Abstract
We aimed to examine the effect of weight change attributable to cessation of cigarette smoking on newly diagnosed metabolic syndrome (MetS). We prospectively followed 5,809 men aged between 40 to 69 years without MetS at baseline in the Health Examinees-Gem (HEXA-G) study up to 4 years. The participants were grouped into continual smokers, quitters with weight gain, quitters without weight change, quitters with weight loss, and never smokers. We constructed multivariable logistic regression models adjusted for sociodemographic factors, health status, and health conditions to estimate the odds of newly diagnosed MetS. During the follow-up, there were 609 cases of newly diagnosed MetS in 5,809 men of the HEXA-G study. After adjustment for potential confounders, the odd ratios (OR) and 95% confidence intervals (95% CI) for MetS were 1.90 (95% CI: 1.43–2.52) in quitters with weight gain, 0.77 (95% CI: 0.60–1.00) in quitters without weight change, and 0.40 (95% CI: 0.28–0.57) in quitters with weight loss compared with continual smokers. Never smokers also had lower odds of MetS (OR = 0.54; 95% CI: 0.42–0.71) compared to continual smokers. Weight management program following smoking cessation may be necessary in clinical practice to reduce worsening of cardiometabolic risk factors related to post-cessation weight gain.
Similar content being viewed by others
Introduction
Although adverse health outcomes of cigarette smoking have been extensively studied1,2,3,4,5,6,7, weight gain after cessation of smoking still remains as one of the leading concerns among current smokers that may discourage them from quitting smoking8,9,10. Abstinence from cigarette smoking can lead to risk reduction of metabolic syndrome (MetS) through multiple pathways11,12, but weight gain after smoking cessation may attenuate the protective association of quitting smoking with MetS13,14. An up-to-date meta-analysis of prospective cohort studies has reported that smoking cessation was associated with 2.61 kg of weight gain as compared with continual smoking4. Since weight gain is also associated with several components of MetS15,16,17, whether weight change associated with smoking cessation counteracts the cardiometabolic benefits of quitting smoking is unclear.
Previous studies on smoking cessation and MetS have shown inconsistent results by the duration of smoking abstinence. One recent study by Song et al. reported that smoking cessation within 3 months is associated with increased risk of developing MetS. In contrast, others have found that smoking cessation is associated with decreased risk of MetS18. However, these studies were restricted to those with nicotine replacement therapy with a relatively small number of patients. Also, these studies did not take weight change attributable to smoking cessation into account. One study of 5,407 men using hospital data found that ex-smokers had elevated risk for incident MetS regardless of weight change compared to non-smokers after 3 years of follow-up19. Findings from these studies are subject to selection bias, misclassification of reference category, inconsistent study design, and follow-up duration.
We aimed to evaluate the association between smoking cessation, 4-year weight change, and newly diagnosed MetS and its components among the middle-aged men. We hypothesized that weight change associated with smoking cessation does not affect the risk reduction of MetS compared to continual smoking, and also investigated this association among quitters by the degree of weight change.
Results
Primary analysis
Among the 5,809 participants of the HEXA-G study included in our analysis, there were 1,325 (22.8%) continual smokers, 576 (9.9%) quitters with weight gain, 1,522 (26.2%), 697 (12.0%) quitters without weight change, and 1,689 (29.1%) never smokers. Of these study population, 609 participants were newly diagnosed with MetS during the follow-up period that lasted up to 4 years. Continual smokers had the highest proportion of 40–49 age group compared to quitters and never smokers. Sociodemographic, health status, and clinical characteristics of the study participants by change in smoking behavior are presented in Table 1.
In the multivariable logistic regression analyses, weight gain following smoking cessation was associated with higher odds of elevated WC (OR = 2.94; 95% CI: 2.17–3.99) and high BP (OR = 1.72; 95% CI: 1.31–2.26). No weight change after smoking cessation was associated with lower odds of high TG (OR = 0.65; 95% CI: 0.54–0.78) and reduced HDL-C (OR = 0.53; 95% CI: 0.38–0.74). Post-cessation weight loss was associated with lower odds of elevated WC (OR = 0.34; 95% CI: 0.20–0.58), high TG (OR = 0.35; 95% CI: 0.27–0.46), and reduced HDL-C (OR = 0.56; 95% CI: 0.37–0.85). As compared with continual smokers, never smokers had lower odds of high TG (OR = 0.41; 95% CI: 0.34–0.50), reduced HDL-C (OR = 0.59; 95% CI: 0.43–0.81), and impaired FSG (OR = 0.66; 95% CI: 0.50–0.87). The association between change in smoking behavior, weight change, and newly diagnosed components of MetS are summarized in Table 2.
