Abstract
We aimed to compare the effectiveness of various local anesthetic methods for controlling prostate biopsy (PBx) related pain using network meta-analysis. Literature searches were performed on PubMed/Medline, Embase, and Cochrane Library up to March 2018. Forty-seven randomized controlled trials, in which the effectiveness of PBx-related pain was investigated using a visual analogue scale after various local anesthetic methods, were included. The local anesthetic methods included intraprostatic local anesthesia (IPLA), intrarectal local anesthesia (IRLA), intravenous sedation (IVS), periprostatic nerve block (PNB), pelvic plexus block (PPB), and spinal anesthesia (SPA). Eight pairwise meta-analyses and network meta-analyses with 21 comparisons were performed. All modalities, except single use of IPLA and IRLA, were more effective than placebo. Our results demonstrate that PNB + IVS (rank 1) and SPA (rank 2) were the most effective methods for pain control. The followings are in order of PPB + IRLA, PNB + IPLA, PPB, PNB + IRLA, IVS, and PNB. In conclusion, the most effective way to alleviate PBx-related pain appears to be PNB + IVS and SPA. However, a potential increase in medical cost and additional risk of morbidities should be considered. In the current outpatient setting, PPB + IRLA, PNB + IPLA, PPB, PNB + IRLA, and PNB methods are potentially more acceptable options.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The introduction of transrectal ultrasonography (TRUS)-guided prostate biopsy (PBx) in 1989 was a significant development in diagnostic methodology for detection of prostate cancer (PCa). This new method quickly became the gold standard for the detection of PCa and remains the method of choice to the present day1. Although many patients can tolerate PBx the procedure can cause severe pain2. A random sampling method is used in PBx, so 10 or more core biopsy recommended to increase the detection rate of PCa3. However, as the number of core increases to achieve better detection rate, the patient experiences more pain. Severe pain can cause the patient to move during the procedure, which can increase the rate of complications and decrease the number of core that can be taken. These factors can potentially decrease accuracy of the final diagnosis4,5. These pitfalls highlight the importance of performing PBx with optimal local anesthesia for patient safety and accurate diagnosis4.
Various types of local anesthesia have been used for TRUS-guided PBx, including the most common methods of intrarectal local anesthesia (IRLA) and periprostatic nerve block (PNB)6. IRLA has been recommended as a non-invasive alternative to relieve pain during PBx7. Periprostatic injection of lidocaine into the neurovascular bundles markedly decreases the discomfort associated with PBx8. In addition to IRLA and PNB, intraprostatic local anesthesia (IPLA), spinal anesthesia (SPA; low dose spinal anesthesia or caudal block), intravenous sedation (IVS), and pelvic plexus block (PPB) are used for pain control during PBx. There are many randomized control trials (RCTs) that compare the effects of these anesthetic methods, alone or in combination, with placebo. However, the question of which method is the most effective way to control pain during PBx remains unanswered.
The purpose of the present study was to compare the effectiveness of various local anesthetic methods for controlling PBx-related pain to determine the best approach. To accomplish this, we performed a systematic review of the published literature and network meta-analysis of the available data.
Results
Systematic review process
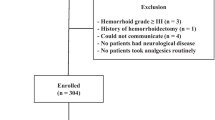
A summary of the analysis results is shown as a PRISMA flow diagram, representing a visual flowchart, in Fig. 1. A total of 1842 articles were found in initial database searches. Of these, 982 articles remained after removing duplicates. After excluding non-English articles, editorials, letters, reviews, case reports, and articles not related to this study by reviewing the titles and abstracts of all available literature, 134 articles remained. Next, a review of full-text articles was performed to evaluate whether they met the inclusion criteria. A total of 47 RCTs1,2,3,4,5,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50 with a total of 5,758 patients were finally included in this study. The characteristics of included studies are displayed in Table 1. All articles were prospective RCTs, and VAS was used to assess pain in all trials. The PNB injection site was the base, with or without apex. The number of PBx core ranged from 6 to 14 across all studies.
Pairwise meta-analysis
IRLA vs. Placebo
Eight studies showed that IRLA demonstrated no statistically significant pain relief compared to placebo (MD: −0.32, 95% CI: −0.72 to 0.09, P = 0.13, Fig. 2A). Heterogeneity among the included studies was observed (P = 0.001; I2 = 70%).
(A) Forest plots comparing IRLA with placebo, (B) Forest plots comparing PNB with IRLA, (C) Forest plots comparing PNB with Placebo, (D) Forest plots comparing PNB + IPLA with PNB, (E) Forest plots comparing PNB + IRLA with IRLA, (F) Forest plots comparing PNB + IRLA with PNB, (G) Forest plots comparing PPB + IRLA with PNB + IRLA, (H) Forest plots comparing SPA with PNB + IRLA IPLA: intraprostatic local anesthesia, IRLA: intrarectal local anesthesia, PNB: periprostatic nerve block, PPB: pelvic plexus block, SPA: spinal anesthesia.
PNB vs. IRLA
Fourteen studies showed that PNB was significantly lower compared to that of IRLA (MD: −1.61, 95% CI: −2.08 to −1.15, P < 0.00001, Fig. 2B). Heterogeneity among the included studies was observed (P < 0.00001; I2 = 88%).
