Abstract
Reference genes are frequently used as normalizers for expression studies despite not being previously verified to present suitable stabilities. Considering the interest that tiger beetles have generated in the past years, resulting in a variety of studies, it is crucial to dispose of a validated reference gene panel for expression studies. Nine candidate genes were tested in Cicindela campestris and Calomera littoralis across several conditions and their transcription levels were assessed with geNorm, NormFinder, BestKeeper and ΔCT method algorithms. Results showed high stabilities across sexes, immune challenge and gonad developmental stages for all genes tested, while body parts comparison presented less constant expression values. Only two genes are sufficient to perform a proper normalization for most of the conditions tested, except for the body parts comparison in C. littoralis, which requires the use of at least three reference genes. On the whole, no universal gene is found to be suitable for all situations, but according to the acceptable range of values, NADH, B-t, Vatpase and ArgKin seem to present the most constant expression stability, indicating their suitability as reference genes in most of the conditions. This is the first report evaluating the stability of housekeeping genes in adephagan beetles.
Similar content being viewed by others

Introduction
Evaluating changes in gene expression has lately been one of the most frequent studies to analyse the effect of physiological responses and therefore the biological differences among populations. Quantitative real-time polymerase chain reaction (RT-qPCR) has recently become the most widely accepted methodology which accurately and with high reproducibility generates expression profiles and allows their interpretation1,2,3,4. Nevertheless, some guidelines must be followed to correctly perform a reliable RT-qPCR analysis5,6,7,8, being essential the use of an internal control so that any other variation across samples derived from handling or cDNA synthesis can be avoided3,7,8,9,10,11. The normalization is then carried out by introducing reference genes known to present a constant expression profile after alterations in a specific condition. Many housekeeping genes (HKG) have been incorrectly described and used as reference genes without previously testing their viability due to the assumption that their role in basic cellular processes grants them a complete and suitable stability. However, it has been established that most of these genes can alter their expression under certain conditions12,13,14,15,16, thus it is crucial to perform a proper validation to determine the suitability of any candidate reference gene for each specific condition.
Several studies validating reference genes have been carried out in the last years, some of them involving insect species. Validation in Bemisia tabaci17 selected genes for biotic and abiotic conditions. Ponton et al.18 checked the suitability of references genes in Drosophila melanogaster after an injury, heat stress and different diets, and Koramutla et al.19 recommended several reference genes in Lipaphis erysimi. Additionally, gene stability was tested in different conditions in two Bombus species20, while Lourenço et al.21 studied the suitability in Apis mellifera. More specifically, reference genes validation has also been carried out in polyphagan beetles. That is the case of Tribolium castaneum22,23,24, for which different ribosomal proteins were found to be the suitable for broad-spectrum expression analyses. Two different species of Coccinellidae25,26, the western corn root worm Diabrotica virgifera virgifera27 and the emerald ash borer Agrilus planipennis28 have also been investigated.
Cicindelids, commonly known as tiger beetles, are one of the most important non-pest species within the suborder Adephaga (Coleoptera), with more than 2500 species described29. They are worldwide distributed with the exception of Tasmania, Antarctica and some remote Oceanic islands, occupying a wide range of habitats30,31, and are considered to be good bioindicators due to their biological and evolutionary characteristics30,32,33,34. Moreover, the genomic study of non-model species, especially with the modern technologies currently available, provides important information to understand the genetic basis of adaptive traits35. Nevertheless, only a few studies have been performed in cicindelids regarding the study of their transcriptome, in relation to their reproduction and innate immune response36,37,38 but there is no information available concerning the stability of HKGs in this group of beetles. The availability of a selection of validated reference genes for genomic and expression analyses will allow us to obtain more reliable results in future studies.
Nine candidate reference genes were evaluated across different conditions in Cicindela campestris and Calomera littoralis in this study, including Arginine Kinase (ArgKin), Myosin regulatory light chain 2 (Myo), Myosin light chain alkali (Myo-Alc), 60 s acidic ribosomal p0 (60Sa), Ribosomal protein S18 (RPS18), Tubulin beta-1 chain (B-t), Vatype proton atpase 16kda proteolipid subunit (Vatpase), NADH dehydrogenase (NADH) and 40 s ribosomal protein s11 (RPS11). The aims of this research are (i) to examine the expression stability of different housekeeping genes in non-model beetle species across sexes, body parts, gonad developmental stages (immature/mature) and immune challenge (ii) to provide a selection of accurate reference genes for future genomic and expression analyses in cicindelids.
