallet, J. L. Diversity in mimicry: paradox or paradigm? Trends Ecol. Evol. 13, 461–466 (1998).
Arenas, L. M. & Stevens, M. Diversity in warning coloration is easily recognized by avian predators. J. Evol. Biol. 30, 1288–1302 (2017).
Richards-Zawacki, C. L., Yeager, J. & Bart, H. P. No evidence for differential survival or predation between sympatric color morphs of an aposematic poison frog. Evol. Ecol. 27, 783–795 (2013).
Rönkä, K. Evolution of signal diversity: predator-prey interactions and the maintenance of warning color polymorphism in the wood tiger moth Arctia plantaginis. Jyväskylä studies in biological and environmental science 339 (2017).
Lawrence, J. P. et al. Weak warning signals can persist in the absence of gene flow. Proc. Natl. Acad. Sci. USA 116, 19037–19045 (2019).
Chiocchio, A., Martino, G., Bisconti, R., Carere, C., Canestrelli D. Shock or jump: deimatic behavior is repeatable and polymorphic in a yellow-bellied toad. bioRxiv 2022.04.29.489992, https://doi.org/10.1101/2022.04.29.489992 (2022).
Koolhaas, J. M., de Boer, S. F., Coppens, C. M. & Buwalda, B. Neuroendocrinology of coping styles: towards understanding the biology of individual variation. Front Neuroendocrinol. 31(3), 307–21 (2010).
Whitfield, C. W., Cziko, A. M. & Robinson, G. E. Gene expression profiles in the brain predict behavior in individual honey bees. Science 302, 296–299 (2003).
Rey, S., Boltana, S., Vargas, R., Roher, N. & Mackenzie, S. Combining animal personalities with transcriptomics resolves individual variation within a wild-type zebrafish population and identifies underpinning molecular differences in brain function. Mol. Ecol. 22, 6100–15 (2013).
Bell, A. M., Bukhari, S. A. & Sanogoc, Y. O. Natural variation in brain gene expression profiles of aggressive and nonaggressive individual sticklebacks. Behavior 153, 1723–1743 (2016).
Lewis, V., Laberge, F. & Heyland, A. Temporal Profile of Brain Gene Expression After Prey Catching Conditioning in an Anuran Amphibian. Front Neurosci 3, 1407 (2020).
Lewis, V., Laberge, F. & Heyland, A. Transcriptomic signature of extinction learning in the brain of the fire-bellied toad, Bombina orientalis. Neurobiol Learn Mem. 184, 107502 (2021).
Harris, R. M., & Hofmann, H. A. Neurogenomics of behavioral plasticity. In Landry, C. R. & Aubin-Horth N. editors. Ecological genomics. (Springer Science, pp. 149–168, 2014).
NCBI Sequence Read Archive https://identifiers.org/ncbi/insdc.sra:SRP337549 (2022)
Ewels, P., Magnusson, M., Lundin, S. & Kaller, M. MultiQC: summarize analysis results for multiple tools and samples in a single report. Bioinformatics 32, 3047–8 (2016).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–20 (2014).
Castrignanò, T. et al. ELIXIR-IT HPC@CINECA: high performance computing resources for the bioinformatics community. BMC Bioinformatics. 21(Suppl 10), 352 (2020).
Bushmanova, E., Antipov, D., Lapidus, A. & Prjibelski, A. D. rnaSPAdes: a de novo transcriptome assembler and its application to RNA-Seq data. Gigascience 8, giz100 (2019).
Simão, F. A., Waterhouse, R. M., Ioannidis, P., Kriventseva, E. V. & Zdobnov, E. M. Busco: Assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31, 3210–3212 (2015).
Li, B. et al. Evaluation of de novo transcriptome assemblies from RNA-Seq data. Genome Biol. 15, 1–21 (2014).
Smith-Unna, R., Boursnell, C., Patro, R., Hibberd, J. M. & Kelly, S. Transrate: Reference-free quality assessment of de novo transcriptome assemblies. Genome Res. 26, 1134–1144 (2016).
Fu, L., Niu, B., Zhu, Z., Wu, S. & Li, W. CD-HIT: Accelerated for clustering the next-generation sequencing data. Bioinformatics 28, 3150–3152 (2012).
