Abstract
Nociceptors, sensory neurons that detect damage or potential damage to the body, are the first stage of communicating noxious stimuli from the periphery to central nervous system (CNS). In this study, long-term potentiation (LTP) in the CNS of the medicinal leech, Hirudo verbana, was examined, taking advantage of the ability to selectively record from nociceptive synapses in this model organism. High frequency stimulation (HFS) of nociceptors produced a persistent increase in synaptic transmission and this LTP was both NMDA receptor-mediated and synapse-specific. Surprisingly, inhibition of NMDA receptors during HFS “uncovered” a persistent form of depression. This long-term depression (LTD) was mediated by the endocannabinoid 2-arachidonoyl glycerol (2-AG) acting on a TRPV (transient receptor potential vanilloid) –like channel. These observations suggest that (1) NMDA receptor mediated LTP is observed in nociceptors across both vertebrate and invertebrate phyla and (2) there may be an interaction between NMDA receptor-mediated and endocannabinoid-mediated forms of synaptic plasticity in nociceptors. Specifically, the NMDA receptor mediated processes may suppress endocannabinoid signaling. Such findings could be significant for understanding cellular mechanisms behind nociceptive sensitization and perhaps their contribution to chronic pain.
Similar content being viewed by others
Introduction
Nociceptors are somatosensory neurons dedicated to detecting damaging or potentially damaging stimuli, which then transmit that information to the rest of the central nervous system (CNS)1. In both invertebrates and vertebrates, nociceptors are distinct from somatosensory neurons that detect and transmit innocuous mechanical and thermal stimuli2,3,4. Activation of nociceptors can elicit motor behaviors designed to withdraw or escape from the noxious stimuli2,5,6,7, but these afferents also activate modulatory circuits that help the animal adapt in its encounter with these stimuli8,9,10. Consequently, changes in the strength of synaptic transmission by nociceptors represent a critical element in how animals respond to future encounters with noxious stimuli. For example, potentiation of nociceptive synapses can contribute to behavioral sensitization that serves to protect the animal from further damage10. Elucidating the cellular mechanisms that mediate potentiation of nociceptive synapses will contribute to our understanding of how animals make adaptive changes following injury. Such information could also be applied to develop new therapeutic approaches in treating pathological forms of nociceptive sensitization in humans, i.e. chronic pain.
Long-term potentiation (LTP) is a form of activity-dependent synaptic strengthening observed practically throughout the CNS and has been documented in vertebrate11 and invertebrate species12,13,14. It is a homosynaptic form of synaptic plasticity and is often mediated by NMDA receptors (NMDARs) that require both glutamate-binding and postsynaptic depolarization in order to become active. One can envision that injury-inducing stimuli could activate nociceptors enough to elicit LTP in nociceptive synapses in the spinal cord and, in fact, NMDAR-mediated LTP (NMDAR-LTP) has been observed in rodent C fiber synapses following repetitive stimulation11,15,16. However, studying LTP in nociceptive synapses in the spinal cord and its contribution to nociceptive sensitization is complicated by a variety of factors. These include the perhaps surprising complexity of the nociceptive and non-nociceptive circuitry in the spinal cord17,18, the fact that different patterns of stimulation are required for LTP in different nociceptive pathways11,15,16 and questions as to whether NMDAR-LTP in spinal nociceptive synapses exhibit synapse specificity similar to what is observed in other regions of the CNS19.
The CNS of Hirudo verbana (the medicinal leech) provides a tractable system to examine LTP in nociceptive synapses. The Hirudo CNS is organized as a chain of ganglia that runs the length of the animal with each ganglion having its own complement of touch- and pressure sensitive neurons as well as bilateral pairs of mechano-sensitive and polymodal nociceptive neurons20,21,22,23. Hirudo nociceptive sensory neurons (N cells) have glutamatergic synaptic input to motor neurons and interneurons responsible for reflexive withdrawal and locomotory escape behaviors6,24,25. These N cells also have input onto serotonergic cells that play a critical role in modulating Hirudo behaviors in the context of learning and responses to stress and hunger26,27,28,29. Hirudo pressure (P) cell and touch (T) cell synapses do undergo LTP that is NMDAR-dependent and synapse specific14,30,31, but LTP in the leech N cell synapses has not been investigated. In this study we examined the capacity of Hirudo N synapses to undergo NMDAR-LTP. We found evidence supporting the presence of LTP in these nociceptive synapses that is NMDAR-dependent and synapse-specific, but also found, to our surprise, evidence of an interaction between NMDAR-mediated synaptic potentiation and endocannabinoid-mediated synaptic depression.
Materials and Methods
Hirudo verbana (3g) were obtained from commercial suppliers (Leeches USA, Westbury, NY and Niagara Leeches, Niagara, NY) and maintained in artificial pond water [0.52 g acquarium salt (Instant Ocean) per liter of H2O] on a 12 hour light/dark cycle at 15 °C. Individual ganglia were dissected and pinned in a recording chamber with constant perfusion of normal Hirudo saline (110 mM NaCl, 5 mM NaOH, 4 mM KCl, 1.8 mM CaCl2, 1 mM MgCl2, and 10 mM HEPES, pH = 7.4) at an approximately 1.5 mL/min.