Results
Selection of reference genes
Nine genes were identified as putative reference genes according to blast searches from both libraries: (i) Arginine Kinase (ArgKin), Myosin regulatory light chain 2 (Myo), Myosin light chain alkali (Myo-Alc) and 60 s acidic ribosomal p0 (60Sa) were obtained from C. littoralis; (ii) Ribosomal protein S18 (RPS18), Tubulin beta-1 chain (B-t), Vatype proton atpase 16kda proteolipid subunit (Vatpase), NADH dehydrogenase (NADH) and 40 s ribosomal protein s11 (RPS11) were identified from C. campestris.
All genes amplified a single fragment with the predicted length (Fig. 1), and were tested for amplification efficiencies in both species to check their stability in expression analyses. Efficiencies ranged between 93.9% and 104.8% in both species (Fig. 2; Table 1), except for RPS11 in C. littoralis and 60Sa in C. campestris which did not amplify correctly with efficiencies outside the acceptable range to be considered as viable for RT-qPCR analyses, and thereby they were discarded for each corresponding species. Melt curves showed the expected single peaks indicating the specificity of the amplifications (Fig. 3).
Agarose gel showing the fragments amplified with all the primers selected for qPCR analyses. All fragments showed predicted lenghts for both species (lit: C. littoralis; cam: C. campestris). Gel is presented uncropped and no posterior image edition and modification in exposure or contrast were made.
Stability of the candidate reference genes
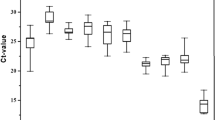
Eight candidate reference genes in each species were analysed by RT-qPCR to validate their stability and therefore their suitability for expression analyses under different conditions. CT values obtained were used as input data for stability analysis with all the four tools. geNorm and NormFinder uses Ct values transformed to logarithmic scale adjusted to the minimum value for each condition, while Bestkeeper and ΔCT method use raw data. All results and stability ranks are shown in Table 2 and Table 3.
geNorm
Two different values are obtained from geNorm analyses. The average expression stability (M) establishes a ranking of genes based on those with the lowest values, considered as the most stable genes for each specific condition. On the other hand, the pairwise variation (V) provides information on the number of genes needed to perform an accurate normalization in expression analyses, and values should be below 0.15 to accept their reliability (Fig. 4).
Optimal number of reference genes for normalization in Cicindela campestris and Calomera littoralis. Pairwise variation Vn/n+1 values of candidate reference genes for the different conditions tested in both tiger beetles species were calculated with geNorm software by analysing the parwise variation (V) between the normalization factors NFN and NFN+1. Values < 0.15 are indicating that additional genes are not necessary for normalization.
In C. campestris, the highest ranked genes for sex differences were B-t and NADH, and Myo was the gene with the lowest stability. However, all studied genes present values below 1 and their stability was very similar for all of them. Results are similar for the gonad development experiment, where NADH and RPS18 are the most stable genes and Myo-Alc the gene with the highest value. Again, all genes showed values below 1. Alternatively, body parts comparison generated values above 1, with Vatpase as the most stable gene and Myo and Myo-Alc presenting values over 1.5, being considered the upper limit for which a gene is not behaving in a stable way. Additionally, Fig. 5 show the stability values of the remaining reference genes at each step after a stepwise exclusion analysis, which showed slightly different results.
C. littoralis differences between sexes showed high stability among all genes tested (M < 1), especially in B-t and Myo-Alc, similar to those values obtained for the infection treatments, being Myo and NADH the genes with the most constant expression. Nevertheless, four of the genes tested were located outside the acceptable values of M for the body parts comparison, with only ArgKin, Vatpase, 60Sa and NADH below 1.5 values.
Regarding pairwise variation analyses, results showed that only two genes were necessary for most of the conditions studied, with the exception of the body parts comparison in C. littoralis, where the analysis is unable to provide an optimal number of genes to normalize the expression of target genes, even when six or seven genes were included in the estimations.
Normfinder
Those genes with the lowest stability values obtained with NormFinder algorithm, based on intra- and inter-group expression variations, are considered to be the most stable reference genes.
Results in C. campestris were equivalent to those generated by geNorm. B-t is established as the most consistent gene and Myo presented the highest stability value for the sexes comparison. Similarly, RPS18 had the lowest value for the gonad development experiment and Myo-Alc was the less stable. Both experiments generated proper stability values for all genes tested. Results for body parts comparison slightly differ from geNorm output. While Myo-Alc was again located as the less constant gene, NADH showed the highest stability, with Vatpase in second place.
All stability values obtained between sexes and infection experiment in C. littoralis were below 0.3 and close to each other, with Myo-Alc and NADH as the most stables genes, respectively. NormFinder considered 60Sa to be the best reference gene for body parts comparisons, even though values were generally higher than previously reported.
Bestkeeper
The Bestkeeper algorithm ranks the candidate references genes by calculating a std dev [±CP] which should not exceed 1 to validate the stability of a gene.
