Abstract
Life history theory predicts that animals often produce fewer offspring of larger size and indicate a stronger trade-off between the number and size of offspring to cope with increasing environmental stress. In order to evaluate this prediction, we tested the life history characteristics of Bufo minshanicus at eight different altitudes on the eastern Tibetan Plateau, China. Our results revealed a positive correlation between female SVL and clutch size or egg size, revealing that larger females produce more and larger eggs. However, high-altitude toads seem to favor more offspring and smaller egg sizes when removing the effect of female SVL, which is counter to theoretical predictions. In addition, there was an overall significantly negative relationship between egg size and clutch size, indicative of a trade-off between egg size and fecundity. Therefore, we suggest that higher fecundity, rather than larger egg size, is a more effective reproductive strategy for this species of anuran living at high-altitude environments.
Similar content being viewed by others
Introduction
Life-history theory suggests that females should be able to optimize their allocation of resources between current breeding efforts and future survival1. Higher altitudes can be stressful environments because temperatures are colder, seasonality is greater, and food availability can fluctuate greatly. For example, there are negative correlations between temperature and altitude2, with rainfall and altitude found to be positively correlated3. High-altitude females are expected to select for producing larger eggs2,3,4,5,6 because larger eggs can lead to larger initial sizes, faster rates of growth, faster developmental rates, or both7,8,9,10,11,12. Yet, a significant increase in egg size may prolong the duration of the embryonic period, which results in increasing total mortality13. In addition, individuals that metamorphose at larger size may be more easily discovered by predators, thus Rollinson and Hutchings (2010)14 suggest that bigger is not always better. However, egg or hatching size may not affect mass at metamorphosis and survival because other ecological factors, such as temperature, density and competition, food level and quality, predation, or a combination of these, can alter the growth and developmental rate of each life stage of amphibians9,15,16,17,18.
The number of offspring produced, and their size, are fundamental life history traits because they are intimately linked with fitness and therefore population viability19. The trade-off between offspring number and size suggests that selection will produce more small eggs or abundant larvae under scarce resources and stressful environments, which is known as r-selection9. The advantage associated with producing smaller eggs is due to the corresponding increase in fecundity9. Conversely, selection will prefer to produce larger eggs or more robust larvae under abundant essential resources and good environmental conditions, which is regarded as K-selection5,20. However, amphibians respond to varying environments by changing egg size or a trade-off between clutch size and egg size18,21.
At higher altitudes, many species have relatively short breeding seasons and active cycles, resulting in slower growth and usually larger size than the same species from warmer areas16,22,23. To date, however, we have a very poor understanding of how maternal investment varies geographically in a high altitude species in response to selective pressure. Thus, we investigated altitudinal variations in Bufo minshanicus life history traits, focusing on a trade-off between egg size and clutch size. So far, the taxonomic status of B. minshanicus has been controversial for over half a century. Fu et al. (2005)24 suggested B. minshanicus and Bufo andrewsi were treated as a single species25, Bufo gargarizans, without subspecies division. However, Fei and Ye (2000)26 treated B. minshanicus as a subspecies of B. gargarizans. In this study, we defined B. minshanicus as a valid species. Bufo minshanicus is a species endemic to high altitude regions of the eastern Tibetan Plateau in China. They are widely distributed in forests, fields, and open alpine marshes from 2500 to 3700 meters (m) altitude and one of the few amphibian species to live at such high altitudes globally (Fei and Ye 2001). Geographically widespread species that occupy many thermal environments provide testable models for understanding the evolution of life-history responses to altitude. As a typical explosive breeder27, Bufo minshanicus exhibits a short breeding season (5–18 days). Females are larger than males28 and clutch size increases with increasing female SVL29,30. Low latitude populations in the eastern Qinghai province select deep swamps as hibernation site, while high latitude populations in the southern Gansu province prefer rabbit or otter holes as hibernating sites. Studies across altitudes provide a good approach to understand life-history evolution in stressful environments. Here, we predict that altitude would be (1) negatively correlated with clutch size, (2) positively associated with egg size, and (3) positively correlated with female SVL. We also tested the prediction that (4) there is a trade-off between egg size and clutch size.
