Abstract
Congenital hypogonadotropic hypogonadism (CHH) is characterized by lack of normal pubertal development due to deficient gonadotropin-releasing hormone (GnRH) secretion or action, and is caused by genetic defects in several genes. Mutations in the CHD7 gene cause CHARGE syndrome (Coloboma of the eye, Heart defects, Atresia of the choanae, Retardation of growth and development, Genital hypoplasia and Ear abnormalities), but have also been found in patients with isolated CHH. The aim of this study was to identify CHD7 mutations in patients with CHH. Fifty Portuguese patients with CHH were screened for mutations in the CHD7 gene by DNA sequencing. Eight (16%) patients had CHD7 rare sequence variants that consisted of six missense (p.Gly388Glu, p.His903Pro, p.Thr1082Ile, p.Val1452Leu, p.Asp1854Gly, and p.Arg2065His) and two synonymous (p.Ser559Ser, and p.Ala2785Ala) mutations. Five of these mutations have never been reported before. Three CHD7 mutations occurred in patients that had mutations in additional CHH-genes. This study uncovered novel genetic variants that expand the known spectrum of mutations associated with CHH. The frequency of CHD7 mutations in this cohort was higher than that of other major CHH-genes and confirms the importance of including CHD7 in the genetic testing of CHH, even in the absence of additional CHARGE features.
Similar content being viewed by others
Introduction
Congenital hypogonadotropic hypogonadism (CHH) is characterized by partial or complete lack of pubertal development, secondary to deficient gonadotropin-releasing hormone (GnRH) induced gonadotropin secretion1. The diagnosis is based on the existence of low levels of sex hormones associated with low or inappropriately normal levels of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). CHH may occur associated with anosmia, a condition referred to as Kallmann syndrome (KS), or may occur without associated olfactory abnormalities, referred to as normosmic CHH (nCHH)1.
Monogenic or oligogenic defects are found in about 50% of patients with CHH, in genes that regulate the embryonic development or migration of GnRH neurons, or the synthesis, secretion or action of GnRH2. Mutations in the ANOS1 (KAL1) (MIM 300836), FGFR1 (KAL2) (MIM 136350) and GNRHR (MIM 138850) genes have been the most frequently reported, but there are now over 30 genes that have been associated with CHH. However, most of these genes are rarely involved in CHH and even the most commonly implicated genes usually account for less than 10% of cases2.
CHARGE syndrome is a rare disorder characterized by a variable combination of congenital anomalies that include Coloboma of the eye, Heart defects, Atresia of the choanae, Retardation of growth and development, Genital hypoplasia and Ear abnormalities3. Heterozygous loss-of-function mutations in the chromodomain helicase DNA-binding protein 7 (CHD7) gene (MIM 608892) are the major cause of CHARGE syndrome4. Hypogonadotropic hypogonadism and olfactory defects, which are the hallmarks of KS, are commonly observed in CHARGE syndrome5 and CHD7 mutations have been identified in patients with isolated CHH (i.e. without additional CHARGE features)6,7,8,9,10,11.
The aim of this study was to determine the frequency of CHD7 mutations in a cohort of patients with isolated CHH.
Material and Methods
Subjects
The study comprised 50 unrelated Portuguese patients with CHH (42 men and 8 women, mean age at diagnosis 21.4 years, range 14–45 years), 22 with KS and 28 with nCHH, recruited by Portuguese clinical endocrine centers. Inclusion criteria were patients with low or inappropriately normal serum FSH, LH and sex steroid levels, failure to enter spontaneous puberty by the age of 18 years or with medically induced puberty below this age, and absence of other pituitary hormone deficiencies. Olfactory function was assessed either by olfaction testing or self-reported by the patients, depending on the clinical center. None of the patients had diagnostic criteria for CHARGE syndrome12. In mutation-positive patients, additional family members were studied, when available. The control population consisted of >200 Portuguese unrelated healthy volunteers who were recruited among blood donors. The study was approved by the local research ethics committee (Faculty of Health Sciences, University of Beira Interior, Ref: CE-FCS-2012-012). Written informed consent was obtained from all subjects and all methods were performed in accordance with the relevant guidelines and regulations.