In C. campestris, Vatpase showed the lowest value when comparing between male and female individuals. Similarly, all genes presented values below 1 for the gonads development comparison, where RPS11 was the most stable gene. RPS11 was also considered as the most stable gene for the body parts comparison.
ArgKin and Myo share the position as the most consistent genes for the sex comparison in C. littoralis, and Vatpase led the reference genes in the infection experiment. While all genes in those two experiments presented values below 1, the opposite occurs when analysing values for the body parts comparison, where all genes showed values above 1. B-t and 60Sa displayed the closest values to 1 and therefore the ones with a higher stability.
ΔC T method
This method uses the standard deviation means of the CT differences between each gene and the rest of them within each condition (Fig. 6). A standard deviation below 1 indicates an appropriate stability.
ΔCT approach to reference gene selection in Cicindela campestris (A–C) and Calomera littoalis (D–F) for each condition tested. ΔCT values are indicated as medians (horizontal lines), 25th to 75th percentile (boxes) and ranges (whiskers). Values in x-axis indicate the means of the standard deviations for each gene comparison. The lowest values correspond to the highest stabilities.
Both sex experiment and gonad development in C. campestris presented all genes with values below 1, but B-t and Myo-Alc were the genes with lower stdv mean, respectively. Contrarily to the previous methods, none of the genes were stable enough for a body part comparison according to ΔCT method. But, for all that, RPS11 with a value of 1.07 and Vatpase with 1.08 were the closest to 1 and therefore the most stables genes.
For C. littoralis the most proper gene for a sex comparison was Myo-Alc, while Myo obtained the lowest value for infection analyses. None of the genes in both analyses showed values above 1. In this case, body parts comparison identified several stable genes, being 60Sa the one with a lower value.
By and large, all four methodologies showed similar results, with slightly variation in some gene positions, especially for the body parts comparison experiment, which took into account four different variables and generated some differences among methods and lower stabilities, mainly those obtained with Bestkeeper and ΔCT method
qPCR experiment
An ubiquitin and defensin genes were tested to check their expression using as reference genes those previously validated in this work (Fig. 7). Changes in expression were observed among different conditions, specially for the body parts comparison, where gonads showed a higher ubiquitin expression than other tissues as thorax or abdomen. For the case of the defensin gene, an over-regulation of almost 3 fold-change in its expression was observed after infection, but no significance was found after the statistical analysis. Sex comparison and gonad development presented a less ubiquitin regulation than other conditions.
Relative expression of Clit-Def gene (control and infected samples) and UBC9 gene (rest of the samples) using the reference genes selected for each condition in both species of tiger beetles. Male, mature, control and head samples are used as normalizer for each experiment. Error bars represent the Standard Error for the Normalized Relative Quantity. Letters are indicating significant differences with a p value < 0.05, while asterisks indicate p values < 0.01.
Discussion
Performing a proper reference gene validation for RT-qPCR experiments is crucial for a correct reliability of the results. Thus, stability of reference genes must be proved for each condition and species used in an experiment to assure an accurate data interpretation7,14.
In this study, nine candidate reference genes are tested for the first time in the tiger beetles species C. campestris and C. littoralis across various conditions. Most stable genes are identified to be used in RT-qPCR for comparisons across sexes, gonads development stages, infection and body parts, using four statistical methods such as geNorm, NormFinder, Bestkeeper and ΔCT method39,40,41,42. There are several studies regarding the validation of reference genes in other insects, but no information has previously been reported in any adephagan beetle species.
All genes tested for the sex comparison in both species showed low stability values to consider them suitable to be used as reference genes. Nevertheless, B-t presented the best values in C. campestris for this condition. Even when the rest of conditions never showed this gene as the most constant it is still inside the acceptable range of stability. B-t is one of the major components of microtubules which function in many cellular processes. Previous works in insects have shown its suitability for developmental stages, tissues and dietary RNAi experiments in Coccinella septempunctata26, developmental stages in Liposcelis Bostrychophila43 and across all the conditions tested in Drosophila melanogaster18, while it has also been tested in Tribolium castaneum24, Bemisia tabaci17, Dibrotica virgifera virgifera27, Apis mellifera44 and two Bombus species20, not being ranked as the optimal gene in most of the cases. Even though Myo and Myo-Alc, related to ATP regulation and cell movement, were allocated as the best genes for sex comparison in C. littoralis, they were proved to be the least recommended genes for most of the conditions tested. That is equivalent to the results obtained by Li et al.17 for biotic and abiotic conditions in Bemisia tabacii. Myo was also considered as a putative reference gene after a viral infection in Spodoptera frugiperda cells45 which is in agreement with the results obtained in this study, but it was discarded due to its high CT values.
