Abstract
Ischemic retinopathy is characterized by ischemia followed by retinal neovascularization (RNV) resulting in visual impairment. Given the role of neuron-secreted growth factors in regulating angiogenesis, we examined how genetic deletion of the neurotrophin receptor; p75NTR can overcome retinal ischemia using oxygen-induced retinopathy (OIR) mouse model. Wildtype (WT) or p75NTR−/− mice pups were subjected to hyperoxia (70% O2, p7-p12) then returned to normal air (relative hypoxia, p12-p17). Vascular alterations were assessed at p12 and p17 time-points. Deletion of p75NTR prevented hyperoxia-associated central vascular cell death (p12) and hypoxia-associated RNV and enhanced central vascular repair (p17). Decreased expression of apoptotic markers; preserved Akt survival signal decreased proNGF were also observed at p12. During hypoxia, deletion of p75NTR maintained VEGF and VEGFR2 activation and restored NGF/proNGF and BDNF/proBDNF levels. Deletion of p75NTR coincided with significant increases in expression and activation of NGF survival receptor, TrkA at basal and hyperoxic condition. Pharmacological inhibition of TrkA using compound K-252a (0.5 μg 1 μl−1/eye) resulted in 2-fold increase in pathological RNV and 1.34-fold increase in central vascular cell death in p75NTR−/− pups. In conclusion, deletion of p75NTR protected against retinal ischemia and prevented RNV, in part, through restoring neurotrophic support and activating TrkA receptor.
Similar content being viewed by others
Introduction
Ischemic retinopathy is a common characteristic of several ocular diseases including diabetic retinopathy, retinopathy of prematurity and retinal vein occlusion1. Ischemic retinopathy is characterized by an initial microvascular degeneration followed by a mal-adaptive pathological retinal neovascularization (RNV), consequent to hypoxia, in an attempt to reinstate metabolic equilibrium; that can result in visual impairment and eventually blindness2. Current therapeutic approaches including intravitreal injection of vascular endothelial growth factor (VEGF) neutralizing therapies are sight-saving, yet hindered by serious concerns such as late intervention, invasiveness and cost-prohibition3. Therefore, there is pressing need to develop new therapeutics that can preserve both neuronal and vascular function in ischemic retinopathy diseases.
Given that retina is a typical neurovascular unit, neurons and glial cells may interact with blood vessels to contribute to pathologic neovascularization by secreting growth factors and guidance cues4,5,6. Nerve growth factor (NGF) is well known for its role in regulating survival, growth and functional maintenance of neuronal cells as well as vasculature. In response to ischemic insult and hypoxia, NGF, among other angiogenic factors, will be released. Several studies showed that NGF mediates survival and angiogenic signal through activation of its high affinity; TrkA receptor7,8,9.
NGF is synthesized and released as precursor, proNGF that normally get cleaved to the mature form. We and others have shown that pro-oxidative milieu can impair maturation of NGF leading to accumulation of proNGF10,11. Contrast for mature form, proNGF has high affinity to neurotrophin death receptor; p75NTR, the first identified member of the tumor necrosis factor (TNF) receptor superfamily that have intracellular death domain12. Several reports demonstrated the action of proNGF, mediating apoptosis via activating p75NTR 13,14,15 and forming a co-receptor with sortilin; a member of Vps10p-domain receptor family16. Nevertheless, the role of proNGF in regulating cell survival and reparative angiogenesis is ill-defined. Few reports showed that proNGF-mediated angiogenic behavior in breast cancer cells17 and retinal endothelial cells18 that was regulated through activation of TrkA but not p75NTR. In the latter study, our group observed that inhibition of p75NTR was associated with enhanced TrkA activation18, suggesting a cross-talk between p75NTR expression and TrkA activation. Despite these significant findings, there is gap in knowledge about the interplay between NGF and proNGF and their receptors p75NTR and TrkA in response to hypoxia and angiogenesis.
In the current study using oxygen-induced retinopathy (OIR) mouse model, a standard model for ischemic retinopathy19, we examined the impact of genetic deletion of p75NTR on vascular cell death as well as pathological neovascularization and reparative angiogenesis. Here, we share the findings that genetic deletion of p75NTR prevented vascular cell death, enhanced central reparative capillary growth and prevented pathological neovascularization in mouse retina. The mechanisms involve restoring levels of NGF and BDNF, preserving VEGF signaling and enhancing TrkA-mediated survival and angiogenic signal. Therefore, we postulate that development of inhibitors against p75NTR receptor can provide potential alternative therapeutic strategy for ischemic retinopathy.
Materials and Methods
Animals
All animal experiments were performed in accordance with relevant guidelines and regulations of “Association for Research in Vision and Ophthalmology” statement for use of animals in ophthalmic and vision research, and were approved by the “Charlie Norwood VA Medical Center Animal Care and Use Committee”, ACORP#16–01–088. The p75NTR, B6.129S4Ngfrtm1Jae/J (p75NTR+/−, exon III knockout mice20) were obtained from Jackson Laboratories (Bar Harbor, Maine, USA) and crossed with C57BL6/J mice (Jackson Laboratories). The WT and p75NTR+/− mice were crossed then back-crossed again to establish a colony of homozygous p75NTR−/− and WT breeders that produced the mice used in the current study.
Oxygen-induced retinopathy (OIR) mouse model
OIR model was performed as previously described by Smith et al.21. Briefly, on postnatal day 7 (p7), WT and p75NTR−/− mice pups were placed along with their dams into a custom-built chamber (Biospherix, Redfield, NY) in which the partial pressure of oxygen was maintained at 70% for 5 days (hyperoxic stage). Pups were then returned to room air (21% oxygen, hypoxic stage) for 5 days (p12-p17). Central capillary drop-out (CDO) and retinal neovascularization (RNV) in OIR-exposed mice pups were performed at p12 and p17 time points, respectively, as described previously21,22 whereas, biochemical assays; Western blotting and PCR were performed at p12 and p14 time points. At the selected time points, mice pups were euthanized in carbon-dioxide chamber (2% flow rate for 5 min) followed by cervical dislocation. One eye was enucleated and fixed in 4% paraformaldehyde overnight to be flat-mounted for morphological studies. For the other eye, retinas were isolated and snap frozen for further biochemical analysis.
Intravitreal injection
Mice were anesthetized by intraperitoneal injection of ketamine (100 mg/kg) xylazine (10 mg/kg) mixture and complete anesthesia was confirmed by loss of reflex to sharp paw pinch. K252a (Sigma, MO, USA; 0.5 μg μL−1/eye) or Dimethyl sulphoxide (DMSO) were injected intravitreally at p12 using a Hamilton syringe with 32-gauge glass capillary. Pups were left to recover (6-hours) from exposure to hyperoxia before intravitreal injection.