Materials and methods
Data collection
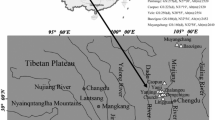
We collected Bufo minshanicus individuals from eight populations (all collected individuals from the same pond during the breeding period were defined as a population, at elevations ranging from 2506 to 3478 m) along the eastern Tibetan Plateau, China. At these sites, annual mean air temperatures decrease with increasing altitude or decreasing latitude, whereas annual total precipitation increases with increasing altitude or decreasing latitude (Table 1). The Gahai-Zecha National Nature Reserve Management Bureau approved this project (approval number GHZCRMB/03-212014), and gave permission for fieldwork. All animals were collected under the guidelines for animal care in China. Handling and processing of toads followed approved protocols from the Animal Scientific Procedures Act 1988 by the State Department of China. A total of 683 amplectant pairs were collected during the peak period of breeding activity from 2009 to 2015 (Table 1; Dataset 1). We transported them to our field lab located close to breeding sites. Each amplexed pair was put into a plastic container (2-liter) until the eggs were deposited. After counting eggs for each clutch and measuring SVL of both sexes to the nearest 1 mm using a ruler, the eggs and adults were returned to the breeding pond.
About 100 eggs were randomly selected from each clutch and placed on a glass plate with calipers to take a digital image. We obtained measurements of individual egg sizes (n = 20–30; ±0.01 mm) excluding the jelly using a tpsdig2 computer software. Clutch volume was calculated by multiplying the number of oviposited eggs with the volume of each egg (Vs = 4/3πr3, where r is the radius). We obtained average ambient temperatures and precipitation from 2009 to 2015 from the Chinese Meteorological Administration (http://www.cma.gov.cn) and the Gansu Gahai-Zecha National Nature Reserve Management Bureau.
Statistical analyses
Clutch size, egg size and clutch volume were analyzed using general linear mixed models (GLMMs) where population was treated as a random factor, and altitude and latitude as covariates. Egg size and clutch size were used as covariates of one another when testing for a trade-off between them. To investigate variation in body size of females, we used GLMMs where female SVL was the dependent variable, latitude and altitude as covariates, and population as a random factor.
In the clutch-by-female analyses, we treated clutch size, egg size and clutch volume as the dependent variable and female SVL, latitude and altitude as covariates. Finally, to investigate the trade-off between egg size and clutch size, we used egg size or clutch size as the dependent variable, the other as the independent variable, and then the other way around. Prior to analyses, we used log-transformed raw data on body size, clutch size, egg size and clutch volume. Meanwhile, we removed one population (3211 m) because only 3 female toads were collected. The analyses were started with a full model, with all possible interactions, and then removing non-significant interactions (P > 0.1, for details, see Räsänen et al. 200821) because interaction effects in many cases may be statistically weaker, but biologically important. Thus, in this case, it would be important to report the effect sizes rather than actual P values. For the R2 values, as we used GLMM with both fixed effects and random factors, it thus was needed to provide the R2 marginal (R2m) as the amount of variation explained by fixed factors, and R2 conditional (R2c) as amount of variation explained by both fixed and random factors. All the analyses were done with IBM SPSS Statistics 20.0 (IBM Corp, Armonk, NY, USA).
Results
Clutch data
Egg size decreased with increasing altitude (F1, 3.863 = 8.397, P = 0.046), but not latitude (F1, 3.879 = 5.357, P = 0.084, R2m = 0.17, R2c = 0.29, Fig. 1), providing weak evidence for correlation between egg sizes and altitudes. Clutch size increased with increasing altitude and latitude (altitude, F1, 3.879 = 10.035, P = 0.035; latitude, F1, 3.856 = 15.186, P = 0.019, R2m = 0.30, R2c = 0.41, Tables 1, 2, Fig. 1). Clutch volume increased with increasing latitude (F1, 3.620 = 14.910, P = 0.022,), but not altitude (F1, 3.702 = 3.636, P = 0.135, R2m = 0.26, R2c = 0.29).
