Abstract
Olfaction in insects is essential for host identification, mating and oviposition, in which olfactory proteins are responsible for chemical signaling. Here, we determined the transcriptomes of male and female adult antennae of Anoplophora chinensis, the citrus longhorned beetle. Among 59,357 unigenes in the antennal assembly, we identified 46 odorant-binding proteins, 16 chemosensory proteins (CSPs), 44 odorant receptors, 19 gustatory receptors, 23 ionotropic receptors, and 3 sensory neuron membrane proteins. Among CSPs, AchiCSP10 was predominantly expressed in antennae (compared with legs or maxillary palps), at a significantly higher level in males than in females, suggesting that AchiCSP10 has a role in reception of female sex pheromones. Many highly expressed genes encoding CSPs are orthologue genes of A. chinensis and Anoplophora glabripennis. Notably, AchiPBP1 and AchiPBP2 showed 100% and 96% identity with AglaPBP1 and AglaPBP2 from A. glabripennis, with similar expression profiles in the two species; PBP2 was highly expressed in male antennae, whereas PBP1 was expressed in all three tissues in both males and females. These results provide a basis for further studies on the molecular chemoreception mechanisms of A. chinensis, and suggest novel targets for control of A. chinensis.
Similar content being viewed by others
Introduction
Anoplophora chinensis (Forster) (Coleoptera: Cerambycidae), taxonomically regarded as a synonym of Anoplophora malasiaca, is an important worldwide quarantine pest, which is native to East Asia, but which has also been introduced to North America and Europe1,2. Although A. chinensis is commonly known as the citrus longhorned beetle, and causes extensive damage to citrus trees in its native oriental area, it is actually a polyphagous xylophage that has been documented on hosts from 40 genera2. The major economic loss associated with A. chinensis results from damage in fruit-tree plantations, whereas the most commonly infested tree genus is Acer 3. Multiple strategies are currently used to control A. chinensis. Identification of pheromones and plant volatiles that are recognized by A. chinensis enables chemical baiting to trap the insects. Behavioral studies suggest that both male and female A. chinensis can emit volatiles that induce mate orientation in the opposite sex4,5, and the contact sex pheromone produced by females can trigger a series of male mating behaviors when it is perceived by the male antennae6,7. An important component of the male attractant pheromone of A. chinensis is 4-(n-heptyloxy)butan-1-ol5. Currently, semiochemically baited traps are being developed to specifically target the Asian long-horned beetle (Anoplophora glabripennis)8, which also has the potential to greatly improve the sensitivity, reliability, and efficiency of detection of populations of A. chinensis.
In insects, olfaction has several vital roles for survival and reproduction, including food selection, mate recognition, location of oviposition sites and predator avoidance9. The olfactory proteins involved in recognition of pheromones and odorants include two binding-protein families (odorant-binding proteins (OBPs) and chemosensory proteins (CSPs)), three receptor families (odorant receptors (ORs), ionotropic receptors (IRs), and gustatory receptors (GRs)), sensory neuron membrane proteins (SNMPs), and even enzymes (pheromone-degrading enzymes (PDEs) and odorant-degrading enzymes (ODEs))9,10. OBPs and CSPs, which are abundant in the lymph of antennal sensilla, are involved in the first step of the recognition of chemical signals. OBPs are small, soluble proteins with a pattern of six conserved cysteine residues11; they are mainly expressed in the antennae in both sexes, and can bind to hydrophobic molecules from the environment and deliver them to the receptors that are located on the membranes of olfactory sensory neurons (OSNs)12,13,14,15. Pheromone-binding proteins (PBPs) and general odorant-binding proteins (GOBPs) are two subclasses of OBPs. PBPs are considered to be involved in recognition of sex pheromones16,17; however, some PBPs cannot distinguish between pheromones and non-pheromones, e.g., SlitPBP of Spodoptera litura (Fabricius)18. Evidence that PBPs specifically bind to a single pheromone component remains elusive; currently, only two PBPs from Batocera horsfieldi 19 and two from A. glabripennis have been identified from transcriptomic sequence data for species in the family Cerambycidae 10. CSPs are characterized by the presence of four cysteine residues, have a low molecular weight (10–16 kDa), are widely expressed in chemosensory organs and non-olfactory organs in insects, and have multiple functions in chemoreception, growth, and development20,21,22.
Chemosensory reception in insects involves two types of olfactory receptor (ORs and IRs), and one type of GR. ORs are seven-transmembrane-domain proteins that detect volatiles and trigger the transduction of chemical signals to electrical signals that are transmitted to the brain23,24. The nonconventional OR previously referred to as OR83b in Drosophila melanogaster, OR2 in Bombyx mori, and OR7 in mosquitoes was recently renamed Orco, which can selectively distinguish between similar odorant chemical structures owing to the selectivity of the highly conserved and insect-specific Orco and ORs25. In addition, BmorOR1 and BmorOR3 expressed in cells located beneath the pheromone-sensitive long sensilla trichodea are pheromone receptor (PR) genes identified in B. mori 26. IRs are a conserved family with a ligand-gated ion-channel domain that evolved from ionotropic glutamate receptors. Mutations in IR84a, IR64a, IR8a, and IR25a of Drosophila are essential for inhibiting odor-evoked neuronal responses27. GRs also have a seven-transmembrane-domain topology, and are mainly expressed in the gustatory organs, such as the mouthparts. Multiple GRs have been implicated in the detection of sweet and bitter tastes and CO2 vapors28,29.
In addition, SNMPs are homologous with the CD36 family, and are membrane proteins that are located in the insect olfactory neurons. To date, two SNMP subfamilies, SNMP1 and SNMP2, have been identified in insects. SNMPs are thought to mediate ligand delivery to receptors, and are expressed late in adult development30,31.
Substantial developments have been achieved in the understanding of odorant detection in insects, providing new prospects for integrated pest management, although information about coleopterous olfaction is sparse, and few olfactory gene families have been reported32,33,34.