Morphological studies
Retinas were dissected and permealized for 15 minutes with 0.3% Triton X-PBS then stained overnight at 4 °C with isolectin; biotinylated griffonia (bandeiraea) simplicifolia lectin I (GSL I, BSL I), (Vector Labs, CA, USA; 1% in 5% normal goat serum in 0.3% Triton X-PBS) followed by incubation with secondary antibody; Texas red® avidin D (Vector labs, CA, USA; 0.5% in 5% normal goat serum in 0.3% Triton X-PBS). Retinal perfusion was assessed by intraperitoneal injection of FITC-dextran (Mol. WT. 2,000,000, Sigma, 50 mg mL−1, 100 μL/pup) 30 minutes prior to sacrifice as described previously23. Lectin-stained and/or FITC-perfused retinas were flat-mounted onto Super-frost/Plus microscope slides (Fisher Scientific, MA, USA) with the photoreceptor side facing down and imbedded in Vectashield mounting media for fluorescence (Vector Labs, CA, USA). Slides were photo-micrographed at 5X using a Zeiss AxioObserver.Z1. Images were assembled into a single file using photoshop software (CS6, Adobe system incorporated). CDO and RNV were quantified as previously described24.
Western blot analysis
Frozen retinas were placed into protein lysis buffer (Millipore) and briefly homogenized. Retinal lysates were centrifuged and 35–50 μg were resolved on an SDS-PAGE gel (4–20% gradient Tris glycine precast gel, BioRad) and electro-blotted to nitrocellulose membranes. Membranes were blocked with 5% milk or BSA in PBS-tween and incubated overnight in 4 °C with the following primary antibodies: p75NTR (kind gift from Dr. Bruce Carter, Department of Biochemistry, Vanderbilt University), Akt (#9272, Cell Signaling), p-Akt (#9275 S, Cell Signaling), cleaved-PARP (#5625, Cell Signaling), total PARP (#9532, Cell Signaling), cleaved caspase-3 (#9664, Cell Signaling), TrkA (#76291, Abcam), phospho-TrkA (#1445, Abcam), NGF (# AN-240, Alomone), proNGF (#ANT-005, Alomone), sortilin (#16640, Abcam), BDNF and proBDNF (SC-546, Santa Cruz), VEGF (#ABS82, Millipore), VEGFR2 (#2472, Cell Signaling), phopho-VEGFR2 (#2474, Cell Signaling), then re-probed with the primary antibodies for the house-keeping proteins; actin (#A5441, Sigma) or tubulin (#ab4074, Abcam) to confirm equal loading. Membranes were incubated with horseradish peroxidase (HRP)-conjugated anti-mouse or HRP-conjugated anti-rabbit secondary antibodies (Millipore) for 2 h at room temperature. The films were scanned with FluorChem™ FC3 (protein simple) and the band intensity was quantified using densitometry software version (Fiji) and expressed as a relative optical density (OD).
Quantitative real-time (RT-PCR)
Retinas samples were processed using (mirVANATM PARISTM Kit) and RNA was purified and quantified as described previously25 following the manufacturer’s instructions. A one-step quantitative RT-PCR kit (Invitrogen) was used to amplify 10 ng retinal mRNA as described previously26,27. PCR primers (listed in Table 1) were obtained from Integrated DNA Technologies (Coralville, IA, USA). Quantitative PCR was conducted using StepOnePlus qPCR system (Applied BioSystems, Life Technologies). The percent expression of various genes was normalized to 18S and expressed relative to WT normoxic controls.
Statistical analysis
All the data are expressed as mean ± SEM. Differences between 2 groups for morphological studies were detected using un-paired Student T-test. Two-way ANOVA was used to assess interaction between two variables; genetic background (WT vs. p75NTR−/−) and OIR exposure (normoxia vs. hyperoxia/hypoxia). Tukey-Kramer post-multiple comparisons was used for significant interactions among various groups. Significance for all tests was determined at α = 0.05, Graphpad Prism, Ver.6.
Results
Hyperoxia but not hypoxia triggered p75NTR expression in oxygen-induced retinopathy (OIR)
We aimed to examine the vascular protective effects of targeting p75NTR in ischemic retinopathy using p75NTR−/−, exon III knockout mice20. OIR is a commonly used model to study ischemic retinopathy as it has two distinctive stages; capillary dropout and ischemia at p12 followed by retinal neovascularization at p17. At postnatal day-7, mice pups were placed in partial pressure of oxygen at 70% for 5 days (hyperoxic stage, p7-p12). At p12, pups were returned to room air (21% oxygen, relative hypoxic stage) for 5 days (hypoxic stage, p12-p17). Exposing the developing retina of WT mice to hyperoxia resulted in significant (1.4-fold) increase in p75NTR expression compared to normoxic controls (Fig. 1A). Next, we examined the expression of p75NTR receptor in WT mice during the hypoxic stage of OIR (p12-p17). As shown in Fig. 1B,C, there was no significant difference in p75NTR expression at p14 or p17 time points. Moreover, we assessed expression of sortilin receptor, a member of Vps10p-domain receptor family, known to be co-receptor for neurotrophin signaling16. As shown in Supplementary Fig. 1, sortilin expression was not affected during hyperoxia at p12 (Supplementary Fig. 1A), or during relative hypoxia at p14 (Supplementary Fig. 1B) in WT pups. In addition, genetic deletion of p75NTR−/− receptor did not impact expression of sortilin under normoxia, during hyperoxia (p7-p12) or during hypoxia (p12-p17).
Hyperoxia, but not hypoxia triggered p75NTR expression in WT-pups in OIR model. (A) Exposing WT pups to 70% oxygen from p7-p12 significantly increased p75NTR expression in WT-pups (*significant compared to WT-controls using two-way ANOVA, p < 0.05, n = 3–4). (B) Western blotting representative and bar graph of p75NTR receptor expression in WT pups by p14 showing no alteration during hypoxic stage of OIR (n = 4–6). (C) Western blotting representative and bar graph of p75NTR receptor expression in WT pups by p17 showing a trend for decreased p75NTR expression that was not statistically significant (n = 4).