There was a negative association between clutch size and egg size, indicating a population-level trade-off between fecundity and egg size (and vice versa; clutch size, b ± SE = −0.03062 ± 0.00816, F1, 723.428 = 14.070, P < 0.001, R2m = 0.22, R2c = 0.28; egg size, b ± SE = −0.61211 ± 0.16551, F1, 725.937 = 13.678, P < 0.001, R2m = 0.32, R2c = 0.41).
Clutch-by-female data
Female body size tended to increase with increasing latitude and altitude, but the difference was not significant (latitude, b ± SE = 0.03954 ± 0.01590, F1, 4.088 = 6.185, P = 0.066; altitude, b ± SE = 0.00007 ± 0.00004, F1, 4.006 = 2.242, P = 0.209, R2m = 0.35, R2c = 0.52, Table 1, Fig. 1).
GLMMs revealed that egg size, clutch size, and clutch volume were associated with female body size (egg size, F1, 665.984 = 4.349, P = 0.037; clutch size, F1, 316.444 = 284.267, P < 0.001; clutch volume, F1, 597.455 = 254.250, P < 0.001, Fig. 2). When removing the effect of female size, egg size decreased significantly with increasing altitude (F1, 3.913 = 9.586, P = 0.037), but not latitude (F1, 4.138 = 4.618, P = 0.096, R2m = 0.20, R2c = 0.30). Clutch volume did not significantly vary with altitude (F1, 1.428 = 0.146, P = 0.752) or latitude (F1, 1.526 = 0.781, P = 0.494, R2m = 0.47, R2c = 0.49), but clutch size increased significantly with increasing altitude (F1, 3.258 = 12.753 P = 0.033) and latitude (F1, 3.247 = 15.689, P = 0.025). The interaction between altitude and latitude was significant (F1, 3.292 = 11.899, P = 0.035, R2m = 0.53, R2c = 0.55, Table 3, Fig. 2) suggesting that clutch size varied inversely with altitude at higher latitudes.
Relationship between altitude or latitude and egg size (a), clutch size (b) and total clutch volume (c) in 7 Bufo minshanicus populations. Solid lines: P < 0.05; dashed line: P > 0.05. Data points are population means. Relative log10 was generated from regression of log10 (egg size) or log10 (clutch size) or log10 (total clutch volume) on log10 (female body size). Statistical details are as in Table 3.
Clutch size was negatively correlated with egg size when accounting for female size within each population, but the correlations for only two populations were significant (both r > −0.24, P < 0.003, Table 1). The clutch-by-female data further showed an overall significant negative relationship between egg and clutch size (and vice versa; clutch size, b ± SE = −0.05014 ± 0.00979, F1, 667.605 = 26.223, P < 0.001, R2m = 0.24, R2c = 0.32; egg size, b ± SE = −0.75571 ± 0.14644, F1, 659.540 = 26.633, P < 0.001, R2m = 0.56, R2c = 0.57), indicative of a trade-off between egg size and clutch size. The slopes of egg size decreased with increasing altitude and latitude, but the difference was not significant (altitude × egg size, F 1, 661.082 = 1.677, P = 0.196; latitude × egg size, F 1, 666.610 = 1.368, P = 0.243).
Discussion
In order to maximize fitness, life history traits vary under different environmental pressures31,32. In this study, our results showed that maternal investment strategies differ among altitudes. When the effect of body size is removed, the clutch data across seven B. minshanicus populations indicated that females are more fecund but produce smaller egg sizes at higher altitudes than at lower altitudes. The clutch-by-female size further showed a population-level trade-off between fecundity and egg size.
However, if the outlier population (at 3211 m) was not removed, the clutch data across eight B. minshanicus populations indicated that egg size and clutch volume tend to decrease or increase with increasing altitude (F1, 4.409 = 4.19, P = 0.104) or latitude (F1, 0.926 = 8.47, P = 0.218), while female SVL increased with increasing latitude (F1, 4.784 = 7.84, P = 0.040). Since those results were not consistent among populations in this study, additional research may be needed with more intensive sampling efforts along more geographical gradients.
