Abstract
This study aims to analyze, in a population of singletons, the potential confounding or modifying effect of noise on the relationship between fetal growth restriction (FGR) or small for gestational age (SGA) and environmental exposure to air pollution. All women with single pregnancies living in one of two medium-sized cities (Besançon, Dijon) and who delivered at a university hospital between 2005 and 2009 were included. FGR and SGA were obtained from medical records. Outdoor residential exposure to nitrogen dioxide (NO2) and particulate matter (PM10) was quantified at the mother’s address at delivery over defined pregnancy periods; outdoor noise exposure was considered to be the annual average daily noise levels in the façade of building (LAeq,24 h). Adjusted odds ratios (ORa) were estimated by multivariable logistic regressions. Among the 8994 included pregnancies, 587 presented FGR and 918 presented SGA. In the two-exposure models, for SGA, the ORa for a 10-µg/m3 increase of PM10 during the two last months before delivery was 1.18, 95%CI 1.00–1.41 and for FGR, these ORa were for the first and the third trimesters, and the two last months before delivery: 0.77 (0.61–0.97), 1.38 (1.12–1.70), and 1.35 (1.11–1.66), respectively. Noise was not associated with SGA or FGR and did not confound the relationship between air pollution and SGA or FGR. These results are in favor of an association between PM10 exposure and fetal growth, independent of noise, particularly towards the end of pregnancy, and of a lack of association between noise and fetal growth.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The question of the influence of environmental conditions during pregnancy on adverse pregnancies outcomes is increasingly studied but remains debated. Among the adverse outcomes of pregnancy, fetal growth abnormalities are associated with poor perinatal outcomes1,2,3,4,5,6 and increase the prevalence of long-term neurodevelopmental, cardiovascular, and endocrine consequences6,7,8,9.
Among urban environmental exposures, air pollution and noise are the most ubiquitous and relatively correlated10.
The exposure to noise induces the release of stress hormones and inflammatory signaling molecules leading to oxidative stress and vascular dysfunction, and the exposure to air pollution has been suspected of increasing oxidative stress and systemic inflammation, and, during pregnancy, it may decrease uterine blood flow and placental fetal exchange, therefore slowing fetal growth8,11,12,13,14,15,16,17,18,19,20.
The association between air pollution and fetal growth disorders has been identified in many of studies in various more and less exposed regions of the world, with different pollutants and different fetal growth indicators measured in newborns at term or not: birth weight (BW), low birth weight (LBW), small for gestational age (SGA), or ultrasound antenatal measurements21,22,23,24,25. Nieuwenhuijsen's review in 2017 identified studies of the effects of noise on adverse pregnancy outcomes including fetal growth18. The authors found low quality evidence for an association between road traffic noise and LBW and SGA. They conclude that good quality studies were needed in various regions, particularly at lower levels of noise, and taking into account confounding factors like air pollution. Indeed, only a few studies have taken into account the effect of the exposure to both air pollution and noise26,27,28,29,30,31,32. The last two studies were published after the Nieuwenhuijsen's review31,32. In London, Smith et al. estimated that 3% of infants born at term with LBW are directly attributable to residential exposure to PM2.5 > 13.8 μg/m3 during pregnancy, and their results suggested little evidence of an independent exposure–response effect of traffic related noise (daytime and night-time road traffic noise) on BW at term32. In 2019, Nieuwenhuijsen et al. studied the influence of the urban exposome on BW in six European birth cohorts and found no evidence of an association between road traffic noise or air pollution and BW and LBW at term in co-exposure analyses31.
Although the adverse effect of air pollution on fetal growth is generally recognized, the influence of noise remains debated, as does the relative contribution of these two types of pollutants and the potential effects of co-exposure on fetal growth. The objective of this study was to explore the potential confounding or modified effect of noise on the relationship between fetal growth and environmental exposure to moderate levels of air pollution.
