Abstract
Parasites alter many traits of their hosts. In particular, parasites known as “manipulative” may increase their probability of transmission by inducing phenotypic alterations in their intermediate hosts. Although parasitic-induced alterations can modify species’ ecological roles, the proximate factors modulating this phenomenon remain poorly known. As temperature is known to affect host–parasite associations, understanding its precise impact has become a major challenge in a context of global warming. Gammarids are ecologically important freshwater crustaceans and serve as intermediate hosts for several acanthocephalan species. These parasites induce multiple effects on gammarids, including alterations of their behavior, ultimately leading to modifications in their functional role. Here, experimental infections were used to assess the effect of two temperatures on several traits of the association between Gammarus pulex and its acanthocephalan parasite Pomphorhynchus laevis. Elevated temperature affected hosts and parasites in multiple ways (decreased host survival, increased gammarids activity, faster parasites development and proboscis eversion). However, behavioral manipulation was unaffected by temperature. These results suggest that predicted change in temperature may have little consequences on the trophic transmission of parasites through changes in manipulation, although it may modify it through increased infection success and faster parasites development.
Similar content being viewed by others
Introduction
Abiotic conditions can strongly influence interspecific interactions and, ultimately, the dynamics of ecological communities. In direct connection with climate change, the understanding of such effect has become a major challenge in recent years1,2,3. Abiotic conditions, including temperature, can, for instance, affect predator–prey interactions and food-web dynamics4,5,6 or competition between species7,8,9,10,11. They can also directly or indirectly affect host–parasite interactions12,13,14, with cascading effects for trophic interactions and ecosystem stability15,16,17. This is all the more relevant in the case of parasite species that infest and modify the phenotype of host species known as ‘ecosystem engineers’, i.e. species that can affect the physical properties of ecosystems18,19,20.
For instance, some recent evidence suggests that changes in temperature can modulate the influence of parasitic infection on both the functional role and the coexistence of crustacean amphipod species17,21,22,23. Crustacean amphipods are widespread throughout a large range of freshwater habitats24,25, in which they play a key ecological role. They represent an important food resource for many species26,27 and are themselves a major predator24,28, capable of modulating the composition of freshwater macroinvertebrates communities28,29. Some species can also directly influence water quality and the recycling of organic matter through their shredder role on dead leaves25,30,31,32. Crustacean amphipods serve as intermediate host for a large variety of helminths with complex life-cycles33. In particular, the association between amphipods and acanthocephalan parasites (thorny-headed worms) has received considerable attention34,35. Acanthocephalans can affect the phenotype of their amphipod hosts in many different ways, through altering their behavior36,37,38, their feeding and predatory activity23,39,40, their immune system41, their energetic reserves42, their fecundity43,44, or their metabolic rate45,46, ultimately leading to modifications in the role of amphipods within ecosystems. In particular, amphipods infected with acanthocephalans tend to be more vulnerable to predation, especially by final hosts of the parasites47,48,49,50,51.
As for other ectotherm species52, temperature has been shown to affect amphipods in several ways, influencing for instance their metabolism53,54,55, growth56, or activity54. Similarly, most parasites are also affected by temperature in diverse ways57. For instance, temperature affects both the development time of acanthocephalan parasites in their intermediate hosts58,59 and their prevalence and abundance in their definitive fish hosts60. This is reflected in field studies having reported seasonal effects in the prevalence and intensity of infection with acanthocephalans in both intermediate and definitive hosts61,62,63,64,65. So far, however, only a limited number of studies have provided evidence for a joint effect of temperature and infection with acanthocephalan parasites on the ecological role of their intermediate host species. For instance, Labaude et al.23 showed that infection with acanthocephalan parasites and temperature have additive effects on the shredding role of crustacean amphipods, whereas Guinnee and Moore66 reported an interaction between temperature and infection with acanthocephalans on the fecundity of insect hosts.
The influence of increased temperature on the phenotypic alterations induced by acanthocephalans on their intermediate hosts has received little attention despite their potential consequences at the ecosystem level. Although the exposition of naturally-infected isopods to different conditions of light and temperature resulted in changes in behavior, no differences were found in the extent of manipulation67. However, isopods used in this study were naturally-infected, such that conditions during parasite development were not controlled. In addition, the effects of temperature and light were not investigated separately, although previous studies showed that the manipulation of gammarids by acanthocephalans depends to some extent on light properties68,69. Temperature was shown to affect significantly the alteration of phototaxis induced by natural infections of the acanthocephalan Pomphorhynchus tereticollis in Gammarus pulex22. However, there was no evidence for an effect of temperature on the use of refuge by infected vs. uninfected gammarids, thus suggesting that temperature may have different effects on different dimensions of manipulation22. Additional studies in which both temperature and parasite infection are controlled experimentally would thus help to better understand how temperature affects host manipulation by acanthocephalan parasites.
Following experimental infections, we investigated the effect of two temperatures on the magnitude of phenotypic alterations induced by P. laevis on G. pulex, including the intensity and timing of manipulation. After parasites reached the cystacanth stage, at which they become infective for the definitive host, the use of refuge by gammarids (a behavior known to be directly involved in parasite trophic transmission47,49) was measured. Other parameters of both hosts and parasites (i.e. host survival and activity level, rapidity of parasites’ proboscis eversion), as well as infection parameters (i.e. infection success, parasite load, speed of parasites development), were also recorded.
Methods
Sampling
Uninfected G. pulex gammarids were collected in a small tributary of the Suzon River (eastern France, 47° 24′ 12.6″ N, 4° 52′ 58.2″ E) in October. Gammarids from this population have been widely used in previous studies for experimental infections, such that the system is now well characterized45,70,71,72,73,74. Previous studies did not show any effect of the sex of gammarids on the extent of behavioral modifications37,73,75,76. However, failure in parasite infection was observed in female gammarids more often than in males73. Therefore, only males were used in this study. Before experimental infections, gammarids were maintained in the laboratory at 15 °C and under a 12:12 light:dark cycle.
The Vouge River (eastern France, 47° 9′ 34.36″ N 5° 9′ 2.50″ E) was chosen to collect parasites from naturally infected chubs (Leuciscus cephalus) in October, as this population of parasites has previously been shown to be highly infective to gammarids in the lab74. We extracted acanthocephalan eggs from adult parasites sampled in the intestines of the fish. Because both P. laevis and P. tereticollis parasites can be found in fish from this river and cannot be distinguished visually, the species of adult parasites was determined using genetic analyses with the method described in Franceschi et al.73. Only eggs from P. laevis were used for experimental infections.
A second sampling of gammarids and fish was conducted in November and December for control experiments (see below).
Experimental infections and treatments
Gammarids were experimentally exposed to P. laevis eggs following the procedure detailed in Franceschi et al.73. Pairs of gammarids that were previously starved for 24 h in glass dishes were exposed for 48 h to 200 parasite eggs (100 eggs per gammarid being a good compromise between a high infection success and a low rate of multiple infections73). Gammarids were then placed in individual glass dishes, and randomly distributed among the different treatments. Control individuals were maintained under the same conditions, without parasite eggs.