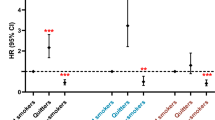
In the analysis of the association of change in smoking behavior and weight change with newly diagnosed MetS, weight gain following smoking cessation was associated with higher odds of MetS (OR = 1.90; 95% CI: 1.43–2.52). In contrast, no weight change (OR = 0.77; 95% CI: 0.60–1.00) and weight loss (OR = 0.40; 95% CI: 0.28–0.57) after smoking cessation was associated with lower odds of MetS. The main analysis of the association between change in smoking behavior, weight change, and newly diagnosed MetS is shown in Table 3. Although statistical significance was attenuated, investigation on subgroup effect, excluding those with family history, stratification by the number of components of MetS at baseline generated similar results compared to the main analysis on the association between change in smoking behavior, weight change, and newly diagnosed MetS. The results of the sensitivity analyses are summarized in Supplemental Table 1.
Secondary analysis
In secondary analyses restricting the study sample to quitters, the percentage of newly diagnosed MetS was the highest among those with weight gain (21.4%). As compared with quitters without weight change, the multivariable adjusted ORs and 95% CIs for newly diagnosed MetS among quitters with weight gain and quitters with weight loss were 2.44 (95% CI: 1.84–3.25) and 0.52 (95% CI: 0.37–0.73), respectively. The results of the secondary analysis is presented in Table 4.
Discussion
In this community-based cohort study comprised of 5,809 middle-aged Korean men without MetS at baseline, 10.5% of the participants were newly diagnosed with MetS during the follow-up period. Weight gain following smoking cessation was significantly associated with elevated WC and impaired FSG, but no weight change and weight loss after smoking cessation were associated with improved TG and HDL-C. Among quitters, those with weight gain of more than 2 kg were more likely to develop metabolic syndrome compared to those with weight gain or loss of less than 2 kg.
One prospective study by Kim et al. comprised of 4,542 Korean men based on records from a single hospital found that ex-smokers had increased risk of MetS despite weight change compared to non-smokers19. In contrast, our study showed that only those with weight gain had higher odds of newly diagnosed MetS among quitters as compared to continual smokers. Additionally, we found that quitters with weight gain had higher odds of elevated WC and impaired FSG while quitters without weight gain had improved levels of TG and HDL-C. While both studies suggest the importance of post-cessation weight management to reduce risk of MetS, our study adds to the evidence for an association of specific components of MetS with smoking cessation by the degree of weight change.
The mechanism by which smoking cessation could reduce MetS despite weight change is not fully understood, but can be explain in part by improvement of lipid profile. Negative health effects of post-cessation weight change may be mitigated by improvements of lipid profile regardless of weight change attributable to quitting smoking. In a follow-up study of 1,955 Japanese male workers, Tamura et al., showed that those who had quit smoking for at least half a year had 2.0 kg of weight gain and worsening effects of BP, FSG, and total cholesterol, but they significantly improved levels of HDL-cholesterol20. Similarly, in another study of 226 Korean men who had quit smoking, quitters with weight gain (≥1.3 kg) had significantly harmful changes of BP, FSG, and cholesterol levels whereas quitters without weight gain (<1.3 kg) had improved levels of cardiometabolic factors21. These studies might have selection bias and small sample size. However, the evidence from these studies suggest that successfully abstaining from cigarette smoking may positively affect MetS as the change of post-cessation weight wanes over time22.
The primary strength of this study is the large sample size from the community-based cohort with verified health examination data through quality control. Moreover, the prospective cohort study design in this study limits the possibility of reverse causality as compared to the studies with cross-sectional design. Assessment of a wide range of known confounding factors associated with the study outcome also strengthens our study. In addition, evaluation of the association between smoking cessation, weight change, and MetS in quitters compared to continual smokers and a secondary analysis among quitters by the degree of weight change further strengthens our investigation. However, there are some limitations of this study that need to be acknowledged. We were not able to include women in this study due to a significantly low proportion of the middle-aged women who are smokers. Since the analytic sample was limited to the middle-aged Korean men who participated in the HEXA-G study, more studies are warranted in young adults, women, and other ethnic groups to test the generalizability of this study. Furthermore, change in smoking behavior was only assessed from the self-reported survey without biochemical assessment. In addition, variables used in our analyses that were derived from the self-reported survey (e.g. smoking status and family history of disease) should be validated in further studies.