Material and methods
Population
For this retrospective study, we included all single pregnancies in women residing in Besançon or in the urban area of Dijon who delivered at the Besançon or Dijon university hospital between 1st January 2005 and 31st December 2009. The Besançon and Dijon university hospitals are level 3 maternities (i.e. with a neonatal intensive care unit). Stillborns and live newborns, whose births occurred after 22 completed weeks of gestation or with birth weight > 500 g, were included. When a woman had several single pregnancies in the study period, only one pregnancy was included after random selection. Hence, the number of women and the number of pregnancies included in the analysis population is the same (N = 8994).
This work is part of the PRECEE program (PREgnancy and Combined Environmental Exposure) and complements results published by Mariet et al. in 2018 on multiple pregnancies, by Barba-Vasseur et al. in 2017 on preterm birth in single pregnancy, and by Brembilla et al. in 2019 on vulnerability markers in pregnant women33,34,35.
Outcomes
Birth weight and fetal growth restriction (FGR) were extracted from the Besançon computerized medical records (DIAMM software (version 7.5, http://www.micro6.fr/mere-enfant.php) developed by the Association of Computerized Users in Pediatrics, Obstetrics and Gynecology36) and from the Burgundy perinatal network records and paper medical records for Dijon. Births were classified as SGA if birth weight was < 10th centile for gestational age (in weeks) and sex. The threshold for the 10th centile of birth weight was estimated in a population of French newborns from single and multiple pregnancies by gestational age and sex36. In order to test for a classification effect, SGA was also defined according to another birth weight standard for gestational age and sex, estimated with data from the 2010 perinatal study37. According to French, British, and Canadian recommendations, FGR was defined as a defect in fetal growth on two antenatal measurements taken two to three weeks apart38,39,40. FGR was retained according to the ICD10 codes in medical records (O36.5, P05.0, P05.1).
Covariables
All variables available in the medical records were analyzed to detect potential confounders: maternal socioeconomic characteristics, obstetrical history, pregnancy complications and newborn characteristics (Supplementary Table S1).
Maternal age was calculated at delivery and dichotomized with a threshold of 35 years old. Maternal smoking during pregnancy was coded as “present” if active smoking was ticked in the medical records. Malnutrition was defined by pre-pregnancy body mass index lower than 18.5 or by the presence of an ICD10 code of malnutrition in medical records (O25, E43, E44). Obesity was defined by pre-pregnancy body mass index higher than 30. Vaginal bleeding in the second and third trimesters referred to an episode of bleeding after 28 weeks of gestation, including placental abruption and bleeding because of placenta previa (ICD10 codes: O45, O46, O441 and P021). Major infant congenital abnormalities corresponded to any major congenital anomalies according to the European network of population-based registries for the epidemiologic surveillance of congenital anomalies41. Infant congenital abnormalities considered for this study were determined before birth or at birth.
The neighborhood socioeconomic level was estimated with a collective socioeconomic index calculated at the geographical scale of the French sub-municipal census block groups (IRIS) defined by the National Institute of Statistic and Economics Studies42. Fifty IRIS in Besançon and 113 in the urban area of Dijon were included; population size ranged between 62 and 4811 inhabitants (mean = 2182 inhabitants). Variables related to family and household, immigration and mobility, employment and income, education and housing were extracted from the 2008 population census database. From among these variables, 39 were selected because of their occurrence in the literature43,44,45. The first component of a principal component analysis (PCA) was used to calculate a standardized socio-economic index following a reduction step. The socioeconomic index was calculated using the R package SesIndexCreatoR43. A value of the socioeconomic index in the last decile was considered as low neighborhood socioeconomic level.
Environmental residential exposures
The residential exposure assessments have been previously described10,33,34,35,46,47,48,49,50. The participants’ addresses at the date of delivery were extracted from CPage software (version 2, https://www.cpage.fr/) using the personal identification number and the date of delivery. This address identified the residential building.