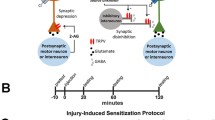
Individual neurons were identified based on their position within the ganglion, size, and electrophysiological properties. Each ganglion contains two bilateral pairs of nociceptive (N) and pressure-sensitive cells and three pairs of light touch sensitive neurons. The N cells are further divided into a lateral N pair that are polymodal nociceptors (Npoly) and a medial pair that are mechanical nociceptors (Nmech; Fig. 1A)21,22,23. One postsynaptic target of the N cells is the longitudinal (L) motor neuron which contributes to the defensive withdrawal reflex elicited by noxious stimulation32. L motor neuron identification was confirmed by recording from the electrically coupled contralateral L motor neuron and observe synchronous activity33. The L cells are located on the dorsal side of the ganglion and the afferent cells are located on the ventral surface. However, it is possible to record from both the L and the Npoly or P cells from the dorsal side of the ganglion given the lateral positions of both these primary afferents. It is not possible to record from the Nmech cell when the ganglion is dorsal side up because this neuron is located medially on the ventral side of the ganglion. Therefore, synaptic recordings were made from the Nmech and anterior pagoda (AP) neurons which also receive input from both types of N cells (Fig. 1A) and are all located on the ventral side of the ganglion. Where they have been compared, the properties of N- or P-to-AP synapses appear to be identical to other synaptic connections made by these sensory cells34,35.
(A) Circuit diagram of neurons recorded in this study. A single body ganglion (1 of 21) is shown. Each ganglion has two pairs of segmental nerves that project to the periphery and a connective nerve that links each ganglion to its anterior and posterior neighbors. All of the neurons shown are actually bilateral pairs, but the contralateral cells for all but the NP have been omitted in this figure for the sake of clarity. In all LTP experiments, HFS was delivered to the lateral N cell (also known as the polymodal N or Np). To test synapse specificity of LTP between bilateral pairs of Np cells, changes in synaptic transmission was compared between the ipsilateral N and the contralateral N signaling onto the L motor neuron. To test synapse specificity of LTP between ipsilateral pairs of N cells, changes in synaptic transmission was compared between the lateral NP and the medial N (also known as the mechanical N cell or Nm) signaling onto the AP neuron. (B) Experimental protocol. Following a pre-test of the N cell EPSP, HFS was applied (see Methods) followed by a 60 min consolidation period and then a post-test measurement of the same N cell synapse.
Drugs used for each experiment were kept as frozen aliquot solutions and then diluted to their final concentration in normal saline just before respective experiment. SB366791 (SB) and tetrahydrolipstatin (THL, also known as Orlistat) were obtained from Tocris/Bio-Techne (Minneapolis, MN) and stocks were made in dimethyl sulfoxide (DMSO). DL-2-Amino-5-phosphonopentanoic acid (AP5; stocks made in normal saline), 1,2-bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid (BAPTA), and DMSO were obtained from Sigma-Aldrich (St. Louis, MO).
Electrophysiological recording techniques have been described in detailed previously36. Briefly, current clamp (bridge balanced) intracellular recordings were carried out using sharp glass microelectrodes (tip resistance 20–35 MΩ) made from borosilicate capillary tubing (1.0 mm OD, 0.75 mm ID; FHC, Bowdoinham, ME) using a horizontal puller (Sutter Instruments P-97; Novato, CA). Microelectrodes were filled with 3 M potassium acetate (KAc). Manual micropositioners (Model 1480; Siskiyou Inc., Grants Pass, OR) were used to impale individual neurons during experiments. Current pulses were delivered to electrodes using a STG 1004 Multi-Channel Systems programmable stimulator (Reutlingen, Germany). Membrane potential data were recorded using a bridge amplifier (BA-1S; NPI, Tamm, Germany) and digitally converted for analysis using a Digidata 1322 A (Molecular Devices, Sunnyvale, CA).
High-frequency stimulation (HFS) of Npoly consisted of 20 trains, 10 action potentials per train at 25 Hz, and a 10 s inter-train interval. EPSP and IR recordings were made prior to (pretest) and 60 min following HFS (Fig. 1B). This procedure has been used in previous experiments to elicit homosynaptic and heterosynaptic forms of LTP14,37. In experiments using BAPTA, 1 mM BAPTA was included in the electrode filling solution and the Ca2+ chelator was iontophoretically injected into the L motor neuron (-1nA holding current for 5 minutes) prior to HFS. For all experiments, electrodes were withdrawn following pre-test recordings and the neurons re-impaled for the post-test recordings approximately 1 hr later. This was done, in part, because prolonged recordings with these sharp microelectrodes results in rundown of EPSP amplitudes likely due to damage to the recorded cell. To insure that changes in synaptic transmission were not the result of changes in post-synaptic input resistance (IR), this was monitored during the pre- and post-test recordings by measuring the membrane potential change during a 500 msec, 1 nA negative current pulse. Only stable recordings (<10% change in IR) were included in the final data analysis. During the pre- and post-tests, EPSPs were elicited at 0.1 Hz and the peak amplitude was calculated from the average of 5–10 EPSPs. EPSP amplitude measurements of the pre- and post-test recordings were normalized and presented as the mean ± SE. Statistical analyses using t-test and one-way analysis of variance (ANOVA) were performed to determine main effects with Student-Newman-Keuls post hoc tests to confirm the ANOVA results. All analyses were carried out with Sigmaplot. All significance was determined at α level of at least P ≤ 0.05.