In this study, we sequenced antennal transcriptomes to identify chemosensory genes in A. chinensis, and used quantitative real-time PCR to assess the expression of all CSP genes and two PBP genes in different tissues of both male and female adults. On the basis of these results, we discuss here the potential functions of the products of these genes in olfactory or other physiological processes.
Results
Transcriptome sequencing and unigene assembly
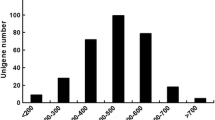
The antennal cDNA libraries of male and female A. chinensis were sequenced using the Illumina HiSeq. 2500 system. A total of 27,706,136 raw reads were produced from male antennae samples, and the Q20 and Q30 base call accuracies were 93.50% and 86.38%, respectively. A total of 24,274,769 raw reads were produced from female antennae samples, and the Q20 and Q30 base call accuracies were 92.65% and 86.04%, respectively (supplementary Table S1). After trimming the adaptor sequences and removing low-quality sequences, blending male and female sequences, splicing, and assembly (using Trinity), we obtained 59,357 unigenes, with an N50 of 1413 bp and a mean length of 673 bp (supplementary Figure S1). The raw reads for A. chinensis have been deposited in the NCBI SRA database (GenBank accession number SRP116677).
Homology analysis and Gene Ontology (GO) annotation
In total, 25,670 unigenes (43.25% of all 59,357 unigenes) were annotated to at least one of the databases using the BLASTx and BLASTn programs with an E-value cut-off of 10 e-5. A total of 17,717 (29.85%), 17,157 (28.90%), 15,824 (26.66%), 8,210 (13.83%), 10,113 (17.04%), 8,646 (14.57%), 24,261 (40.87%), and 9,266 (15.61%) unigenes from A. chinensis were annotated using the Nr, Pfam, KOG, COG, Swiss-Prot, KEGG, eggNOG, and GO databases, respectively (supplementary Table S2). Homology searches against the Nr database showed that A. chinensis antennal transcriptomes shared the highest homology (55%) with sequences from Tribolium castaneum, followed by Dendroctonus ponderosae (16%) and Acyrthosiphon pisum (2%).
GO annotation was obtained with BLAST2GO. Only 9,266 of the 25,670 A. chinensis antennal unigenes (36.1%) were assigned to specific GO terms within the top-level categories ‘biological process’, ‘cellular component’, and ‘molecular function’. The most highly represented terms within these categories were ‘metabolic process’, ‘cellular process’, and ‘single-organism process’. In each of the three GO categories, cell aggregation, metallochaperone activity, and virion part were minimum (Fig. 1).
Identification of putative OBPs
We identified 46 transcripts encoding putative OBPs in A. chinensis antennal transcriptomes. Among these genes, 23 had complete open reading frames (ORFs) ≥ 400 bp that included sequences encoding signal peptides. Based on the number and location of the conserved cysteines, 12 of the 23 full-length AchiOBPs lacked the conserved cysteines C2 and C5 (see supplementary Figure S2). AchiOBP3 have additional cysteines located downstream of the conserved C6 residue in addition to the six conserved cysteines (see supplementary Figure S3). Two of these proteins were identified as PBPs (see supplementary Table S3). Remarkably, using BLASTx to analyze gene homology, we found that the sequence identity between PBP1 from A. chinensis (AchiPBP1) and PBP1 from A. glabripennis (AglaPBP1) is 100%. Almost all of the matching genes encoded OBPs (or PBPs), but AchiOBP16 showed similarity with the gene from B. horsfieldi that encodes Minus-C odorant-binding protein 2. More than one A. chinensis gene exhibited a best match with the same species and gene sequence in the database. For example, four unigenes (c13216.graph_c0, c13631.graph_c0, c18867.graph_c0, and c24613.graph_c1) exhibited best matches with A. glabripennis ARH65462.1 (see supplementary Table S3).
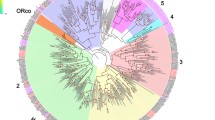
The FPKM (Fragments Per Kilobase of transcript per Million mapped reads) values indicated that AchiOBP11 had the highest expression in the female antennae, whereas AchiOBP27 had the highest expression in the male antennae. A phylogenetic tree shows the evolutionary relationships between insect OBP genes (Fig. 2). AchiPBP1 and AchiPBP2 have high homology with AglaPBP1 and AglaPBP2, respectively. AchiOBP3, which encodes a protein with two more cysteine residues than other A. chinensis OBPs, clustered with Plus-C OBPs, including those from Ips typographus, whereas 9 OBP genes (AchiOBP2, AchiOBP5, AchiOBP8, AchiOBP12, AchiOBP25, AchiOBP30, AchiOBP32, AchiOBP33, and AchiOBP41) that encode proteins lacking two cysteine residues formed part of a Minus-C clade (Fig. 2).
Neighbor-joining phylogenetic tree of candidate odorant-binding proteins (OBPs) The tree was constructed with MEGA5.0 with bootstrap support based on 1,000 replications, and only bootstrap values ≥0.5 are shown at the corresponding nodes. The scale bar represents 2.0 substitutions per site. A. chinensis sequences are in red, and the protein names and sequences of the 165 OBPs that were used in this analysis are listed in S8 Table.
Identification of putative CSPs
Among 16 putative CSP transcripts, five sequences had full-length ORFs (Table 1). The sequences of 11 A. chinensis CSPs had >80% similarity with other species’ CSPs. The FPKM values indicated that AchiCSP11 had the highest expression in the female antennae, whereas AchiCSP10 had the highest expression in the male antennae. The A. chinensis CSPs were scattered in the phylogenetic tree, and most clustered with AglaCSPs (see supplementary Figure S4).
Identification of putative ORs
Among 44 putative OR genes identified in the A. chinensis antennal transcriptomes, 10 unigenes had full-length ORFs encoding proteins longer than 328 amino acids (see supplementary Table S4). Three of the sequences (AchiOR11, AchiOR39, and AchiOR42) encoded seven-transmembrane-domain proteins. AchiOR10, AchiOR24, and AchiOR41 all matched Colaphellus bowringi ALR72551.1, and AchiOR9, AchiOR22, and AchiOR25 all matched C. bowringi ALR72569.1. Notably, AchiOR35 had 97% similarity with M. alternatus AIX97092.1, and had the highest level of expression in antennae (both male and female), according to FPKM. In a phylogenetic tree, AchiOR35 clustered in the Orco clade (Fig. 3).