PNB vs. Placebo
Ten studies showed that PNB significantly reduced pain compared with placebo (MD: −2.04, 95% CI: −2.77 to −1.31, P < 0.00001, Fig. 2C). Heterogeneity among the included studies was observed (P < 0.00001; I2 = 90%).
PNB + IPLA vs. PNB
Five studies showed that PNB + IPLA significantly reduced pain compared with PNB (MD: −0.97, 95% CI: −1.33 to −0.60, P < 0.00001; Fig. 2D). Heterogeneity among the included studies was not observed (P = 0.001, I2 = 78%).
PNB + IRLA vs. IRLA
Four studies showed that PNB + IRLA significantly reduced pain compared with IRLA (MD: −1.75, 95% CI: −2.97 to −0.53, P = 0.005; Fig. 2E). Heterogeneity among the included studies was observed (P < 0.00001; I2 = 96%).
PNB + IRLA vs. PNB
Seven studies were included in the analysis that compared PNB + IRLA with PNB (Fig. 2F). Combined PNB + IRLA resulted in significantly lower VAS scores than PNB alone (MD: −0.61, 95% CI: −1.09 to −0.13, P = 0.01). Heterogeneity among the included studies was observed (P < 0.00001; I2 = 87%).
PPB + IRLA vs. PNB + IRLA
Two studies showed that PPB + IRLA significantly reduced pain compared with PNB + IRLA (MD: −1.09, 95% CI: −1.29 to −0.90, P < 0.00001; Fig. 2G). Heterogeneity among the included studies was not observed (P = 0.96; I2 = 0%).
SPA vs. PNB + IRLA
Three studies showed that although SPA seems to have reduced pain compared with PNB + IRLA, the result was not statistically significant (MD: −1.47, 95% CI: −2.73 to −0.21, P = 0.02; Fig. 2H). Heterogeneity among the included studies was observed (P < 0.00001; I2 = 97%).
Bayesian framework network meta-analysis
The network meta-analysis was performed with 47 studies. Eleven nodes (10 local anesthetic methods plus placebo) and 21 comparisons in the network plot of evidence are shown in Fig. 3. The width of each line is proportional to the number of trials comparing every pair of treatments, and the size of every node is proportional to the number of randomized participants.
Network plot of evidence of all trials. The width of the lines is proportional to the number of trials comparing every pair of treatments, and the size of every node is proportional to the number of randomized participants. IPLA: intraprostatic local anesthesia, IRLA: intrarectal local anesthesia, IVS: intravenous sedation, PNB: periprostatic nerve block, PPB: pelvic plexus block, SPA: spinal anesthesia.
The network meta-analysis results are described in Fig. 4. Compared to placebo, PNB + IVS, SPA, PPB + IRLA, PNB + IPLA, PPB, PNB + IRLA, IVS, and PNB were significantly more effective on PBx-related pain control. Neither IPLA nor IRLA showed a significant difference in VAS compared to placebo. The relative effect plot confirmed these findings (Fig. 5). The rankings of the local anesthetic methods are similarly presented in Fig. 6. PNB + IVS and SPA were ranked first and the second, respectively, followed by PPB + IRLA, PNB + IPLA, PPB, PNB + IRLA, IVS, PNB, IPLA, and IRLA. There were 15 node-splitting models. With the exception of two models (PNB vs. PPB and PPB vs. placebo), 95% CrIs of inconsistency factors included zero and P-values of > 0.05 for the comparison between direct and indirect effects in all other node-splitting models.
Relative effect table of local anesthetic method’s efficacy for pain control related prostate biopsy. Comparisons between treatments should be read from left to right, and the estimate is in the cell in common between the column-defining treatment and the row-defining treatment. For efficacy in local anesthesia, mean differences (MDs) less than 0 favor the column-defining treatment. Bold indicates statistical significance. IPLA: intraprostatic local anesthesia, IRLA: intrarectal local anesthesia, IVS: intravenous sedation, PNB: periprostatic nerve block, PPB: pelvic plexus block, SPA: spinal anesthesia.
There were 15 node-splitting models. With the exception of two models (PNB vs. PPB and PPB vs. placebo), 95% CrIs of inconsistency factors included zero and P-values of > 0.05 for the comparison between direct and indirect effects in all other node-splitting models. There were no significant inconsistencies in results in this analysis.
Quality assessment and qualitative risk of bias
The risk of bias graph and assessment are summarized in Figs 7 and 8. There are three main sources of bias in the included trials. The first is insufficient number of participants in some trials, which may make it difficult to demonstrate the effect of local anesthesia. The second is that the number of biopsies taken was not identical for every patient in the trials. This variable may also affect the VAS score.
The results of GRADE quality assessment of direct evidence of each pairwise treatment comparison are shown in Table 2. Of the eight comparisons, certainty was low in six and very low in two.