Results
The first experiments were to assess whether HFS of a single nociceptor could elicit LTP that was synapse specific. L motor neurons receive synaptic input from both contralateral and ipsilateral afferents and the morphology of both the motor neuron and sensory cell arborizations permits synaptic contacts on both sides of the ganglion24,38. HFS of a Npoly cell did result in substantial potentiation of the EPSP elicited in the ipsilateral L motor neuron recorded 1 hr later (Fig. 2A; ANOVA F2,14 = 60.95; post-hoc HFS vs. control p < 0.001). No potentiation was observed in the synapse made by contralateral Npoly onto the same postsynaptic L cell (Figs 1A and 2A; post-hoc p > 0.05). A separate set of experiments was carried to examine whether HFS of one nociceptor would influence synaptic transmission by an ipsilateral nociceptive afferent onto the same postsynaptic target. Specifically, these effects were examined in the Npoly and Nmech synapses onto a shared AP neuron that was ipsilateral relative to both nociceptors (Fig. 1A). Again, HFS of the lateral Npoly produced significant potentiation 1 hr later compared to the medial Nmech, in which no potentiation was observed (Fig. 2B; t1,8 = 2.58, p < 0.05). Together these results demonstrate that HFS on a single nociceptor elicits homosynaptic LTP that is synapse specific.
Synapse specificity of LTP in Hirudo nociceptive synapses. (A) HFS elicited substantial potentiation in the N-to-L synapse that underwent LFS (HFS synapse), but no change in EPSP size was observed in the synapse made by the contralateral N cell onto the same postsynaptic target (Contralateral synapse). No change in the N-to-L EPSP was observed in experiments in which the HFS was omitted (Control). (B) In experiments in which both N cells were ipsilateral to each other, HFS of the lateral N cell elicited potentiation in that nociceptor’s synapses, but no potentiation was observed in the synapse made by the ipsilateral N cell (the Nm) onto the same postsynaptic target.
Next, the potential role of NMDARs during N-cell LTP was examined. HFS was delivered in the presence of 100 µM AP5 which has been shown to block LTP in Hirudo and Aplysia synapses13,14,31,39. AP5 delivered alone (no HFS) had no persistent effect on the nociceptive synapses (Fig. 3). No LTP was observed in N-to-L synapses that received HFS in the presence of AP5. Surprisingly, a statistically significant decrease in the N-to-L EPSP was observed in these HFS + AP5 synapses (Fig. 3; F2,14 = 58.23, post-hoc for AP5 vs. AP5 + HFS p < 0.001). Furthermore, the synapse made by the contralateral N cell onto the same postsynaptic L motor neuron, which was unchanged by HFS in normal saline (Fig. 2A), was depressed in the AP5 + HFS condition (Fig. 3; post-hoc for AP5 vs. Contralateral AP5 + HFS p < 0.001). A comparison of the contralateral synapses tested in the AP5 + HFS (Fig. 3) condition versus the contralateral synapses tested in the HFS + saline condition also reveal a statistically significant difference (Fig. 2A; t = 9.76, p < 0.0001). These finding indicate that (1) LTP in the N-to-L synapse is mediated by NMDARs, (2) blocking NMDAR activity during HFS uncovers a form of long-term depression (LTD) in the activated synapse and (3) this same HFS produced heterosynaptic LTD in the synapses made by the contralateral N cell onto the same postsynaptic target.
LTD was observed in the HFS + AP5 conditions. Experiments were repeated in which synaptic transmission by contralateral N cells onto a shared postsynaptic target were measured (see Fig. 1A), but in the presence of AP5. In these conditions, LTD was observed in the N cell synapse that received HFS. LTD was also observed in the contralateral synapse that did not receive HFS. AP5 alone does not alter EPSP magnitude in these experiments.
What mechanism might explain the appearance of LTD in synapses that receive HFS in the presence of a NMDAR antagonist? Low frequency stimulation (LFS) of afferents with input onto the L motor neuron elicits LTD in N-to-L synapses that is heterosynaptic and mediated by endocannabinoid signaling. Specifically, LFS (1 Hz stimulation for 15 mins) elicits synthesis of the endocannabinoid 2-arachidonoyl glycerol (2-AG) in the postsynaptic L motor neuron, which then acts in a retrograde manner on TRPV-like channels on the N cell to produce synaptic depression6,36,40. Perhaps, blocking NMDAR activity during HFS of afferent-to-L inputs is permissive to a form of 2-AG/TRPV signaling that is normally inhibited by HFS. To test this hypothesis, we first tested the effects of HFS plus AP5 in synapses in which the DAG lipase inhibitor THL (10 µM; DAG lipase is responsible for 2-AG synthesis) was iontophoretically injected into the L motorneuron (HFS + AP + THL). No change in EPSP amplitude was observed in the HFS + AP5 + THL group indicating that the synaptic depression previously observed in the HFS + AP5 condition had been blocked (Fig. 4; F8,42 = 10.92, p < 0.001; HFS + AP5 vs. HFS + AP5 + THL post-hoc p < 0.01). Injection of THL into the L cell with AP5, but without HFS had no effect on EPSP amplitude.