Neighbor-joining phylogenetic tree of candidate odorant receptors (ORs) The tree was constructed with MEGA5.0 with bootstrap support based on 1,000 replications, and only bootstrap values ≥ 0.5 are shown at the corresponding nodes. The scale bar represents 2.0 substitutions per site. A. chinensis sequences are in red, and the protein names and sequences of the 155 ORs that were used in this analysis are listed in S8 Table.
Identification of putative IRs
Among 23 putative IR genes identified in the antennal transcriptomes, four (AchiIR6, AchiIR9, AchiIR10, and AchiIR19) had complete ORFs encoding ≥567 amino acids, indicating they were intact full-length unigenes (see supplementary Table S5). The BLASTX searches indicated that AchiIR4, AchiIR5, and AchiIR10 all matched Phyllotreta striolata ANQ46493.1. The FPKM values indicated that AchiIR10 had the highest expression in both male and female antennae, whereas there was no identifiable expression of AchiIR18 or AchiIR22 in female antennae. The IR phylogenetic tree showed segregation between D. melanogaster genes and others. AchiIR15 clustered with IR8a sequences from other insects, and AchiIR11 clustered with IR25a sequences (Fig. 4).
Neighbor-joining phylogenetic tree of candidate ionotropic receptors (IRs). The tree was constructed with MEGA5.0 with bootstrap support based on 1,000 replications, and only bootstrap values ≥ 0.5 are shown at the corresponding nodes. The scale bar represents 2.0 substitutions per site. A. chinensis sequences are in red, and the protein names and sequences of the 125 IRs that were used in this analysis are listed in S8 Table.
Identification of putative GRs
Among 19 putative GR genes, four (AchiGR2, AchiGR6, AchiGR11, and AchiGR19) had complete ORFs encoding ≥357 amino acids, indicating they were almost full-length genes (see supplementary Table S6). AchiGR2 and AchiGR11 encoded seven-transmembrane-domain proteins. All of the AchiGRs had low levels of expression in the antennae of both females and males. A phylogenetic analysis suggested that the highly similar AchiGR2, AchiGR6, AchiGR8, and AchiGR11 were members of the sugar-receptor subfamily, whereas AchiGR4 was related to known CO2-receptor genes (Fig. 5).
Neighbor-joining phylogenetic tree of candidate gustatory receptors (GRs). The tree was constructed with MEGA5.0 with bootstrap support based on 1,000 replications, and only bootstrap values ≥ 0.5 are shown at the corresponding nodes. The scale bar represents 2.0 substitutions per site. A. chinensis sequences are in red, and the protein names and sequences of the 106 GRs that were used in this analysis are listed in S8 Table.
Identification of putative SNMPs
Among three putative AchiSNMP genes in the antennal transcriptomes, AchiSNMP1 and AchiSNMP3 had complete ORFs encoding 529 and 561 amino acids respectively and were full-length genes (see supplementary Table S5). The SNMP genes clustered into two clades, with AchiSNMP1 and AchiSNMP3 in the SNMP1 family, and AchiSNMP2 in the SNMP2 family (Fig. 6).
Neighbor-joining phylogenetic tree of candidate sensory neuron membrane proteins (SNMPs). The tree was constructed with MEGA5.0 with bootstrap support based on 1,000 replications, and only bootstrap values ≥ 0.5 are shown at the corresponding nodes. The scale bar represents 0.08 substitutions per site. A. chinensis sequences are in red, and the protein names and sequences of the SNMPs that were used in this analysis are listed in S8 Table.
Homologous CSPs in A. chinensis and A. glabripennis
Sequence comparison between A. chinensis and A. glabripennis identified 12 pairs of homologous CSPs whose sequence similarities were higher than 90% (Fig. 7B). Four CSPs (AchiCSP4, AchiCSP5, AchiCSP6, and AchiCSP14) were A. chinensis-specific (Fig. 7A). The alignment length of CSPs from A. chinensis and A. glabripennis was longer than 400 bp, with fewer than 25 bp of mismatches (see supplementary Table S7).
Pairwise comparison of the CSPs from A. chinensis and A. glabripennis. A: neighbor-joining tree of the CSPs from A. chinensis and A. glabripennis. The tree was constructed with MEGA5.0 with bootstrap support based on 1,000 replications. B: sequence identity percentages of the homologous CSPs from the two beetles.
Tissue expression analysis of CSPs and PBPs
We verified AchiCSP and AchiPBP expression in antennae and surveyed the expression patterns of 16 AchiCSPs and two AchiPBPs in three chemosensory tissues (antennae, legs, and maxillary palps) in both females and males using RT-PCR (Fig. 8). The bands of five CSPs (AchiCSP1, AchiCSP8, AchiCSP9, AchiCSP11, and AchiCSP16) were relatively intense not only in antennae, but also in the other tissues. Although the bands of AchiCSP2, AchiCSP3, AchiCSP4, AchiCSP5, and AchiCSP13 were very weak or undetectable, this may be due to the relatively low expression levels of these genes, which is consistent with their low abundances (FPKM < 10) (Table 1). PBP2 was only expressed in antennae in both females and males.
A. chinensis CSPs and PBPs transcript levels in different tissues as evaluated by RT-PCR. FA, female antennae; MA, male antennae; FL, female leg; ML, male leg; FM, female maxillary palp; MM, male maxillary palp; NC: no template control. GAPDH used as a reference gene for each cDNA template. the similar intensity of GAPDH bands among different tissues indicates the use of equal template concentrations.