In this study, female SVL was not significantly positive correlated with latitude or altitude. This result was not consistent with previous evidence considering six populations28. The shorter seasonal period of activity, weaker digestion and lower energy of prey may limit larger body size for appearing at high altitudes or latitudes. Moreover, females living in higher-altitudes must store enough energy during the short activity period in order to survive the harsh and extended winter, which may become more difficult for larger individuals. Therefore, Chen et al. (2013)4 suggested that larger females at high altitudes may be costly.
Life history theory suggests that organisms should allocate the limited energy between growth and development, thus creating a trade-off between growth and development1. The clutch data across seven B. minshanicus populations revealed that clutch attributes (clutch size, egg size, and clutch volume) were positively correlated with female size, revealing that larger females produce more eggs, larger eggs and increase reproductive effort. In fact, most amphibians and reptiles, in general, exhibit this relationship between body size and reproductive output. Thus, it is attributed to positive fecundity selection, where female body size is larger for larger clutches33. Like most ectotherms with indeterminate growth, female B. minshanicus fit the von Bertalanffy’s model28, which describes rapid somatic growth for earlier stages followed by slower growth thereafter. This implies that a larger fraction of energy would be devoted to reproduction as individuals become older34,35, thus resulting in age-specific reproductive output.
In some anurans, larger clutches in high-altitude or latitude populations are due to the larger size of high-altitude or latitude females16,22,36,37. When removing the effect of female body size, females at high altitudes produce relatively fewer clutches, but larger eggs than conspecifics from low altitudes5,16,23,38. However, in this study, after controlling for the effect of female size, higher altitude females had higher fecundity and smaller egg sizes with similar reproductive effort than lower altitudes females, thus the endemic plateau toad has evolved a different breeding strategy. This result was consistent with a previous study39, which showed that the phrynosomatid lizard Phrynosoma cornutum had smaller females at higher latitudes that produced larger clutches but similarly sized eggs when compared to larger females at lower latitudes and removing the effect of female size. It is well known that rainfall can directly affect the abundance of food, and may indirectly affect fecundity by reducing fat stores, which is necessary for development of a holistic egg number40. Females may have more resources for reproduction and clutch production if food availability is higher and physiological stress is lower. Interestingly, in this study, rainfall increases with increasing altitude, indicating higher food availability at higher altitudes. In this case, we speculated that resource may not limit clutch production at higher altitudes. Meanwhile, this species selects rabbit or otter holes as hiding and hibernating sites in the field. In this case, they reduce chances of predation and protect against extreme temperature fluctuations, especially in summer and winter. Additionally, disease and predation tend to decline when increasing altitude41. Thus, we speculate that this species does not allocate as much energy to survive the harsh and extended winter, and suggest females living at high altitudes likely allocate more energy for producing clutches. Similarly, Dobzhansky (1950)42 suggested that r-selection strategies were more likely to appear in changing environments (e.g. temperate and high altitude regions), whereas K-selection more likely to be found in relatively stable situations (e.g. tropics). As a consequence, larger clutch size is regarded as an attribute that populations have evolved through time as an adaptation to their general environment and their ecological niche43,44.
Life-history theory emphasizes that high-altitude females are expected to select for the production larger eggs2,3,4,5,6 because larger eggs or offspring will lead to larger initial sizes, faster growth speeds, higher developmental speeds, or both7,8,9,10,11,12. However, our results showed egg size across seven B. minshanicus populations decreased with increasing altitude, which provide support for the notion that life-history strategies respond to environmental conditions. Similarly, in some instances smaller eggs are better45 or some studies report no significant difference between egg size and altitude46,47. Other studies found that some species with small eggs will increase the number of offspring48, especially in the uncertain larval environment. Additionally, small eggs have a relatively large surface-to-volume ratio and require relatively little oxygen during embryonic development49. In this case, this animal may better be able to adapt to the anoxic environment of the Tibetan Plateau.