LTD observed in the HFS + AP5 conditions require endocannabinoid signaling. LTD was blocked in experiments where the HFS + AP5 treatment also included either injection of THL into the L motor neuron (HFS + AP5 + THL) or injection of SB366791 into the N cell (HFS + AP5 + SB). In experiments where HFS was omitted, neither AP5 + THL nor AP5 + SB produce any change in the N-to-L EPSP. In experiments in which HFS was delivered in the presence of 2-AG, no potentiation was observed, indicating that N cell synapses that received HFS were still sensitive to 2-AG. Postsynaptic injection of BAPTA prior to HFS prevented both LTP and LTD of the nociceptive synapses.
Next, we examined the role of the TRPV-like receptor. TRPV1 can act as an endocannabinoid receptor in mammals and a TRPV-like channel has been proposed to have a similar function in invertebrates4,41,42. In these experiments, we utilized SB366791, a selective inhibitor of mammalian TRPV1 channels that acts as a competitive antagonist at the intracellular capsaicin binding site, but also blocks TRPV1 activation by other stimuli43. In Hirudo, SB366791 has been shown to block the ability of 2-AG to elicit LTD in N cell synapses and also blocks LFS-elicited depression at these synapses6,36. In the current experiments the SB366791was iontophoretically injected into the N cell prior to HFS in ganglia that were also treated with AP5 (HFS + AP5 + SB). No change in EPSP amplitude was observed in the HFS + AP5 + SB group indicating that the synaptic depression previously observed in the HFS + AP5 condition had been blocked as when the presynaptic TRPV-like channel was inhibited (Fig. 4; HFS + AP5 vs. HFS + AP5 + SB post-hoc p < 0.01). Injection of SB366791 in the N cell with AP5, but without HFS had no effect on EPSP amplitude.
2-AG normally elicits significant depression in synapses that have not undergone HFS6,36,40,44. Therefore, the ability of synapses that had undergone HFS to respond to 2-AG was examined to determine whether these synapses were still sensitive to this endocannabinoid. 100 µM 2-AG was bath-applied to ganglia during HFS and then the EPSP amplitude was tested 60 mins later. In these experiments, neither potentiation nor depression of the EPSP was observed. This suggests that the potentiation induced by the HFS was offset by the depression induced by 2-AG, with the end result being no change in EPSP amplitude.
Finally, the potential role of intracellular Ca2+ was examined since increases in intracellular postsynaptic Ca2+ are required for both NMDAR-LTP in other Hirudo synapses and endocannabinoid-mediated LTD (eCB-LTD) in nociceptive synapses30,31,44. In experiments where 1 mM BAPTA was iontophoretically injected into the L motor neuron, HFS failed to elicit any change in the EPSP amplitude (Fig. 4) indicating that there was no LTP (HFS vs. HFS + BAPTA post-hoc p < 0.001) nor LTD (HFS + AP5 vs. HFS + BAPTA post-hoc p < 0.05). Injection of BAPTA into the L motor neuron without HFS had no effect on EPSP amplitude. These findings suggest that postsynaptic Ca2+ signaling is required for both NMDAR-LTP and the eCB-LTD that is uncovered when LTP is blocked.
Discussion
How activity-driven processes potentiate synaptic transmission by nociceptors is of critical interest since such changes are likely to contribute to behavioral sensitization to nociceptive stimuli10. LTP mediated by NMDARs, previously shown to be an important modulator of synaptic signaling in other regions of the CNS, has been considered a potential candidate for contributing to nociceptive sensitization45,46. Indeed, NMDAR-LTP has been observed in nociceptive synapses in the mammalian spinal cord, although different nociceptive pathways appear to require different patterns of activity to elicit LTP15,47. There has been some question in the mammalian literature as to whether NMDAR-LTP, which is synapse specific in other regions of the CNS, contributes to nociceptive sensitization since this can include both sensitization at the site of injury (primary hyperalgesia) and away from the site of injury (secondary hyperalgesia)48,49. The present study using Hirudo shows that nociceptive synapses are capable of NMDAR-LTP that is synapse specific. This was observed for both contralateral pairs of nociceptors and ipsilateral pairs that converge onto a common postsynaptic target. Earlier studies in Hirudo and in Aplysia (a marine mollusk) have also demonstrated NMDAR-mediated LTP in afferent synapses that is synapse specific14,50,51.