Some of the most used genes in identification of reference genes studies are the ribosomal proteins, which are involved in the cellular process of translation. Three of them have been tested in this work but only RPS18 was used for both species because RPS11 primer efficiencies in C. littoralis were not good enough to allow its use in expression studies, and exactly the opposite case occurred for 60Sa. Their best stability is observed in the gonads development comparison in C. campestris as both RPS18 and RPS11 were top-ranked by all four algorithms. RPS11 was ranked among the most stables genes for the body parts comparison in C. campestris, contrary to RPS18 which was not placed among the top-ranked genes in either both species. Previous studies in insects showed good stabilities of ribosomal proteins in developmental and tissue comparisons23,28,46,47,48. Nevertheless, RPS18, which was the most constant gene after bacterial infection in Apis mellifera44, showed the least stable values in C. littoralis after the treatment. This highlights the necessity of a proper validation to find the optimal reference gene for each species and specific condition. Additionally, several ribosomal proteins were also validated in T. castaneum after a fungal challenge22, RPS9 was proved to be suitable for RNAi experiments27, and RPS18 and RPL13 were selected among the best reference genes in the mustard aphid Lipaphis erysimi19. Lastly, 60Sa was only used for C. littoralis conditions and its best stability value is shown for body parts comparison.
NADH plays a key role in cellular respiration and was selected as one of the most stable genes across all conditions tested by most of the methods used. It is one of the best genes together with the ribosomal proteins for gonad development comparison and was considered by geNorm and NormFinder as the most constant gene in C. littoralis after the infection and for the body parts comparison. Despite of not being the top-ranked gene in the other conditions tested, it still presents good stability values within the acceptable range. Li et al.17 verified its usefulness in Bemisia tabaci for abiotic factor, but it also presented good values for some biotic factor such as developmental stages. In addition, NADH was top-ranked as one of the most stables gene for developmental stages and tissue comparison in Coccinella septempunctata26 and exhibited acceptable values in Coleomegilla maculata25.
ArgKin, known to function as an energy and ATP regulator, has not been a common reference gene in previous studies, some of them not presenting it as a top-ranked gene for expression analyses in insects25,26,47. Alternatively, it was found to be suitable for studies in two Bombus species by Horňáková et al.20. In this study, it was neither ranked as the most stable gene for any of the conditions tested nor among the worst. It displayed a constant stability across all treatments with most of its values within an acceptable stability range, which may be indicating that, not being the best gene for any condition tested, it could be used as reference genes in several situations along with other validated genes. A similar situation occurs in the case of Vatpase, protein that acts as a proton pumb in cell membranes. Its best stability is displayed for body parts and sex comparisons in C. campestris but most of the time is ranked in the middle of the gene distribution. Previous studies in Bicyclus anynana49, Coleomegilla maculate25 and Coccinella septempunctata26 support these results, as Vatpase was never positioned among the most constant genes.
Stability values for both species body parts comparisons were considerably higher than those obtained for the rest of conditions. Moreover, no genes were obtained which presented acceptable values according to ΔCT method in C. campestris, and that was also the case for C. littoralis with BestKeeper results. That is likely due to the complexity of all body parts and tissue, which were divided into head, thorax, abdomen and gonads, each of them composed by different tissues which could be generating a higher variability in the expression levels of all genes.
Regarding the optimal number of genes for normalization in expression experiments, the results from the pair-wise variation of the normalization factors (NFN and NFN+1) analysis in geNorm highlighted the need of at least two different genes for most of the conditions tested. The exception was the body parts comparison experiment in C. littoralis, for which no value was obtained below the Vn/n+1 0.15 cut-off, even when the number of genes considered for the analysis was increased. For that case, according to the geNorm manual, the recommended number of genes for normalization should be three, as has previously considered by other authors such as Lu et al.47 or Koramutla et al.19. On the other hand, Ling & Salvaterra50 pointed that the optimal number of reference genes should be given according to the minimal Vn/n+1 value, which in this case also corresponds to three genes.
All reference genes selected after these analyses were tested in an actual qPCR experiment to complete their validation. A defensin gene was tested in C. littoralis after an infection, which seemed to increase its expression as it was showed in a previous work37, being in accordance to other studies performed for defensin genes in insects51,52. On the other hand, the ubiquitin UBC9 was tested for the rest of the conditions in both species, showing significant differences in the body parts comparison, primarily in gonads, as was previously checked in a different work38 and in other insects such as Drosophila53, which confirmed the relation of this gene with cicindelids reproductive system.