Deletion of p75NTR attenuated hyperoxia-increase in proNGF and decrease in NGF
We have previously shown that, exposure to hyperglycemia disturbed homeostasis of NGF by accumulation of its precursor; proNGF at the expense of the mature form; NGF11,28. Similar observation was noticed in OIR-exposed WT pups, where, early hyperoxic stage resulted in significant (1.7-fold) increase in proNGF expression (Fig. 2A) whereas; NGF showed mild decrease (Fig. 2B) compared to normoxic controls. Imbalance in both proNGF and NGF was ameliorated with genetic deletion of p75NTR (Fig. 2A,B). Blotting the ratio of NGF/proNGF showed 50% decrease in WT pups after exposure to hyperoxia while the ratio was preserved in p75NTR−/− retinas, as shown in Fig. 2C. These results suggest that hyperoxia shifted the homeostasis of NGF signaling towards apoptotic pathway of proNGF/p75NTR at the expense of survival pathway; NGF signaling pathway.
Deletion of p75NTR attenuated hyperoxia-induced increase in proNGF and decrease in NGF. (A) Representative Western blotting and scatter graph showing significant increase in proNGF protein levels in retinas from WT, but not in p75NTR−/− in response to hyperoxia. (*significant compared to the rest of the groups using two-way ANOVA, p < 0.05, n = 6–8). (B) Representative Western blotting and scatter graph showing a tendency of NGF protein level to be decreased in WT (P = 0.07), but preserved-level in p75NTR−/− during hyperoxia. (C) Bar graph showing significant decrease in of NGF/proNGF ratio in retinas from WT, but not p75NTR−/− pups in response to hyperoxia (*significant compared to WT normoxic controls, using two-way ANOVA, p < 0.05, n = 6–8).
Deletion of p75NTR preserved survival and attenuated hyperoxia-mediated apoptosis
Exposure to hyperoxia tended to decrease the activation of the survival kinase; Akt by 30% in retinas of WT pups, but was preserved in p75NTR−/− mice (Fig. 3A). Hyperoxia resulted in increased total and cleaved PARP in retinas from WT pups (Fig. 3B,C). Furthermore, retinas from p75NTR−/− pups showed ~50% lower levels of both total-PARP (Fig. 3B) and cleaved-PARP (Fig. 3C) compared to their WT littermates either at normoxic or hyperoxic conditions.
Deletion of p75NTR preserved survival signal and attenuated hyperoxia-mediated apoptosis. (A) Representative Western blotting and bar graph showing modest decrease in Akt activation (Y308) in response to hyperoxia at p12 retinal lysates of WT and p75NTR−/− mice (n = 5–7). (B) Representative Western blotting and bar graph of total PARP expression in p12 retinal lysates of WT and p75NTR−/− mice (*significant using two-way ANOVA, p < 0.05, n = 7). (C) Representative Western blotting and bar graph of cleaved PARP expression in p12 retinal lysates of WT and p75NTR−/− mice (*significant using two-way ANOVA, p < 0.05, n = 4–6).
Deletion of p75NTR attenuated hyperoxia-induced capillary dropout at p12
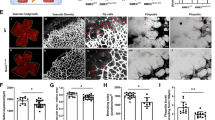
By p12, retinal ischemia and vaso-obliteration evident by central capillary dropout (CDO) reach its maximum in OIR model. Indeed, hyperoxia caused marked vaso-obliteration and CDO, illustrated by the central encircled area (Fig. 4A) in retinas from WT pups but not in p75NTR−/− pups (Fig. 4B). Statistical analysis showed significant reduction by 34% in CDO in p75NTR−/− pups compared to their WT littermates (Fig. 4C). These vascular manifestations coincided with increased expression of proNGF, p75NTR and apoptotic markers in WT pups but not p75NTR−/− during hyperoxia.
Deletion of p75NTR attenuated hyperoxia-induced retinal central vaso-obliteration. (A,B) Representative images of Isolectin GS-stained retinal flat mounts of WT and p75NTR−/− pups showing capillary drop-out (CDO) illustrated by encircled area at the center at p12 (magnification is 5X). (C) Bar graph and statistical analysis using unpaired student T-test for CDO showing significant reduction of CDO in p75NTR−/− compared to WT pups in response to hyperoxia (*significant compared to WT-hyperoxic group, p < 0.05, n = 8–12). (Hyper) hyperoxia-exposed group, (Norm) normoxic controls, (CDO): central capillary dropout.
Relative hypoxia triggers retinal neovascularization in WT but not p75NTR−/− at p17
At p12, pups that were exposed to hyperoxia were returned to room air (21% oxygen, relative hypoxic stage) for 5 days (p12-p17). Relative hypoxia triggered significant pathological retinal neovascularization in the mid-periphery of WT retinas, growing towards the vitreous (encircled in Fig. 5A by dotted white line). Additionally, relative hypoxia impaired intra-retinal reparative angiogenesis towards the central avascular area in WT-pups (Fig. 5A, encircled area at the center). Deletion of p75NTR receptor significantly attenuated hypoxia-induced pathological RNV by 42% compared to WT-pups (Fig. 5B,C). More importantly, Deletion of p75NTR stimulated reparative angiogenesis as indicated by growth of capillaries toward the central retina and decreasing central avascular area by ~32% compared to WT-pups during hypoxic stage of OIR model (Fig. 5B,D).
Deletion of p75NTR attenuated hypoxia-induced neovascularization and enhances reparative angiogenesis. (A,B) Representative images of GS-isolectin stained retinal flat mounts of WT and p75NTR−/− pups at p17, showing pathological retinal neovascularization (RNV, encircled by dotted white line) and central avascular area (encircled area at the center). Images were taken on 5X magnification. Bar graph of RNV (C) and central avascular area (D) showing significant decrease in both manifestations in p75NTR−/− pups by p14 during hypoxic stage of OIR (*significant using unpaired student T-test, p < 0.05, n = 7–14). Hypo: relative hypoxia-exposed groups, RNV: retinal neovascularization.
Hypoxia triggered VEGF expression and VEGFR2 activation in both WT and p75NTR knockout
Expression of vascular endothelial growth factor (VEGF), a well-studied angiogenic factor in OIR model is triggered by hypoxia and correlates with RNV29. Therefore, we investigated impact of deleting p75NTR on VEGF level and its signal. At p14, hypoxia resulted in significant 2.2-fold increase in VEGF mRNA expression in WT pups (Supplementary Fig. 2A) and 1.53-fold increase in VEGF protein expression in retinas of WT pups as compared to normoxic controls (Fig. 6A). Deletion of p75NTR receptor tended to alter but did not reach statistical significance the basal expression of VEGF mRNA or protein in normoxia. Yet upon hypoxia exposure, retinas from p75NTR−/− pups showed tendency of increased mRNA expression of VEGF that was not statistically significant (Supplementary Fig. 2A), and significant increase by 1.6-fold in VEGF protein as compared to normoxic controls (Fig. 6A). Next, we examined activation of VEGF receptor-2 (VEGFR2) known to mediate angiogenic signal. Exposure to relative hypoxia resulted in an increase in VEGFR2 activation that did not reach statistical analysis in retinas from WT pups compared to normoxic controls (Fig. 6B). Interestingly, retinas from p75NTR−/− pups exhibited significant activation of VEGFR2 at both basal and hypoxic conditions compared to WT-normoxic controls (Fig. 6B).