That synapse-specific NMDAR-LTP occurs in primary nociceptors synapses is not inconsistent with the presence of both primary and secondary nociceptive sensitization in invertebrates or hyperalgesia in mammals. Noxious stimuli that elicit homosynaptic LTP in primary nociceptors can also initiate heterosynaptic modulatory processes that contribute to sensitization52. Furthermore, the homosynaptic processes are in a position to contribute to primary (or site-specific) sensitization while heterosynaptic processes can contribute to generalized or secondary sensitization via a wide variety of modulatory mechanisms10. In one recent examples of such a process, induction of LTP in rodent C fiber synapses was found to stimulate the release of cytokines and D-serine by astrocytes and/or microglia which produced heterosynaptic potentiation in non-activated nociceptive synapses16. Interestingly, in Hirudo, the same HFS of N cells that produced NMDAR-LTP in nociceptive synapses also produced a heterosynaptic potentiation in pressure (P) cell synapses37. In this situation, potentiation of the P cell synapses was due to endocannabinoid-mediated depression of GABAergic input to P cells, resulting in a persistent form of disinhibition. These findings were replicated in semi-intact preparations in which HFS of N cells in the same segment or noxious stimuli delivered several segments away produced endocannabinoid-mediated potentiation of P cell synapses and sensitization of reflexive withdrawal behaviors elicited by P cell activation53.
It is surprising that AP5 treatment during N cell HFS resulted not only in blocking LTP, but also uncovering LTD in these activated nociceptive synapses and a potential mechanism for how these two forms of plasticity interact is presented in Fig. 5. This LTD was blocked by inhibitors of 2-AG synthesis and TRPV channel function, similar to the eCB-LTD observed in N cell synapses following LFS of the non-nociceptive T cells6,36,40. One feature that both LTP and LTD share is activation of the same postsynaptic cell, the L motor neuron. In the case of eCB-LTD, pharmacological studies have shown that the L motor neuron is the site of 2-AG synthesis6,40. Similarly in the present studies, injection of THL into the L motor neuron also blocked the LTD observed in synapses that received HFS in the presence of the NMDAR antagonist AP5. Another similarity was that injection of the TRPV inhibitor SB366791 into the N cell also blocked both LFS-induced LTD and depression observed following HFS in the presence of AP5. In previous studies eCB-LTD produced by LFS was heterosynaptic, with depression observed at both the active and inactive synapses that converge onto a common postsynaptic target (in this case the L motor neuron)40. This heterosynaptic feature was also observed in the eCB-LTD observed in the HFS + AP5 conditions. LTD was observed both at the N cell synapse that underwent HFS and by the synapse made by the inactive, contralateral N cell onto the same postsynaptic target. To summarize, the synaptic depression observed when inhibiting NMDARs during HFS is indistinguishable from the eCB-LTD produced by LFS in past experiments.
Proposed model of interaction between synaptic modulatory processes. High frequency stimulation (HFS) of an N cell activates postsynaptic NMDARs (in this case in the L motor neuron) that lead to LTP, but only in the activated synapse (i.e. a homosynaptic effect). This LTP is Ca2+-dependent, consistent with previous studies of NMDAR-mediated LTP in other Hirudo mechanosensory synapses14,30,31. This same increase in Ca2+ is thought to inhibit DAG lipase (DAGL) –mediated 2-AG synthesis, which can also be initiated by synaptic activity36,44. It is not clear if this inhibition of DAGL is due to a direct effect of Ca2+ or through Ca2+-dependent activation of an intermediary, e.g. a protein kinase. If NMDAR activity is blocked, then 2-AG synthesis is permitted, which in turn elicits eCB-LTD in both the stimulated and control N cell inputs (i.e. a homo- and heterosynaptic effect).
These results suggest that whether nociceptive synapses undergo NMDAR-LTP or eCB-LTD is determine by the pattern of activity, with LFS producing eCB-LTD and HFS preferentially producing NMDAR-LTP, but also being capable of eliciting eCB-LTD. In previous experiments using LFS, no evidence of LTP was observed when LFS-induced eCB-LTD was blocked using either pre-synaptic THL or postsynaptic SB366791 injections6,36,44. This suggests that only HFS has the capacity to engage both NMDAR- and eCB-mediated forms of synaptic plasticity. Based on our BAPTA results, both forms of synaptic plasticity require postsynaptic Ca2+ signaling in agreement with earlier studies30,44. Nevertheless, the activation of NMDARs not only elicits synaptic potentiation, but also actively inhibits eCB-LTD in these synapses. Exactly what intracellular mechanisms mediated NMDAR-LTP in N cell synapses is unknown at this time, however in Hirudo P cell synapses, NMDAR-LTP requires, in addition to increases in intracellular Ca2+, activation of protein kinases including CamKII30,31. CamKII has been shown to phosphorylate mammalian forms of the 2-AG synthesizing enzymes, DAG lipase, resulting in an inhibition of 2-AG synthesis that did reduce eCB-mediated synaptic depression54. It would be interesting to examine not only the potential role of CaMKII, but also whether different levels of Ca2+ signaling selectively activate NMDAR-LTP vs. eCB-LTD. However, an important caveat of this potential mechanism is that it is not known if Hirudo DAG lipase is similarly regulated by CaMKII. Also studies using other kinases have been shown that they actually increase DAG lipase activity55. Alternative mechanisms include regulation of DAG lipase via interactions with Homer or palmitoylation55 or regulation at a site independent of DAG lipase, e.g., the 2-AG metabolizing enzyme, MAG lipase. All of these processes imply that HFS-induced inhibition of eCB-LTD is acting at the level of the induction of this form of synaptic depression (a postsynaptic effect). However, it is also possible that inhibition is occurring at the level of expression of synaptic depression (a presynaptic effect). While it is not possible from the current experiments to distinguish between effects of induction vs. expression of depression, the fact that synapses that underwent HFS were still sensitive to exogenous application of 2-AG would seem to suggest that NMDAR-LTP does not alter the ability of the N cells to undergo endocannabinoid-mediated depression.