Discussion
PCa is the most frequently diagnosed cancer worldwide in men, with approximately 1.1 million new cases being diagnosed each year51. Assuming that the current PBx standard of using 10–14 biopsy cores detects prostate cancer in up to 44% of patients52, then PBx can be expected to be performed in approximately 2.5 million cases a year worldwide. Recently, various types of local anesthetic methods have been proposed to reduce the discomfort and pain associated with PBx22. Consequently, there is ongoing research to identify the perfect method for reduction of pain. Therefore, we performed a systematic review and network meta-analysis of RCTs published to date to determine the most effective anesthetic methods for pain control. Although similar meta-analyses6,7,53,54,55,56 have been performed previously, a new analysis was needed to incorporate the results of new trials that have been published on this topic. Moreover, previous meta-analyses only made direct comparisons, whereas we included multiple comparisons with more local anesthetic techniques (specifically, IVS, SPA, and PPB). Our study was an arm-based network meta-analysis with 11 arms (IPLA, IRLA, IVS, PNB, PPB, SPA, PNB + IPLA, PNB + IRLA, PNB + IVS, PPB + IRLA, and placebo).
The most commonly used local anesthetic methods for PBx are IRLA and PNB. National Comprehensive Cancer Network guidelines state that local lidocaine injection is more efficacious in reducing pain during probe insertion, whereas PNB reduces pain during the biopsy itself57. European Urological Association guidelines recommend PNB as the standard of care58. IRLA is a convenient local anesthesia method that causes only a little discomfort to patients54. The most commonly used agent is lidocaine alone or in combination with other molecules (prilocaine, nifedipine, dimethyl sulfoxide, etc.)59. Our results show that IRLA with lidocaine gel alone or in combination with other molecules could not significantly reduce the pain during the PBx, which is consistent with results from previous meta-analyses7,53,54. IRLA alone was the worst ranked method in this analysis other than placebo. Nash et al. were the first to describe the use of PNB before PBx in 19968. Subsequently, many trials2,9,12,21,22,25,28,30,34,36,37,40,42 and meta-analyses7,54,55 have suggested that injection of local anesthetic around the nerve bundles might provide the best pain control during PBx. The results of our study also show that PNB significantly reduced pain compared with IRLA and placebo and are consistent with results from previous meta-analyses7,54,55,56. However, PNB alone had a probability of being ranked just seventh in effectiveness in the current study.
Combining PNB with IPLA or with IRLA results in a more extensive pain control effect53. In 2005, Mutaguchi et al.60 reported IPLA was a new local anesthesia technique for anesthetizing the prostate by blocking all sensory nerves from the posterior and anterior. Our results show that PNB with either IPLA or IRLA significantly reduced pain compared with placebo and had intermediate probability of being ranked fourth and sixth, respectively. However, IPLA alone did not significantly reduce the pain during the PBx. Recently, IPLA was shown to extend the time of PBx, but also provide improved pain control compared to PNB59. Nonetheless, more RCTs are required to justify the single use of IPLA in general.
Three recent trials investigated the effect of IVS for pain control during PBx14,31,50. Atta et al.14 conducted IVS by slow infiltration (3–5 min) of diazepam (5 mg) just before probe insertion and found that this IVS approach significantly decreases discomfort and fear of insertion of the probe and alleviates patient anxiety. In addition, PNB + IVS displays a significantly lower VAS score during PBx. Mazdak et al.31 performed IVS by infiltration of midazolam (25 μg/kg), fentanyl (2 μg/kg), and ketamine (1 mg/kg) through an antecubital vein at 5 min before taking the biopsies. They reported that the differences in VAS score between PNB + IRLA and IVS are not statistically significant. Additionally, they reported SPA significantly reduces pain compared with IVS. Turgut et al.50 injected midazolam (0.07–0.1 mg/kg) intravenously 5–10 min before the procedure. They showed that IVS displays a significantly lower VAS score compared with placebo, whereas no significant difference is observed between IVS and PNB. Our results show that IVS alone had no pain control effects compared with PNB alone, whereas combined PNB + IVS significantly reduced pain compared with PNB + IRLA. PNB + IVS had the highest probability of being ranked first. Nevertheless, IVS has a risk of respiratory and cardiovascular depression, and the use of anesthetic staff has cost implications50. Only three trials were analyzed in the aforementioned studies, so more RCTs should be performed for more accurate analysis of the effect of IVS.
PPB is also used to control biopsy pain. Wu et al.47 first described PPB using 5 mL of 1% lidocaine directly lateral to the tip of the seminal vesicles under grey-scale ultrasonography guidance. They found that this method does not reduce biopsy-related pain. Conversely, Akpinar et al.10 conducted a trial comparing PPB (using 2 mL of 2% lidocaine), to PNB at the same dosage and concentration. The authors reported that PPB provides superior analgesia to PNB because it acts on the prostate proximally. They suggested that the reasons for their divergent results are not clear but may be caused by the use of Doppler ultrasonography and larger cohorts unlike the study of Wu et al.47. Color Doppler ultrasonography is helpful for identifying the entire pelvic plexus with no influence of the left lateral decubitus position59. Another two studies reported that PPB + IRLA has significantly more effective pain control than PNB + IRLA17,24. Although the currently available guidelines do not mention the use of PPB, our study suggests the possibility that PPB might be superior to PNB. This concept requires more RCTs to confirm this possibility. Moreover, this procedure should be conducted only by skilled practitioners under the guidance of color Doppler ultrasonography, a requirement that may represent a limitation for this method.