To investigate the effect of temperature during the development of parasites, 1,200 gammarids were exposed to parasite eggs, and 420 individuals were maintained as control. Immediately following the exposure, gammarids were allocated to two temperature treatments: normal temperature of 14 °C—which is naturally experienced by gammarids in the field during summer—and increased temperature—corresponding to an addition of 3 °C consistent with scenarios of climate change77,78,79 (Fig. 1).
Because the development time of parasites is highly dependent on temperature58,59, parasites were expected to reach the cystacanth stage later at 14 °C compared to 17 °C. Consequently, gammarids at 14 °C would spend more time in the laboratory before being tested. Therefore, a second experimental infection was conducted to control for the effect of time spent by gammarids in the laboratory (Fig. 1). Additional gammarids from the first sampling (October) were maintained for 40 days at 17 °C before being exposed to parasite eggs sampled in December (n = 650), or kept as controls (n = 120). In parallel, 90 freshly sampled gammarids (second sampling, November) were experimentally infected with the same parasite eggs, and 30 individuals were used as controls. Following the second infection, all gammarids were maintained in individual glass dishes at 17 °C (see Fig. 1).
All individuals were maintained under a 12:12 light:dark cycle, and were fed ad libitum with conditioned elm leaves, plus one additional frozen chironomid larvae once every 2 weeks, ensuring improved survival of gammarids45. Water was changed once every 2 weeks, using an oxygenated mix of water from the Suzon River and dechlorinated, UV-treated tap water.
Monitoring
All gammarids were inspected on a daily basis to record the death of any individual. Individuals that were exposed to parasites were dissected immediately after their death to determine their infection status (number of parasites and their development stage). In addition, 150 control and 150 gammarids exposed to parasite eggs were randomly selected at each temperature at the beginning of the experiment. Individuals found dead among them were then measured from body height at the level of the fourth coxal plate80 using a microscope and Lucia G 4.81 software (Prague, Czech Republic).
Following detection of the first parasites at the advanced acanthella stage (> 1,000 µm) upon dissection, all individuals were checked daily under a binocular microscope to monitor the exact date of the switch between the acanthella stage (ovoid shape, translucent orange color) and the cystacanth stage (spherical and more pronounced opaque color81). Behavioral tests were then conducted (see below).
At the end of the experiments, we measured and dissected all gammarids that were used in behavioral tests, including control individuals. Although the presence of P. laevis has not been recorded in the Suzon River, gammarids from this population can be naturally infected with other acanthocephalan parasite species (Echinorhynchus truttae and Polymorphus minutus), as well as other macro-parasites such as Cyathocephalus truncatus (Cestoda). Such infected individuals were removed from the data set.
Measurement of refuge use
The use of refuges by gammarids was recorded on three consecutive occasions (hereafter referred as “rounds”) for all infected individuals: 1 day, 8 days, and 16 days after the cystacanth stage was detected. We tested control individuals in a similar fashion in parallel. Gammarids were placed in individual boxes (10.5 × 16 cm) filled with 250 ml of water, containing a refuge that consisted of a saucer terracotta pot (8.5 cm of diameter) cut in half, with a 1 cm hole in the convex part (see Dianne et al.82). After 10 min of acclimatization following the introduction of gammarids, we recorded the position of each individual every 2 min for 60 min. For each observation, a score of zero was given to individuals that were outside of the refuge, and one for individuals inside it. Summed scores at the end of each round thus ranged from 0 (always outside the refuge) to 30 (always inside) for each individual.
As expected, the development of parasites in gammarids maintained at 14 °C was longer than that of gammarids maintained at 17 °C. We therefore tested whether the timing and intensity of manipulation depended on the absolute time that parasites spent into their hosts rather than on their development stage, as well as the effect of the time spent by gammarids in the laboratory (both tests and their results are detailed in the Supplementary material 1).
All gammarids were tested at their acclimatization temperature. In order to be able to perform tests at different temperatures in one single room, test boxes were placed in water baths, with surrounding water constantly recirculated through a temperature control device (Tank TK-1000 Chiller, Teco, Ravenna, Italy; see Labaude et al.23).
Measurement of activity
The activity level of individual gammarids was tested 3 days after the second round of refuge use tests (i.e. 11 days after the detection of the cystacanth stage for infected individuals). The apparatus consisted of a 10-cm diameter glass dish containing a smaller dish (6-cm diameter) preventing the gammarid to go in the centre of the larger glass dish, thus forming a 2-cm wide annulus. To limit vertical movements of the individual, the apparatus was filled with only 1 cm of water. Lines were traced under the glass dish, intersecting in their centre, thus dividing the annulus into eight equally large zones. After 5 min of acclimatization following the introduction of the gammarid in the device, the behavior of the individual was video-recorded from above for 5 min. The activity level of the individual was expressed as the number of lines that it crossed during 5 min.
Rapidity of proboscis eversion
The rapidity of cystacanth parasites to evert their proboscis was measured in old cystacanths (between 20 and 30 days after their detection) extracted from gammarids previously tested for their behavior. The eversion of the proboscis, which allows parasites to attach to the intestine wall of their fish host, is a crucial step for a parasite to complete its life cycle and incurs a physiological cost as an active process83,84. Thus, latency to evert the proboscis can be used as a proxy to estimate parasite stamina85. Immediately following the dissection of their hosts, each cystacanth parasite was carefully placed in a 96-well microplate. The eversion of cystacanth proboscis is known to occur in reaction to a component of fish bile86. Therefore, 30 µl of bile extracted several months earlier from European chubs, Squalius cephalus, frozen for conservation since then, and diluted 30 times with water, were added to each microplate well. The microplate was immediately covered with aluminium foil to limit evaporation, and with an opaque box to ensure darkness (see Perrot-Minnot et al.85). Each microplate, containing no more than 20 cystacanths, was then checked every 5 min under a dissecting microscope, with reduced light, and quickly replaced in the dark. We recorded the time needed for each parasite to start to evert their proboscis. The temperature was kept at 15 °C for all measurements of proboscis eversion, regardless of the treatment group.
Effect of acclimatization temperature on prevalence
In the experiments previously described, all gammarids were exposed to parasite eggs at the same temperature (15 °C), before being maintained at 14 °C or 17 °C. Therefore, we could not draw any conclusion about the effect of temperature on the success of parasite infection. Indeed, although temperature might affect the success of establishment of parasites, it is also known to modify the consumption rate of gammarids23,87, thus possibly affecting their probability of consuming parasite eggs and getting infected at different temperatures. Therefore, to measure the effect of temperature on the success of parasite infection, 100 gammarids from the second sampling (November) were acclimatized for 3 weeks at each of the two temperatures (14 °C or 17 °C), in individual glass dishes. We then exposed gammarids to parasite eggs collected in December, following the protocol described above, at their acclimatization temperature (Fig. 1). Conditions were kept similar during parasite development. All gammarids were dissected immediately after their death or at the end of the experiment to determine their infection status.
Statistical analyses
The survival of gammarids was analyzed using Cox regressions. First, the effects of infection status (control vs infected) and temperature were analyzed. A second Cox regression was performed using only infected individuals to investigate the effect of parasite load (one, two, or more than two parasites per gammarid) and temperature. Once parasites were large enough to be detected upon dissection, individuals that were exposed to parasite eggs in which no parasite developed were removed from the analyses.