Deletion of p75NTR preserved hypoxia-induced VEGF expression and VEGFR2 activation. (A) Representative Western blotting and bar graph of retinal VEGF protein expression from WT and p75NTR−/− pups exposed to OIR at p14. Two-way ANOVA showed significant effect of hypoxia increasing VEGF both in WT and p75NTR−/− pups (*significant compared to WT normoxic group, p < 0.05, n = 6–12). (B) Representative Western blotting and bar graph of pVEGFR2 compared to its total protein VEGFR2 level. Two-way ANOVA showed marked gene deletion effect where p75NTR−/− showed significant increase in pVEGFR2 under normoxia or hypoxia compared to WT-normoxic controls (*significant compared to WT normoxic group using, p < 0.05, n = 4–5).
Deletion of p75NTR attenuated hypoxia-induced increase in proNGF and preserved NGF/proNGF ratio
At p14, relative hypoxia caused significant (1.45-fold) increase of proNGF in retinas from WT-pups compared to normoxic controls (Fig. 7A). In contrast, hypoxia did not significantly alter NGF levels in WT-pups compared to normoxic controls (Fig. 7B). Deletion of p75NTR prevented hypoxia-induced increase in retinal proNGF level and significantly increased NGF levels (1.4-fold) compared to p75NTR−/− and WT normoxic controls (Fig. 7A,B). Next, we investigated impact of hypoxia and p75NTR deletion on mRNA expression of NGF. As shown in Supplementary Fig. 2B, exposure to hypoxia significantly decreased NGF expression in retinas from WT-mice. In contrast, p75NTR gene deletion did not alter expression of NGF under normoxia or hypoxia compared to WT-normoxic controls. These results suggest that the observed increase of proNGF in response to hypoxia and the increase in NGF levels associated with p75NTR deletion are post-transcriptional. Blotting the ratio of retinal NGF/proNGF protein showed that hypoxia caused significant 40% decrease in NGF/proNGF in retinas from WT pups compared to normoxic controls but not in p75NTR−/− pups (Fig. 7C). Similar results showing that hypoxia altered the levels and ratio between the precursor and mature form of the brain derived neurotrophic factor (BDNF) were observed. As shown in Supplementary Fig. 3, hypoxia triggered significant increase in retinal proBDNF level in WT-pups but not in p75NTR−/−. In response to hypoxia, the ratio of BDNF/proBDNF was significantly decreased in WT pups but not in p75NTR−/−.
Deletion of p75NTR attenuated hypoxia-induced increase in proNGF and restored ratio of NGF/proNGF. (A) Representative Western blotting and scatter graph for proNGF showing that hypoxia caused significant increase in proNGF levels in retinas from WT-pups but not p75NTR−/− compared to normoxic control at p14. (B) Representative Western blotting and scatter graph for NGF showing that hypoxia caused significant increase in NGF level in retinas from p75NTR−/−, but not WT compared to their normoxic controls. (C) Bar graph of NGF/proNGF ratio and Two-way ANOVA statistical analysis showing significant decrease in retinal NGF/proNGF ratio in WT-pups during hypoxia while p75NTR−/− pups maintained balanced ratio of NGF/proNGF (*significant using Two-way ANOVA, p < 0.05, n = 5–7).
Deletion of p75NTR enhanced the NGF survival receptor TrkA expression and activation at p12
TrkA is a tyrosine kinase receptor that selectively binds NGF to mediate survival and angiogenic signal (reviewed in15). We assessed the expression and activation pattern of TrkA during the two stages of OIR. Two-way ANOVA analysis showed significant effect of hyperoxia where it decreased mRNA expression of TrkA by ~40% in retinas from WT-pups and 50% from p75NTR−/− compared to corresponding normoxic controls (Fig. 8A). Two-way ANOVA analysis showed significant gene effect where deletion of p75NTR receptor significantly increased mRNA expression of retinal TrkA by ~5-fold compared to WT-pups at normoxia and 2.5-fold increase upon hyperoxia compared to WT retinas (Fig. 8A). Next, we evaluated protein expression of TrkA receptor during hyperoxic stage of OIR (Fig. 8B,C). Two-way ANOVA analysis showed significant effect of deleting p75NTR gene where retinas from p75NTR−/− pups showed significant increase in TrkA protein level compared to WT under normoxia and hyperoxia (Fig. 8B,C). Remarkably, Two-way ANOVA analysis showed significant effect of hyperoxia on TrkA activation. As shown in Fig. 8B,D, retinas from p75NTR−/− pups exposed to hyperoxia showed significant TrkA activation compared to p75NTR−/− normoxic controls and to retinas from WT-pups exposed to normoxia and hyperoxia. These results of increased TrkA expression and activation in p75NTR−/− coincided with improved NGF/proNGF and vaso-protective effects observed at the ischemic stage of OIR at p12. Next, we examined impact of hypoxia; the second stage of OIR and p75NTR deletion on TrkA expression and activation. At p14, there was no significant impact of relative hypoxia or gene deletion on TrkA receptor mRNA or protein expression compared to normoxic controls (Supplementary Fig. 4). Interestingly, Two-way ANOVA analysis showed a significant impact of hypoxia on p-TrkA (Y490) activation where retinas from both WT and p75NTR hypoxic pups tended to show higher TrkA activation (1.4-fold and 1.7-fold) compared to their normoxic controls but it did not reach statistical significance (Fig. 8E,F). These results indicate that genetic deletion of p75NTR exerted vascular protective effect via preserving and enhancing TrkA cell survival signal during hyperoxic-ischemic phase more than angiogenic signal during hypoxic-angiogenic phase.
Deletion of p75NTR enhanced expression and activation of NGF survival receptor; TrkA. (A) Quantitative real-time PCR of TrkA gene expression in p12 WT and p75NTR−/− exposed to OIR showing significant increase in basal mRNA expression in p75NTR−/− retinas compared to the other groups (*significant using Two-Way ANOVA, p < 0.05, n = 6). (B,C) Representative Western blotting and bar graph statistical analysis of TrkA expression by p12 showing significant gene effect and significant increase in TrkA expression in p75NTR−/− pups compared to their corresponding WT-controls under normoxia and hyperoxia (*significant using Two-way ANOVA, p < 0.05, n = 6–9). (B,D) Representative Western blotting and bar graph statistical analysis of pTrkA activation by p12 showing an overall significant gene effect and a significant increase in pTrkA activation (Y490) in p75NTR−/− pups exposed to hyperoxia compared to the rest of the groups (*significant using Two-way ANOVA, p < 0.05, n = 6–9). (E,F) Representative Western blotting and bar graph statistical analysis of pTrkA activation by p14 showing an overall significant effect of hypoxia (#) to increase activation of pTrkA (Y490) in p75NTR−/− pups, however there was no significant difference between individual groups (n = 6–9).