These findings are compelling because they show an entirely novel mechanism by which activity sufficient to potentiate nociceptive synapses also actively suppresses processes that depress nociceptive synapses. From a functional perspective, these results suggest that injury-induced nociceptive sensitization involves not only strengthening afferent signaling pathways, but also “turning off” modulatory processes that could limit or reverse such sensitization. In the case of endocannabinoid-based modulatory processes this could include stress-mediated analgesia56,57 or anti-nociceptive modulation produced by repetitive activation on non-nociceptive afferents6,58. This interaction between endogenous pro-nociceptive and anti-nociceptive forms of modulation could also play a role in chronic pain. That is, suppression of endogenous anti-nociceptive forms of modulation may play a critical role in what makes pain pathologically persistent, in parallel with the well-recognized, pro-nociceptive forms of modulation. Under normal conditions, these endogenous, anti-nociceptive forms of modulation may provide a homeostatic role that ultimately ameliorates or even actively reverses the physiological processes that mediate nociceptive sensitization. However, under chronic pain conditions, these corrective factors may be disrupted, potentially allowing pro-nociceptive processes to persist and even expand. Consequently, there is a critical need to understand the cellular mechanisms that mediate this interaction between pro- and anti-nociceptive modulatory processes given that this may lead to novel and effective approaches in treating chronic pain.
References
Woolf, C. J. & Ma, Q. Nociceptors–noxious stimulus detectors. Neuron 55, 353–364, https://doi.org/10.1016/j.neuron.2007.07.016 (2007).
Tobin, D. M. & Bargmann, C. I. Invertebrate nociception: behaviors, neurons and molecules. Journal of neurobiology 61, 161–174, https://doi.org/10.1002/neu.20082 (2004).
Perl, E. R. Ideas about pain, a historical view. Nat Rev Neurosci 8, 71–80, https://doi.org/10.1038/nrn2042 (2007).
Burrell, B. D. Comparative biology of pain: What invertebrates can tell us about how nociception works. J Neurophysiol 117, 1461–1473, https://doi.org/10.1152/jn.00600.2016 (2017).
Skljarevski, V. & Ramadan, N. M. The nociceptive flexion reflex in humans–review article. Pain 96, 3–8 (2002).
Yuan, S. & Burrell, B. D. Nonnociceptive afferent activity depresses nocifensive behavior and nociceptive synapses via an endocannabinoid-dependent mechanism. Journal of Neurophysiology 110, 2607–2616, https://doi.org/10.1152/jn.00170.2013 (2013).
Hwang, R. Y. et al. Nociceptive neurons protect Drosophila larvae from parasitoid wasps. Current biology: CB 17, 2105–2116, https://doi.org/10.1016/j.cub.2007.11.029 (2007).
Heinricher, M. M., Tavares, I., Leith, J. L. & Lumb, B. M. Descending control of nociception: Specificity, recruitment and plasticity. Brain Res Rev 60, 214–225, https://doi.org/10.1016/j.brainresrev.2008.12.009 (2009).
Walters, E. T. & Moroz, L. L. Molluscan memory of injury: evolutionary insights into chronic pain and neurological disorders. Brain Behav Evol 74, 206–218, https://doi.org/10.1159/000258667 (2009).
Sandkuhler, J. Models and mechanisms of hyperalgesia and allodynia. Physiol Rev 89, 707–758, https://doi.org/10.1152/physrev.00025.2008 (2009).
Liu, X. & Sandkuhler, J. Characterization of long-term potentiation of C-fiber-evoked potentials in spinal dorsal horn of adult rat: essential role of NK1 and NK2 receptors. J Neurophysiol 78, 1973–1982 (1997).
Malenka, R. C. & Bear, M. F. LTP and LTD: An embarrassment of riches. Neuron 44, 5–21 (2004).
Murphy, G. G. & Glanzman, D. L. Mediation of classical conditioning in Aplysia californica by long-term potentiation of sensorimotor synapses. Science (New York, N.Y.) 278, 467–471 (1997).
Burrell, B. D. & Sahley, C. L. Multiple forms of long-term potentiation and long-term depression converge on a single interneuron in the leech CNS. The Journal of neuroscience: the official journal of the Society for Neuroscience 24, 4011–4019, https://doi.org/10.1523/JNEUROSCI.0178-04.2004 (2004).
Ikeda, H. et al. Synaptic amplifier of inflammatory pain in the spinal dorsal horn. Science (New York, N.Y.) 312, 1659–1662, https://doi.org/10.1126/science.1127233 (2006).
Kronschläger, M. et al. Gliogenic LTP spreads widely in nociceptive pathways. Science (New York, N.Y.) 354, 1144–1148 (2016).
Arcourt, A. & Lechner, S. G. Peripheral and spinal circuits involved in mechanical allodynia. Pain 156, 220–221, https://doi.org/10.1097/01.j.pain.0000460818.62406.38 (2015).