We used nominal logistic regressions to investigate the success of infection (i.e. the proportion of gammarids harboring at least one parasite among those exposed to the infection), and Mann–Whitney U tests to compare the parasite load between the groups as well as the time needed for parasites to reach the cystacanth stage.
Scores for refuge use were analyzed as repeated measures using the ‘nparLD’ R software package. This function is suitable for nonparametric analyses of right-censured longitudinal data, allowing the decrease in sample size along time, due to individuals’ death88. First, the effects of temperature (14 °C vs 17 °C), infection status (control vs infected) and their interaction were investigated along time (rounds of measurements: 1 day, 8 days and 16 days after parasites reached the cystacanth stage). A second analysis was conducted on infected individuals only, with temperature, parasite load, their interaction, and rounds as factors. For each analysis, ‘ANOVA-type statistics’ were performed, followed by post-hoc pair-comparisons when suitable (see Noguchi et al.88 for details). Because parasites developed faster at high temperature, the size of gammarids that were killed following behavior tests was measured later at 14 °C compared to 17 °C. We therefore suspected that gammarids may have had more time to make an additional molt at 14 °C than at 17 °C, and thus could be larger. An ANOVA confirmed that the size of gammarids was globally larger at 14 °C (F399,1 = 5.84, P = 0.016). However, within each temperature, we found no general correlation between refuge scores and the size of gammarids, neither at 14 °C (Spearman correlation, rho = 0.064, P = 0.056) or at 17 °C (rho = 0.05, P = 0.21). This suggests that size does not influence this behavior. Therefore, to avoid confusion between size and temperature in our analyses, the size of individuals was not considered in between-temperatures analyses of refuge score.
The activity level of gammarids was investigated using a linear model (ANOVA), followed by Tukey post-hoc tests.
Latency time to evert the proboscis was investigated using a Generalized Linear Model with a Poisson distribution corrected for over-dispersion. We tested for the effects of temperature, parasite load, the time needed for each parasite to reach the cystacanth stage, and their interactions. Spearman correlations were used to test if there was a link between how fast parasites were everting their proboscis and their ability to manipulate the behavior of their hosts (scores of refuge use and activity level) at each temperature.
Statistical analyses were performed using JMP version 10.0.0 software (SAS Institute, Cary, NC, USA) and R version 3.1.1 software (R Foundation for Statistical Computing, Vienna, Austria). For each analysis described above, all factors and their second order interactions were first entered in the models. Non-significant factors or interactions were then removed.
Ethical statement
The animals were captured, transported and handled in accordance with article L.436-9 of the Environment Code, with the authorization of the Prefectural Decree of Côte-D'or no. 77 from the 11/02/2019. The fish for parasites samplings were killed after being anesthetized, in accordance with French regulations on the ethics of animal testing (decree 2013-118).
Results
Effect of temperature on survival
Cox regression on gammarids from the first exposure (Global model: Likelihood-Ratioχ2 = 208.97, df = 2, P < 0.0001) showed that survival was significantly higher at 14 °C compared to 17 °C (LR-χ2 = 188.56, df = 1, P < 0.0001, Fig. 2). Control individuals also survived significantly better than infected individuals (LR-χ2 = 34.88, df = 1, P < 0.0001, Fig. 2). The virulence of parasites did not differ between temperatures, as indicated by the absence of significant interaction between temperature and infection. When considering infected individuals only, no effect of parasite load on the survival of gammarids was found, and only temperature remained in the model (LR-χ2 = 144.09, df = 1, P < 0.0001).
Survival of gammarids according to infection status (control or infected by P. laevis parasites) and temperature (14 °C or 17 °C). Time 0 was considered as the day on which gammarids were exposed to parasite eggs. Arrows indicate the average day of switching of parasites between acanthella and cystacanth stages at each temperature.
Effect of temperature on infection parameters
Overall, 53.8% of individuals that were exposed to parasite eggs were successfully infected. When gammarids were exposed to eggs at the same temperature before being distributed among the two temperature treatments, there was no difference between the two temperatures in both the success of infection (LR-χ2 = 0.003, df = 1, P = 0.96) and the parasite load (mean ± standard deviation = 1.67 ± 1.04 parasites per gammarid, Mann–Whitney U test: Z = 1.06, P = 0.29). Development time of parasites (from exposure to cystacanth stage) was however significantly longer at 14 °C (mean ± standard deviation = 89.95 ± 2.49 days) than at 17 °C (57.27 ± 1.39 days; Mann–Whitney U test, Z = − 14.69, P < 0.0001; Fig. 2).
When gammarids were acclimatized for 3 weeks at the two temperatures before exposure to parasite eggs, infection success tended to be higher at 17 °C (84.9%; n = 86) than at 14 °C (74%; n = 77), although the difference was not significant (LR-χ2 = 2.97, df = 1, P = 0.08). More parasites developed in gammarids at 17 °C (mean ± standard deviation = 4.58 ± 2.76 parasites per gammarid) compared to 14 °C (mean ± standard deviation = 3.86 ± 2.55 parasites per gammarid), although this difference was only very close to significant (Mann–Whitney U test, Z = − 1.96, P = 0.05).
Effect of temperature on the behavior of gammarids
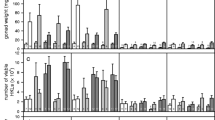
ANOVA-type results from the nparLD model showed that temperature did not influence the use of refuges by gammarids (Statistic = 0.63, df = 1, P = 0.43; Fig. 3), justifying the removal of temperature from the model. The remaining model showed that infection status, time (behavioral rounds), and the interaction between these two factors significantly influenced refuge use (Table 1). The use of refuges decreased with time for infected individuals, whereas it increased for control ones (Fig. 3).
Scores of refuge use by gammarids (high scores refer to more time inside the refuge) according to infection status (infected or control) and temperature (14 °C or 17 °C), measured during three rounds: 1 day (C + 1), 8 days (C + 8) and 16 days (C + 16) after detection of cystacanth stages. Scores range from 0 (individuals always outside the refuge) to 30 (individuals always inside the refuge). Thick lines represent the medians, boxes represent the upper and lower quartiles, and dotted lines represent the upper and lower deciles. Sample sizes are given above each plot.
When considering only infected individuals to investigate the effect of parasite load (one, two or more than two parasites), temperature had again no effect on the use of refuges (Statistic = 1.19, df = 1, P = 0.27) and was thus removed from the analysis. Although the remaining model confirmed that infected gammarids decreased their use of refuges over time, we found that parasite load and its interaction with time also significantly affected the use of refuges by gammarids (Table 2). Paired comparisons showed that, overall, refuge use was lower in gammarids infected with one or two parasites compared to gammarids infected with more than two parasites (Fig. 4, pair-comparisons 2 and 3 in Table 2). In addition, while all groups reached similar scores on the third round, their dynamics was different across time (this difference being significant only between gammarids harboring one and those harboring more than two parasites). In particular, the switch from refuge use to refuge avoidance happened later in gammarids with more than two worms (Fig. 4, pair-comparison 2 in Table 2).