TrkA activity is required for the vaso-protective effects observed in p75NTR−/− but not WT
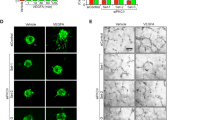
To examine the specific contribution of TrkA activation in vascular protection observed in p75NTR−/− mice, we used compound K-252a, a staurosporine analog that is potent inhibitor of various Trk receptors30. K-252a was dissolved in dimethyl sulfoxide (DMSO) and administered via intra-vitreal route (0.5 μg μl−1/eye) at p12. Animals were perfused with FITC-dextran 30 minutes before sacrifice to examine capillary perfusion and central avascular area (Fig. 9A,B, Supplementary Fig. 5A,B) and after sacrifice at p17, retinas were flat-mounted and stained with Isolectin-GS to examine RNV (Fig. 9D,E, Supplementary Fig. 5C-D). WT data showed that treatment with K-252a did not significantly alter central avascular area (Supplementary Fig. 5A,B) or RNV (Supplementary Fig. 5C,D) compared to DMSO-injected controls. In contrast, injection of K-252a into retinas of p75NTR−/− pups resulted in significant 1.34-fold increase in central avascular area (Fig. 9B,C) and 2-fold increase in RNV (Fig. 9E,F) when compared to DMSO-injected controls.
TrkA activity is required for the vaso-protective effects observed in p75NTR−/− pups. (A,B) Representatives of p17 retinal flat mounts of FITC-perfused p75NTR−/− pups exposed to OIR and receiving DMSO or K252a (0.5 μg μL−1/eye) showing central avascular area encircled in the center by white line, 5X magnification. (C) Statistical analysis of central capillary dropout (CDO) area showing significant increase of CDO in p75NTR−/− pups that received intravitreal injection of K252a compared to DMSO-receiving p75NTR−/− group or WT-pups that received K252a. There was no significant difference between WT-pups that received K252a compared to DMSO injection. (*Significant using Two-Way ANOVA, p < 0.05, n = 5–7). (D,E) Representatives of the same p75NTR−/− retinal flat mounts stained with GS-isolectin to illustrate retinal neovascularization (RNV) at retinal mid-periphery (encircled by dotted white line, 5X magnification). (F) Statistical analysis using two-way ANOVA showing significant increase of RNV in p75NTR−/− pups that received intravitreal injection of K252a compared to DMSO-injected WT or p75NTR−/− groups. There was no significant difference between WT-pups that received K252a compared to DMSO injection. (*Significant using Two-Way ANOVA, p < 0.05, n = 4–7).
Discussion
Retinal ischemia and the subsequent secretion of angiogenic factors including VEGF and NGF are thought to be common precursors to retinal neovascularization. While targeting VEGF has proven effective clinically in preventing macular edema and RNV, it deprives the retina from neuroprotective factors31. Therefore, the challenge remains to identify strategies to vascularize the ischemic retina and to maintain homeostasis of retinal function as a neurovascular unit. The findings from this study support targeting p75NTR as a novel and effective vascular protective strategy for ischemic retinopathy as follows: A. Deletion of the p75NTR reduced hyperoxia-mediated vaso-obliteration, hypoxia-induced pathological neovascularization and enhanced reparative angiogenesis in OIR model (Figs 4, 5). B. In response to hyperoxic-ischemic insult, deletion of p75NTR decreased apoptotic signal, preserved survival signal by decreasing proNGF and enhancing NGF and TrkA expression and activation (Figs 2, 3, 8). C. In response to hypoxic-angiogenic phase, deletion of p75NTR increased revascularization of central retina by maintaining VEGF/VEGFR2 activation and restoring balance between NGF and BDNF and their corresponding precursors; proNGF and proBDNF (Figs 6, 7; Suppl. Fig. 3). D. Vascular protection observed in p75NTR−/− was blunted by inhibition of NGF survival receptor; TrkA receptor at p12 (Figs 8, 9). These findings support the notion that modulating p75NTR exert vascular protection by two-fold mechanism: one by inhibiting proNGF/p75NTR apoptotic signal and one by activating NGF/TrkA survival and angiogenic signal via the cross-talk between neurotrophin receptors; p75NTR and TrkA.
Growing body of evidence support the role of neurons in regulating angiogenesis by secreting growth factors and guidance cues (reviewed in32). NGF and BDNF are members of the neurotrophin family that can exert neuroprotective, vascular protective and angiogenic effects. As adaptive response to overcome ischemia, NGF will be released, initially as a precursor, proNGF that normally get cleaved to its mature form12. We and others have shown that pro-oxidative milieu can impair maturation of NGF leading to accumulation of proNGF and activation of p75NTR pathway in the diabetic retina and ischemic heart10,11. Here, we show that exposure of WT pups to hyperoxia, a well-documented condition of oxidative and nitrative stress33,34 triggered the accumulation of proNGF compared to normoxic-controls. It is interesting that the ratio between NGF to proNGF was significantly reduced in hyperoxia that coincided with upregulation of its receptor p75NTR and vascular cell death at p12 in WT. The apoptotic action of p75NTR receptor via forming a co-receptor with sortilin is well-documented in both neuro- and microvascular degeneration (reviewed in15. While there was no change in sortilin expression in response to OIR exposure or p75NTR gene deletion, we observed upregulation of p75NTR protein expression at p12, a time point for maximum central vascular death in WT retina. In contrast, a recent study using similar OIR model showed that expression of p75NTR receptor did not change in response to hyperoxia (p8, p10 and p12), yet it showed significant increase in response to hypoxia at p14 and p1714. Nevertheless, our results came in agreement with reports showing increased expression of p75NTR in ischemic retina models including ischemia induced by elevated intra-ocular pressure35, and in an inherited retinal degeneration model, Royal College of Surgeons rats, that was associated with progressive capillary dropout and subretinal neovascularization36.