Two pollutants related to road traffic were studied. NO2 is a gaseous pollutant known to be the main indicator of road traffic51. Particulate matters (PM10) are also generated by road traffic and residential heating and were chosen because of their significant impact on human health and climate52,53. NO2 and PM10 exposure assessments were calculated at each mother’s building considering a 50 m radius buffer centered on the building centroid. The NO2 and PM10 levels were calculated using a two-step emission and diffusion modeling. Air pollutant emissions were calculated from road traffic data using CIRCUL'AIR software (version 2.51, www.atmo-grandest.eu), developed and used by all approved French Air Quality Monitoring Agencies (AASQA) (COPERT IV European standard methodology). AASQA’s pollution emission inventory was used to assess air pollutant emissions related to all activity sectors, especially heating, industries or agriculture. Air pollutant concentration was estimated 2 m above ground on a 25 m grid with reinforced gridding around the axes of emission, using the ADMS-Urban software (version 3.1.6, http://www.cerc.co.uk/) for diffusion modeling. ESRI ArcGIS software (version 10.1, https://www.esrifrance.fr/) was used for spatial interpolation to increase the spatial resolution of the ADMS output. Air pollutant concentration expressed in micrograms per cubic meter (μg/m3) was thus calculated at a 4m2 (2 m × 2 m) raster. The validity of the 2 m result was estimated on the basis of data from four, two-week-long measurement campaigns carried out during autumn and winter 2010 as well as spring and summer 2011. Measurements were based on 863 passive samplers and the nine AASQA air pollution measurement stations for NO2 (ATMO Franche-Comté and Atmosf’Air Bourgogne). Validation statistics (r2) range from 0.64 to 0.69. The validity of the PM10 models were verified using the city's fixed air-quality monitoring network. Monthly maps of air pollutant concentration were established from January 2004 to December 2009 using hourly meteorological data and background pollution levels to account for the seasonal variations in air pollutant concentrations. Using the monthly maps, time-weighted average air pollutant exposure was assessed over the following defined pregnancy periods: first, second and third trimester, entire pregnancy and two months before delivery.
Noise levels were calculated in accordance with the European Commission’s Environmental Noise Directive 2002/49/CE using an environmental noise prediction model with MithraSIG software (version 3.7, https://www.geomod.fr/fr/geomatique-modelisation-3d/mithrasig/)54. The following four types of noise sources were considered: road traffic, rail traffic, pedestrian streets, and fountains. Individual aircraft noise data were not available for the 2005–2009 period (military airport located within the city limits of Dijon). Women living in the area exposed to aircraft noise (according to the urban unit noise exposure plan) were not considered in this study. Theoretical noise levels were calculated in front of each building façade on each floor. Measurement campaigns were used for validation (76 points). The validation was good with a Spearman correlation coefficient at 0.81 (p < 0.01). For each woman, the average building noise levels in front of the entire façade were calculated using the following five indices: the daily equivalent A-weighted total noise level, LAeq,24 h for principal analysis; day equivalent A-weighted total noise level LAeq,day (6:00–18:00 h); evening equivalent A-weighted noise level LAeq,evening (18:00–22:00 h); night equivalent A-weighted total noise level LAeq,night (22:00–6:00 h); and combined day-evening-night A-weighted total noise level Lden, with evening and night exposures penalized by 5 and 10 dB, respectively, for sensitivity analyses. Moreover, source-specific indices for LAeq,24 h were calculated for road and rail traffic related noise for sensitivity analyses.
Statistics
The association between environmental exposures (NO2, PM10 and LAeq,24 h) and SGA or FGR was estimated with simple and multiple logistic regression analyses, in single, and two-exposure models (air pollution with noise), where SGA or FGR were taken as binary outcomes in the models. Departure from the assumption of linearity was tested by introducing a polynomial function of the environmental exposure variables into the models. The ORs were adjusted for: term in gestational weeks, maternal age older than 35 years at delivery, low neighborhood socioeconomic level, maternal smoking during pregnancy, malnutrition and obesity, nulliparity, gestational hypertension, diabetes, assisted reproductive techniques, vaginal bleeding in the second and third trimesters, infection, and major infant congenital abnormalities. Because of the non-random distribution of missing data, a missing data class was used for categorical variables in multivariate analysis. Only two adjustment factors of the model had missing data: malnutrition and obesity (n = 219) and maternal smoking during pregnancy (n = 196), affecting only 2.4% of pregnancies. Potential interactions between air pollution and noise indices were assessed in models where at least one exposure was significant (p-value < 0.15). The interaction was tested using the p-value associated with the term from the regression analysis and comparing the Akaike Information Criterion (AIC) of models with and without the interaction term. Sensitivity analyses were conducted using four noise indices (LAeq,day, LAeq,evening, LAeq,night, Lden), two source-specific noise indices (road and rail traffic related noise LAeq,24 h), and the other birth weight standard to define SGA as an outcome variable37. Maternal age at delivery and neighborhood socioeconomic level were also considered for adjustment in continuous form, or with a 2nd or 3rd order polynomial. Adjustment for year-season of conception was also considered in sensitivity analysis. Finally, a last analysis was conducted restricted to the subgroup of live births. Statistical analyses were performed with SAS software (version 9.4, SAS Institute, Cary, NC: https://www.sas.com/).