Scores of refuge use by infected gammarids (high scores refer to more time inside the refuge) according to parasite load (one, two or more than two parasites per gammarid), measured during three rounds: 1 day (C + 1), 8 days (C + 8) and 16 days (C + 16) after the detection of cystacanth stages. Scores range from 0 (individuals always outside the refuge) to 30 (individuals always inside the refuge). Thick lines represent the medians, boxes represent the upper and lower quartiles, and dotted lines represent the upper and lower deciles. Sample sizes are given above each plot.
Temperature significantly affected the level of activity of gammarids (ANOVA F3,500 = 29.05, P < 0.0001). Individuals were significantly more active at 17 °C compared to 14 °C (F1,500 = 77.14, P < 0.0001, Fig. 5). Although infection status alone was not significant (F1,500 = 0.89, P = 0.35), its interaction with temperature also influenced gammarids activity level (F1,500 = 7.99, P = 0.005). Tukey’s HSD post hoc tests showed that the activity of infected individuals was significantly higher than that of control individuals at 14 °C (P = 0.0019), but not at 17 °C (P = 0.26; Fig. 5).
Activity level of gammarids according to infection status (infected by P. laevis cystacanths or control) and temperature (14 °C and 17 °C). Level of activity is given by a score corresponding to the number of zones entered during 5 min in an annulus-shaped arena. Mean values and 95% confidence intervals are indicated. Sample sizes are given above each bar. Significant differences are indicated by different letters (Tukey’s HSD post hoc tests; P < 0.05).
Effect of temperature on parasites stamina: latency time to evert the proboscis
The Generalized Linear Model (χ2 = 6.13, df = 2, P = 0.047) showed that proboscis eversion, considered as a proxy to estimate parasite stamina85, was slightly but significantly faster for parasites that developed in hosts maintained at 17 °C (median and interquartile range = 40 [35; 45] min) compared to hosts maintained at 14 °C (median and interquartile range = 45 [40; 50] min, LR-χ2 = 5.57, df = 1, P = 0.018). In addition, there was a negative relation between the time taken by parasites to reach the cystacanth stage and the latency time to evert their proboscis, with eversion starting sooner for parasites that took more time to develop (LR-χ2 = 4.97, df = 1, P = 0.026).
Spearman correlations showed that proboscis eversion was faster in parasites inducing a less intense manipulation in the third round of the refuge use test, although the correlation was significant only at 17 °C (Table 3). No other behavior of gammarids was correlated with the rapidity of proboscis eversion of their parasites (Table 3).
Discussion
Higher temperature resulted in decreased survival and increased activity in gammarids, as well as in faster cystacanth development and quicker proboscis eversion in parasites. An increased parasite load with increased temperature during exposition of amphipod hosts to parasite eggs was also observed. Despite all these effects, neither the timing nor the intensity of manipulation (as assessed by refuge use) was affected by temperature once the parasites had reached the cystacanth stage.
As expected based on previous studies58,59, the time of development of parasites was much longer (57%) at 14 °C compared to 17 °C, with a remarkable synchrony within temperatures, all parasites switching between the acanthella and cystacanth stages within a few days at each temperature. Because of this difference, gammarids at 14 °C spent more time in laboratory conditions than those at 17 °C before being tested for behavior. In addition, we conducted behavioral tests while parasites were at the same development stage, but not at the same age. However, control tests allowed us to discard these two potentially confounding parameters (the control tests and their results are detailed in the Supplementary material 1). First, all other parameters being equal, the amount of time spent by gammarids in the laboratory had no influence on infection dynamics, in terms of development time of parasites, infection success, parasite load and behavioral manipulation. Only activity was reduced in gammarids that spent more time under laboratory conditions. Second, gammarids tested at the same absolute parasite age (in days) showed differences in their behavior consistent with the idea that manipulation is linked to the parasite stage73, and not to the time that parasites did spend in gammarids. Indeed, gammarids infected with acanthella stages behaved significantly differently than gammarids infected with cystacanth stages of the same age.
In addition to the parasite’s development time, temperature also modified other infection parameters in our study. When exposure to parasite eggs occurred at different temperatures with hosts previously acclimatized at these temperatures, infection success was slightly higher at 17 °C compared to 14 °C and more parasites per host developed at 17 °C compared to 14 °C in these conditions, although these two differences were only marginally significant. These results agree with those found in Squalius cephalus, a definitive fish host of P. laevis in which a higher probability of infection and a higher parasite load was found at 22 °C compared to 18 °C60. The fact that a difference was observed only when exposure to parasite eggs occurred at different temperatures suggests that the effect of temperature on these two parameters was due to a higher consumption of eggs rather than a higher success of establishment of the parasites in their hosts. This hypothesis is supported by several studies that have reported a positive effect of temperature on food consumption by gammarids23,87,89, which may have affected their probability of consuming parasite eggs in the present study.
The latency of parasite to evert their proboscis, a parameter used as a proxy for parasite stamina85, was also affected by temperature, with parasites that developed at 17 °C starting the eversion sooner after the adding of fish bile compared to those which developed at 14 °C. During this test, the temperature was similar between the two groups, such that only the temperature experienced during parasite development could affect their proboscis eversion. This result, along with the faster development of parasites inside their hosts, may stem from an increased metabolism of parasites at 17 °C compared to 14 °C. The observed negative correlation between the rapidity of proboscis eversion and the magnitude of manipulation in infected gammarids from the first exposure suggests that the two phenomena might not be independent. However, the correlation was very weak and significant only at 17 °C for the third behavior round. Such negative correlation, if confirmed, could underlie a trade-off between parasite stamina and manipulation. Similarly, Maure et al.90 found a negative correlation between the fecundity of the parasitic wasp Dinocampus coccinellae and the length of the period of manipulation of its beetle host. Such correlations were not found in other studies45,70, although not directly tested, and the existence of such trade-offs thus needs to be further investigated.
In parallel to its effect on parasite traits, temperature also affected certain traits of the hosts. First, the survival of gammarids was decreased at high temperature, as already shown in other studies55,56, while infection with parasites also led to a higher mortality of gammarids. However, the influence of parasites on host survival was not affected by temperature.
Activity level, a parameter tightly linked to metabolism in gammarids54, was also affected by temperature. As expected, gammarids were globally more active at high temperature54,91. This difference could however be partly linked to the fact that gammarids kept at low temperature were tested after a longer time in laboratory conditions, as the second part of the experiment showed that a longer maintenance led to a decrease in the activity level of gammarids. In parallel, infection with acanthocephalan parasites has been shown to increase the activity of gammarids compared to that of uninfected ones in several studies92,93,94, although contradictory results were also reported72,95,96. Interestingly, our results suggest that abiotic conditions could be responsible for such contradictions. Indeed, infected individuals were significantly more active than control individuals only at 14 °C. On the contrary, although this difference was never significant, average activity level was slightly higher in control individuals compared to infected ones at 17 °C, in all our experimental infections.