SPA is an ideal anesthetic for anorectal surgery1 and had a highly probability of being ranked second in our study. Four of the included trials investigated the PBx pain control effect of SPA1,31,34,46. We classified the caudal block in one trial46 into one kind of SPA in which caudal block was accomplished using 20 mL of 1.2% lidocaine. Other trials used SPA with a low dose of 0.5% bupivacaine (0.5–1.5 mL) at the L3-5 level. They reported SPA significantly decreases pain during the procedure1,31,34,46. Moreover, there was no significant difference in anesthesia time or pain due to anesthesia itself compared with PNB. Nonetheless, SPA requires the presence of an anesthesiologist, which adds to the overall cost of the procedure and reduces the likelihood of SPA being practiced in an outpatient setting. Other drawbacks to SPA include a potential to cause a spinal headache or backache, systemic blood pressure or heart rate changes, and risk of infection into the subarachnoid space1.
To the best of our knowledge, ours is the first network meta-analysis to evaluate the optimal local anesthetic for controlling PBx-related pain in order to help clinicians select an appropriate anesthetic method for patients undergoing PBx. Our meta-analysis results suggest that various local anesthetic modalities, alone or in combination, were significantly more effective than placebo in alleviating pain for TRUS-guided PBx. Of these methods, PNB + IVS and SPA seem to be the most effective way to control PBx-related pain. However, several medical and socioeconomic factors should be considered because SPA and IVS are undoubtedly associated with increases of medical cost, need for anesthesiologist, need for extra medical personnel for monitoring patients’ recovery, longer hospital stay, and additional risk of morbidities, etc. In the current outpatient setting, PPB + IRLA, PNB + IPLA, PPB, PNB + IRLA, and PNB are acceptable modalities, but it is still uncertain whether PPB can be superior to PNB. Using the results of this study, clinicians could select the most effective method by providing a choice of anesthetic methods and discussing the methods with patients based on the patient’s personality, medical condition, and financial status, and the capabilities of the hospital.
We performed only the GRADE assessment of the direct comparison without assessing of indirect evidences. However, there are the GRADE Working Group approach for accessing the quality of treatment effect estimates from network meta-analysis61. Salanti et al. also suggested an approach to determining confidence in the output of a network meta-analysis62. They have emphasized the importance of assessing the quality and confidence of each pairwise comparison (containing direct, indirect or mixed evidence) and the ranking estimates. The judgments of the evidence of the network meta-analysis may differ from those of the evidence of the pairwise meta-analysis. Since most of the risk of bias for included studies was unclear, all of the GRADE risk-of-bias items were assessed as serious. This resulted in low and very low quality of all evidence due to the low quality of the studies included. Low level of GRADE assessment means that further research is highly likely to have a significant impact on our confidence in estimating effects and changes in estimates. Very low of GRADE assessment means that estimates of effects are highly uncertain. There was a discrepancy in the placebo arm among the included studies. For examples, some trials3,23,32,35 performed periprostatic saline injection, but others5,9,18,19,29,37 instilled placebo solutions into the rectum. Moreover, there was also no agreement in the type or dose of anesthetic in the same group. A major practical disadvantage of the VAS is that subsequent distance measurements are required63. Patients have difficulty finding the point that best describes the pain they feel. In other words, the VAS can be measured higher than actually talking about pain in words64,65. It may be easy to interpret the category of modest VAS meaningfully. Although VAS is may useful for the comparison of pain between the two groups, the interpretation of a single VAS may be subjective63. Network meta-analysis assumes that some treatment arms can be grouped in a similarity of rationality and procedure. Although there is added potential for combining studies that are not adequately similar, this assumption allows us to group treatment arms together as one node in the network66. Further high-quality RCTs are needed to overcome these drawbacks, to validate this result, and to find the optimal anesthetic method for PBx.
In conclusion, our meta-analysis suggests that various local anesthetic method, alone or in combination, were more effective in alleviating pain for TRUS-guided PBx than placebo. Of these anesthetic methods, PNB + IVS and SPA seem to be the most effective way to control PBx-related pain. Unfortunately, increase of medical cost and additional risk of morbidity must be considered before using these methods. In the current outpatient setting, PPB + IRLA, PNB + IPLA, PPB, PNB + IRLA, and PNB are the more acceptable options. Although the currently available guidelines do not mention the use of PPB, our study suggests that PPB might be a promising alternative to PNB. Further high-quality RCTs are needed to validate these results and clarify the optimal anesthetic method.
Material and Method
This systematic review was registered in PROSPERO (CRD42018092602).
Literature search
We performed literature searches on PubMed/Medline, Embase, Cochrane Library, Web of science, Scopus, and Clinical Key up to March 2018. Only published trials in English were included. Conference and meeting abstracts were excluded even if they met the eligibility criteria. The search terms used included ‘transrectal’, ‘prostate’, ‘biopsy’, ‘intraprostatic’, ‘periprostatic’, ‘intrarectal’, ‘spinal’, ‘anesthesia’, ‘block’, ‘pelvic plexus’, and relevant variants. A total of 1842 possible articles, of which 982 articles remained after removing duplicates, were found using these literature searches. Two authors (DKK and YSH) reviewed the titles and abstracts in consideration of the inclusion criteria and reviewed the independently identified papers. In cases of disagreement, the reviewers decided by agreement on whether or not to include the article.