To confirm the RT-PCR results, fluorescence quantitative real-time PCR was conducted to determine the expression patterns of CSPs and PBPs in different tissues. In these analyses, expression of all AchiCSP genes in the tested tissues was consistent with the RT-PCR results. AchiCSP10 and AchiCSP12 had higher expression in the antennae than elsewhere, and AchiCSP10 expression in the male antennae was highly significantly different from expression in other tissue types. AchiPBP2 had a significantly higher level of expression in the antennae than in other tissues, and also a significantly higher level of expression in the male antennae than in the female antennae (Fig. 9).
Pheromone-binding protein (PBPs) and Chemosensory protein (CSPs) transcript levels in different tissues of A. chinensis FA, female antennae; MA, male antennae; FL, female leg; ML, male leg; FM, female maxillary palp; MM, male maxillary palp. Fold changes are relative to transcript levels in MM. Bars with different letters are significantly different from each other (a, b, c, d, p < 0.05; A, B, p < 0.01). N/A indicates that the transcript level was too low to measure. NA means the transcript level too low to measure.
Discussion
Olfactory genes have been studied in 20 species of Coleoptera to date, but only in three species of Cerambycidae (M. alternatus 35, B. horsfieldi 19, and A. glabripennis 10). Here, we identified numerous olfactory genes via sequencing using the Illumina Hiseq™ 2500 platform to analyze the antennal transcriptome of A. chinensis as a step toward understanding olfactory processing in this and related species. In the transcriptome sets, we identified 59,357 unigenes with a mean length of 673 bp, a minimum length 201 bp, and a maximum length 10,205 bp, indicating the high quality and depth of sequencing at the transcriptome level. Homology searches against the Nr database showed that A. chinensis antennal transcriptomes share high homology with sequences from T. castaneum (55%) and D. ponderosae (16%), possibly due in part to the identification of many genes, including olfactory genes, in the T. castaneum 32 and D. ponderosae 36 genomes.
We identified 46 candidate OBP gene sequences, 16 CSPs, 44 ORs, 19 GRs, 23 IRs, and 3 SNMPs from the antennae of A. chinensis. By comparison, 40 OBPs, 12 CSPs, 37 ORs, 11 GRs, 4 IRs, 2 SNMPs, and 1 ODE were previously identified in A. glabripennis 10. We did not identify any ODE genes, suggesting that they are lowly expressed in A. chinensis and that some are species-specific. Functional characterization of these genes from the two species may help to explain host selection. Evidence of the possibility requires us further functional research of two species in future. Our results contribute to the understanding of the vital role of olfactory proteins and olfactory processing in A. chinensis and related species.
An OBP typically has from 120 to 150 amino acids and a mass of approximately 14 kDa, including a signal peptide37, and 23 putative A. chinensis OBPs conformed to these criteria. A phylogenetic tree grouped OBP sequences into several categories. Classic OBPs, which include the PBP-GOBP group, are characterized by a conserved six-cysteine-residue pattern. AchiOBP3 encodes a protein with two additional cysteine residues and one proline, making it the Plus-C OBP of A. chinensis, which grouped together with the Plus-C OBPs from other insect species. Another 9 AchiOBPs were missing two of the conserved cysteines (C2 and C5), and were members of the Minus-C class. The Plus-C and Minus-C subfamilies may contribute to the diverse roles of OBPs in different biological processes38. Most of the AchiOBPs had a high level of homology with AglaOBPs, and as the closely related species A. chinensis and A. glabripennis have the same host trees, we speculate that these OBPs recognize the same volatiles that are emitted by the hosts. Notably, AchiPBP1 had 100% identity with AglaPBP1, and AchiPBP2 had 96% identity with AglaPBP2, and these genes had the same expression profiles in the two insect species. Both A. chinensis and A. glabripennis contain 4-(n-heptyloxy) butanol in the attractant pheromones produced by the males, and the high level of similarity between the AchiPBPs and AglaPBPs suggests that they capture the same pheromones. AchiPBP2 had a high level of expression in the antennae, suggesting a putative role for this protein in the detection of the female sex pheromones, as with PBPs from Cydia pomonella and Hyphantria cunea 39,40, and as with AglaPBP2. AchiPBP1 was equally expressed in both sexes (similar to PxylPBP2 of Plutella xyllotella 41). Female moths are capable of detecting and monitoring both female and male sex pheromones42,43. AchiPBP1 expression in females suggests that it might be associated with recognition of male-produced pheromone. In female A. glabripennis, five sex-pheromone components have been identified: (Z)-9-tricosene; (Z)-9-pentacosene; (Z)-7-pentacosene; (Z)-9-heptacosene; and (Z)-7-heptacosene44. In female A. chinensis, different sex-pheromone components have been identified: (Z)-18-heptacosen-10-one; (18Z,21Z)-heptacosa-18,21-dien-10-one; and (18Z,21Z, 24Z)-heptacosa-18,21,24-trien-10- one6,7. The mechanism by which PBPs with similar structures distinguish a variety of pheromone components is not yet known, and the function of A. chinensis PBPs should be studied further to determine their roles in pheromone recognition.
The 16 CSP genes predicted in the A. chinensis transcriptome is more than the 12 CSP-encoding genes identified in A. glabripennis, but less than the 19 genes in M. alternatus and D. helophoroides 45, or the 20 genes in T. castaneum 32. In a phylogenetic analysis, all CSPs of D. melanogaster were contained in a single clade, whereas those of A. chinensis and A. glabripennis were divided into several different branches, as has previously been observed for the CSP genes of Mamestra brassicae 10,46. CSPs are more highly conserved than OBPs, and are widely expressed in different parts of the insect body. AchiCSP12 clustered with AglaCSP7; both genes are highly expressed in the antennae and could be involved in chemoreception. AchiCSP10 was almost exclusively expressed in male antennae, and similarly biased expression has previously been observed for SinfCSP19 of Sesamia inferens 47. Palps are primarily responsible for sensing the trail pheromone in A. glabripennis 48. Similar to AglaCSP3, AglaCSP6, AglaCSP10, and AglaCSP12. expression of AchiCSP1, AchiCSP7, AchiCSP11, and AchiCSP14 was enriched in maxillary palps, which might be related to the delivery of these semiochemicals. CSPs are likely involved in carrying semiochemicals in the moth M. brassicae 49. In addition, some CSPs were highly expressed in the legs of A. chinensis, and might participate in other physiological processes besides chemoreception in this species. AchiCSP1, AchiCSP8, AchiCSP10, AchiCSP11, AchiCSP16, and their homologous genes (AglaCSP5, AglaCSP1, AglaCSP9, AglaCSP2, and AglaCSP6) were highly expressed. We can test the functions of these homologous gene pairs, which avoids the high cost associated with functional analysis of every CSP as well as the bias and incompleteness associated with the random selection of CSPs.