Abraira, V. E. et al. The Cellular and Synaptic Architecture of the Mechanosensory Dorsal Horn. Cell 168, 295–310.e219, https://doi.org/10.1016/j.cell.2016.12.010 (2017).
Latremoliere, A. & Woolf, C. J. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. The journal of pain: official journal of the American Pain Society 10, 895–926, https://doi.org/10.1016/j.jpain.2009.06.012 (2009).
Burrell, B. D. Leech Mechanosensation. Oxford Research Encyclopedia of Neuroscience (Interactive Factory), https://doi.org/10.1093/acrefore/9780190264086.013.179 (2017).
Nicholls, J. G. & Baylor, D. A. Specific modalities and receptive fields of sensory neurons in CNS of the leech. Journal of Neurophysiology 31, 740–756 (1968).
Pastor, J., Soria, B. & Belmonte, C. Properties of the nociceptive neurons of the leech segmental ganglion. Journal of Neurophysiology 75, 2268–2279 (1996).
Blackshaw, S. E., Nicholls, J. G. & Parnas, I. Physiological responses, receptive fields and terminal arborizations of nociceptive cells in the leech. J Physiol 326, 251–260 (1982).
Nicholls, J. G. & Purves, D. Monosynaptic chemical and electrical connexions between sensory and motor cells in the central nervous system of the leech. J. Physiol 209, 647–667 (1970).
Brodfuehrer, P. D. & Friesen, W. O. From stimulation to undulation: a neuronal pathway for the control of swimming in the leech. Science (New York, N.Y.) 234, 1002–1004, https://doi.org/10.1126/science.234.4779.1002 (1986).
Velazquez-Ulloa, N. et al. Convergence of mechanosensory inputs onto neuromodulatory serotonergic neurons in the leech. J Neurobiol. 54, 604–617 (2003).
Gaudry, Q., Ruiz, N., Huang, T., Kristan, W. B. III. & Kristan, W. B. Jr. Behavioral choice across leech species: chacun a son gout. The Journal of experimental biology 213, 1356–1365, https://doi.org/10.1242/jeb.039495 (2010).
Sahley, C. L. Serotonin depletion impairs but does not eliminate classical conditioning in the leech Hirudo medicinalis. Behav.Neurosci. 108, 1043–1052 (1994).
Burrell, B. D. & Sahley, C. L. Serotonin mediates learning-induced potentiation of excitability. J Neurophysiol 94, 4002–4010, https://doi.org/10.1152/jn.00432.2005 (2005).
Grey, K. B. & Burrell, B. D. Co-induction of LTP and LTD and its regulation by protein kinases and phosphatases. J Neurophysiol 103, 2737–2746, https://doi.org/10.1152/jn.01112.2009 (2010).
Li, Q. & Burrell, B. D. Associative, bidirectional changes in neural signaling utilizing NMDA receptor- and endocannabinoid-dependent mechanisms. Learning & memory (Cold Spring Harbor, N.Y.) 18, 545–553, https://doi.org/10.1101/lm.2252511 (2011).
Shaw, B. K. & Kristan, W. B. Jr. The whole-body shortening reflex of the medicinal leech: motor pattern, sensory basis, and interneuronal pathways. J Comp Physiol [A] 177, 667–681 (1995).
Stuart, A. E. Physiological and morphological properties of motoneurones in the central nervous system of the leech. J.Physiol 209, 627–646 (1970).
Gaudry, Q. & Kristan, W. B. Jr. Behavioral choice by presynaptic inhibition of tactile sensory terminals. Nat Neurosci 12, 1450–1457, https://doi.org/10.1038/nn.2400 (2009).
Higgins, A., Yuan, S., Wang, Y. & Burrell, B. Differential modulation of nociceptive versus non-nociceptive synapses by endocannabinoids. Molecular Pain 9, 26 (2013).
Yuan, S. & Burrell, B. D. Endocannabinoid-Dependent Long-Term Depression in a Nociceptive Synapse Requires Coordinated Presynaptic and Postsynaptic Transcription and Translation. Journal of Neuroscience 33, 4349–4358, https://doi.org/10.1523/Jneurosci.3922-12.2013 (2013).
Wang, Y. & Burrell, B. D. Differences in chloride gradients allow for three distinct types of synaptic modulation by endocannabinoids. J Neurophysiol 116, 619–628, https://doi.org/10.1152/jn.00235.2016 (2016).
Muller, K. J. & McMahan, U. J. The shapes of sensory and motor neurones and the distribution of their synapses in ganglia of the leech: a study using intracellular injection of horseradish peroxidase. Proceedings of the Royal Society of London. Series B, Biological sciences 194, 481–499 (1976).
Lin, X. Y. & Glanzman, D. L. Hebbian induction of long-term potentiation of Aplysia sensorimotor synapses: partial requirement for activation of an NMDA-related receptor. Proc Biol Sci. 255, 215–221 (1994).
Yuan, S. & Burrell, B. D. Endocannabinoid-dependent LTD in a nociceptive synapse requires activation of a presynaptic TRPV-like receptor. J Neurophysiol 104, 2766–2777, https://doi.org/10.1152/jn.00491.2010 (2010).