Trial inclusion criteria and exclusion criteria
The eligibility of a study was evaluated by the PICOS (participants, interventions, comparators, outcomes, and study design) approach and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines67. Study population was defined as patients who underwent TRUS-guided PBx, and intervention was defined as local anesthesia. The comparator was defined as placebo or anesthetic method. The outcomes were biopsy pain scores that were measured using a visual analogue scale (VAS). Inclusion criteria were as follows: (1) RCT, (2) human research, (3) patients underwent TRUS-guided PBx, (4) use of local anesthesia, and (5) reported outcome values (VAS: mean and standard deviation could be calculated).
Data extraction
Two independent authors (DKK and YSH) extracted the data using a predesigned form. Any conflicts in extracted data between the two authors were resolved via consensus. Extracted data included the first author, publication year, study design, treatment arms, number of patients, local anesthetic method and anesthetics, inclusion and exclusion criteria, number of PBx core, type of pain scale, and outcome. The main outcome was the biopsy pain score measured by VAS.
Study quality assessments and quality of evidence
The risk of bias was evaluated in individual studies using tools recommended in recent meta-analysis guidelines that rate various aspects of RCT design and implementation68. Risk of bias was assessed for random sequence generation, allocation concealment, blinding of participants and researchers, blinding of outcome assessment, incomplete outcome data, selective reporting, and others.
The Grading of Recommendations, Assessments, Developments, and Evaluation (GRADE) was used to provide a systematic approach to the evaluation of the quality of evidence and strength of recommendations69. Criteria for consideration included assessment of methodology, precision and consistency of results, directness, and risk of publication bias. Based on five criteria, we assessed only direct evidence of pairwise meta-analysis by classifying the quality of evidence as one of four levels (i.e., high, moderate, low, and very low).
Statistical analysis
A pairwise meta-analysis was performed on the comparison of a minimum of two studies. Outcomes are reported as a combination of the weighted mean difference (MD) with 95% confidence interval (CI) and the P-value. The pooled MD with 95% CIs expresses the difference in the size of the intervention effect. Statistical heterogeneity between trials was evaluated by Chi-square heterogeneity tests. The I2 statistic was also calculated to measure discrepancies between clinical trials. Either a P-value of < 0.05 for the Cochran Q statistic or an I2 statistic of > 50% indicated significant heterogeneity between trials68.
To indirectly compare the effect of each local anesthetic method on the endpoint (VAS), we conducted a network meta-analysis using a Bayesian hierarchical random effects model for continuous outcomes. This model estimates treatment-specific effects and effect differences. Pooled estimates were obtained using the Markov Chains Monte Carlo method in which each chain has 20,000 simulations and the first 5,000 simulations are discarded as burn-in. We modeled the continuous outcomes for every local anesthetic method of all trials and quantified the association between MDs with 95% credible intervals (CrIs) among studies (CrIs can be regarded as similar to conventional CIs). The selection of a random effects model for reported outcomes was based on the deviance information criteria. The random effects model is a measure of model fit that penalizes model complexity70. The node-splitting method was applied for computing the inconsistency of the model. The results of node-splitting analysis are considered to show no significant inconsistency when 95% CIs of inconsistency factors include zero or when there is a large probability value (P-value > 0.05) for the comparison between direct and indirect effects71. The relative effects were also assessed visually using the relative effects table and plots. Probability values were summarized and are reported as a rank probabilities table and plot.
We conducted the pairwise meta-analysis using Review Manager v.5.1 (The Nordic Cochrane Center, The Cochrane Collaboration, Copenhagen, Denmark, 2008). The network meta-analyses were conducted using R 3.4.3 (R development Core Team, Vienna, http://www.R-project.org) with the ‘GEMTC’ packages.
References
Kucur, M. et al. Selective Low-Dose Spinal Anesthesia for Transrectal Prostate Biopsy: A Prospective and Randomized Study. J Endourol. 29, 1412–1417 (2015).
Izol, V., Soyupak, B., Seydaoglu, G., Aridogan, I. A. & Tansug, Z. Three different techniques for administering analgesia during transrectal ultrasound-guided prostate biopsy: a comparative study. Int Braz J Urol. 38, 122–128 (2012).
Ingber, M. S., Ibrahim, I., Turzewski, C., Hollander, J. B. & Diokno, A. C. Does periprostatic block reduce pain during transrectal prostate biopsy? A randomized, placebo-controlled, double-blinded study. Int Urol Nephrol. 42, 23–27 (2010).
Giannarini, G. et al. Combination of perianal-intrarectal lidocaine-prilocaine cream and periprostatic nerve block for pain control during transrectal ultrasound guided prostate biopsy: a randomized, controlled trial in J Urol. 181, 585–591, discussion 591–583 (2009).