A total of 44 OR genes were identified in the A. chinensis antennal transcriptomes, which is similar to the 43 genes in I. typographus, fewer than the 49 in D. ponderosae 34 and the 57 in M. caryae 33, and considerably fewer than the 110 in T. castaneum 50, but more than the 37 in A. glabripennis 10 and the 20 in Tenebrio molitor 17. In a phylogenetic analysis, AchiOR35 clustered with Orco genes51, particularly those encoding Orco orthologues in T. castaneum. AchiOR35 could be the Orco in A. chinensis. A total of 23 candidate IR genes were identified. Phylogenetic analysis suggests that AchiIR15 and AchiIR11 are the IR8a and IR25a genes, respectively, of A. chinensis. These IR subtypes have essential roles in tuning IR sensory cilia targeting and IR-based sensory channels52.
SNMPs belong to two subfamilies, SNMP1 and SNMP253,54. In a phylogenetic analysis, AchiSNMP1 and AchiSNMP3 clustered into the SNMP1 subfamily, and AchiSNMP2 clustered into SNMP2. Members of the insect SNMP1 subfamily are expressed in the pheromone-sensitive ORNs; however, the SNMP2 proteins are expressed in the supporting cells, rather than the ORNs54. We also identified 19 GR genes, which is more than the 11 identified in the A. glabripennis antennal transcriptome10, the six identified in I. Typographus, and the two identified in D. Ponderosae 34. A phylogenetic analysis suggested that AchiGR4 might have a role in CO2 detection, whereas four AchiGRs clustered with DmelGRs that encode sugar receptors. Sugar recognition is thought to be involved in host-plant selection and egg-laying behavior of codling moth (C. pomonella) females55,56. Further research is required to determine the precise roles of the SNMP and GR proteins in A. chinensis.
Materials and Methods
Insect and tissue collection
Adult A. chinensis (Forster)were collected from host trees (Melia azedarach L.) at Fuzhou, Fujian Province, China, and reared in our laboratories at 25 ± 1 °C. Antennae from two males and two females were excised and stored in RNAlater (Ambion, Austin, TX, USA) at −80 °C until RNA extraction. All operations were performed according to ethical guidelines to minimize pain and discomfort to the insects.
Total RNA extraction, cDNA library construction, and Illumina sequencing
Total RNA was extracted from two female antennae and two male antennae using TRIzol reagent (Ambion) and the RNeasy Plus Mini Kit (No. 74134; Qiagen, Hilden, Germany), following the manufacturers’ instructions. RNA quantity was detected using the NanoDrop 8000 (Thermo, Waltham, MA, USA). RNA of male and female antennae was used to construct the cDNA library respectively. The cDNA library construction and Illumina sequencing of samples were performed at CapitalBio Corporation (Beijing, China). mRNA samples were purified and fragmented using the TruSeq RNA Sample Preparation Kit v2-Set A (No. RS-122-2001; Illumina, San Diego, CA, USA). Random hexamer primers were used to synthesize the first-strand cDNA, followed by synthesis of the second-strand cDNA using buffer, dNTPs, RNase H, and DNA polymerase I at 16 °C for 1 h. and the products were amplified by PCR and quantified precisely using the Qubit DNA Br Assay Kit (Q10211; Invitrogen, Carlsbad, CA, USA). After adenylation of 3′ ends of DNA fragments, NEBNext Adaptor with hairpin loop structure were ligated to prepare for hybridization. In order to select cDNA fragments of preferentially 150 ~ 200 bp in length, the library fragments were purified with AMPure XP system (Beckman Coulter, Beverly, USA). Then 3 μl USER Enzyme (NEB, USA) was used with size-selected, adaptor-ligated cDNA at 37 °C for 15 min followed by 5 min at 95 °C before PCR. Then PCR was performed with Phusion High-Fidelity DNA polymerase, Universal PCR primers and Index (X) Primer. At last, PCR products were purified (AMPure XP system) and library quality was assessed on the HiSeq. 2500 platform.
Assembly and functional annotation
All raw reads were processed to remove low-quality and adaptor sequences by Trimmomatic (http://www.usadellab.org/cms/index.php?page=trimmomatic), Clean reads assembly was carried out with the short-read assembly program Trinity (Version: r2014-04-13) with the default parameters after combined the male and female clean reads. The largest alternative splicing variants in the Trinity results were called unigenes. The annotation of unigenes was performed by NCBI BLASTx searches against the Nr protein database, with an E-value threshold of 1e-5. The blast results were then imported into the Blast2GO pipeline for GO annotation. The longest ORF of each unigene was determined by the NCBI ORF Finder tool (http://www.ncbi.nlm.nih.gov/gorf/gorf.html). The level of expression of unigenes was indicated using FPKM values, which were calculated by RSEM (RNA-Seq by Expectation-Maximization)57.
Identification of chemosensory genes
Putative unigenes involved in olfaction in A. chinensis were confirmed by analysis with the BLASTX program. All candidate OBP, CSP, OR, GR, IR, and SNMP genes were manually checked by tBLASTn in NCBI online.