Ethics
This study was approved by the French National Advisory Committee for the Treatment of Information in Health Research (CCTIRS) (registration number 15.292, April 9th 2015) and by the French data protection authority (CNIL) (registration number DR-2015-736, December 24th 2015). All methods were carried out in accordance with the ethical standards of CNIL and the Declaration of Helsinki. The requirement for patient informed consent was waived by the CNIL because of the retrospective nature of the study. A letter of information was sent to each eligible participant, and 20 families refused to participate. They were excluded from the study. All records were anonymized prior to analysis.
Consent to participate
The requirement for patient informed consent was waived by the CNIL because of the retrospective nature of the study. So, in accordance with the CNIL recommendations, a letter of information was sent to each eligible family and those who refused to participate where not included.
Results
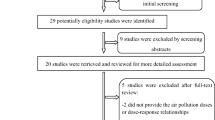
Among the 10 570 single deliveries which occurred in the Besançon or Dijon university hospital from women over 18 years old and living in the defined study area, 8994 pregnancies were included in the study. Twenty pregnancies were excluded because the family opposed the use of their medical data, 103 due to incorrect address (wrong or unrecognizable recorded street names or anonymous childbirth) and 1453 pregnancies were excluded because the same woman had several pregnancies over the study period.
Among the 8994 pregnancies, 587 had FGR and 918 had SGA. Five hundred and five (55.0%) fetuses with SGA did not have FGR; and 172 (29.4%) fetuses with FGR did not have SGA. SGA and FGR were significantly associated (p-value < 0.0001, Chi-square test). The pregnancy and newborn characteristics are presented in Table 1.
The median NO2 concentration considering a 50 m radius buffer during the entire pregnancy was 23.2 µg/m3; the minimum exposure was 7.4 µg/m3 and the maximum exposure was 51.6 µg/m3 (Supplementary Figure S1, Supplementary Table S2). The median PM10 concentration considering a 50 m radius buffer during the entire pregnancy was 18.5 µg/m3; the minimum exposure was 11.9 µg/m3 and the maximum exposure was 31.5 µg/m3 (Supplementary Figure S2, Supplementary Table S2). The median daily equivalent A-weighted total noise level was 55.5 dB; the minimum exposure was 33.2 dB and the maximum exposure was 76.9 dB (Supplementary Figure S3, Supplementary Table S2). The Pearson correlation coefficients for environmental exposure were: 0.56 between LAeq,24 h and NO2 exposure during the entire pregnancy, 0.28 between LAeq,24 h and PM10 exposure during the entire pregnancy, and 0.28 between NO2 and PM10 exposure during the entire pregnancy (p-value < 10–3) (Supplementary Table S3).
We found no departure from the assumption of linearity of pregnancy outcomes with noise and air pollution (Supplementary Figure S4).