Genetic studies
Genomic deoxyribonucleic acid (DNA) was extracted from peripheral blood leucocytes using previously described methods13. Patients had already been screened for mutations in the ANOS1, FGFR1 and GNRHR genes, resulting in the discovery of three mutations in ANOS114, six mutations in FGFR115 and six mutations in GNRHR16. All 50 patients were subsequently screened for mutations in the CHD7 gene by polymerase chain reaction (PCR) amplification of the coding exons and exon-intron boundaries, and bi-directional sequencing using CEQ DTCS sequencing kit (Beckman Coulter, Fullerton, CA, USA) and an automated capillary DNA sequencer (GenomeLab TM GeXP, Genetic Analysis System, Beckman Coulter). Primer sequences for CHD7 were previously described by Song et al.17, except for the primer sequence of exons 2.4 and 3, which were designed using Primer 3 Plus18. Genomic sequence variants identified in patients were searched in the Exome Aggregation Consortium (ExAC) population variant database19, to assess their frequency in the general population. Variants found to be absent in the ExAC database or with frequencies <0.1% were further screened in a panel of at least 200 healthy Portuguese volunteers (400 alleles), using allele-specific PCR or sequence-specific restriction enzymes, to exclude the possibility that they represented common polymorphisms in the Portuguese population. Variants were considered to be pathogenic when they were simultaneously found to have an ExAC population frequency <0.1%, to be absent in the Portuguese control population, and to have a deleterious effect predicted by at least one of four bioinformatic programs (SIFT20, PolyPhen-221, Mutation Taster22 or Human Splicing Finder23). Sequence variant nomenclature followed standard guidelines24 and was based on the cDNA reference sequence for the CHD7 gene (GenBank accession NM_017780.3). Patients with pathogenic variants in CHD7 were further screened for mutations in additional genes related to the hypothalamic–pituitary–gonadal axis (PROKR2, PROK2, FGF8, GNRH1, KISS1R, TAC3 and TACR3), to identify possible cases of oligogenicity (primer sequences and PCR conditions are available upon request).
Results
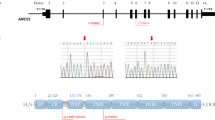
Sequence analysis of the entire coding region and exon–intron boundaries of CHD7 revealed 13 heterozygous variants that had frequencies <0.1% in the ExAC population database. Five of these variants were detected in normal Portuguese controls (p.Thr689Thr, 2 of 408 alleles; p.Lys729Glu, 1 of 436 alleles; c.2613 + 4 C > T, 1 of 454 alleles; p.Gly1479Gly, 1 of 398 alleles; p.Pro2072Pro, 1 of 450 alleles) and were excluded from further analysis. The remaining eight variants were found to be absent in the Portuguese control population and were predicted to cause changes in protein function, by at least one bioinformatic prediction program (Table 1). Thus, 16% (8 out of 50) of patients with CHH were considered to have pathogenic variants in CHD7. Mutations occurred in both KS (4/22, 18%) and nCHH (4/28, 14%). Mutations consisted of six missense (c.1163 G > A, p.Gly388Glu; c.2708 A > C, p.His903Pro; c.3245 C > T, p.Thr1082Ile; c.4354 G > T, p.Val1452Leu; c.5561 A > G, p.Asp1854Gly; c.6194 G > A, p.Arg2065His) and two synonymous (c.1677G > A, p.Ser559Ser; c.8355 C > T, p.Ala2785Ala) variants (Fig. 1). Conservation analysis revealed that, with the exception of p.Gly388Glu, all missense variants occurred at amino acids that were highly conserved across species (Supplemental Table 1).
CHD7 mutations identified in patients with CHH. Arrows represent index cases, filled symbols represent affected individuals, open symbols represent unaffected individuals, squares denote men, circles denote women, oblique lines through symbols represent deceased individuals. Filled squares within squares represent individuals reported to have heart defects associated with polydactyly. Patients from families 3, 5 and 8 had mutations in additional genes, thus representing cases of oligogenicity. Genotypes for additional family members, when available, are presented beside each individual (+, wild-type allele; −, mutated allele; n/a, not available for genetic studies). The position of each heterozygous mutation on the DNA sequence is indicated by an asterisk.