Sequence and phylogenetic analysis
Amino acid sequences were deduced by analysis on the ExPASy portal (http://web.expasy.org/translate/). Putative N-terminal signal peptides of candidate OBP and CSP proteins were predicted on the SignalP4.1 Server (http://www.cbs.dtu.dk/services/SignalP/). Transmembrane domains of candidate OR, IR, GR, and SNMP proteins were predicted using the TMHMM server v3.0 (http://www.cbs.dtu.dk/services/TMHMM/). After amino acid sequence alignment using ClustalX (1.83), phylogenetic trees were constructed using the neighbor-joining method in MEGA 5.0 with default settings and 1,000 bootstrap replications58, and then the dendrograms were colored and arranged in FigTree v1.4.3. Phylogenetic tree analyses of AchiOBPs, AchiCSPs, AchiORs, AchiIRs, AchiGRs, and AchiSNMPs were based on the amino acid sequences encoded by the putative chemosensory genes, and on sequences identified from other insects. The protein names and gene accession numbers are indicated in supplementary Table S6.
Tissue expression analysis of OBP and CSP genes
The expression patterns of CSP and PBP genes in both male and female tissues (antenna, leg, and maxillary palp) were analyzed by were analyzed by RT-PCR and fluorescence quantitative real-time PCR. Antennae, leg and maxillary palps were collected from four adult A. chinensis of both male and female and one total RNA was extracted following the methods described above. Equal amounts of RNA from each tissue were used to synthesize cDNA by the PrimeScript RT Reagent Kit with gDNA Eraser (No. RR047A; TaKaRa, Shiga, Japan). Gene-specific primers were designed using Primer3 (http://bioinfo.ut.ee/primer3-0.4.0/) (see supplementary Table S7). The RT-PCR was performed under following conditions: 95 °C for 2 min, followed by 35 cycles of 95 °C for 30 sec, 55 °C for 30 sec, 72 °C for 1 min, and a final extension for 10 min at 72 °C. PCR products were analyzed on 1.5% agarose gel and visualized after staining with ethidium bromide. In additional, the PCR products were selected and verified by DNA sequencing. To reach reproducibility, each sample was examined at least three times with three biological samples.
qPCR analysis was performed on the Bio-Rad CFX96 PCR System (Bio-Rad, Hercules, CA, USA). GAPDH from A. chinensis was used as a reference gene. each PCR reaction (25 µl) contained 12.5 µl of SYBR Premix Ex Taq II, 1 µl of each primer (10 mM), 2 µl of sample cDNA (2.5 ng of RNA), and 8.5 µl of dH2O (sterile distilled water). qPCR cycling parameters were 95 °C for 30 s, followed by 40 cycles of 95 °C for 5 s and 60 °C for 30 s, and a final gradient from 65 to 95 °C in increments of 0.5 °C for 5 s each, to generate the melting curves. Negative controls without a template were included in each experiment. To examine reproducibility, each qPCR reaction for each tissue was performed in three technical replicates and three biological replicates. Bio-Rad CFX Manager (version 3.1.1517.0823) was used to normalize expression based on ΔΔCq values, with male maxillary palps in analyze mode as control samples, with relative expression calculated by the 2−ΔΔCT method(the amplification efficiency for 17 genes was equal to 100%)59. The comparative analyses for every gene among six tissue types were assessed by a one-way nested analysis of variance (ANOVA), followed by Tukey’s honestly significance difference (HSD) tests implemented in SPSS Statistics 18.0. Values are presented as means ± SE.
Availability of data and materials
All supporting data is included within the article and its additional files. The raw reads for A. chinensis were submitted in the NCBI SRA database under the GenBank accession number SRP116677; the entire transcriptome assembly were deposited in the NCBI TSA database under the GenBank accession number GFXC00000000; the olfactory protein gene sequences were submitted to Genbank, accession number is MF975370-MF975415, MF975416-MF975431, MF975432-MF975474, MF975475-MF975492, MF975493-MF975514 and MF975515-MF975517.
Ethics approval and consent to participate
The citrus longhorned beetle Anoplophora chinensis (Coleoptera: Cerambycidae) is an important worldwide quarantine pest in China, which collections were made with the direct permission of Fujian forestry bureau. It’s not included in the “List of Endangered and Protected Animals in China”. All operations were performed according to ethical guidelines in order to minimize pain and discomfort to the insects.
Authors’ information
Jingzhen Wang: master candidate; major: forest protection; study direction: insect molecular biology. Ping Hu: PhD candidate; major: forest protection; study direction: insect molecular biology and insect chemical ecology. Peng Gao: PhD candidate; major: forest protection; study direction: insect molecular biology and insect chemical ecology. Jing Tao: PhD, study direction: insect molecular biology. Youqing Luo: The Yangtze River Scholar Professor, study direction: Pest Control and forest ecology.
References
Lingafelter, S. W. & Hoebeke, E. R. Revision of the genus Anoplophora (Coleoptera: Cerambycidae) (2002).
Haack, R. A., Hérard, F., Sun, J. & Turgeon, J. J. Managing Invasive Populations of Asian Longhorned Beetle and Citrus Longhorned Beetle: A Worldwide Perspective. Annual Review of Entomology 55, 521–546 (2009).
Hérard, F. et al. Anoplophora species in Europe: infestations and management processes 1. Bulletin Oepp/eppo Bulletin 36, 470–474 (2006).
Fukaya, M. et al. Effects of male body size on mating behavior and female mate refusal in the white-spotted longicorn beetle, Anoplophora malasiaca (Thomson) (Coleoptera: Cerambycidae). Applied Entomology & Zoology 39, (731–737 (2004).
Hansen, L. et al. Identification of a Male-Produced Pheromone Component of the Citrus Longhorned Beetle, Anoplophora chinensis. Plos One 10, e0145355 (2015).
Wang, Q. Evidence for a contact female sex pheromone in Anoplophora chinensis (Forster) (Coleoptera: Cerambycidae: Laminae). Coleopterists Bulletin 52, 363–368 (1999).
Yasui, H. et al. Ketone components in the contact sex pheromone of the white-spotted longicorn beetle, Anoplophora malasiaca, and pheromonal activity of synthetic ketones. Entomologia Experimentalis Et Applicata 107, 167–176 (2003).
Nehme, M. E. et al. Development and Evaluation of a Trapping System for Anoplophora glabripennis (Coleoptera: Cerambycidae) in the United States. Environmental Entomology 43, 1034 (2014).