Galosi, A., Minardi, D., Dell’atti, L., Yehia, M. & Muzzonigro, G. Tolerability of prostate transrectal biopsies using gel and local anesthetics: results of a randomized clinical trial in. J Endourol. 19, 738–743 (2005).
Yang, Y. et al. The Efficiency and Safety of Intrarectal Topical Anesthesia for Transrectal Ultrasound-Guided Prostate Biopsy: A Systematic Review and Meta-Analysis. Urol Int. 99, 373–383 (2017).
Tiong, H., Liew, L., Samuel, M., Consigliere, D. & Esuvaranathan, K. A meta-analysis of local anesthesia for transrectal ultrasound-guided biopsy of the prostate. Prostate Cancer Prostatic Dis. 10, 127 (2007).
Nash, P. A., Bruce, J. E., Indudhara, R. & Shinohara, K. Transrectal ultrasound guided prostatic nerve blockade eases systematic needle biopsy of the prostate. J Urol. 155, 607–609 (1996).
Adamakis, I. et al. Pain during transrectal ultrasonography guided prostate biopsy: a randomized prospective trial comparing periprostatic infiltration with lidocaine with the intrarectal instillation of lidocaine-prilocain cream. World J Urol. 22, 281–284 (2004).
Akpınar, H., Tüfek, İ., Atuğ, F., Esen, E. H. & Kural, A. R. Doppler ultrasonography-guided pelvic plexus block before systematic needle biopsy of the prostate: a prospective randomized study. Urology. 74, 267–271 (2009).
Aktoz, T. et al. ‘Multimodal’ approach to management of prostate biopsy pain and effects on sexual function: efficacy of levobupivacaine adjuvant to diclofenac sodium - a prospective randomized trial. Andrologia. 42, 35–40 (2010).
Alavi, A. S., Soloway, M. S., Vaidya, A., Lynne, C. M. & Gheiler, E. L. Local anesthesia for ultrasound guided prostate biopsy: a prospective randomized trial comparing 2 methods. J Urol. 166, 1343–1345 (2001).
Anastasi, G. et al. Transrectal ultrasound (TRUS) guided prostate biopsy: Three different types of local anesthesia. Arch Ital Urol Androl. 88, 308–310 (2016).
Atta, H., Mostafa, M. F. & Shalaby, M. Which is better for pain reduction during transrectal ultrasound-guided biopsy of the prostate: Intravenous diazepam, local periprostatic nerve block, or combination? controlled randomized study. Saudi J Anaesth. 12, 16–21 (2018).
Bingqian, L., Peihuan, L., Yudong, W., Jinxing, W. & Zhiyong, W. Intraprostatic local anesthesia with periprostatic nerve block for transrectal ultrasound guided prostate biopsy. J Urol. 182, 479–483, discussion 483–474 (2009).
Cam, K., Sener, M., Kayikci, A., Akman, Y. & Erol, A. Combined periprostatic and intraprostatic local anesthesia for prostate biopsy: a double-blind, placebo controlled, randomized trial. J Urol. 180, 141–144, discussion 144–145 (2008).
Cantiello, F. et al. Pelvic plexus block is more effective than periprostatic nerve block for pain control during office transrectal ultrasound guided prostate biopsy: a single center, prospective, randomized, double arm study. J Urol. 188, 417–421 (2012).
Cevik, I., Ozveri, H., Dillioglugil, O. & Akda, A. Lack of effect of intrarectal lidocaine for pain control during transrectal prostate biopsy: a randomized prospective study. Eur Urol. 42, 217–220 (2002).
Chang, S. S., Alberts, G., Wells, N., Smith, J. A. Jr. & Cookson, M. S. Intrarectal lidocaine during transrectal prostate biopsy: results of a prospective double-blind randomized trial. J Urol. 166, 2178–2180 (2001).
Goluza, E. et al. Lidocaine suppository for transrectal ultrasound-guided biopsy of the prostate: a prospective, double-blind, randomized study. Urol Int. 86, 315–319 (2011).
Gurbuz, C. et al. Visual pain score during transrectal ultrasound-guided prostate biopsy using no anaesthesia or three different types of local anaesthetic application. Scand J Urol Nephrol. 44, 212–216 (2010).
Hiros, M., Selimovic, M., Spahovic, H., Sadovic, S. & Spuzic-Celic, E. Transrectal ultrasound-guided prostate biopsy, periprostatic local anesthesia and pain tolerance. Bosnian journal of basic medical sciences. 10, 68–72 (2010).
Inal, G. et al. Effect of periprostatic nerve blockade before transrectal ultrasound-guided prostate biopsy on patient comfort: a randomized placebo controlled study. Int J Urol. 11, 148–151 (2004).
Jindal, T. et al. Transrectal ultrasonography (TRUS)-guided pelvic plexus block to reduce pain during prostate biopsy: a randomised controlled trial. BJU Int. 115, 892–896 (2015).
Kravchick, S. et al. Comparison of different local anesthesia techniques during TRUS-guided biopsies: a prospective pilot study. Urology. 65, 109–113 (2005).
Kumar, A., Griwan, M. S., Singh, S. K., Sen, J. & Pawar, D. S. Is periprostatic nerve block a gold standard in case of transrectal ultrasound-guided prostate biopsy? Urol Ann. 5, 152–156 (2013).