In this community-based cohort study comprised of middle-aged men, we found that weight gain after smoking cessation was associated with higher odds of elevated WC and impaired FSG as compared with continual smoking. Quitters who gained substantial amount of weight after abstinence from smoking were more likely to develop MetS compared to the quitters without weight change. Overall, our results indicate the importance of weight control after smoking cessation to reduce the burden of MetS. Accordingly, intervention programs for weight management in clinical practice may be necessary for those who attempt to quit smoking.
Methods
Study population
We examined data on the participants of the Health Examinees-Gem (HEXA-G) study conducted in the Republic of Korea. The HEXA-G is a community-based multicenter study database comprised of Korean adults aged between 40 and 69 years who visited study centers for health examinations between 2004 and 2013. The HEXA-G study excluded the dataset from the sites that only participated in the pilot study, less than 2 years of follow-up, or did not pass quality control for biospecimen. The HEXA-G study has been used for research purpose previously23,24,25,26, and the design and validity of the study has been described in detail elsewhere27,28. For this study, we recruited the male participants with complete information on smoking status without MetS who were followed up to 4 years from the baseline. Female participants were not included due to the low proportion of those who reported themselves as current smokers at baseline (less than 5%). Among the 6,138 participants without MetS at baseline, we excluded those that met the following conditions: (1) missing data on smoking status (n = 36); (2) missing information on potential confounders (n = 184); (3) extremely low or high energy intake (<800 or ≥4,000 kcal/day) (n = 185). Finally, a total of 5,809 men were included in the analytic sample.
Ethical approval and informed consent
All participants provided informed consent before enrollment to the HEXA-G study, and the dataset are publicly open only for research purpose upon request. The Institutional Review Board (IRB) of the Seoul National University Hospital, which complies with the principles of the Declaration of Helsinki, approved this study (IRB no: 1503-103-657).
Assessment of change in smoking behavior and weight change
Based on the self-reported questionnaire and weight change from the baseline and follow-up health examination records of the HEXA-G study, we categorized the study participants as continual smokers, quitters with weight gain, quitters without weight change, quitters with weight loss, and never smokers. Post-cessation weight change category (stable, gain or loss of ≤2 kg; gain, gain of >2 kg, and loss, loss of <2 kg) was based on the previously reported average weight change attributable to smoking cessation in the middle-aged Korean men21.
Assessment of metabolic syndrome
We adopted criteria from the National Cholesterol Education Program Adult Treatment Panel III (NCEP-ATP III)29 modified for the Asian population for the assessment of MetS in the HEXA-G study. For each participant, meeting three or more of the following conditions was defined as the MetS: (1) elevated waist circumference (WC) ≥ 90 cm; (2) high triglyceride (TG), ≥150 mg/dL or taking antihyperlipidemic drugs; (3) reduced high-density lipoprotein cholesterol (HDL-C), ≤40 mg/dL; (4) high blood pressure (BP), systolic BP ≥ 130 mmHg, diastolic BP ≥ 85 mmHg or taking antihypertensive medication; and (5) impaired fasting serum glucose (FSG), ≥100 mg/dL or taking antihyperglycemic agents.
Assessment of potential confounders
In the HEXA-G study, information on sociodemographic factors (age, education level, and household income) was obtained from the self-reported survey. Age was categorized as 40–49 years, 50–59 years, and ≥60 years. Education level was grouped into elementary, middle/high school, college or above based on the final academic degree. Household income was divided into quartiles. We also assessed lifestyle factors (physical activity, alcohol consumption, and energy intake) from the survey data. Alcohol consumption was classified into non-drinker, abstainer (those who reported to have gradually reduce amount of alcohol consumption over time), and regular drinker (those who reported to regularly consume at least one alcoholic beverage per week). Regular exercise was defined as those who reported to routinely exercise until sweating on regular basis. Total energy intake per day (kcal/day) was estimated from converting the types and amount of servings consumed per day into macro and micro nutrient intake (grams per day) from different food categories in the food frequency questionnaire. BMI was calculated from dividing weight in kilograms (kg) by height in meters (m) squared (kg/m2). Data on uric acid and high sensitivity C-reactive protein were derived from the laboratory assessment, and information on family history of disease (hypertension, type2 diabetes, and hyperlipidemia) was obtained from the responses of the self-reported questionnaire.