Leal, W. S. Odorant reception in insects: roles of receptors, binding proteins, and degrading enzymes. Annual Review of Entomology 58, 373 (2013).
Hu, P., Wang, J., Cui, M., Tao, J. & Luo, Y. Antennal transcriptome analysis of the Asian longhorned beetle Anoplophora glabripennis. Sci Rep 6, 26652 (2016).
Leal, W. S., Nikonova, L. & Peng, G. Disulfide structure of the pheromone binding protein from the silkworm moth, Bombyx mori. Febs Letters 464, 85 (1999).
Krieger, J. & Breer, H. Olfactory reception in invertebrates. Science 286, 720–723 (1999).
Sánchez-Gracia, A., Vieira, F. G. & Rozas, J. Molecular evolution of the major chemosensory gene families in insects. Heredity 103, 208 (2009).
Chang, H. et al. Pheromone binding proteins enhance the sensitivity of olfactory receptors to sex pheromones in Chilo suppressalis. Scientific Reports 5, 13093 (2015).
Wang, R. et al. Identification and expression profile analysis of odorant binding protein and chemosensory protein genes in Bemisia tabaci MED by head transcriptome. Plos One 12, e0171739 (2017).
Prestwich, G. D. & Du, G. Pheromone-Binding Proteins, Pheromone Recognition, and Signal Transduction in Moth Olfaction. (1997).
Liu, S. et al. Identification of candidate chemosensory genes in the antennal transcriptome of Tenebrio molitor (Coleoptera: Tenebrionidae). Comparative Biochemistry & Physiology Part D Genomics & Proteomics 13, 44–51 (2015).
Liu, N. Y., Liu, C. C. & Dong, S. L. Functional differentiation of pheromone-binding proteins in the common cutworm Spodoptera litura. Comparative Biochemistry & Physiology Part A Molecular & Integrative Physiology 165, 254–262 (2013).
Li, H., Zhang, A., Chen, L. Z., Zhang, G. & Wang, M. Q. Construction and analysis of cDNA libraries from the antennae of Batocera horsfieldi and expression pattern of putative odorant binding proteins. Journal of Insect Science 14, 57 (2014).
Jin, X. et al. Expression and immunolocalisation of odorant-binding and chemosensory proteins in locusts. Cellular & Molecular Life Sciences Cmls 62, 1156–1166 (2005).
Maleszka, J., Forêt, S., Saint, R. & Maleszka, R. RNAi-induced phenotypes suggest a novel role for a chemosensory protein CSP5 in the development of embryonic integument in the honeybee (Apis mellifera). Development Genes and Evolution 217, 189–196 (2007).
Xue, W. et al. Identification and Expression Analysis of Candidate Odorant-Binding Protein and Chemosensory Protein Genes by Antennal Transcriptome of Sitobion avenae. Plos One 11, e0161839 (2016).
Benton, R., Sachse, S., Michnick, S. & Vosshall, L. Atypical membrane topology and heteromeric function of Drosophila odorant receptors in vivo - art. no. e20. Plos Biology 4, 240–257 (2012).
Wicher, D. et al. Drosophila odorant receptors are both ligand-gated and cyclic-nucleotide-activated cation channels. Nature 452, 1007 (2008).
Vosshall, L. B. & Hansson, B. S. A unified nomenclature system for the insect olfactory coreceptor. Chemical Senses 36, 497 (2011).
Grosse-Wilde, E., Gohl, T., Bouché, E., Breer, H. & Krieger, J. Candidate pheromone receptors provide the basis for the response of distinct antennal neurons to pheromonal compounds. European Journal of Neuroscience 25, 2364–2373 (2007).
Grosjean, Y. et al. An olfactory receptor for food-derived odours promotes male courtship in Drosophila. Nature 478, 236–240 (2011).
Clyne, P. J., Warr, C. G. & Carlson, J. R. Candidate Taste Receptors in Drosophila. Science 287, 1830 (2000).
Poudel, S., Kim, Y., Yun, T. K. & Lee, Y. Gustatory receptors required for sensing umbelliferone in Drosophila melanogaster. Insect Biochemistry & Molecular Biology 66, 110–118 (2015).
Rogers, M. E., Krieger, J. & Vogt, R. G. Antennal SNMPs (sensory neuron membrane proteins) of Lepidoptera define a unique family of invertebrate CD36-like proteins. Journal of Neurobiology 49, 47 (2001).
Jin, X., Ha, T. S. & Smith, D. P. SNMP is a signaling component required for pheromone sensitivity in Drosophila. Proceedings of the National Academy of Sciences of the United States of America 105, 10996 (2008).
Richards, S. et al. The genome of the model beetle and pest Tribolium castaneum. Nature 452, 949–955 (2008).
Mitchell, R. F. et al. Sequencing and characterizing odorant receptors of the cerambycid beetle Megacyllene caryae. Insect Biochemistry & Molecular Biology 42, 499–505 (2012).
Andersson, M. N. et al. Antennal transcriptome analysis of the chemosensory gene families in the tree killing bark beetles, Ips typographus and Dendroctonus ponderosae (Coleoptera: Curculionidae: Scolytinae). BMC Genomics 14, 198 (2013).
Gao, X. & Wang, M. Q. A cDNA library from the antenna of Monochamus alternatus Hope and binding properties of odorant-binding proteins. Journal of Applied Entomology 139, 229–236 (2015).
Keeling, C. I. et al. Draft genome of the mountain pine beetle, Dendroctonus ponderosae Hopkins, a major forest pest. Genome Biology 14, R27 (2013).
Briand, L., Nespoulous, C., Huet, J. C. & Pernollet, J. C. Disulfide pairing and secondary structure of ASP1, an olfactory-binding protein from honeybee (Apis mellifera L). Journal of Peptide Research 58, 540–545 (2002).
Xu, Y. L. et al. Large-scale identification of odorant-binding proteins and chemosensory proteins from expressed sequence tags in insects. BMC Genomics 10, 632 (2009).