Altogether, these results suggest that the metabolism of both hosts and parasites was accelerated at 17 °C compared to 14 °C. In contrast, no difference was observed according to temperature in the behavior of gammarids in terms of refuge use, neither in the timing of manipulation nor in its intensity. As already stated before, refuge use tended to increase with time for control individuals, while infected individuals decreased their use of refuges through time45,97. These two trends were observed at both temperatures, with the same progressive manipulation of infected individuals. Only the number of parasites per hosts was found to influence the use of refuges. Manipulation was delayed in gammarids harboring more than two parasites. However, this phenomenon might result from our protocol, that could not control for competition between parasites of different stages within the same host. Indeed, the behavior of individuals was tested as soon as one cystacanth was detected through gammarids cuticle. Variation in the growth of parasites sharing the same host has been shown70, and a small asynchrony is thus expected to occur in the exact day of switching to the cystacanth stage in gammarids with multiple acanthocephalan infection. It is thus likely that multi-infected gammarids still harbored acanthella parasites when they were first tested, or parasites at an earlier cystacanth stage in the second test. The manipulation of gammarids by parasites at the cystacanth stage is known to be reduced by the presence of acanthella parasites within the host97, a stage known to enhance the anti-predatory behaviors of their hosts rather than reducing it49, thus potentially explaining the delayed manipulation observed in our study.
The absence of any effect of temperature on manipulation, in terms of use of refuges, suggests that manipulation, at least on this dimension, might not be plastic, contrary to what has been previously proposed45. Indeed, as the survival of gammarids decreased at high temperature, we would expect ideally adapted parasites to adopt a strategy to increase their chances of being transmitted before the death of their hosts, such as a faster manipulation98. Interestingly, in a similar study investigating the effect of host nutritional condition, Labaude et al.45 also found that, although the survival of gammarids was reduced by a poor diet, along with effects on other metabolic traits, the amount of host resources had no effect on P. laevis manipulation of G. pulex.
Nevertheless, temperature might have affected parasite manipulation in a way that could not be detected in our study. First, tests occurred in the absence of any clue from predators. Although such conditions are sufficient to induce alterations in behavior, the differences between control and infected individuals might have been exacerbated by the presence of a predator odour38,47,99,100,101. Second, variations induced by temperature might be associated to other behaviors than the use of refuges. Indeed, the seasonal variation of manipulation observed by Franceschi et al.71 in gammarids infected with acanthocephalans was evidenced while assessing phototaxis, whereas the use of refuges was not tested. A recent study investigating the effects of a shorter acclimatization time at different temperatures on control and gammarids naturally infected by P. tereticollis showed a significant effect of temperature on the phototaxis of both infected and control gammarids, but not on their use of refuges22. By contrast, Benesh et al.67 showed that different experimental conditions of light and temperature, chosen to mimic seasonal differences, altered the use of refuges of both infected and uninfected isopods. However, the difference of behavior between infected and uninfected individuals remained similar under each experimental condition, although the two parameters were not investigated separately. On the contrary, they found that this difference varied among isopods collected at different seasons. A seasonal effect of manipulation was also documented in gammarids infected by acanthocephalan parasites71, although the mechanisms explaining such seasonality were not identified. Our study supports the hypothesis made by Benesh et al.67 who suggested that seasonal changes in manipulation might not be caused by proximal abiotic conditions. In other studies, only light properties were shown to affect manipulation68,69, while other factors such as the quantity of resources available did not affect manipulation either45. Thus, seasonality in manipulation could depend upon other parameters. We might expect such variation to rely on an internal clock with a genetic basis, although the mechanisms responsible for such timing need to be investigated. In this case, global change might alter the seasonal distribution and/or the diet of definitive fish hosts and ultimately lead to a maladaptation of the degree of manipulative efforts of parasites.
Although temperature did not plastically affect the manipulation of acanthocephalan parasites in our study, indirect effects are likely to occur102. Indeed, temperature was shown to be linked to leaf consumption by gammarids23,87,89, ultimately leading to higher infection success and parasite load. Such an effect was also observed in the definitive hosts of parasites60. In addition, parasites developed faster at high temperature. Other conditions, such as the availability of resources known to modulate parasite load in their gammarid hosts45, are also likely to be affected by temperature. Altogether, these effects might lead to modifications of the intensity of infection, known to influence manipulation73, as well as in the prevalence of acanthocephalan parasites in gammarid populations, thus, provided that prevalence is high enough, modifying behaviors at the population level.
To our knowledge, this study is the first to investigate the effect of temperature on the timing and intensity of behavioral changes using experimental infections. Our results provide solid evidence that temperature might affect many parameters of host–parasite associations, with no direct effect on the extent of certain important traits of behavioral manipulation. In addition to an increase of temperature as tested here, climate changes might also lead to an increase in the frequency of extreme climatic events, leading to more fluctuations in temperature regimes103. In particular, the effect of sudden increases of temperature at a higher temperature than the one tested in this study would be interesting to investigate. Finally, although temperature might not be directly responsible for changes in the behavior of gammarids, further studies are needed to investigate the generality of its effect in other host–parasite associations, before concluding about how its interaction with manipulative parasites might alter the functional role of gammarids within food webs. To that end, it would be worth considering other development stages of parasites, as well as the behavior of the definitive host, in more integrative experiments. The relevance of studies focusing on the impact of global changes on manipulative parasites has already been suggested102, and other host–parasite systems should also be investigated.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Deguines, N., Brashares, J. S. & Prugh, L. R. Precipitation alters interactions in a grassland ecological community. J. Anim. Ecol. 86, 262–272 (2017).
Bernabé, T. N. et al. Warming weakens facilitative interactions between decomposers and detritivores, and modifies freshwater ecosystem functioning. Glob. Change Biol. 24, 3170–3186 (2018).
Boulangeat, I., Svenning, J. C., Daufresne, T., Leblond, M. & Gravel, D. The transient response of ecosystems to climate change is amplified by trophic interactions. Oikos 127, 1822–1833 (2018).
Salt, J. L., Bulit, C., Zhang, W., Qi, H. & Montagnes, D. J. S. Spatial extinction or persistence: Landscape–temperature interactions perturb predator–prey dynamics. Ecography (Cop.) 40, 1177–1186 (2017).
Zhang, L., Takahashi, D., Hartvig, M. & Andersen, K. H. Food-web dynamics under climate change. Proc. R. Soc. B 284, 20171772 (2017).
Campanati, C., Dupont, S., Williams, G. A. & Thiyagarajan, V. Differential sensitivity of larvae to ocean acidification in two interacting mollusc species. Mar. Environ. Res. 141, 66–74 (2018).
Woehler, E., Patterson, T. A., Bravington, M. V., Hobday, A. J. & Chambers, L. E. Climate and competition in abundance trends in native and invasive Tasmanian gulls. Mar. Ecol. Prog. Ser. 511, 249–263 (2014).
Frizzi, F., Bartalesi, V. & Santini, G. Combined effects of temperature and interspecific competition on the mortality of the invasive garden ant, Lasius neglectus: A laboratory study. J. Therm. Biol. 65, 76–81 (2017).
Grainger, T. N., Rego, A. I. & Gilbert, B. Temperature-dependent species interactions shape priority effects and the persistence of unequal competitors. Am. Nat. 191, 197–209 (2018).
Friesen, O. C., Poulin, R. & Lagrue, C. Parasite-mediated microhabitat segregation between congeneric hosts. Biol. Lett. 14, 20170671 (2018).
Srinivasan, U., Elsen, P. R., Tingley, M. W. & Wilcove, D. S. Temperature and competition interact to structure Himalayan bird communities. Proc. R. Soc. B Biol. Sci. 285, 20172593 (2018).