Statistical analysis
For the primary analysis, we chose continual smokers as the reference group as recommended by the U.S Surgeon General’s report in 199030 to assess the effect of smoking cessation on MetS. We used multivariable logistic regression models to compute adjusted odds ratio (OR) and 95% confidence intervals (95% CI) to assess the association between smoking cessation, weight change, and components of MetS. We calculated age-adjusted and multivariable adjusted (adjusted for age, education level, household income, physical activity, alcohol consumption, energy intake, and BMI) ORs and 95% CIs for elevated WC, high TG, reduced HDL-C, high BP, and impaired FSG. BMI was not adjusted when computing the adjusted OR (95% CI) for elevated WC due to the high collinearity between the two variables. In addition to the variables included in the multivariable model, weight change was further adjusted as a continuous variable to examine the effect of weight change attributable to smoking cessation. For the component of MetS, participants who had already developed the metabolic alternation at the baseline were removed from the analytic sample. Similarly, we used multivariable logistic regression model adjusted for age, education level, household income, number of components of metabolic syndrome at baseline, physical activity, alcohol consumption, energy intake, body mass index, uric acid, high sensitivity C-reactive protein, family history of hypertension, family history of type 2 diabetes, and family history of hyperlipidemia to assess the relationship between smoking cessation and odds of newly diagnosed MetS. To further test whether weight change from smoking cessation alters this association, we adjusted weight change as a continuous variable in the multivariable model. For sensitivity analyses, we assessed subgroup effects by stratifying the analysis into different categories of age, education level, household income, BMI, and number of MetS components at baseline. Also, we excluded those with family history of type 2 diabetes, hypertension, and hyperlipidemia to examine whether the results remained consistent after exclusion of those with family history of disease related to MetS. In secondary analysis, we examined the association of weight change following smoking cessation only among quitters. For this analysis, we chose quitters with stable weight change (gain or loss of ≤2 kg) as the reference group, and compared the odds of newly diagnosed MetS in quitters with weight gain (gain of >2 kg) and weight loss (loss of <2 kg). All p-values were two-sided, and we considered p-value < 0.05 as statistically significant. We used SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) for data collection and analyses.
Data Availability
No additional data available. Only authorized researchers had access to the HEXA-G data according to the data sharing policy of Korea Centers for Disease Control and Prevention.
References
Carter, B. D. et al. Smoking and mortality—beyond established causes. New England journal of medicine 372, 631–640 (2015).
Jain, P., Danaei, G., Robins, J. M., Manson, J. E. & Hernán, M. A. Smoking cessation and long-term weight gain in the Framingham Heart Study: an application of the parametric g-formula for a continuous outcome. European journal of epidemiology 31, 1223–1229 (2016).
Mons, U. et al. Impact of smoking and smoking cessation on cardiovascular events and mortality among older adults: meta-analysis of individual participant data from prospective cohort studies of the CHANCES consortium. bmj 350, h1551 (2015).
Pan, A., Wang, Y., Talaei, M., Hu, F. B. & Wu, T. Relation of active, passive, and quitting smoking with incident type 2 diabetes: a systematic review and meta-analysis. The lancet Diabetes & endocrinology 3, 958–967 (2015).
Sun, K., Liu, J. & Ning, G. Active smoking and risk of metabolic syndrome: a meta-analysis of prospective studies. PloS one 7, e47791 (2012).
Doll, R., Peto, R., Boreham, J. & Sutherland, I. Mortality in relation to smoking: 50 years’ observations on male British doctors. Bmj 328, 1519 (2004).
Jacobs, D. R. et al. Cigarette smoking and mortality risk: twenty-five–year follow-up of the seven countries study. Archives of Internal Medicine 159, 733–740 (1999).
Iida, M. Weight Gain After Smoking Cessation and Atherosclerotic Low-Density Lipoprotein Marker. Journal of atherosclerosis and thrombosis 23, 1257–1258 (2016).
Tian, J., Venn, A., Otahal, P. & Gall, S. The association between quitting smoking and weight gain: a systemic review and meta‐analysis of prospective cohort studies. Obesity reviews 16, 883–901 (2015).
Williamson, D. F. et al. Smoking cessation and severity of weight gain in a national cohort. New England Journal of Medicine 324, 739–745 (1991).
Harris, K. K., Zopey, M. & Friedman, T. C. Metabolic effects of smoking cessation. Nature reviews. Endocrinology 12, 299 (2016).
Matsushita, Y. et al. Associations of smoking cessation with visceral fat area and prevalence of metabolic syndrome in men: the Hitachi health study. Obesity 19, 647–651 (2011).