Deletion of p75NTR receptor significantly attenuated vascular cell death as assessed by attenuated capillary dropout area at p12 (Fig. 4). Our group has previously shown that vaso-obliteration at p12 is associated with increased expression of apoptotic markers including; cleaved caspase-3 and PARP as well as decreased activation of Akt25,37. Deletion of p75NTR-attenuated vascular cell death coincided with preserved activation of the survival Akt pathway as well as attenuated expression of apoptotic markers, cleaved and total-PARP (Fig. 3). Deletion of p75NTR reduced pathological retinal neovascularization (tufts) and stimulated reparative angiogenesis evident by smaller central avascular area by p17 (Fig. 5). These results lend further support to recent studies using molecular or pharmacological inhibitor of p75NTR that show vascular protection in different models of ischemic retinopathy14,38. Modulating VEGF expression was proposed as a potential contributor to vascular protection observed in p75NTR−/− mice pups38. As expected, hypoxia triggered significant increase in gene and protein expression of VEGF in WT-pups (Suppl. Figs 2A, 6A). This corroborates with previous findings22,23,38. Deletion of p75NTR did not reduce, but rather sustained hypoxia-induced VEGF expression and VEGFR2 activation (Fig. 6). These results support prior findings that gene delivery of p75NTR impaired neovascularization and blood flow recovery in diabetic mouse with hind limb ischemia through depression of VEGF39, however our results came in contrast to the results of another study by Le Moan N et al.38, which showed that genetic deletion of p75NTR receptor resulted in decreased stabilization of HIF-1α and VEGF expression by p17 in OIR model38. Our results are further supported by prior work showing that treatments that sustain VEGF/VEGFR2 activation stimulate reparative angiogenesis in addition to preventing retinal neovascularization in OIR model23.
Exposure to relative hypoxia did not alter p75NTR expression at p14 or p17, a time point of maximum retinal pathological neovascularization (Fig. 1). Our results came in agreement with a prior study showing that hypoxia stimulates shedding of p75NTR rather than modulating its expression38. Moreover, exposure to relative hypoxia resulted in significant increase in proNGF and proBDNF at p14 in WT. In agreement, vitreal samples from patients with proliferative diabetic retinopathy showed significantly low levels of mature NGF11,28 and BDNF40,41. Clinically, preterm infants who experienced proliferative retinopathy had decreased serum levels of BDNF compared to full-term42,43. Down regulation of neurotrophins; NGF, BDNF, NT-3 and GDNF was also reported in an ischemic rat retina model44. Deletion of p75NTR receptor significantly decreased the levels of proNGF and preserved the level of mature NGF (Fig. 2) at p12, an effect that was sustained at p14 (Fig. 7). Same results were also found for BDNF where, deletion of p75NTR preserved the ratio of mature BDNF level to the level of the precursor form; proBDNF (Supplementary Fig. 3). Our results lend further support to prior studies showing that silencing p75NTR expression or genetic deletion of p75NTR corroborates with restoring balance of NGF to proNGF ratio13,45, as well as balance of neurotrophin-3 (NT-3) to proNT-3 ratio46.
One of the cardinal findings of the current study is the observation that deletion of p75NTR preserved the ratio between mature NGF and its precursor proNGF under both hyperoxic-ischemic phase and hypoxic-angiogenic phase (Figs 2, 7). Restoring the balance of NGF/proNGF coincided with significant increases in expression as well as activation of TrkA receptor in p75NTR−/− pups by p12 (Fig. 8). The crosstalk between downplaying p75NTR and enhancing TrkA signal was maintained throughout the neovascularization phase of OIR, however to lesser extent (Figs 7, 8). The cross-talk between p75NTR and TrkA has been demonstrated by several groups. We and others have shown that upregulation of p75NTR expression concurred with inhibition of Trk-Y490 phosphorylation26,27,47. Treatments that enhanced TrkA-activation was associated with inverse effects on p75NTR expression26 or activation48. The current findings lend further support to prior reports showing that downregulation of p75NTR enhanced activation of TrkA pathway13,18,45,49. However, this is the first study that demonstrated shifting the balance by decreasing p75NTR and increasing TrkA in retinal angiogenesis model. Previous literature identified clear angiogenic effects of NGF via activation of TrkA in models of angiogenesis7,50. Other studies identified proangiogenic effects of proNGF via activation of TrkA in vitro and in vivo17,18,51. Therefore, our results support the hypothesis that deletion of p75NTR receptor preserved a balanced ratio of NGF/proNGF that can dictate the extent of TrkA survival and angiogenic signal and afford vascular protection. Next, we examined the impact of blocking TrkA receptor using compound K-252a, a general and potent inhibitor of Trk receptors30 that was delivered intravitreally at the end of ischemic phase at p12 (Fig. 9). Treatment with K-252a blunted the protective effects including amelioration of central avascular area and retinal neovascularization observed during hypoxic stage of OIR in p75NTR−/− pups. We believe that this is the first report that demonstrates direct involvement of TrkA to mediate vascular protection observed by genetic deletion of p75NTR receptor in both ischemic and angiogenic phases of OIR model.
Conclusion
Here, we demonstrate a number of unexplored pathways that unraveled novel pathways by which the genetic deletion of p75NTR receptor can contribute to vascular protection including; preserved NGF and BDNF signal and sustained VEGF/VEGFR2 activation that together maintain healthy retinal vasculature. Deletion of p75NTR can impact TrkA expression and activation, suggesting that the cross talk between these two receptors can be utilized not only to down regulate one of them but also to up regulate the other receptor endogenously. These findings support neurotrophins as viable therapeutic targets to combat retinal diseases with aberrant angiogenesis.
References
Cringle, S. J. & Yu, D. Y. Oxygen supply and consumption in the retina: implications for studies of retinopathy of prematurity. Doc Ophthalmol 120, 99–109, https://doi.org/10.1007/s10633-009-9197-2 (2010).
Cheung, N. & Wong, T. Y. Diabetic retinopathy and systemic vascular complications. Prog Retin Eye Res 27, 161–176, https://doi.org/10.1016/j.preteyeres.2007.12.001 (2008).
Antonetti, D. A., Klein, R. & Gardner, T. W. Diabetic retinopathy. N Engl J Med 366, 1227–1239, https://doi.org/10.1056/NEJMra1005073 (2012).
West, H., Richardson, W. D. & Fruttiger, M. Stabilization of the retinal vascular network by reciprocal feedback between blood vessels and astrocytes. Development 132, 1855–1862, https://doi.org/10.1242/dev.01732 (2005).
Nakamura-Ishizu, A. et al. The formation of an angiogenic astrocyte template is regulated by the neuroretina in a HIF-1-dependent manner. Dev Biol 363, 106–114, https://doi.org/10.1016/j.ydbio.2011.12.027 (2012).