Franke, F., Armitage, S. A. O., Kutzer, M. A. M., Kurtz, J. & Scharsack, J. P. Environmental temperature variation influences fitness trade-offs and tolerance in a fish–tapeworm association. Parasit. Vectors 10, 252 (2017).
Castano-Vazquez, F., Martinez, J., Merino, S. & Lozano, M. Experimental manipulation of temperature reduce ectoparasites in nests of blue tits Cyanistes caeruleus. J. Avian Biol. 49, UNSP e01695 (2018).
Paull, S. H. & Johnson, P. T. J. How temperature, pond-drying, and nutrients influence parasite infection and pathology. EcoHealth 15, 396–408 (2018).
Larsen, M. H. & Mouritsen, K. N. Temperature–parasitism synergy alters intertidal soft-bottom community structure. J. Exp. Mar. Bio. Ecol. 460, 109–119 (2014).
Marcogliese, D. J. The distribution and abundance of parasites in aquatic ecosystems in a changing climate: More than just temperature. Integr. Comp. Biol. 56, 611–619 (2016).
Mouritsen, K. N., Sørensen, M. M., Poulin, R. & Fredensborg, B. L. Coastal ecosystems on a tipping point: Global warming and parasitism combine to alter community structure and function. Glob. Change Biol. 24, 4340–4356 (2018).
Hatcher, M. J., Dick, J. T. A. & Dunn, A. M. Diverse effects of parasites in ecosystems: Linking interdependent processes. Front. Ecol. Environ. 10, 186–194 (2012).
Repetto, M. & Griffen, B. D. Physiological consequences of parasite infection in the burrowing mud shrimp, Upogebia pugettensis, a widespread ecosystem engineer. Mar. Freshw. Res. 63, 60–67 (2012).
Boze, B. G. V. & Moore, J. The effect of a nematode parasite on feeding and dung-burying behavior of an ecosystem engineer. Integr. Comp. Biol. 54, 177–183 (2014).
Laverty, C. et al. Temperature rise and parasitic infection interact to increase the impact of an invasive species. Int. J. Parasitol. 47, 291–296 (2017).
Labaude, S., Cézilly, F. & Rigaud, T. Temperature-related intraspecific variability in the behavioral manipulation of acanthocephalan parasites on their gammarid hosts. Biol. Bull. 232, 82–90 (2017).
Labaude, S., Rigaud, T. & Cézilly, F. Additive effects of temperature and infection with an acanthocephalan parasite on the shredding activity of Gammarus fossarum (Crustacea: Amphipoda): The importance of aggregative behavior. Glob. Chang. Biol. 23, 1415–1424 (2017).
MacNeil, C., Dick, J. T. A. & Elwood, R. W. The trophic ecology of freshwater Gammarus spp. (crustacea:amphipoda): Problems and perspectives concerning the functional feeding group concept. Biol. Rev. 72, 349–364 (1997).
Piscart, C., Genoel, R., Doledec, S., Chauvet, E. & Marmonier, P. Effects of intense agricultural practices on heterotrophic processes in streams. Environ. Pollut. 157, 1011–1018 (2009).
Degani, G., Bromley, H. J., Ortal, R., Netzer, Y. & Harari, N. Diets of rainbow trout (Salmo gairdneri) in a thermally constant stream. Vie Milieu 37, 99–103 (1987).
Friberg, N. et al. The effect of brown trout (Salmo trutta L.) on stream invertebrate drift, with special reference to Gammarus pulex L. Hydrobiologia 294, 105–110 (1994).
Kelly, D. W., Dick, J. T. A. & Montgomery, W. I. The functional role of Gammarus (Crustacea, Amphipoda): Shredders, predators, or both?. Hydrobiologia 485, 199–203 (2002).
Piscart, C., Bergerot, B., Laffaille, P. & Marmonier, P. Are amphipod invaders a threat to regional biodiversity?. Biol. Invasions 12, 853–863 (2010).
Constable, D. & Birkby, N. J. The impact of the invasive amphipod Dikerogammarus haemobaphes on leaf litter processing in UK rivers. Aquat. Ecol. 50, 273–281 (2016).
Foucreau, N., Puijalon, S., Hervant, F. & Piscart, C. Effect of leaf litter characteristics on leaf conditioning and on consumption by Gammarus pulex. Freshw. Biol. 58, 1672–1681 (2013).
Maltby, L., Clayton, S. A., Wood, R. M. & McLoughlin, N. Evaluation of the Gammarus pulex in situ feeding assay as a biomonitor of water quality: Robustness, responsiveness, and relevance. Environ. Toxicol. Chem. 21, 361–368 (2002).
Benesh, D. P., Lafferty, K. D. & Kuris, A. A life cycle database for parasitic acanthocephalans, cestodes, and nematodes. Ecology 98, 882–882 (2017).
Crompton, D. W. T. & Nickol, B. B. Biology of the Acanthocephala (Cambridge University Press, Cambridge, 1985).
Bakker, T. C. M., Frommen, J. G. & Thünken, T. Adaptive parasitic manipulation as exemplified by acanthocephalans. Ethology https://doi.org/10.1111/eth.12660 (2017).
Bethel, W. M. & Holmes, J. C. Altered evasive behavior and responses to light in amphipods harboring acanthocephalan cystacanths. J. Parasitol. 59, 945–956 (1973).
Bauer, A., Trouvé, S., Grégoire, A., Bollache, L. & Cézilly, F. Differential influence of Pomphorhynchus laevis (Acanthocephala) on the behaviour of native and invader gammarid species. Int. J. Parasitol. 30, 1453–1457 (2000).
Kaldonski, N., Perrot-Minnot, M.-J. & Cézilly, F. Differential influence of two acanthocephalan parasites on the antipredator behaviour of their common intermediate host. Anim. Behav. 74, 1311–1317 (2007).
McCahon, C. P., Brown, A. F. & Pascoe, D. The effect of the acanthocephalan Pomphorhynchus laevis (Müller 1776) on the acute toxicity of cadmium to its intermediate host, the amphipod Gammarus pulex (L.). Arch. Environ. Contam. Toxicol. 17, 239–243 (1988).
Médoc, V., Piscart, C., Maazouzi, C., Simon, L. & Beisel, J.-N. Parasite-induced changes in the diet of a freshwater amphipod: Field and laboratory evidence. Parasitology 138, 537–546 (2011).
Cornet, S., Franceschi, N., Bauer, A., Rigaud, T. & Moret, Y. Immune depression induced by acanthocephalan parasites in their intermediate crustacean host: Consequences for the risk of super-infection and links with host behavioural manipulation. Int. J. Parasitol. 39, 221–229 (2009).
Plaistow, S. J., Troussard, J.-P. & Cézilly, F. The effect of the acanthocephalan parasite Pomphorhynchus laevis on the lipid and glycogen content of its intermediate host Gammarus pulex. Int. J. Parasitol. 31, 346–351 (2001).
Bollache, L., Rigaud, T. & Cézilly, F. Effects of two acanthocephalan parasites on the fecundity and pairing status of female Gammarus pulex (Crustacea: Amphipoda). J. Invertebr. Pathol. 79, 102–110 (2002).