Zygmunt, P. M. et al. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature 400, 452–457, https://doi.org/10.1038/22761 (1999).
Zygmunt, P. M. et al. Monoacylglycerols activate TRPV1–a link between phospholipase C and TRPV1. PLoS One 8, e81618, https://doi.org/10.1371/journal.pone.0081618 (2013).
Gunthorpe, M. J. et al. Identification and characterisation of SB-366791, a potent and selective vanilloid receptor (VR1/TRPV1) antagonist. Neuropharmacology 46, 133–149 (2004).
Yuan, S. & Burrell, B. D. Long-term depression of nociceptive synapses by non-nociceptive afferent activity: role of endocannabinoids, Ca(2)+, and calcineurin. Brain Res 1460, 1–11, https://doi.org/10.1016/j.brainres.2012.04.030 (2012).
Price, T. J. & Inyang, K. E. Commonalities between pain and memory mechanisms and their meaning for understanding chronic pain. Prog Mol Biol Transl Sci 131, 409–434, https://doi.org/10.1016/bs.pmbts.2014.11.010 (2015).
Kim, S. J. & Linden, D. J. Ubiquitous plasticity and memory storage. Neuron 56, 582–592, https://doi.org/10.1016/j.neuron.2007.10.030 (2007).
Sandkuhler, J., Chen, J. G., Cheng, G. & Randic, M. Low-frequency stimulation of afferent Adelta-fibers induces long-term depression at primary afferent synapses with substantia gelatinosa neurons in the rat. The Journal of neuroscience: the official journal of the Society for Neuroscience 17, 6483–6491 (1997).
Sandkuhler, J. Central sensitization versus synaptic long-term potentiation (LTP): a critical comment. The journal of pain: official journal of the American Pain. Society 11, 798–800, https://doi.org/10.1016/j.jpain.2010.05.002 (2010).
Latremoliere, A. & Woolf, C. J. Synaptic plasticity and central sensitization: author reply. The journal of pain: official journal of the American Pain. Society 11, 801–803, https://doi.org/10.1016/j.jpain.2010.06.006 (2010).
Lin, X. Y. & Glanzman, D. L. Long-term potentiation of Aplysia sensorimotor synapses in cell culture: regulation by postsynaptic voltage. Proc Biol Sci. 255, 113–118 (1994).
Murphy, G. G. & Glanzman, D. L. Cellular analog of differential classical conditioning in Aplysia: disruption by the NMDA receptor antagonist DL-2-amino-5-phosphonovalerate. The Journal of neuroscience: the official journal of the Society for Neuroscience 19, 10595–10602 (1999).
Walters, E. T. Site-specific sensitization of defensive reflexes in Aplysia: a simple model of long-term hyperalgesia. The Journal of neuroscience: the official journal of the Society for Neuroscience 7, 400–407 (1987).
Wang, Y. & Burrell, B. D. Endocannabinoid-mediated potentiation of nonnociceptive synapses contributes to behavioral sensitization. Journal of Neurophysiology 119, 641–651, https://doi.org/10.1152/jn.00092.2017 (2018).
Shonesy, B. C. et al. CaMKII regulates diacylglycerol lipase-alpha and striatal endocannabinoid signaling. Nat Neurosci 16, 456–463, https://doi.org/10.1038/nn.3353 (2013).
Reisenberg, M., Singh, P. K., Williams, G. & Doherty, P. The diacylglycerol lipases: structure, regulation and roles in and beyond endocannabinoid signalling. Philos Trans R Soc Lond B Biol Sci 367, 3264–3275, https://doi.org/10.1098/rstb.2011.0387 (2012).
Hohmann, A. G. et al. An endocannabinoid mechanism for stress-induced analgesia. Nature 435, 1108–1112, https://doi.org/10.1038/nature03658 (2005).
Nyilas, R. et al. Molecular architecture of endocannabinoid signaling at nociceptive synapses mediating analgesia. The European journal of neuroscience 29, 1964–1978, https://doi.org/10.1111/j.1460-9568.2009.06751.x (2009).
Yang, F. et al. Activation of cannabinoid CB1 receptor contributes to suppression of spinal nociceptive transmission and inhibition of mechanical hypersensitivity by Abeta-fiber stimulation. Pain 157, 2582–2593, https://doi.org/10.1097/j.pain.0000000000000680 (2016).
Acknowledgements
The authors thank the anonymous reviewers who provided constructive input on the initially submitted manuscript. Supported by grants from the National Science Foundation (IOS-1051734; B. Burrell), the National Institute of Neurological Disorders And Stroke (R01 NS092716, B. Burrell; F31 NS074473, S. Yuan) and the Medical Student Research Program at the University of South Dakota Sanford School of Medicine (S. Yuan).
Author information
Authors and Affiliations
Contributions
B.D.B. and S.Y. designed the experiments. S.Y. carried out the experiments and data analysis. B.D.B. and S.Y. wrote the manuscript and prepared the figures.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yuan, S., Burrell, B.D. Interaction between NMDA Receptor- and Endocannabinoid-Mediated Modulation of Nociceptive Synapses. Sci Rep 9, 1373 (2019). https://doi.org/10.1038/s41598-018-37890-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-37890-z
- Springer Nature Limited