For SGA, the crude OR associated with a 5 dB increase of total building LAeq,24 h was 0.97 (95% CI 0.91–1.03) (Table 2). The adjusted OR remained similar in single and two-exposure models. The crude OR associated with a 10 µg/m3 increase in NO2 exposure during the first, second and third trimester, the entire pregnancy and the two months before delivery were 0.94 (95% CI 0.86–1.02), 0.96 (95% CI 0.87–1.04), 0.95 (95% CI 0.87–1.04), 0.94 (95% CI 0.86–1.03), and 0.96 (95% CI 0.88–1.05), respectively. The adjusted OR remained similar in single-exposure models and in two-exposure models with total building LAeq,24 h. The crude OR associated with a 10 µg/m3 increase in PM10 exposure during the first, second and third trimester, the entire pregnancy and the two months before delivery were 1.02 (95% CI 0.86–1.22), 1.17 (95% CI 0.99–1.39), 1.21 (95% CI 1.03–1.43), 1.29 (95% CI 1.00–1.66), and 1.20 (95% CI 1.02–1.41), respectively. The adjusted OR in single-exposure models decreased slightly and became non-significant. In the two-exposure models with total building LAeq,24 h, only the adjusted OR associated with a 10 µg/m3 increase in PM10 exposure during the two months before delivery was significant: 1.18 (95% CI 1.00–1.40). Sensitivity analyses were conducted using the birth weight standard of Ego (Cf. Material and methods). The proportion of pregnancies with SGA was 12.4%. Sensitivity analyses led to OR estimates close to the main analysis.
When considering FGR, the crude OR associated with a 5 dB increase of total building LAeq,24 h was 0.98 (95% CI 0.90–1.06) (Table 2). The adjusted OR remained similar in single and two-exposure models. The crude OR associated with a 10 µg/m3 increase in NO2 exposure during the first, second and third trimester, the entire pregnancy and the two months before delivery were 0.93 (95% CI 0.83–1.03), 1.00 (95% CI 0.90–1.11), 1.03 (95% CI 0.93–1.15), 0.98 (95% CI 0.88–1.10), and 1.04 (95% CI 0.93–1.16), respectively. The adjusted OR remained similar in single and two-exposure models with values close to 1. The crude OR associated with a 10 µg/m3 increase in PM10 exposure during the first, second and third trimester, the entire pregnancy and the two months before delivery were 0.78 (95% CI 0.63–0.98), 1.02 (95% CI 0.82–1.26), 1.39 (95% CI 1.14–1.70), 1.05 (95% CI 0.77–1.43), and 1.35 (95% CI 1.11–1.63), respectively. The adjusted OR remained similar in single and two-exposure models.
For SGA, the OR for single-exposure adjusted models for an increase of an IQR were 0.95 (95% CI 0.87–1.05) for total building LAeq,24 h, 0.92 (95% CI 0.84–1.02) for NO2 exposure during the third trimester, and 1.08 (95% CI 0.99–1.18) for PM10 exposure during the third trimester, respectively. For FGR, they were 0.97 (95% CI 0.87–1.09) for building LAeq,24 h, 1.01 (95% CI 0.89–1.14) for NO2 exposure during the third trimester, and 1.16 (95% CI 1.05–1.29) for PM10 exposure during the third trimester, respectively.
The two centers differed on characteristics of the pregnancy, for example: low neighborhood socioeconomic level (16.2 for Besançon versus 13.4% for Dijon), malnutrition (9.1% for Besançon versus 6.6% for Dijon), prematurity (7.3% for Besançon versus 8.8% for Dijon), or small for gestational age (11.8% for Besançon versus 9.1% for Dijon), but not on FGR (7.0% for Besançon versus 6.2% for Dijon). Environmental exposures were significantly different between the two centers: in Besançon, noise and NO2 levels were lower, but PM10 levels were higher (Supplementary Table S4). Adjustment for the maternity of delivery has been tested and stratified analyses on the maternity of delivery have been performed. In the adjusted analysis, OR associated with total noise exposure remained similar and did not change the OR associated with air pollution exposure in the two-exposure models. The results were also similar in the stratified analyses. For example, for FGR, OR associated with a 5 dB increase of total building LAeq,24 h was 0.93 (95% CI 0.82–1.05) for Besançon and 1.03 (95% CI 0.92–1.15) for Dijon, and OR associated with a 10 µg/m3 increase in PM10 exposure during the third trimester was 1.33 (95% CI 1.00–1.75) for Besançon and 1.30 (95% CI 0.93–1.81) for Dijon in single exposure models. And OR associated with a 10 µg/m3 increase in PM10 exposure during the third trimester were 1.41 (95% CI 1.06–1.88) for Besançon and 1.31 (95% CI 0.93–1.85) for Dijon in two exposure models.