Davidson, N. M. & Oshlack, A. Corset: enabling differential gene expression analysis for de novo assembled transcriptomes. Genome Biol. 15(7), 410 (2014).
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: a fast spliced aligner with low memory requirements. Nat. Methods. 12, 357–360 (2015).
Pertea, M., Kim, D., Pertea, G., Leek, J. T. & Salzberg, S. L. Transcript-level expression analysis of RNA-seq experiments with HISAT, StringTie and Ballgown. Nat. Protoc. 11, 1650–67 (2016).
Signal, B., & Kahlke, T. Borf: Improved ORF prediction in de novo assembled transcriptome annotation. https://www.biorxiv.org/content/10.1101/2021.04.12.439551v1 (2021).
Tang, S., Lomsadze, A., Borodovsky, M. Identification of protein coding regions in RNA transcripts. Nucleic Acids Res. 43 (2015).
Buchfink, B., Xie, C. & Huson, D. Fast and sensitive protein alignment using DIAMOND. Nat. Methods. 12, 59–60 (2015).
Hunter, S. et al. InterPro: the integrative protein signature database. Nucleic Acids Res. 37(Database Issue), D211–5 (2009).
Chiocchio, A. et al. Brain de novo transcriptome assembly of a toad species showing polymorphic anti-predatory behavior. Project description: figshare https://doi.org/10.6084/m9.figshare.c.5696179 (2022).
Chiocchio, A. et al. Brain de novo transcriptome assembly of a toad species showing polymorphic anti-predatory behavior. Assembly: figshare https://doi.org/10.6084/m9.figshare.16945270 (2022).
Chiocchio, A. et al. Brain de novo transcriptome assembly of a toad species showing polymorphic anti-predatory behavior. Annotation: figshare https://doi.org/10.6084/m9.figshare.16945264 (2022).
Chiocchio, A. et al. Brain de novo transcriptome assembly of a toad species showing polymorphic anti-predatory behavior. Comparison with Bombina orientalis transcriptome: figshare https://doi.org/10.6084/m9.figshare.20319633 (2022).
Acknowledgements
We are grateful to Michela Paoletti for her support during the laboratory procedures and to Jessica Di Martino for her work on the transcriptome annotation. We acknowledge the CINECA for the availability of high-performance computing resources and the ELIXIR-ITA HPC@CINECA initiative for providing HPC resources to our projects: (1) name of the call “Call ELIXIR-ITA CINECA (2020–2021)”, P.I. Andrea Chiocchio, name of the project “ELIX4_chiocchi”; (2) name of the call “Call ELIXIR-ITA CINECA (2021–2022)”, P.I. Tiziana Castrignanò, name of the project “ELIX4_castrign2”. This study was supported by grants from the Italian Ministry for Education, University and Research (Prin project: 2017KLZ3MA), and from the Aspromonte National Park.
Author information
Authors and Affiliations
Contributions
D.C. conceived and financed the study; A.C. e D.C. designed the experiment; A.C., R.B. and G.M. performed sample collection and preparation; A.C. coordinated the RNA extraction and sequencing; T.C. designed and coordinated the bioinformatic analysis; P.L. and T.C. performed reads quality assessment, reads alignment on transcriptome, transcriptome annotation and validation; A.C., P.L. and T.C. wrote the manuscript; D.C., T.C., A.C., R.B., P.L. and G.M. reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chiocchio, A., Libro, P., Martino, G. et al. Brain de novo transcriptome assembly of a toad species showing polymorphic anti-predatory behavior. Sci Data 9, 619 (2022). https://doi.org/10.1038/s41597-022-01724-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41597-022-01724-5
- Springer Nature Limited
This article is cited by
-
HPC-T-Annotator: an HPC tool for de novo transcriptome assembly annotation
BMC Bioinformatics (2024)
-
Integrated de novo transcriptome of Culex pipiens mosquito larvae as a resource for genetic control strategies
Scientific Data (2024)
-
De novo transcriptome assembly of an Antarctic nematode for the study of thermal adaptation in marine parasites
Scientific Data (2023)
-
De novo transcriptome assembly and annotation for gene discovery in Salamandra salamandra at the larval stage
Scientific Data (2023)