Although some studies suggest that trade-offs between clutch size and egg size in amphibians seldom occurs4,50,51,52,53, some previous findings5,9,54,55 support the existence of a trade-off. In this study, although intra-population-level trade-off between fecundity and egg size was uncommon, there was a significant inter-population-level trade-off between fecundity and egg size, suggesting that females producing greater numbers will produce smaller eggs to compensate for the energy put into the greater numbers, which are consistent with the general principle of MacArthur and Wilson’s theory of r-versus K-selection in populations56,57. Moreover, higher altitudes influenced investment in egg size, as suggested by the fact that there were stronger negative effects on egg size than on clutch size and reproductive output. Our data also showed that the strength of the trade-off between clutch size and egg size was not different among altitudes and latitudes. However, theoretical53,58 and empirical9,21,54 studies showed that harsh environments promote larger maternal investment in per-individual offspring to improve survival of individual offspring. For B. minshanicus, at higher altitudes, the advantage associated with producing smaller eggs because of increasing fecundity, which potentially maximizes the number of offspring surviving to reproduction10. Therefore, different environmental pressures will lead to geographical variation in life history traits aimed at maximizing fitness31,32. Bufo minshanicus have evolved a different life history strategy to adapt their environments, which may be inconsistent with our expectations. On the Tibetan Plateau, it appears as though more fecundity and smaller egg size are traits that enhance fitness for this animal.
References
Roff, D. A. Life-history evolution. Sunderland, Sinauer Associates (2002).
McGinley, M. A., Temme, D. H. & Geber, M. A. Parental investment in offspring in variable environments: theoretical and empirical considerations. Am. Nat. 130, 370–398 (1987).
Einum, S. & Fleming, I. A. Maternal effects of egg size in brown trout (Salmo trutta): norms of reaction to environmental quality. P. Roy. Soc. B-Biol. Sci. 266, 2095–2100 (1999).
Chen, W., Tang, Z. H., Fan, X. G., Wang, Y. & Pike, D. A. Maternal investment increases with altitude in a frog on the Tibetan Plateau. J. Evol. Biol. 26, 2710–2715 (2013).
Liao, W. B., Lu, X. & Jehle, R. Altitudinal variation in maternal investment and trade-offs between egg size and clutch size in the Andrew’s toad. J. Zool. 293, 84–91 (2014).
Liao, W. B. & Lu, X. A comparison of reproductive output of the Omei Treefrog (Rhacophorus omeimontis) between high and low elevations. Anim. Biol. 61, 263–276 (2011).
Kuramoto, M. Correlations of quantitative parameters of fecundity in amphibians. Evolution 32, 287–296 (1978).
Crump, M. L. Intraclutch egg size variability in Hyla crucifer (Anura: Hylidae). Copeia 1984, 302–308 (1984).
Roff, D. A. The evolution of life histories. Chapman and Hall, New York, New York, USA (1992).
Stearns, S. C. The evolution of life histories. Oxford University Press, Oxford, UK (1992).
Kaplan, R. H. & King, E. G. Egg size is a developmentally plastic trait: evidence from long term studies in the frog Bombina orientalis. Herpetologica 53, 149–165 (1997).
Einum, S. & Fleming, I. A. Highly fecund mothers sacrifice offspring survival to maximise fitness. Nature 405, 565–567 (2000).
Nussbaum, R. A. Parental care and egg size in salamanders: an examination of the safe harbor hypothesis. Res. Popul. Ecol. 29, 27–44 (1987).
Rollinson, N. & Hutchings, J. A. Why does egg size increase with maternal size? Effects of egg size and egg density on offspring phenotypes in Atlantic salmon (Salmo salar). Evol. Ecol. Res. 12, 949–960 (2010).
Kaplan, R. H. The implications of ovum size variability for offspring fitness and clutch size within several populations of salamanders (Ambystoma). Evolution 34, 51–64 (1980).
Berven, K. A. The genetic basis of altitudinal variation in the wood frog Rana sylvatica. I. An experimental analysis of life history traits. Evolution 36, 962–983 (1982).
Licht, L. E. Comparative life history features of the western spotted frog, Rana pretiosa, from low- and high- elevation populations. Can. J. Zool. 53, 1254–1258 (1975).
Berven, K. A. & Chadra, B. G. The relationship among egg size, density and food level on larval development in the wood frog (Rana sylvatica). Oecologia 75, 67–72 (1988).