The p.Ser559Ser, p.Arg2065His and p.Ala2785Ala variants, have been previously identified in patients with CHARGE syndrome25. The remaining five variants have not yet been reported in patients with either CHH or CHARGE syndrome.
In six of the eight families, it was possible to determine the parental origin of the CHD7 mutation and in all six cases the mutation was transmitted by a parent who did not have CHARGE symptoms or a history of delayed puberty (Fig. 1). In three of these cases, patients presented mutations in additional CHH genes, namely one digenic mutation (CHD7/FGFR1) and two trigenic mutations (CHD7/FGFR1/PROKR2), thus representing cases of oligogenic inheritance (Fig. 1).
All CHD7 variants were submitted to the CHD7 mutation database at www.chd7.org.
The clinical characteristics of patients with identified CHD7 pathogenic variants are summarized in Table 2.
Discussion
The overall prevalence of CHD7 mutations in this cohort of Portuguese patients with CHH was 16% (8 out of 50), with a similar distribution between the KS and nCHH forms. This is a high prevalence when compared to the contribution of other genes that have been historically considered as priorities in the genetic study of CHH, namely the ANOS1 (KAL1), FGFR1, and GNRHR genes26. Indeed, previous studies in the Portuguese population have shown that the ANOS1 (KAL1), FGFR1, and GNRHR genes are mutated in only 7.1%, 12.0%, and 12.5% of cases, respectively14,15,16. Studies in other populations have also shown an important contribution of CHD7 mutations in the aetiology of CHH, with frequencies ranging from 5.2% to 19.0%, of cases6,7,8,9,10,11. Thus, the CHD7 gene is becoming increasingly recognised as one of the most commonly mutated genes in CHH.
Although CHD7 mutations can cause both CHARGE syndrome and isolated CHH, it is likely that this variable phenotypic expression is related to the severity of the CHD7 mutations, as mutations in CHARGE syndrome are typically highly deleterious protein-truncating mutations, whereas CHD7 mutations in isolated CHH are typically missense8. Accordingly, in our CHH patients, all identified CHD7 mutations were either missense (n = 6) or synonymous (n = 2).
The eight CHD7 mutations identified in this study were found to be absent or very rare (<0.1%) in the ExAC population database19, absent in Portuguese controls, and predicted to be damaging by structure- and sequence-based bioinformatics programs20,21,22,23. Three of these mutations (one missense and two synonymous) have already been reported in patients with CHARGE syndrome25, and the remaining five missense mutations have not yet been reported in patients with either CHH or CHARGE syndrome. The missense mutations occurred at highly conserved amino acids. Furthermore, the p.His903Pro, p.Thr1082Ile and p.Val1452Leu mutations are located in chromodomain 2, the SNF2 domain and the helicase domain, respectively, which play important roles in the chromatin remodelling activity of the CHD7 protein6. The remaining missense mutations occurred outside these catalytic domains but were also predicted to affect protein function. The mechanisms by which the synonymous mutations exert their effect were not possible to determine. However, synonymous mutations - sometimes called ‘silent’ mutations - are widely acknowledged to be able to cause changes in protein expression, conformation and function, and have been implicated in several diseases27. It should be noted however that according to more stringent classification criteria recommended by the American College of Medical Genetics and Genomics (ACMG)28, only the p.Thr1082Ile variant is considered “likely pathogenic” and the remaining are considered “variants of uncertain significance”.
Three patients with CHD7 mutations presented additional defects in known CHH-genes. Thus, these represent cases of oligogenic inheritance, which is a frequent genetic finding in CHH29. Oligogenicity might explain some cases of incomplete penetrance, where carriers of a CHD7 mutation only express CHH in the presence of other mutated genes.