Tian, Z. & Zhang, Y. Molecular characterization and functional analysis of pheromone binding protein 1 from Cydia pomonella (L.). Insect Molecular Biology 25, 769–777 (2016).
Zhang, L. W. et al. Analysis of the Antennal Transcriptome and Insights into Olfactory Genes in Hyphantria cunea (Drury). Plos One 11 (2016).
Sun, M., Liu, Y. & Wang, G. Expression patterns and binding properties of three pheromone binding proteins in the diamondback moth, Plutella xyllotella. Journal of Insect Physiology 59, 46 (2013).
Krieger, J. et al. Genes encoding candidate pheromone receptors in a moth (Heliothis virescens). Proceedings of the National Academy of Sciences of the United States of America 101, 11845–11850 (2004).
Bober, R. & Rafaeli, A. Gene-silencing reveals the functional significance of pheromone biosynthesis activating neuropeptide receptor (PBAN-R) in a male moth. Proceedings of the National Academy of Sciences of the United States of America 107, 16858–16862 (2010).
Hoover, K. et al. Sex-Specific Trail Pheromone Mediates Complex Mate Finding Behavior in Anoplophora glabripennis. Journal of Chemical Ecology 40, 169–180 (2014).
Wang, J. et al. Analysis of chemosensory gene families in the beetle Monochamus alternatus and its parasitoid Dastarcus helophoroides. Comparative Biochemistry & Physiology Part D Genomics & Proteomics 11, 1–8 (2014).
Jacquinjoly, E., Vogt, R. G., Francois, M. C. & Nagnanle, M. P. Functional and expression pattern analysis of chemosensory proteins expressed in antennae and pheromonal gland of Mamestra brassicae. Chemical Senses 26, 833–844 (2001).
Zhang, Y. N., Ye, Z. F., Yang, K. & Dong, S. L. Antenna-predominant and male-biased CSP19 of Sesamia inferens is able to bind the female sex pheromones and host plant volatiles. 536, 279–286 (2013).
Graves, F., Baker, T. C., Zhang, A., Keena, M. & Hoover, K. Sensory Aspects of Trail-Following Behaviors in the Asian Longhorned Beetle. Anoplophora glabripennis. Journal of Insect Behavior 29, 1–14 (2016).
Jacquin-Joly, E., Vogt, R. G., Francois, M. C. & Nagnan-Le, M. P. Functional and expression pattern analysis of chemosensory proteins expressed in antennae and pheromonal gland of Mamestra brassicae. Chemical Senses 26, 833 (2001).
Engsontia, P. et al. The red flour beetle’s large nose: An expanded odorant receptor gene family in Tribolium castaneum. Insect Biochemistry & Molecular Biology 38, 387 (2008).
HS, W., KB, L., S, Z., Y. Z., C. & J, Y. Identification of candidate chemosensory genes by transcriptome analysis in Loxostege sticticalis Linnaeus. PloS one 12, e0174036 (2017).
Abuin, L. et al. Functional architecture of olfactory ionotropic glutamate receptors. Neuron 69, 44–60 (2011).
Rogers, M. E., Steinbrecht, R. A. & Vogt, R. G. Expression of SNMP-1 in olfactory neurons and sensilla of male and female antennae of the silkmoth Antheraea polyphemus. Cell and Tissue Research 303, 433–446 (2001).
Liu, C., Zhang, J., Liu, Y., Wang, G. & Dong, S. Expression of SNMP1 and SNMP2 genes in antennal sensilla of Spodoptera exigua (Hubner). Archives of Insect Biochemistry &. Physiology 85, 114–126 (2014).
Zhang, H. J. et al. Topological and Functional Characterization of an Insect Gustatory Receptor. Plos One 6, e24111 (2011).
Lombarkia, N. & Derridj, S. Resistance of apple trees to Cydiapomonella egg-laying due to leaf surface metabolites. Entomologia Experimentalis Et Applicata 128, 57–65 (2008).
Trapnell, C. et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nature Biotechnology 28, 511–515 (2010).
Saitou, N. N. M. & Nei, M. C. Saitou N, Nei M. The Neighbor-Joining Method-a New Method for Reconstructing Phylogenetic Trees. Mol Biol Evol 4: 406–425. 4, 406–425 (1987).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Acknowledgements
We are thankful to Rong Wang (Fujian Agricultural and Forestry university) and Lili Ren for help in collecting of A. chinensis. We also acknowledge Lu Gan and Kejia Liu for help with the bioinformatic analysis. This work was supported by the Fundamental Research Funds for the Central Universities(No.2016ZCQ07).
Author information
Authors and Affiliations
Contributions
J.Z.W. J.T. and Y.Q.L. conceived and designed the experiments. J.Z.W. and P.H. performed the sample collection. J.Z.W. P.H. and P.G. Performed the experiments. J.Z.W. P.H. and P.G. analyzed the data. P.H. and P.G. contributed reagents /materials/analysis tools. J.Z.W. and J.T. wrote the paper.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, J., Hu, P., Gao, P. et al. Antennal transcriptome analysis and expression profiles of olfactory genes in Anoplophora chinensis . Sci Rep 7, 15470 (2017). https://doi.org/10.1038/s41598-017-15425-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-15425-2
- Springer Nature Limited
This article is cited by
-
Antennal transcriptome analysis of olfactory genes and tissue expression profiling of odorant binding proteins in Semanotus bifasciatus (cerambycidae: coleoptera)
BMC Genomics (2022)
-
Antennal transcriptome analysis of olfactory genes and characterizations of odorant binding proteins in two woodwasps, Sirex noctilio and Sirex nitobei (Hymenoptera: Siricidae)
BMC Genomics (2021)
-
Chemical Ecology of the Asian Longhorn Beetle, Anoplophora glabripennis
Journal of Chemical Ecology (2021)
-
Antennal transcriptome analyses and olfactory protein identification in an important wood-boring moth pest, Streltzoviella insularis (Lepidoptera: Cossidae)
Scientific Reports (2019)
-
Systemic and sex-biased regulation of OBP expression under semiochemical stimuli
Scientific Reports (2018)