On the whole, this work identifies nine candidate reference genes and validates their suitability as normalizers in expression studies in two different species of tiger beetles, and later used in a relative expression experiment with two target genes which showed variation in their expression. As has previously concluded in several studies, this work proves the absence of a single and universal reference gene for both species and all conditions tested, and gene validations must be performed for every species and experiment before any expression analysis. Based on the results obtained with all four algorithms, the recommended genes are displayed in Table 4. Nevertheless, some genes seem to present a better stability across samples and species, and in most of the cases all genes showed values within the acceptable range of stability, considering them suitable as well. That is the case of NADH and, to a lesser extent, ArgKin, Vatpase and B-t. Other genes were top-ranked for some specific conditions but were discarded for other, such as RPS18 or Myo-Alc for gonad development and sex comparison, respectively. This work is the first reference gene validation in cicindelids, an ecologically important group of predaceous beetles, and provides useful information to perform future genomic studies in other coleopteran species, either based on the concrete results of selected genes or on the procedures to carry out a proper validation to perform reliable expression studies.
Methods
Insects
All individuals used in this study were captured directly from field, and subsequently treated, harvested and stored for experimental analyses. C. campestris imagoes were collected from Laguna del Arquillo (Albacete, Spain) while C. littoralis were collected from Laguna de Pétrola (Albacete, Spain).
Total RNA extraction and cDNA synthesis
Three different conditions were tested for C. campestris: (i) male and female beetle comparisons; (ii) four different body parts including head, thorax, abdomen and gonads; (iii) developmental stage of their gonads, discerned based on the description reported by Paarmann54 and previously used by Rodríguez-García et al.38. The first two conditions were also tested for C. littoralis, with an additional new condition: infection of beetles with 1 μl of 1 mg/ml lipopolysaccharides (LPS) of Escherichia coli as previously performed by Rodríguez-García et al.37 and harvested after 12 hours of infection. Control samples were injected with 1 μl of insect saline solution. Each condition was replicated three times and each replicate was pooled with 3 different individual or body parts.
Insects and tissues dissected were stored in RNAlater (Qiagen, Crawley, UK) at −20 °C until RNA isolation. Total RNA from samples was extracted using TRIzol reagent (Life Technologies) following the manufacturer’s protocol, and isolated RNA was treated using a TURBO DNA-free Kit (Ambion, Life Technologies) to remove DNA contamination. RNA presence and quality was checked on a 1.5% agarose gel and quantified by NanoDrop 1000 from Thermo Fisher Scientific® (Waltham, MA, USA). First strand cDNA was obtained using PrimeScriptTM RT Reagent Kit (Takara Bio Inc., Japan) and store at −20 °C.
ESTs analysis and selection of candidate HKGs
ESTs libraries from C. campestris55, C. littoralis36 and other unpublished sequences from C. littoralis were analysed in order to identify putative HKGs. Blast searches and contig annotations performed with Blast2go v2.5.0 software56 revealed nine genes known to function as housekeeping genes in other insect species, all of which were selected for further analyses.
Real-time quantitative PCR
Primers for reverse transcription quantitative PCR (RT-qPCR) analyses designed with Primer Express 3 software (Applied Biosystems®) are shown in Table 1. A RT-PCR was performed and checked in an agarose gel to prove the single amplification and the predicted length of each gene (95 °C for 2 min; 40 cycles at 95 °C for 15 s, 60 °C for 30 s and 72 °C for 1 min; 72 °C for 5 min). Gene amplification efficiencies were calculated for all genes and both species using a serial dilution pool of cDNA with the quantification standard curve method, repeated in triplicate and carried out on a StepOnePlus™ Real-Time PCR System using SYBR® Green (Applied Biosystems®). Those genes with good efficiencies were used for RT-qPCR experiments on the same equipment with 1.5 µl of the same starting concentration of cDNA for each sample. All PCR conditions were as follows: one cycle at 95 °C for 2 min and 40 cycles at 95 °C for 15 s and 60 °C for 30 s. Experiments were performed with three biological and three technical replicates. Non template controls were used as negative control and melting curves analysis were carried out to assure the specificity of the amplifications.
Additionally, two target genes were amplified using the same instrument and conditions to perform a relative expression analysis using the reference genes previously selected for C. campestris (sex: B-t and Vatpase; gonad development: RPS18 and NADH; body parts: Vatpase and RPS11) and C. littoralis (sex: Myo and Myo-Alc; infection: NADH and B-t; body parts: NADH, 60Sa and ArgKin). Both target genes were amplified as described in previous works with previously described primers37,38. UBC9, an ubiquitin gene related to reproduction and sexual activity, was tested for all conditions and species with the exception of the infection in C. littoralis, for which a member of the defensin family (Clit-Def) was used to check changes in the expression.