Lardies, M. A., Carter, M. J. & Bozinovic, F. Dietary effects on life history traits in a terrestrial isopod: the importance of evaluating maternal effects and trade-offs. Oecologia 138, 387–395 (2004).
Kaplan, R. H. & Phillips, P. C. Ecological and developmental context of natural selection: maternal effects and thermally induced plasticity in the frog Bombina orientalis. Evolution 60, 142–156 (2006).
Räsänen, K., Söderman, F., Laurila, A. & Merilä, J. Geographic variation in maternal investment: acidity affects egg size and fecundity in Rana arvalis. Ecology 89, 2553–2562 (2008).
Pettus, D. & Angleton, G. M. Comparative reproductive biology of montane and piedmont chorus frogs. Evolution 21, 500–507 (1967).
Kozlowska, M. Differences in the reproductive biology of mountain and lowland common frogs Rana temporaria L. Acta Biol. Cracov.Zool. 14, 17–32 (1971).
Fu, J. et al. Phylogeographic analysis of the Bufo gargarizans species complex: a revisit. Mol. Phylogenet. Evol. 37, 202–213 (2005).
Frost, D. R. Amphibian Species of the World: an Online Reference. Version 6.0 (accessed on June 8, 2015). Electronic Database accessible at, http://research.amnh.org/herpetology/amphibia/index.html. American Museum of Natural History, New York, USA (2015).
Fei, L. & Ye, C. The Colour Handbook of the Amphibians of Sichuan. Chinese Forestry Press, Beijing (in Chinese) (2000).
Wells, K. D. The Ecology and Behavior of Amphibians. University of Chicago Press (2007).
Yu, T. L., Wang, D. L., Busam, M. & Yao, H. D. Altitudinal variation in body size in Bufo minshanicus supports Bergmann’s rule. Evol. Ecol. 33, 449–460 (2019).
Yu, T. L. & Lu, X. Lack of male mate choice in the Minshan’s toad (Bufo gargarizans minshanicus). North-West. J, Zool. 9, 1211–126 (2013).
Yu, T. L., Dang, Q. K. & Chen, J. B. Bufo gargarizans minshanicus males exhibit size-dependent mate choice but lack sex recognition: an experimental approach. Herpetol. J. 24, 197–199 (2014).
Lüddecke, H. Variation and trade-off in reproductive output of the Andean frog Hyla labialis. Oecologia 130, 403–410 (2002).
Morrison, C. & Hero, J. M. Geographic variation in life-history characteristics of amphibians: a review. J. Anim. Ecol. 72, 270–279 (2003).
Pincheira-Donoso, D. & Hunt, J. Fecundity selection theory: concepts and evidence. Biol. Rev. 92, 341–356 (2017).
Jørgensen, C. B. Growth and reproduction, In Environmental physiology of the amphibians: 439–467. Feder, M. E. & Burggren, W. W. (Eds). Chicago: University Chicago Press (1992).
Czarnoleski, M. & Kozlowski, J. Do Bertalanffy’s growth curves result from optimal resource allocation. Ecol. Lett. 1, 5–7 (1998).
Cummins, P. Temporal and spatial variation in egg size and fecundity in Rana temporaria. J. Anim. Ecol. 55, 303–316 (1986).
Tilley, S. G. Life histories and comparative demography of two salamander populations. Copeia 1980, 806–821 (1980).
Liao, W. B., Luo, Y., Lou, S. L., Lu, D. & Jehle, R. Geographic variation in life-history traits: growth season affects age structure, egg size and clutch size in Andrew’s toad (Bufo andrewsi). Front. Zool. 13, 6 (2016).
Hughes, D. F., Meshaka, W. E. Jr, Lieb, C. S. & Pechmann, J. H. Latitudinal variation in life history reveals a reproductive advantage in the Texas Horned Lizard (Phrynosoma cornutum). Copeia 107, 736–747 (2019).
Heatwole, H. & Taylor, J. Ecology of Reptiles. Surrey Beatty and Sons Pty Ltd, Chipping Norton (1987).