Dezfuli, B. S., Lui, A., Giovinazzo, G. & Giari, L. Effect of Acanthocephala infection on the reproductive potential of crustacean intermediate hosts. J. Invertebr. Pathol. 98, 116–119 (2008).
Labaude, S., Cézilly, F., Tercier, X. & Rigaud, T. Influence of host nutritional condition on post-infection traits in the association between the manipulative acanthocephalan Pomphorhynchus laevis and the amphipod Gammarus pulex. Parasit. Vectors 8, 403 (2015).
Rumpus, A. E. & Kennedy, C. R. The effect of the acanthocephalan Pomphorhynchus laevis upon the respiration of its intermediate host, Gammarus pulex. Parasitology 68, 271–284 (1974).
Perrot-Minnot, M.-J., Kaldonski, N. & Cézilly, F. Increased susceptibility to predation and altered anti-predator behaviour in an acanthocephalan-infected amphipod. Int. J. Parasitol. 37, 645–651 (2007).
Hindsbo, O. Effects of Polymorphus (Acanthocephala) on colour and behaviour of Gammarus lacustris. Nature 238, 333 (1972).
Dianne, L. et al. Protection first then facilitation: A manipulative parasite modulates the vulnerability to predation of its intermediate host according to its own developmental stage. Evolution 65, 2692–2698 (2011).
Lagrue, C., Kaldonski, N., Perrot-Minnot, M.-J., Motreuil, S. & Bollache, L. Modification of hosts’ behavior by a parasite: Field evidence for adaptive manipulation. Ecology 88, 2839–2847 (2007).
Kaldonski, N., Perrot-Minnot, M.-J., Motreuil, S. & Cézilly, F. Infection with acanthocephalans increases the vulnerability of Gammarus pulex (Crustacea Amphipoda) to non-host invertebrate predators. Parasitology 135, 627–632 (2008).
Gillooly, J. F., Brown, J. H., West, G. B., Savage, V. B. & Charnov, E. L. Effects of size and temperature on metabolic rate. Science 293, 2248–2251 (2001).
Roux, C. & Roux, A. L. Température et métabolisme respiratoire d’espèces sympatriques de gammares du groupe pulex (Crustacés, Amphipodes). Ann. Limnol. 3, 3–16 (1967).
Issartel, J., Hervant, F., Voituron, Y., Renault, D. & Vernon, P. Behavioural, ventilatory and respiratory responses of epigean and hypogean crustaceans to different temperatures. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 141, 1–7 (2005).
Foucreau, N., Cottin, D., Piscart, C. & Hervant, F. Physiological and metabolic responses to rising temperature in Gammarus pulex (Crustacea) populations living under continental or Mediterranean climates. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 168, 69–75 (2014).
Moenickes, S. et al. From population-level effects to individual response: Modelling temperature dependence in Gammarus pulex. J. Exp. Biol. 214, 3678–3687 (2011).
Barber, I., Berkhout, B. W. & Ismail, Z. Thermal change and the dynamics of multi-host parasite life cycles in aquatic ecosystems. Integr. Comp. Biol. 56, 561–572 (2016).
Olson, R. E. & Pratt, I. The life cycle and larval development of Echinorhynchus lageniformis Ekbaum, 1938 (Acanthocephala: Echinorhynchidae). J. Parasitol. 57, 143–149 (1971).
Tokeson, J. P. E. & Holmes, J. C. The effects of temperature and oxygen on the development of Polymorphus marilis (Acanthocephala) in Gammarus lacustris (Amphipoda). J. Parasitol. 68, 112–119 (1982).
Sheath, D. J., Andreou, D. & Britton, J. R. Interactions of warming and exposure affect susceptibility to parasite infection in a temperate fish species. Parasitology 143, 1340–1346 (2016).
VanCleave, H. J. Seasonal distribution of some acanthocephala from fresh-water hosts. J. Parasitol. 2, 106–110 (1916).
Muzzall, P. M. & Rabalais, F. C. Studies on Acanthocephalus jacksoni Bullock, 1962 (Acanthocephala: Echinorhynchidae). I. Seasonal periodicity and new host records. Proc. Helminthol. Soc. Wash. 42, 31–34 (1975).
Brown, A. F. Seasonal dynamics of the acanthocephalan Pomphorhynchus laevis (Muller, 1776) in its intermediate and preferred definitive hosts. J. Fish Biol. 34, 183–194 (1989).
Rauque, C. A. & Semenas, L. Infection pattern of two sympatric acanthocephalan species in the amphipod Hyalella patagonica (Amphipoda: Hyalellidae) from Lake Mascardi (Patagonia, Argentina). Parasitol. Res. 100, 1271–1276 (2007).
Wali, A. et al. Distribution of helminth parasites in intestines and their seasonal rate of infestation in three freshwater fishes of Kashmir. J. Parasitol. Res. https://doi.org/10.1155/2016/8901518 (2016).
Guinnee, M. A. & Moore, J. The effect of parasitism on host fecundity is dependent on temperature in a cockroach-acanthocephalan system. J. Parasitol. 90, 673–677 (2004).
Benesh, D. P., Hasu, T., Seppälä, O. & Valtonen, E. T. Seasonal changes in host phenotype manipulation by an acanthocephalan: Time to be transmitted?. Parasitology 136, 219–230 (2009).
Perrot-Minnot, M.-J., Maddaleno, M., Balourdet, A. & Cézilly, F. Host manipulation revisited: No evidence for a causal link between altered photophobia and increased trophic transmission of amphipods infected with acanthocephalans. Funct. Ecol. 26, 1007–1014 (2012).
Benesh, D. P., Duclos, L. M. & Nickol, B. B. The behavioral response of amphipods harboring Corynosoma constrictum (Acanthocephala) to various components of light. J. Parasitol. 91, 731–736 (2005).
Dianne, L., Bollache, L., Lagrue, C., Franceschi, N. & Rigaud, T. Larval size in acanthocephalan parasites: Influence of intraspecific competition and effects on intermediate host behavioural changes. Parasit. Vectors 5, 166 (2012).
Franceschi, N. et al. Co-variation between the intensity of behavioural manipulation and parasite development time in an acanthocephalan–amphipod system. J. Evol. Biol. 23, 2143–2150 (2010).
Perrot-Minnot, M.-J., Sanchez-Thirion, K. & Cézilly, F. Multidimensionality in host manipulation mimicked by serotonin injection. Proc. R. Soc. B Biol. Sci. 281, 20141915 (2014).
Franceschi, N., Bauer, A., Bollache, L. & Rigaud, T. The effects of parasite age and intensity on variability in acanthocephalan-induced behavioural manipulation. Int. J. Parasitol. 38, 1161–1170 (2008).
Franceschi, N. et al. Variation between populations and local adaptation in acanthocephalan-induced parasite manipulation. Evolution 64, 2417–2430 (2010).
Cézilly, F., Grégoire, A. & Bertin, A. Conflict between co-occuring manipulative parasites; an experimental study of the joint influence of two acanthocephalan parasites on the behaviour of Gammarus pulex. Parasitology 120, 625–630 (2000).