Data analysis
The raw CT data were obtained with 7500 software v2.0.5 (Applied Biosystems®), and gene expression and stability were later analysed using four different tools and algorithms: geNorm39, NormFinder40, BestKeeper41 and ΔCT method42.
geNorm calculates the average expression stability values (M) as the average pairwise of variation of one of the genes with all control genes present in the experiment. The most stable genes are those with the lowest M values. At the same time, it provides the optimal number of reference genes needed for normalization in expression measurements studies. NormFinder algorithm estimates the overall expression variation of the candidate reference genes and also the variation between sample subgroups of the same set. The stability value provides information about the reference genes, with lower values meaning higher stability, as occurs with geNorm algorithm. Both geNorm and NormFinder use quantities transformed to a linear scale as input. Similarly, the main principle of Bestkeeper is that reference genes should present the same expression patterns across samples. Thus, this software uses raw CT values to determine the best correlated reference genes and combines them into an index. The lower the standard deviation values obtained are the more stability present the reference genes tested. Finally, ΔCT method also uses raw CT values and compares them between pairs of genes within each sample to rank them considering the standard deviation mean for each reference gene.
For qPCR analyses with both target genes, gene expression was obtained considering all genes efficiencies as described by Pfaffl57. To obtain the statistical significance, a Student’s t-test was performed for sex comparison, gonad development and infection analyses and a one-way ANOVA with a Tukey’s HSD post-hoc test for body parts comparison with the software GraphPad Prism version 5.00 (GraphPad Software, San Diego California USA, www.graphpad.com).
References
Heid, C. A., Stevens, J., Livak, K. J. & Williams, P. M. Real time quantitative PCR. Genome Res. 6, 986–994 (1996).
Bustin, S. A. Absolute quantification of mRNA using real-time reverse transcription polymerase chain reaction assays. Journal of molecular endocrinology 25, 169–193 (2000).
Bustin, S. A. Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J. Mol. Endocrinol. 29, 23–39 (2002).
VanGuilder, H. D., Vrana, K. E. & Freeman, W. M. Twenty-five years of quantitative PCR for gene expression analysis. Biotechniques 44, 619 (2008).
Ginzinger, D. G. Gene quantification using real-time quantitative PCR: an emerging technology hits the mainstream. Exp. Hematol. 30, 503–512 (2002).
Derveaux, S., Vandesompele, J. & Hellemans, J. How to do successful gene expression analysis using real-time PCR. Methods 50, 227–230 (2010).
Thellin, O. et al. Housekeeping genes as internal standards: use and limits. J. Biotechnol. 75, 291–295 (1999).
Bustin, S. & Nolan, T. Talking the talk, but not walking the walk: RT‐qPCR as a paradigm for the lack of reproducibility in molecular research. Eur. J. Clin. Invest. 47, 756–774 (2017).
Stürzenbaum, S. R. & Kille, P. Control genes in quantitative molecular biological techniques: the variability of invariance. Comp. Biochem. Physiol. B-Biochem. Mol. Biol. 130, 281–289 (2001).
Huggett, J., Dheda, K., Bustin, S. & Zumla, A. Real-time RT-PCR normalisation; strategies and considerations. Genes Immun. 6, 279–284 (2005).
Shakeel, M., Rodriguez, A., Tahir, U. B. & Jin, F. Gene expression studies of reference genes for quantitative real-time PCR: an overview in insects. Biotechnol. Lett. 40, 227–236 (2018).
Suzuki, T., Higgins, P. J. & Crawford, D. R. Control selection for RNA quantitation. Biotechniques 29, 332–337 (2000).
Tricarico, C. et al. Quantitative real-time reverse transcription polymerase chain reaction: normalization to rRNA or single housekeeping genes is inappropriate for human tissue biopsies. Anal. Biochem. 309, 293–300 (2002).
Radonić, A. et al. Guideline to reference gene selection for quantitative real-time PCR. Biochem. Biophys. Res. Commun. 313, 856–862 (2004).
Dheda, K. et al. The implications of using an inappropriate reference gene for real-time reverse transcription PCR data normalization. Anal. Biochem. 344, 141–143 (2005).
Ruan, W. & Lai, M. Actin, a reliable marker of internal control? Clin. Chim. Acta 385, 1–5 (2007).
Li, R. et al. Reference gene selection for qRT-PCR analysis in the sweetpotato whitefly, Bemisia tabaci (Hemiptera: Aleyrodidae). PLoS One 8, e53006 (2013).
Ponton, F., Chapuis, M. P., Pernice, M., Sword, G. A. & Simpson, S. J. Evaluation of potential reference genes for reverse transcription-qPCR studies of physiological responses in Drosophila melanogaster. J. Insect Physiol. 57, 840–850 (2011).
Koramutla, M. K., Aminedi, R. & Bhattacharya, R. Comprehensive evaluation of candidate reference genes for qRT-PCR studies of gene expression in mustard aphid, Lipaphis erysimi (Kalt). Sci. Rep. 6, 25883, https://doi.org/10.1038/srep25883 (2016).