Interactions between noise and air pollution do not improve the adequacy of the models. Furthermore, no synergistic or antagonist effects were observed in the models with the interaction term.
Sensitivity analyses led to similar results when considering road or rail traffic related noise exposure, maternal age or neighborhood socioeconomic level in continuous variable with 1st, or a 2nd or a 3rd order polynomial, or restricted to the subgroup of live births (Supplementary Tables S5, S6, S7, S8, and S9). In the sensitivity analysis with adjustment for year-season of conception, the OR for the association between first trimester PM10 concentration and FGR became non-significant and the other estimates did not been change (Supplementary Table S10).
Discussion
Beyond studies with high levels of noise and air pollution, the situation with low levels of air pollution makes it possible to better assess the effect of noise on the relationship between air pollution and fetal growth disorders, avoiding a potential masking effect by high levels of air pollution. The systematic two-exposure modeling allowed to explore for confounding of noise on the relationship between air pollution and fetal growth. In our study, we first confirmed the previously observed association between moderate exposure to PM10 and fetal growth disorders, especially towards the end of pregnancy. Our results are also in favor of a lack of association between environmental noise exposure and fetal growth disorders in singletons. Finally, our data suggest that moderate exposure to noise has no confounding or modifying effect on the relationship between fetal growth and environmental exposure to the air pollution analyzed.
Air pollution, noise and fetal growth
In a context of moderate levels of air pollution, our results are in favor of an independent association between air pollution, and of a lack of association between noise and NO2 exposure and fetal growth disorders.
Air pollution and fetal growth
Most of the studies that found that air pollution (NO2 or PM10) had a negative impact on fetal growth (SGA or FGR) were conducted on more polluted areas than in our study (1.5 to twofold more for NO2 and 2 to threefold more for PM10)21,55,56,57,58,59,60. Our results confirm that exposure to PM10 at the end of the pregnancy has a negative impact on FGR in a moderately polluted area. However, it could not be excluded that the study of finer particles would have led to different results (data were unfortunately not available for our study area). A study conducted in Vancouver, Canada, where the levels of NO2 were comparable to those of our study area, also described a negative impact of NO2 on SGA61. Other studies found no associations between NO2 and SGA27,62,63,64,65. The OR in our study ranged between 0.92 and 0.96 and were not significant. We conducted a precedent study in a population of multiple pregnancies (twins and triplets) and found no association between NO2 exposure and SGA with non-significant OR ranged between 0.78 and 0.88, but an association between NO2 exposure and FGR with OR ranged between 1.42 and 1.5235.
Noise and fetal growth
In agreement with our results, a recent meta-analysis conducted by Dzhambov and Lercher found no association between noise and SGA (OR for an increase of 10 dB(A) = 1.02; 95% CI 0.86, 1.21) (I2 = 90%)66. This meta-analysis was performed on five populations in four cohort or cross-sectional studies27,29,32,67. However, levels of noise exposure were moderate in our study and associations between noise and fetal growth disorders could be plausible at higher levels of exposure.
Multiexposure to noise and air pollution, and fetal growth
Our study is one of the rare studies exploring co-exposure to environmental noise and air pollution. Other studies have not specifically explored the influence of noise on the relationship between air pollution and fetal growth disorders, and have used different methods in contexts different from ours. In the metropolitan area of Vancouver, Canada, Gehring et al. found an association between all transportation noise exposure and SGA. The observed association between NO2 exposure and SGA differed according to the assessment of exposure: the OR associated with an IQR increase was 0.98 (95% CI 0.96–1.01) when a land-use regression model was used and 1.10 (95% CI 1.06–1.15) for the inversed distance weighting method. In co-pollutant models, only noise was associated with SGA29. In two surveys in the Alpine area, a positive association between noise and SGA was revealed in two-exposure model with adjustment for NO2 exposure, which was not associated with SGA in the first survey. In the second survey, noise was negatively associated with SGA though this association disappeared with adjustment for NO2 exposure67. In London, Smith et al. found no association between NO2, PM10, noise and SGA in one and co-pollutant models32.