Wang, Y. J., Ji, W. H., Zhao, W., Yu, N. N. & Liu, N. F. Geographic variation in clutch and egg size for the lizard Phrynocephalus przewalskii (Squamata: Agamidae). Asian Herpetol. Res. 2, 97–102 (2011).
Dobzhansky, T. Evolution in the tropics. Am. Sci. 38, 208–221 (1950).
Crump, M. L. Reproductive strategies in a tropical anuran community. University of Kansas Museum of Natural History Miscellaneous Publications 61, 1–68 (1974).
Fitch, H. S. Variation in clutch and litter sizes in New World repltiles. University of Kansas Museum of Natural History Miscellaneous Publications 7, 61–70 (1985).
Parichy, D. M. & Kaplan, R. H. Maternal investment and developmental plasticity: functional consequences for locomotor performance on hatchling frog larvae. Funct. Ecol. 9, 606–617 (1995).
Ryser, J. Comparative life histories of a low- and a high-elevation population of the common frog Rana temporaria. Amphibia–Reptilia 17, 183–195 (1996).
Morrison, C. & Hero, J. M. Geographic variation in life history characteristics of amphibians in mid-eastern Australia: reproductive traits. Proceedings of the Frogs in the Community Symposium (ed. A. Nattrass), pp. 52–61. Environment Australia, Canberra (2002).
Kuramoto, M. Embryonic temperature adaptation in development rate of frogs. Physiol. Zool. 48, 360–366 (1975).
Einum, S., Hendry, A. P. & Fleming, I. A. Egg-size evolution in aquatic environments: does oxygen availability constrain size? P. Roy. Soc. London B 269, 2325–2330 (2002).
Tejado, M. Absence of the trade-off between the size and number of offspring in the natterjack toad (Bufo calamita). Oecologia 90, 294–296 (1992).
Laugen, A., Laurila, A. & Merila, J. Maternal and genetic contributions to geographical variation in Rana temporaria larval life-history traits. Biol. J. Linn. Soc. 76, 61–70 (2002).
Castellano, S., Cucco, M. & Giacoma, C. Reproductive investment of female green toads (Bufo viridis). Copeia 2000, 659–664 (2004).
Dziminski, M. A. & Alford, R. A. Patterns and fitness consequences of intra clutch variation in egg provisioning intropical Australian frogs. Oecologia 146, 98–109 (2005).
Lloyd, D. G. Selection of offspring size at independence and other size-versus-number strategies. Am. Nat. 129, 800–817 (1987).
Räsänen, K., Laurila, A. & Merilä, J. Maternal investment in egg size: environment- and population-specific effects on offspring performance. Oecologia 142, 546–553 (2005).
MacArthur, R. H. & Wilson, E. O. The Theory of Island Biogeography. Princeton University Press, Princeton, NJ (1967).
Pianka, E. R. On r- and K-Selection. Am. Nat. 104, 592–597 (1970).
Smith, C. C. & Fretwell, S. D. The optimal balance between size and number of offspring. Am. Nat. 108, 499–506 (1974).
Acknowledgements
We are very grateful to the staff of the Gahai-Zecha National Nature Reserve for assistance in the field,as well as to Denita M. Weeks and David Pike for checking the language of this manuscript. The study was funded by the Emergency Management Program of National Natural Science Foundation of China, Grant/Award Number: 31741019; Natural Science Foundation of Henan Province of China, Grant/Award Number: 182300410019; Foundation for University Key Teacher of Henan province, Grant/Award Number: 2016GGJS-098 and Program for Innovative Research Team (in Science and Technology) in universities of Henan Province, Grant/Award Number: 17IRTSTHN019. The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Contributions
T.L.Y. conceived the ideas and designed methodology; T.L.Y. and Y.H.D. collected the data; T.L.Y. analyzed the data; T.L.Y. wrote the manuscript. All authors read and approved the final manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, T.L., Deng, Y.H. Geographic variation in maternal investment and trade-offs between egg size and clutch size in an endemic toad of the Qinghai-Tibet Plateau. Sci Rep 10, 6838 (2020). https://doi.org/10.1038/s41598-020-63635-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-63635-y
- Springer Nature Limited