Lee, H. Y. et al. Effect of Intraprostatic Local Anesthesia During Transrectal Ultrasound Guided Prostate Biopsy: Comparison of 3 Methods in a Randomized, Double-Blind, Placebo Controlled Trial. J Urol. 178, 469–472 (2007).
Lee, Y. I., Seo, I. Y., Jeong, H. J. & Rim, J. S. Comparison of two local anesthestic methods for transrectal ultrasound guided prostate biopsy: periprostatic injection of lidocaine and rectal instillation of lidocaine gel. Korean J Urol. 45, 423–427 (2004).
Leung, S. et al. Intrarectal administration of lidocaine gel versus plain lubricant gel for pain control during transrectal ultrasound-guided extensive 10-core prostate biopsy in Hong Kong Chinese population: prospective double-blind randomised controlled trial. Hong Kong Med J. 12, 103–107 (2006).
Mallick, S. et al. Which anaesthesia should be recommended for prostate biopsy? West Indian Med J. 54, 135–138 (2005).
Mazdak, H., Abtahi, A. M., Momeni, F. & Izadpanahi, M. H. A comparison of pain control and complications using three different ways of anesthesia in patients undergoing transrectal ultrasound-guided prostate biopsy. J Res Med Sci. 23, 17 (2018).
Nambirajan, T. et al. Efficacy and safety of peri-prostatic local anaesthetic injection in trans-rectal biopsy of the prostrate: A prospective randomised study. Surgeon. 2, 221–224 (2004).
Noh, D. H. et al. The effects of combination of perianal-intrarectal lidocaineprilocaine cream and periprostatic nerve block for pain control during transrectal ultrasound guided biopsy of the prostate: A randomized, controlled trial. Int Urogynecol J Pelvic Floor Dysfunct. 22, S250–S251 (2011).
Obi, A. O., Okafor, V. U. & Nnodi, P. I. Prospective randomized trial of spinal saddle block versus periprostatic lignocaine for anesthesia during transrectal prostate biopsy. Urology. 77, 280–285 (2011).
Ozden, E., Yaman, O., Gogus, C., Ozgencil, E. & Soygur, T. The optimum doses of and injection locations for periprostatic nerve blockade for transrectal ultrasound guided biopsy of the prostate: a prospective, randomized, placebo controlled study. J Urol. 170, 2319–2322 (2003).
Raber, M. et al. Topical prilocaine-lidocaine cream combined with peripheral nerve block improves pain control in prostatic biopsy: results from a prospective randomized trial. Eur Urol. 53, 967–975 (2008).
Raber, M., Scattoni, V., Roscigno, M., Rigatti, P. & Montorsi, F. Perianal and intrarectal anaesthesia for transrectal biopsy of the prostate: a prospective randomized study comparing lidocaine‐prilocaine cream and placebo. BJU Int. 96, 1264–1267 (2005).
Rodriguez, A., Kyriakou, G., Leray, E., Lobel, B. & Guillé, F. Prospective study comparing two methods of anaesthesia for prostate biopsies: apex periprostatic nerve block versus intrarectal lidocaine gel: review of the literature. Eur Urol. 44, 195–200 (2003).
Seckiner, I., Sen, H., Erturhan, S. & Yagci, F. A prospective, randomized controlled study comparing lidocaine and tramadol in periprostatic nerve blockage for transrectal ultrasound-guided prostate biopsy. Urology. 78, 257–260 (2011).
Singh, S., Griwan, M. & Kumar, A. Comparison of efficacy of periprostatic nerve block versus combination of periprostatic nerve block and intraprostatic anaesthesia during transrectal ultrasound guided prostate biopsy. Urology. 78, S308 (2011).
Skriapas, K., Konstantinidis, C., Samarinas, M., Xanthis, S. & Gekas, A. Comparison between lidocaine and glyceryl trinitrate ointment for perianal-intrarectal local anesthesia before transrectal ultrasonography-guided prostate biopsy: a placebo-controlled trial. Urology. 77, 905–908 (2011).
Song, S. H., Kim, J. K., Song, K., Ahn, H. & Kim, C. S. Effectiveness of local anaesthesia techniques in patients undergoing transrectal ultrasound-guided prostate biopsy: a prospective randomized study. Int J Urol. 13, 707–710 (2006).
Szlauer, R., Paras, L. & Fink, K. G. Addition of lidocaine suppositories to periprostatic nerve block enhances pain control in prostate biopsies: a placebo-controlled randomized trial. Urol Int. 84, 413–417 (2010).
Trucchi, A. et al. Local anesthesia reduces pain associated with transrectal prostatic biopsy. Urol Int. 74, 209–213 (2005).
Vanni, A. P., Schaal, C. H., Costa, R. P. & Sala, F. C. Is the periprostatic anesthetic blockade advantageous in ultrasound-guided prostate biopsy? Int Braz J Urol. 30, 114–118 (2004).
Wang, N., Fu, Y., Ma, H., Wang, J. & Gao, Y. Advantages of caudal block over intrarectal local anesthesia plus periprostatic nerve block for transrectal ultrasound guided prostate biopsy. Pak J Med Sci. 32, 978–982 (2016).