Moran, E. P. et al. Neurovascular cross talk in diabetic retinopathy: Pathophysiological roles and therapeutic implications. Am J Physiol Heart Circ Physiol 311, H738–749, https://doi.org/10.1152/ajpheart.00005.2016 (2016).
Salis, M. B. et al. Nerve growth factor supplementation reverses the impairment, induced by Type 1 diabetes, of hindlimb post-ischaemic recovery in mice. Diabetologia 47, 1055–1063, https://doi.org/10.1007/s00125-004-1424-5 (2004).
Wang, J. et al. NGF increases VEGF expression and promotes cell proliferation via ERK1/2 and AKT signaling in Muller cells. Mol Vis 22, 254–263 (2016).
Vera, C., Tapia, V., Vega, M. & Romero, C. Role of nerve growth factor and its TRKA receptor in normal ovarian and epithelial ovarian cancer angiogenesis. J Ovarian Res 7, 82, https://doi.org/10.1186/s13048-014-0082-6 (2014).
Siao, C. J. et al. ProNGF, a cytokine induced after myocardial infarction in humans, targets pericytes to promote microvascular damage and activation. J Exp Med 209, 2291–2305, https://doi.org/10.1084/jem.20111749 (2012).
Ali, T. K. et al. Diabetes-induced peroxynitrite impairs the balance of pro-nerve growth factor and nerve growth factor, and causes neurovascular injury. Diabetologia 54, 657–668, https://doi.org/10.1007/s00125-010-1935-1 (2011).
Huang, E. J. & Reichardt, L. F. Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci 24, 677–736, https://doi.org/10.1146/annurev.neuro.24.1.677 (2001).
Shanab, A. Y., Mysona, B. A., Matragoon, S. & El-Remessy, A. B. Silencingp75(NTR) prevents proNGF-induced endothelial cell death and development of acellular capillaries in rat retina. Mol Ther Methods Clin Dev 2, 15013, https://doi.org/10.1038/mtm.2015.13 (2015).
Barcelona, P. F. et al. p75NTR and Its Ligand ProNGF Activate Paracrine Mechanisms Etiological to the Vascular, Inflammatory, and Neurodegenerative Pathologies of Diabetic Retinopathy. J Neurosci 36, 8826–8841, https://doi.org/10.1523/JNEUROSCI.4278-15.2016 (2016).
Elshaer, S. & El-Remessy, A. Implication of the neurotrophin receptor p75NTR in vascular diseases: beyond the eye. Expert Review of Ophthalmology 12, 149–158 (2017).
Nykjaer, A. et al. Sortilin is essential for proNGF-induced neuronal cell death. Nature 427, 843–848, https://doi.org/10.1038/nature02319 (2004).
Demont, Y. et al. Pro-nerve growth factor induces autocrine stimulation of breast cancer cell invasion through tropomyosin-related kinase A (TrkA) and sortilin protein. J Biol Chem 287, 1923–1931, https://doi.org/10.1074/jbc.M110.211714 (2012).
Elshaer, S. L. et al. Pronerve growth factor induces angiogenesis via activation of TrkA: possible role in proliferative diabetic retinopathy. J Diabetes Res 2013, 432659, https://doi.org/10.1155/2013/432659 (2013).
Cunha-Vaz, J. & Bernardes, R. Nonproliferative retinopathy in diabetes type 2. Initial stages and characterization of phenotypes. Prog Retin Eye Res 24, 355–377, https://doi.org/10.1016/j.preteyeres.2004.07.004 (2005).
Lee, K. F. et al. Targeted mutation of the gene encoding the low affinity NGF receptor p75 leads to deficits in the peripheral sensory nervous system. Cell 69, 737–749 (1992).
Smith, L. E. et al. Oxygen-induced retinopathy in the mouse. Invest Ophthalmol Vis Sci 35, 101–111 (1994).
Abdelsaid, M. A., Matragoon, S. & El-Remessy, A. B. Thioredoxin-interacting protein expression is required for VEGF-mediated angiogenic signal in endothelial cells. Antioxid Redox Signal 19, 2199–2212, https://doi.org/10.1089/ars.2012.4761 (2013).
Shanab, A. Y. et al. Candesartan stimulates reparative angiogenesis in ischemic retinopathy model: role of hemeoxygenase-1 (HO-1). Angiogenesis 18, 137–150, https://doi.org/10.1007/s10456-014-9451-4 (2015).
Banin, E. et al. T2-TrpRS inhibits preretinal neovascularization and enhances physiological vascular regrowth in OIR as assessed by a new method of quantification. Invest Ophthalmol Vis Sci 47, 2125–2134, https://doi.org/10.1167/iovs.05-1096 (2006).
Abdelsaid, M. A., Matragoon, S., Ergul, A. & El-Remessy, A. B. Deletion of thioredoxin interacting protein (TXNIP) augments hyperoxia-induced vaso-obliteration in a mouse model of oxygen induced-retinopathy. PLoS One 9, e110388, https://doi.org/10.1371/journal.pone.0110388 (2014).
Al-Gayyar, M. M. et al. Epicatechin blocks pro-nerve growth factor (proNGF)-mediated retinal neurodegeneration via inhibition of p75 neurotrophin receptor expression in a rat model of diabetes [corrected]. Diabetologia 54, 669–680, https://doi.org/10.1007/s00125-010-1994-3 (2011).
Ali, T. K., Matragoon, S., Pillai, B. A., Liou, G. I. & El-Remessy, A. B. Peroxynitrite mediates retinal neurodegeneration by inhibiting nerve growth factor survival signaling in experimental and human diabetes. Diabetes 57, 889–898, https://doi.org/10.2337/db07-1669 (2008).
Mysona, B. A. et al. Imbalance of the nerve growth factor and its precursor as a potential biomarker for diabetic retinopathy. Biomed Res Int 2015, 571456, https://doi.org/10.1155/2015/571456 (2015).
Ozaki, H. et al. Blockade of vascular endothelial cell growth factor receptor signaling is sufficient to completely prevent retinal neovascularization. Am J Pathol 156, 697–707, https://doi.org/10.1016/S0002-9440(10)64773-6 (2000).
Knusel, B. & Hefti, F. K-252 compounds: modulators of neurotrophin signal transduction. J Neurochem 59, 1987–1996 (1992).
Saint-Geniez, M. et al. Endogenous VEGF is required for visual function: evidence for a survival role on muller cells and photoreceptors. PLoS One 3, e3554, https://doi.org/10.1371/journal.pone.0003554 (2008).
Wilson, A. & Sapieha, P. Neurons and guidance cues in retinal vascular diseases. Oncotarget 7, 9618–9619, https://doi.org/10.18632/oncotarget.7413 (2016).