Our patients with CHD7 mutations did not undergo detailed radiological investigations to detect hypoplasia/agenesis of semicircular canals or of olfactory bulbs and tracts, which are currently major criteria for CHARGE syndrome30,31. Thus, we cannot exclude that patients have light forms of CHARGE syndrome. Other studies have shown that a subset of patients with apparently isolated CHH, in whom a CHD7 defect was demonstrated, were subsequently found to exhibit multiple CHARGE features and reclassified as having CHARGE syndrome32,33,34. Interestingly, three of our patients with CHD7 mutations had minor CHARGE features30,31, namely hearing deficit, renal anomalies and intellectual disability, respectively, but insufficient for a clinical diagnosis of CHARGE syndrome. It remains to be determined if these were coincidental findings or the result of the CHD7 mutations.
Our results should be viewed with caution as in vitro and in vivo functional tests are not readily available for CHD735, and therefore it was not possible to confirm the damaging effect of the observed CHD7 variants. In addition, a limited number of CHH-genes was analysed by Sanger sequencing and it remains to be determined if a more comprehensive genetic analysis (e.g. through whole exome sequencing) would uncover additional cases of oligogenicity and explain the incomplete penetrance observed in some families. Finally, CHH is clinically heterogeneous and sometimes overlaps with constitutional delay of growth and puberty (CDGP)36. Our study included patients with a wide range of ages and we cannot exclude the possibility that some of the younger patients, who underwent medically induced puberty, may represent cases of CDGP or mild forms of CHH that would have eventually developed spontaneous late puberty. Therefore, our results may not be directly comparable with those of other studies that used different inclusion criteria.
In conclusion, our study identified a high frequency of CHD7 mutations in patients with CHH and uncovered novel genetic variants that expand the known spectrum of mutations associated with CHH.
References
Boehm, U. et al. Expert consensus document: European Consensus Statement on congenital hypogonadotropic hypogonadism–pathogenesis, diagnosis and treatment. Nat Rev Endocrinol 11, 547–564, https://doi.org/10.1038/nrendo.2015.112 (2015).
Kim, S. H. Congenital Hypogonadotropic Hypogonadism and Kallmann Syndrome: Past, Present, and Future. Endocrinology and metabolism 30, 456–466, https://doi.org/10.3803/EnM.2015.30.4.456 (2015).
Pagon, R. A., Graham, J. M. Jr., Zonana, J. & Yong, S. L. Coloboma, congenital heart disease, and choanal atresia with multiple anomalies: CHARGE association. The Journal of pediatrics 99, 223–227 (1981).
Vissers, L. E. et al. Mutations in a new member of the chromodomain gene family cause CHARGE syndrome. Nat Genet 36, 955–957, https://doi.org/10.1038/ng1407 (2004).
Balasubramanian, R. & Crowley, W. F. Jr. Reproductive endocrine phenotypes relating to CHD7 mutations in humans. Am J Med Genet C Semin Med Genet 175, 507–515, https://doi.org/10.1002/ajmg.c.31585 (2017).
Kim, H. G. et al. Mutations in CHD7, encoding a chromatin-remodeling protein, cause idiopathic hypogonadotropic hypogonadism and Kallmann syndrome. Am J Hum Genet 83, 511–519, https://doi.org/10.1016/j.ajhg.2008.09.005 (2008).
Balasubramanian, R. et al. Functionally compromised CHD7 alleles in patients with isolated GnRH deficiency. Proc Natl Acad Sci USA 111, 17953–17958, https://doi.org/10.1073/pnas.1417438111 (2014).
Marcos, S. et al. The prevalence of CHD7 missense versus truncating mutations is higher in patients with Kallmann syndrome than in typical CHARGE patients. J Clin Endocrinol Metab 99, E2138–2143, https://doi.org/10.1210/jc.2014-2110 (2014).
Wang, Y., Gong, C., Qin, M., Liu, Y. & Tian, Y. Clinical and genetic features of 64 young male paediatric patients with congenital hypogonadotropic hypogonadism. Clin Endocrinol (Oxf) 87, 757–766, https://doi.org/10.1111/cen.13451 (2017).