Wu, C., Carter, H., Naqibuddin, M. & Fleisher, L. Effect of local anesthetics on patient recovery after transrectal biopsy. Urology. 57, 925–929 (2001).
Yun, T. J. et al. Does the intrarectal instillation of lidocaine gel before periprostatic neurovascular bundle block during transrectal ultrasound guided prostate biopsies improve analgesic efficacy? A prospective, randomized trial. J Urol. 178, 103–106 (2007).
Yurdakul, T., Taspinar, B., Kilic, O., Kilinc, M. & Serarslan, A. Topical and long-acting local anesthetic for prostate biopsy: a prospective randomized placebo-controlled study. Urol Int. 83, 151–154 (2009).
Turgut, A., Ergun, E., Koşar, U., Koşar, P. & Özcan, A. Sedation as an alternative method to lessen patient discomfort due to transrectal ultrasonography-guided prostate biopsy. Eur J Radiol. 57, 148–153 (2006).
Torre, L. A. et al. Global cancer statistics, 2012. CA Cancer J Clin. 65, 87–108 (2015).
Patel, A. R. & Jones, J. S. Optimal biopsy strategies for the diagnosis and staging of prostate cancer. Curr Opin Urol. 19, 232–237 (2009).
Yan, P., Wang, X.-Y., Huang, W. & Zhang, Y. Local anesthesia for pain control during transrectal ultrasound-guided prostate biopsy: a systematic review and meta-analysis. J Pain Res. 9, 787 (2016).
Li, M. et al. Local anesthesia for transrectal ultrasound-guided biopsy of the prostate: A meta-analysis. Sci Rep. 7, 40421 (2017).
Hergan, L., Kashefi, C. & Parsons, J. K. Local anesthetic reduces pain associated with transrectal ultrasound-guided prostate biopsy: a meta-analysis. Urology. 69, 520–525 (2007).
Richman, J. M. et al. Efficacy of periprostatic local anesthetic for prostate biopsy analgesia: a meta-analysis. Urology. 67, 1224–1228 (2006).
Carroll, P. R. et al. NCCN guidelines insights: prostate cancer early detection, version 2.2016. J Natl Compr Canc Netw. 14, 509–519 (2016).
Mottet, N. et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 71, 618–629 (2017).
Maccagnano, C. et al. Anaesthesia in transrectal prostate biopsy: which is the most effective technique? Urol Int. 87, 1–13 (2011).
Mutaguchi, K. et al. Local anesthesia during 10 core biopsy of the prostate: Comparison of 2 methods. J Urol. 173, 742–745 (2005).
Puhan, M. A. et al. A GRADE Working Group approach for rating the quality of treatment effect estimates from network meta-analysis. BMJ. 349, g5630 (2014).
Salanti, G., Del Giovane, C., Chaimani, A., Caldwell, D. M. & Higgins, J. P. Evaluating the quality of evidence from a network meta-analysis. PLoS One. 9, e99682 (2014).
Klimek, L. et al. Visual analogue scales (VAS): Measuring instruments for the documentation of symptoms and therapy monitoring in cases of allergic rhinitis in everyday health care: Position Paper of the German Society of Allergology (AeDA) and the German Society of Allergy and Clinical Immunology (DGAKI), ENT Section, in collaboration with the working group on Clinical Immunology, Allergology and Environmental Medicine of the German Society of Otorhinolaryngology, Head and Neck Surgery (DGHNOKHC). Allergo J Int. 26, 16–24 (2017).
Duncan, G. H., Bushnell, M. C. & Lavigne, G. J. Comparison of verbal and visual analogue scales for measuring the intensity and unpleasantness of experimental pain. Pain. 37, 295–303 (1989).
Joyce, C. R., Zutshi, D. W., Hrubes, V. & Mason, R. M. Comparison of fixed interval and visual analogue scales for rating chronic pain. Eur J Clin Pharmacol. 8, 415–420 (1975).
Lumley, T. Network meta‐analysis for indirect treatment comparisons. Stat Med. 21, 2313–2324 (2002).
Moher, D. et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 4, 1 (2015).
Higgins, J. P. et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 343, d5928 (2011).
Guyatt, G. H. GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 336, 924–926 (2008).
Caldwell, D. M., Ades, A. & Higgins, J. Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ. 331, 897 (2005).
Lu, G. & Ades, A. Assessing evidence inconsistency in mixed treatment comparisons. J Am Stat Assoc. 101, 447–459 (2006).
Author information
Authors and Affiliations
Contributions
D.K.K., B.H.C. and K.S.C. designed this study and prepared the manuscript. D.K.K., J.Y.L., J.H.J., Y.S.H. and K.C.K. collected and analyzed the clinical data. D.K.K., Y.S.H. and K.S.L. performed statistical analysis. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, D.K., Lee, J.Y., Jung, J.H. et al. What is the most effective local anesthesia for transrectal ultrasonography-guided biopsy of the prostate? A systematic review and network meta-analysis of 47 randomized clinical trials. Sci Rep 9, 4901 (2019). https://doi.org/10.1038/s41598-019-41412-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-41412-w
- Springer Nature Limited