Bauer, A., Haine, E. R., Perrot-Minnot, M.-J. & Rigaud, T. The acanthocephalan parasite Polymorphus minutus alters the geotactic and clinging behaviours of two sympatric amphipod hosts: the native Gammarus pulex and the invasive Gammarus roeseli. J. Zool. 267, 39–43 (2005).
Xu, Y., Castel, T., Richard, Y., Cuccia, C. & Bois, B. Burgundy regional climate change and its potential impact on grapevines. Clim. Dyn. 39, 1613–1626 (2012).
Gunn, J. & Crumley, C. L. Global energy balance and regional hydrology: A Burgundian case study. Earth Surf. Process. Landforms 16, 579–592 (1991).
Rowell, D. P. A scenario of European climate change for the late twenty-first century: Seasonal means and interannual variability. Clim. Dyn. 25, 837–849 (2005).
Bollache, L., Gambade, G. & Cézilly, F. The influence of micro-habitat segregation on size assortative pairing in Gammarus pulex (L.) (Crustacea, Amphipoda). Arch. für Hydrobiol. 147, 547–558 (2000).
Dezfuli, B. S., Zanini, N., Reggiani, G. & Rossi, R. Echinogammarus stammen (Amphipoda) as an intermediate host for Pomphorhynchus laevis (Acanthocephala) parasite of fishes from the river Brenta. Bolletino di Zool. 58, 267–271 (1991).
Dianne, L., Perrot-Minnot, M.-J., Bauer, A., Guvenatam, A. & Rigaud, T. Parasite-induced alteration of plastic response to predation threat: increased refuge use but lower food intake in Gammarus pulex infected with the acanothocephalan Pomphorhynchus laevis. Int. J. Parasitol. 44, 211–216 (2014).
Hammond, B. A. the proboscis mechanism of Acanthocephalus ranae. J. Exp. Biol. 45, 203–213 (1966).
Taraschewski, H. Host–parasite interactions in Acanthocephala: A morphological approach. Adv. Parasitol. 46, 1–179 (2000).
Perrot-Minnot, M.-J., Gaillard, M., Dodet, R. & Cézilly, F. Interspecific differences in carotenoid content and sensitivity to UVB radiation in three acanthocephalan parasites exploiting a common intermediate host. Int. J. Parasitol. 41, 173–181 (2011).
Kennedy, C. R., Broughton, P. F. & Hine, P. M. The status of brown and rainbow trout, Salmo trutta and S. gairdneri as hosts of the acanthocephalan, Pomphorhynchus laevis. J. Fish Biol. 13, 265–275 (1978).
Foucreau, N., Piscart, C., Puijalon, S. & Hervant, F. Effects of rising temperature on a functional process: Consumption and digestion of leaf litter by a freshwater shredder. Fundam. Appl. Limnol./Arch. für Hydrobiol. 187, 295–306 (2016).
Noguchi, K., Gel, Y. R., Brunner, E. & Konietschke, F. nparLD: An R software package for the nonparametric analysis of longitudinal data in factorial experiments. J. Stat. Softw. 50, 1–23 (2012).
Pellan, L., Médoc, V., Renault, D., Spataro, T. & Piscart, C. Feeding choice and predation pressure of two invasive gammarids, Gammarus tigrinus and Dikerogammarus villosus, under increasing temperature. Hydrobiologia 781, 43–54 (2015).
Maure, F. et al. The cost of a bodyguard. Biol. Lett. 7, 843–846 (2011).
Maazouzi, C., Piscart, C., Legier, F. & Hervant, F. Ecophysiological responses to temperature of the ‘killer shrimp’ Dikerogammarus villosus: Is the invader really stronger than the native Gammarus pulex?. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 159, 268–274 (2011).
Maynard, B. J., Wellnitz, T. A., Zanini, N., Wright, W. G. & Dezfuli, B. S. Parasite-altered behavior in a crustacean intermediate host: Field and laboratory studies. J. Parasitol. 84, 1102–1106 (1998).
Dezfuli, B. S., Maynard, B. J. & Wellnitz, T. A. Activity levels and predator detection by amphipods infected with an acanthocephalan parasite, Pomphorhynchus laevis. Folia Parasitol. (Praha) 50, 129–134 (2003).
Stone, C. F. & Moore, J. Parasite-induced alteration of odour responses in an amphipod-acanthocephalan system. Int. J. Parasitol. 44, 969–975 (2014).
Jacquin, L., Mori, Q., Pause, M., Steffen, M. & Medoc, V. Non-specific manipulation of gammarid behaviour by P. minutus parasite enhances their predation by definitive bird hosts. PLoS ONE 9, e101684 (2014).
Thünken, T. et al. Impact of olfactory non-host predator cues on aggregation behaviour and activity in Polymorphus minutus infected Gammarus pulex. Hydrobiologia 654, 137–145 (2010).
Dianne, L. et al. Intraspecific conflict over host manipulation between different larval stages of an acanthocephalan parasite. J. Evol. Biol. 23, 2648–2655 (2010).
Thomas, F., Brown, S. P., Sukhdeo, M. V. K. & Renaud, F. Understanding parasite strategies: A state-dependent approach?. Trends Parasitol. 18, 387–390 (2002).
Baldauf, S. A. et al. Infection with an acanthocephalan manipulates an amphipod’s reaction to a fish predator’s odours. Int. J. Parasitol. 37, 61–65 (2007).
Durieux, R., Rigaud, T. & Médoc, V. Parasite-induced suppression of aggregation under predation risk in a freshwater amphipod. Sociality of infected amphipods. Behav. Process. 91, 207–213 (2012).
Lewis, S. E., Hodel, A., Sturdy, T., Todd, R. & Weigl, C. Impact of acanthocephalan parasites on aggregation behavior of amphipods (Gammarus pseudolimnaeus). Behav. Process. 91, 159–163 (2012).
Labaude, S., Rigaud, T. & Cézilly, F. Host manipulation in the face of environmental changes: Ecological consequences. Int. J. Parasitol. Parasit. Wildl. 4, 442–451 (2015).
Rahmstorf, S. & Coumou, D. Increase of extreme events in a warming world. PNAS 108, 17905–17910 (2011).
Acknowledgements
We thank Aude Balourdet, Alexandre Bauer and Sébastien Motreuil for technical assistance. This work was funded by the Agence Nationale de la recherche (ANR, Grant ANR-13-BSV7-0004-01 to TR), the PhD Grant to SL from the Ministère de l'Education Nationale, de l'Enseignement Supérieur et de la Recherche and was supported by the French “Investissements d'Avenir” program, project ISITE-BFC INVACLIM.
Author information
Authors and Affiliations
Contributions
T.R., F.C. and S.L. conceived the study. S.L. and L.D.M. performed the experiments and collected the data. S.L., L.D.M. and T.R. performed the statistical analyses and F.C. helped in their interpretation. The manuscript was written by S.L. and F.C. and critically revised by T.R. and L.D.M. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Labaude, S., Cézilly, F., De Marco, L. et al. Increased temperature has no consequence for behavioral manipulation despite effects on both partners in the interaction between a crustacean host and a manipulative parasite. Sci Rep 10, 11670 (2020). https://doi.org/10.1038/s41598-020-68577-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-68577-z
- Springer Nature Limited