Stevenson, L. et al. Reduced nitro-oxidative stress and neural cell death suggests a protective role for microglial cells in TNFalpha−/− mice in ischemic retinopathy. Invest Ophthalmol Vis Sci 51, 3291–3299, https://doi.org/10.1167/iovs.09-4344 (2010).
Suwanpradid, J., Rojas, M., Behzadian, M. A., Caldwell, R. W. & Caldwell, R. B. Arginase 2 deficiency prevents oxidative stress and limits hyperoxia-induced retinal vascular degeneration. PLoS One 9, e110604, https://doi.org/10.1371/journal.pone.0110604 (2014).
Wei, Y. et al. Enhanced protein expressions of sortilin and p75NTR in retina of rat following elevated intraocular pressure-induced retinal ischemia. Neurosci Lett 429, 169–174, https://doi.org/10.1016/j.neulet.2007.10.012 (2007).
Shen, W. et al. Systemic administration of erythropoietin inhibits retinopathy in RCS rats. PLoS One 9, e104759, https://doi.org/10.1371/journal.pone.0104759 (2014).
Abdelsaid, M. A. et al. Early intervention of tyrosine nitration prevents vaso-obliteration and neovascularization in ischemic retinopathy. J Pharmacol Exp Ther 332, 125–134, https://doi.org/10.1124/jpet.109.157941 (2010).
Le Moan, N., Houslay, D. M., Christian, F., Houslay, M. D. & Akassoglou, K. Oxygen-dependent cleavage of the p75 neurotrophin receptor triggers stabilization of HIF-1alpha. Mol Cell 44, 476–490, https://doi.org/10.1016/j.molcel.2011.08.033 (2011).
Caporali, A. et al. Neurotrophin p75 receptor (p75NTR) promotes endothelial cell apoptosis and inhibits angiogenesis: implications for diabetes-induced impaired neovascularization in ischemic limb muscles. Circ Res 103, e15–26, https://doi.org/10.1161/circresaha.108.177386 (2008).
Ola, M. S., Nawaz, M. I., El-Asrar, A. A., Abouammoh, M. & Alhomida, A. S. Reduced levels of brain derived neurotrophic factor (BDNF) in the serum of diabetic retinopathy patients and in the retina of diabetic rats. Cell Mol Neurobiol 33, 359–367, https://doi.org/10.1007/s10571-012-9901-8 (2013).
Kaviarasan, K. et al. Low blood and vitreal BDNF, LXA4 and altered Th1/Th2 cytokine balance are potential risk factors for diabetic retinopathy. Metabolism 64, 958–966, https://doi.org/10.1016/j.metabol.2015.04.005 (2015).
Rao, R. et al. Brain-derived neurotrophic factor in infants < 32 weeks gestational age: correlation with antenatal factors and postnatal outcomes. Pediatr Res 65, 548–552, https://doi.org/10.1203/PDR.0b013e31819d9ea5 (2009).
Hellgren, G. et al. Proliferative retinopathy is associated with impaired increase in BDNF and RANTES expression levels after preterm birth. Neonatology 98, 409–418, https://doi.org/10.1159/000317779 (2010).
Guo, X. J. et al. Dysregulation of neurotrophic and inflammatory systems accompanied by decreased CREB signaling in ischemic rat retina. Exp Eye Res 125, 156–163, https://doi.org/10.1016/j.exer.2014.06.003 (2014).
Mohamed, R., Shanab, A. Y. & El-Remessy, A. B. Deletion of the Neurotrophin Receptor p75NTR Prevents Diabetes-Induced Retinal Acellular Capillaries in Streptozotocin-Induced Mouse Diabetic Model. J Diabetes Metab Disord Control 4 https://doi.org/10.15406/jdmdc.2017.04.00129 (2017).
Shen, W., Zhu, L., Lee, S. R., Chung, S. H. & Gillies, M. C. Involvement of NT3 and P75(NTR) in photoreceptor degeneration following selective Muller cell ablation. J Neuroinflammation 10, 137, https://doi.org/10.1186/1742-2094-10-137 (2013).
Ostendorf, G. M. Naturopathy and alternative medicine–definition of the concept and delineation. Offentl Gesundheitswes 53, 84–87 (1991).
Barcelona, P. F. & Saragovi, H. U. A Pro-Nerve Growth Factor (proNGF) and NGF Binding Protein, alpha2-Macroglobulin, Differentially Regulates p75 and TrkA Receptors and Is Relevant to Neurodegeneration Ex Vivo and In Vivo. Mol Cell Biol 35, 3396–3408, https://doi.org/10.1128/MCB.00544-15 (2015).
Mysona, B. A. et al. Modulation ofp75(NTR) prevents diabetes- and proNGF-induced retinal inflammation and blood-retina barrier breakdown in mice and rats. Diabetologia 56, 2329–2339, https://doi.org/10.1007/s00125-013-2998-6 (2013).
Liu, X. et al. Neuronal-driven angiogenesis: role of NGF in retinal neovascularization in an oxygen-induced retinopathy model. Invest Ophthalmol Vis Sci 51, 3749–3757, https://doi.org/10.1167/iovs.09-4226 (2010).
Bradshaw, R. A. et al. NGF and ProNGF: Regulation of neuronal and neoplastic responses through receptor signaling. Adv Biol Regul 58, 16–27, https://doi.org/10.1016/j.jbior.2014.11.003 (2015).
Acknowledgements
This work is supported by pre-doctoral fellowship from American Heart Association (15PRE22830019) to Sally Elshaer, a pre-doctoral scholarship from Egyptian Educational Bureau to Sally Elshaer (GM-1000) and RO-1-EY-022408 to Azza El-Remessy. This material is the result of work supported with resources and the use of facilities at the Charlie Norwood VA medical center, Augusta, GA. We would like to thank Dr. Bruce Carter (Dept. of Biochemistry, Vanderbilt University) for providing p75NTR antibody. The contents do not represent the views of the U.S. Department of Veterans Affairs or NIH funding.
Author information
Authors and Affiliations
Contributions
Sally L. Elshaer planned and performed the experiments, analyzed the data and wrote the manuscript. This study was designed, coordinated by Azza B. El-Remessy as the principal investigator, providing conceptual and technical guidance for all aspects of the project in addition to editing the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elshaer, S.L., El-Remessy, A.B. Deletion of p75NTR prevents vaso-obliteration and retinal neovascularization via activation of Trk- A receptor in ischemic retinopathy model. Sci Rep 8, 12490 (2018). https://doi.org/10.1038/s41598-018-30029-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-30029-0
- Springer Nature Limited