Horňáková, D., Matoušková, P., Kindl, J., Valterová, I. & Pichová, I. Selection of reference genes for real-time polymerase chain reaction analysis in tissues from Bombus terrestris and Bombus lucorum of different ages. Anal. Biochem. 397, 118–120 (2010).
Lourenço, A. P., Mackert, A., dos Santos Cristino, A. & Simões, Z. L. P. Validation of reference genes for gene expression studies in the honey bee. Apis mellifera, by quantitative real-time RT-PCR. Apidologie 39, 372–385 (2008).
Lord, J. C., Hartzer, K., Toutges, M. & Oppert, B. Evaluation of quantitative PCR reference genes for gene expression studies in Tribolium castaneum after fungal challenge. J. Microbiol. Methods 80, 219–221 (2010).
Toutges, M. J., Hartzer, K., Lord, J. & Oppert, B. Evaluation of reference genes for quantitative polymerase chain reaction across life cycle stages and tissue types of Tribolium castaneum. J. Agric. Food Chem. 58, 8948–8951 (2010).
Sang, W., He, L., Wang, X. P., Zhu-Salzman, K. & Lei, C. L. Evaluation of reference genes for RT-qPCR in Tribolium castaneum (Coleoptera: Tenebrionidae) under UVB stress. Environ. Entomol. 44, 418–425 (2015).
Yang, C. et al. Selection of reference genes for RT-qPCR analysis in a predatory biological control agent, Coleomegilla maculata (Coleoptera: Coccinellidae). Sci. Rep. 5, 18201 (2015).
Yang, C. et al. Selection of Reference Genes for RT-qPCR Analysis in Coccinella septempunctata to Assess Un-intended Effects of RNAi Transgenic Plants. Front. Plant Sci. 7, 1672 (2016).
Barros Rodrigues, T. et al. Validation of reference housekeeping genes for gene expression studies in western corn rootworm (Diabrotica virgifera virgifera). PLoS One 9, e109825 (2014).
Rajarapu, S. P., Mamidala, P. & Mittapalli, O. Validation of reference genes for gene expression studies in the emerald ash borer (Agrilus planipennis). Insect Sci. 19, 41–46 (2012).
Pearson, D. L. & Cassola, F. A quantitative analysis of species descriptions of tiger beetles (Coleoptera: Cicindelidae), from 1758 to 2004, and notes about related developments in biodiversity studies. Coleops. Bull. 59, 184–193 (2005).
Pearson, D. L. Biology of tiger beetles. Annu. Rev. Entomol. 33, 123–147 (1988).
Cassola, F. & Pearson, D. L. Global patterns of tiger beetle species richness (Coleoptera: Cicindelidae): their use in conservation planning. Biol. Conserv. 95, 197–208 (2000).
Pearson, D. L. Tiger beetles as indicators for biodiversity patterns in Amazonia. Res. Explor. 8, 116–117 (1992).
Pearson, D. L. & Cassola, F. World‐wide species richness patterns of tiger beetles (Coleoptera: Cicindelidae): Indicator taxon for biodiversity and conservation studies. Conserv. Biol. 6, 376–391 (1992).
Rodríguez, J. P., Pearson, D. L. & Barrera, R. R. A test for the adequacy of bioindicator taxa: are tiger beetles (Coleoptera: Cicindelidae) appropriate indicators for monitoring the degradation of tropical forests in Venezuela? Conserv. Biol. 83, 69–76 (1998).
Elmer, K. R. & Meyer, A. Adaptation in the age of ecological genomics: insights from parallelism and convergence. Trends Ecol. Evol. 26, 298–306 (2011).
Rodríguez-García, M. J., Machado, V. & Galián, J. Identification and characterisation of putative seminal fluid proteins from male reproductive tissue EST libraries in tiger beetles. BMC Genomics 16, 391 (2015).
Rodríguez-García, M. J., García-Reina, A., Machado, V. & Galián, J. Identification, structural characterisation and expression analysis of a defensin gene from the tiger beetle Calomera littoralis (Coleoptera: Cicindelidae). Gene 589, 56–62 (2016).
Rodríguez-García, M. J., García-Reina, A., Machado, V. & Galián, J. Characterisation and expression analysis of UBC9 and UBS27 genes in developing gonads of cicindelids (Coleoptera: Cicindelidae). Comp. Biochem. Physiol. B-Biochem. Mol. Biol. 202, 75–82 (2016).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3, research0034–1 (2002).
Andersen, C. L., Jensen, J. L. & Ørntoft, T. F. Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer res. 64, 5245–5250 (2004).