Ayers, K. L. et al. Variants in congenital hypogonadotrophic hypogonadism genes identified in an Indonesian cohort of 46,XY under-virilised boys. Hum Genomics 11, 1, https://doi.org/10.1186/s40246-017-0098-2 (2017).
Aoyama, K. et al. Molecular genetic and clinical delineation of 22 patients with congenital hypogonadotropic hypogonadism. J Pediatr Endocrinol Metab 30, 1111–1118, https://doi.org/10.1515/jpem-2017-0035 (2017).
Hefner, M. A. & Fassi, E. Genetic counseling in CHARGE syndrome: Diagnostic evaluation through follow up. Am J Med Genet C Semin Med Genet 175, 407–416, https://doi.org/10.1002/ajmg.c.31589 (2017).
Lemos, M. C. & Regateiro, F. J. N-acetyltransferase genotypes in the Portuguese population. Pharmacogenetics 8, 561–564 (1998).
Goncalves, C. I., Fonseca, F., Borges, T., Cunha, F. & Lemos, M. C. Expanding the genetic spectrum of ANOS1 mutations in patients with congenital hypogonadotropic hypogonadism. Hum Reprod 32, 704–711, https://doi.org/10.1093/humrep/dew354 (2017).
Goncalves, C. et al. Novel FGFR1 mutations in Kallmann syndrome and normosmic idiopathic hypogonadotropic hypogonadism: evidence for the involvement of an alternatively spliced isoform. Fertil Steril 104, 1261–1267 e1261, https://doi.org/10.1016/j.fertnstert.2015.07.1142 (2015).
Goncalves, C. I. et al. GNRHR biallelic and digenic mutations in patients with normosmic congenital hypogonadotropic hypogonadism. Endocr Connect 6, 360–366, https://doi.org/10.1530/EC-17-0104 (2017).
Song, M. H. et al. CHD7 mutational analysis and clinical considerations for auditory rehabilitation in deaf patients with CHARGE syndrome. PLoS One 6, e24511, https://doi.org/10.1371/journal.pone.0024511 (2011).
Untergasser, A. et al. Primer3Plus, an enhanced web interface to Primer3. Nucleic Acids Res 35, W71–74, https://doi.org/10.1093/nar/gkm306 (2007).
ExAC. Exome Aggregation Consortium. http://exac.broadinstitute.org (2018).
Ng, P. C. & Henikoff, S. SIFT: Predicting amino acid changes that affect protein function. Nucleic Acids Res 31, 3812–3814 (2003).
Adzhubei, I. A. et al. A method and server for predicting damaging missense mutations. Nat Methods 7, 248–249, https://doi.org/10.1038/nmeth0410-248 (2010).
Schwarz, J. M., Rodelsperger, C., Schuelke, M. & Seelow, D. MutationTaster evaluates disease-causing potential of sequence alterations. Nat Methods 7, 575–576, https://doi.org/10.1038/nmeth0810-575 (2010).
Desmet, F. O. et al. Human Splicing Finder: an online bioinformatics tool to predict splicing signals. Nucleic Acids Res 37, e67, https://doi.org/10.1093/nar/gkp215 (2009).
den Dunnen, J. T. & Antonarakis, S. E. Nomenclature for the description of human sequence variations. Hum Genet 109, 121–124 (2001).
Bartels, C. F., Scacheri, C., White, L., Scacheri, P. C. & Bale, S. Mutations in the CHD7 gene: the experience of a commercial laboratory. Genet Test Mol Biomarkers 14, 881–891, https://doi.org/10.1089/gtmb.2010.0101 (2010).
Semple, R. K. & Topaloglu, A. K. The recent genetics of hypogonadotrophic hypogonadism - novel insights and new questions. Clin Endocrinol (Oxf) 72, 427–435, https://doi.org/10.1111/j.1365-2265.2009.03687.x (2010).
Sauna, Z. E. & Kimchi-Sarfaty, C. Understanding the contribution of synonymous mutations to human disease. Nat Rev Genet 12, 683–691, https://doi.org/10.1038/nrg3051 (2011).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17, 405–424, https://doi.org/10.1038/gim.2015.30 (2015).