Takayama, S. et al. Smoking Cessation without Educational Instruction Could Promote the Development of Metabolic Syndrome. Journal of Atherosclerosis and Thrombosis, 40063 (2017).
Nakanishi, N., Takatorige, T. & Suzuki, K. Cigarette smoking and the risk of the metabolic syndrome in middle-aged Japanese male office workers. Industrial health 43, 295–301 (2005).
Shah, R. V. et al. Visceral adiposity and the risk of metabolic syndrome across body mass index: the MESA Study. JACC: Cardiovascular Imaging 7, 1221–1235 (2014).
Alberti, K. G. M., Zimmet, P. & Shaw, J. The metabolic syndrome—a new worldwide definition. The Lancet 366, 1059–1062 (2005).
Després, J.-P., Lemieux, I. & Alméras, N. In Overweight and the Metabolic Syndrome 137–152 (Springer, 2006).
Song, Y.-M., Chang, W.-D., Hsu, H.-Y. & Chen, M.-D. A short-term smoking cessation may increase the risk of developing metabolic syndrome. Diabetes & Metabolic Syndrome: Clinical Research & Reviews 9, 135–137 (2015).
Kim, B. J. et al. Association of smoking status, weight change, and incident metabolic syndrome in men: a 3-year follow-up study. Diabetes Care 32, 1314–1316 (2009).
Tamura, U. et al. Changes in weight, cardiovascular risk factors and estimated risk of coronary heart disease following smoking cessation in Japanese male workers: HIPOP-OHP study. Journal of atherosclerosis and thrombosis 17, 12–20 (2010).
Yoon, C., Goh, E., Park, S. M. & Cho, B. Effects of smoking cessation and weight gain on cardiovascular disease risk factors in Asian male population. Atherosclerosis 208, 275–279 (2010).
Filozof, C., Pinilla, F. & Fernández‐Cruz, A. Smoking cessation and weight gain. Obesity reviews 5, 95–103 (2004).
Shin, S. et al. Egg Consumption and Risk of Metabolic Syndrome in Korean Adults: Results from the Health Examinees Study. Nutrients 9, 687 (2017).
Ahn, Y. et al. Validation and reproducibility of food frequency questionnaire for Korean genome epidemiologic study. European journal of clinical nutrition 61, 1435 (2007).
Yang, J. J. et al. What are the major determinants in the success of smoking cessation: results from the health examinees study. PloS one 10, e0143303 (2015).
Jung, S. J. et al. Body mass index at age 18–20 and later risk of spontaneous abortion in the Health Examinees Study (HEXA). BMC pregnancy and childbirth 15, 228 (2015).
Group, H. E. S. The Health Examinees (HEXA) study: rationale, study design and baseline characteristics. Asian Pacific journal of cancer prevention: APJCP 16, 1591–1597 (2015).
Kim, Y., Han, B.-G. & Group, K. Cohort profile: the Korean genome and epidemiology study (KoGES) Consortium. International journal of epidemiology 46, e20–e20 (2016).
Grundy, S. et al. Detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). Circulation-Hagertown 106, 3143 (2002).
Novello, A. C. Surgeon General’s report on the health benefits of smoking cessation. Public Health Reports 105, 545 (1990).
Acknowledgements
Kyuwoong Kim and Seulggie Choi received a scholarship from the BK21-plus education program provided by the National Research Foundation of Korea. This study was supported by the National Genome Research Institute, Korea Centers for Disease Control and Prevention, and by a grant from the Seoul National University Hospital. We would like to extend our gratitude to the participants of the HEXA-G study group. This research was also supported by Basic ScienceResearch Program through the National ResearchFoundation (NRF) funded by the Ministry of Education [2017R1D1A1B03033721] in the Republic ofKorea.
Author information
Authors and Affiliations
Contributions
K.K. contributed to the study design, data analysis, interpretation, and wrote the first draft of the manuscript. S.C., J.L., J.C., A.S., S.K.P. and D.K. were involved in the study design, data analysis, and interpretation of the analysis. All authors were involved in the critical revision of the manuscript for intellectual contents under the supervision of S.M.P. All authors had complete access to the dataset used in this study and are responsible for the accuracy of the collection and analysis of the data.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, K., Choi, S., Lee, JK. et al. Weight change after smoking cessation and incident metabolic syndrome in middle-aged Korean men: an observational study. Sci Rep 9, 3103 (2019). https://doi.org/10.1038/s41598-019-39811-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39811-0
- Springer Nature Limited