Pfaffl, M. W., Tichopad, A., Prgomet, C. & Neuvians, T. P. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol. Lett. 26, 509–515 (2004).
Silver, N., Best, S., Jiang, J. & Thein, S. L. Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol Biol 7, 33 (2006).
Jiang, H. B., Liu, Y. H., Tang, P. A., Zhou, A. W. & Wang, J. J. Validation of endogenous reference genes for insecticide-induced and developmental expression profiling of Liposcelis bostsrychophila (Psocoptera: Liposcelididae). Mol. Biol. Rep 37, 1019–1029 (2010).
Scharlaken, B. et al. Reference gene selection for insect expression studies using quantitative real-time PCR: The head of the honeybee. Apis mellifera, after a bacterial challenge. J. Insect Sci. 8, 1–10 (2008).
Salem, T. Z., Allam, W. R. & Thiem, S. M. Verifying the stability of selected genes for normalization in Q PCR experiments of Spodoptera frugiperda cells during AcMNPV infection. PloS one 9, e108516 (2014).
Fu, W. et al. Exploring valid reference genes for quantitative real-time PCR analysis in Plutella xylostella (Lepidoptera: Plutellidae). Int. J. Biol. Sci. 9, 792 (2013).
Lu, Y. et al. Evaluation and validation of reference genes for SYBR Green qRT-PCR normalization in Sesamia inferens (Lepidoptera: Noctuidae). J. Asia-Pac. Entomol. 18, 669–675 (2015).
Shi, X. Q. et al. Validation of reference genes for expression analysis by quantitative real-time PCR in Leptinotarsa decemlineata (Say). BMC Res. Notes 6, 93 (2013).
Arun, A., Baumlé, V., Amelot, G. & Nieberding, C. M. Selection and Validation of Reference Genes for qRT-PCR Expression Analysis of Candidate Genes Involved in Olfactory Communication in the Butterfly Bicyclus anynana. PloS one 10, e0120401 (2015).
Ling, D. & Salvaterra, P. M. Robust RT-qPCR data normalization: validation and selection of internal reference genes during post-experimental data analysis. PloS one 6, e17762 (2011).
Kaneko, Y., Tanaka, H., Ishibashi, J., Iwasaki, T. & Yamakawa, M. Gene expression of a novel defensin antimicrobial peptide in the silkworm. Bombyx mori. Biosci. Biotechnol. Biochem. 72, 2353–2361 (2008).
Seufi, A. M., Hafez, E. E. & Galal, F. H. Identification, phylogenetic analysis and expression profile of an anionic insect defensin gene, with antibacterial activity, from bacterial-challenged cotton leafworm, Spodoptera littoralis. BMC Mol. Biol. 12, 47 (2011).
Hashiyama, K., Shigenobu, S. & Kobayashi, S. Expression of genes involved in sumoylation in the Drosophila germline. Gene Expr. Patterns 9, 50–53 (2009).
Paarmann, W. Annual periodicity of the polyvoltine ground beetle Pogonus chalceus Marsh (Coleoptera, Carabidae) and its control by environmental factors. Zool. Anz. 196, 150–160 (1976).
Theodorides, K., De Riva, A., Gómez‐Zurita, J., Foster, P. G. & Vogler, A. P. Comparison of EST libraries from seven beetle species: towards a framework for phylogenomics of the Coleoptera. Insect Mol. Biol. 11, 467–475 (2002).
Conesa, A. et al. Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21, 3674–3676 (2005).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res. 29, e45–e45 (2001).
Acknowledgements
This work has been funded by project 19908-GERM-15 of the Fundación Seneca (Gobierno Regional de Murcia, Spain).
Author information
Authors and Affiliations
Contributions
A.G.R. participated in the conception and design of the study, performed the laboratory work, data analysis and drafted the manuscript. M.J.R. took part in the laboratory work and qPCR analysis and helped to draft the ending version of the manuscript. J.G. participated in the conception, design and coordination of the study and helped to produce the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
García-Reina, A., Rodríguez-García, M.J. & Galián, J. Validation of reference genes for quantitative real-time PCR in tiger beetles across sexes, body parts, sexual maturity and immune challenge. Sci Rep 8, 10743 (2018). https://doi.org/10.1038/s41598-018-28978-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-28978-7
- Springer Nature Limited
This article is cited by
-
Reference genes to study the sex-biased expression of genes regulating Drosophila metabolism
Scientific Reports (2024)
-
Identifying optimal reference genes for gene expression studies in Eurasian spruce bark beetle, Ips typographus (Coleoptera: Curculionidae: Scolytinae)
Scientific Reports (2022)
-
Selection of reference genes for normalization of RT-qPCR data in gene expression studies in Anthonomus eugenii Cano (Coleoptera: Curculionidae)
Scientific Reports (2020)