Sykiotis, G. P. et al. Oligogenic basis of isolated gonadotropin-releasing hormone deficiency. Proc Natl Acad Sci USA 107, 15140–15144, https://doi.org/10.1073/pnas.1009622107 (2010).
Hale, C. L., Niederriter, A. N., Green, G. E. & Martin, D. M. Atypical phenotypes associated with pathogenic CHD7 variants and a proposal for broadening CHARGE syndrome clinical diagnostic criteria. American journal of medical genetics. Part A 170A, 344–354, https://doi.org/10.1002/ajmg.a.37435 (2016).
Legendre, M. et al. Phenotype and genotype analysis of a French cohort of 119 patients with CHARGE syndrome. Am J Med Genet C Semin Med Genet 175, 417–430, https://doi.org/10.1002/ajmg.c.31591 (2017).
Xu, C. et al. Evaluating CHARGE syndrome in congenital hypogonadotropic hypogonadism patients harboring CHD7 variants. Genet Med 20, 872–881, https://doi.org/10.1038/gim.2017.197 (2018).
Jongmans, M. C. et al. CHD7 mutations in patients initially diagnosed with Kallmann syndrome–the clinical overlap with CHARGE syndrome. Clin Genet 75, 65–71, https://doi.org/10.1111/j.1399-0004.2008.01107.x (2009).
Bergman, J. E. et al. The results of CHD7 analysis in clinically well-characterized patients with Kallmann syndrome. J Clin Endocrinol Metab 97, E858–862, https://doi.org/10.1210/jc.2011-2652 (2012).
van Ravenswaaij-Arts, C. & Martin, D. M. New insights and advances in CHARGE syndrome: Diagnosis, etiologies, treatments, and research discoveries. Am J Med Genet C Semin Med Genet 175, 397–406, https://doi.org/10.1002/ajmg.c.31592 (2017).
Cassatella, D. et al. Congenital hypogonadotropic hypogonadism and constitutional delay of growth and puberty have distinct genetic architectures. Eur J Endocrinol 178, 377–388, https://doi.org/10.1530/EJE-17-0568 (2018).
Acknowledgements
This work was supported by the Portuguese Foundation for Science and Technology (PTDC/SAU-GMG/098419/2008) and by “Programa Operacional do Centro, Centro 2020” through the funding of the ICON project (Interdisciplinary Challenges On Neurodegeneration; CENTRO-01-0145-FEDER-000013)”. The authors are grateful to the following clinicians who contributed with patient samples and data: Ana Saavedra (Porto), Ana Varela (Porto), António Garrão (Lisboa), Carla Baptista (Coimbra), Carla Meireles (Guimarães), Carolina Moreno (Coimbra), Catarina Limbert (Lisboa) Cíntia Correia (Porto), Cláudia Nogueira (Porto), Duarte Pignatelli (Porto), Eduardo Vinha (Porto), Filipe Cunha (Porto), Francisco Carrilho (Coimbra), Luísa Barros (Coimbra), Luísa Cortez (Lisboa), Margarida Bastos (Coimbra), Maria João Oliveira (Porto), Mariana Martinho (Penafiel), Miguel Melo (Coimbra), Nuno Vicente (Coimbra), Patrícia Oliveira (Coimbra), Paula Freitas (Porto), Raquel Martins (Porto), Selma Souto (Porto), Susana Gama (Famalicão), and Teresa Martins (Coimbra).
Author information
Authors and Affiliations
Contributions
C.I.G., J.M.O. and M.C.L. conceived and designed the study. C.I.G. and F.M.P. performed the genetic studies of the patients. J.M.A., D.C., F.F., S.M., O.M. and B.D.P. collected samples and acquired clinical data of the patients with mutations. C.I.G. and M.C.L. drafted the article and all authors revised it critically for important intellectual content and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gonçalves, C.I., Patriarca, F.M., Aragüés, J.M. et al. High frequency of CHD7 mutations in congenital hypogonadotropic hypogonadism. Sci Rep 9, 1597 (2019). https://doi.org/10.1038/s41598-018-38178-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-38178-y
- Springer Nature Limited