Other studies analyze the association between noise, air pollution and (term-) birth weight or (term-) LBW or birth weight26,27,28,30,31. Two ecological time-series studies in Madrid, Spain, assessed the impact of PM2.5, NO2, O3, noise, and temperature on LBW and found an impact on LBW of: (1) daily diurnal noise at onset of gestation, in the second trimester and in the week of birth itself, and of NO2 in the second trimester, in all live singleton births for the first study; (2) only of PM2.5 in non-preterm births for the second study26,28. A cohort study in Barcelona, Spain, based on singleton term births found no effect of noise but an effect of PM2.5 and PM10 in the third trimester on LBW27. In a study on the Danish National Birth Cohort, no association between road traffic noise and NO2 on birth weight was found in children born at term in two exposure models30. Finally, a study grouping six European birth cohorts found no association between NO2, PM10, PM2.5, road traffic noise and LBW31.
Potential biases, study limits and strengths
The negative association between PM10 exposure during the first trimester and FGR could be explained by the combined effects of three factors: the effect of the exposure on fetal growth, the seasonal variations of the exposure, and the existence of a gestational period of sensitivity to exposure. In fact, PM10 levels vary in a 1-year cycle and are higher in the colder season than in warmer season due to anthropic activities (transport and heating). The seasonal variations are higher for PM10 than NO2. The duration of a pregnancy is 9 months. So when the first trimester of a pregnancy is in winter, then the third trimester will be in summer; PM10 levels will be higher in the first trimester than in the third. Conversely, when the first trimester of a pregnancy is in summer, the third will be in winter; PM10 levels will be higher in the third trimester than in the first. This is consistent with the sensitivity analysis adjusted for year-season of conception where the OR for the first trimester became non-significant. The mechanism of placental hypoperfusion intervenes especially at the end of pregnancy on fetal growth. In the hypothesis of an impact of PM10 on fetal growth, women with a third trimester in winter may experience more frequently FGR than women with a third trimester in summer. Higher PM10 levels will be associated with the occurrence of FGR, the observed OR may be greater than one for the third trimester exposure. Conversely, for women with a first trimester in the summer associated with lower PM10 levels, the observed OR may be lower than one for the first trimester exposure. This could potentially lead to seasonality bias. Moreover, as suggested by Hao et al. and by Raz et al., we cannot exclude a selection bias called live-birth bias at the beginning of the pregnancy due to early abortions that can be caused by exposure to air pollution because they were not detectable in our study and therefore were not included68,69. Raz et al. described two types of live-birth bias: (1) the selection of a group among whom those with high exposure to air pollution could have low exposure to other risk factors for FGR (or higher exposure to protective factors), or (2) the selection of a group among whom those with high exposure to air pollution could have lower susceptibility to the effects of other risk factors for FGR than those with lower air-pollution exposure.
Compared to the single pregnancies recorded in the 2010 French perinatal study, our study sample presented just slightly adverse outcomes70. In fact, the two maternities included are level 3 maternity units where complicated pregnancies are more closely monitored. But the two public university hospitals included in the study are also obstetrical primary care hospitals. So because of their immediate proximity for women living in the studied urban areas, the effect of the reference status of the two maternities is limited.
SGA and FGR seem to be the most relevant indicators of fetal growth abnormalities. In fact, the sexually dimorphic differences in growth of the fetus is mediated by the sex specific function of the human placenta71 and SGA and FGR take into account the sex of the fetus. They were defined differently. On one hand, SGA is defined as a weight lower than the 10th centile of weight for gestational age and sex. This indicator was objectively determined from the French perinatal network reference of birth weight for sex and gestational age36. When it was compared with another reference; the associations were similar. On the other hand, FGR was established from the ICD10 codes listed in medical records. FGR is defined as a defect in fetal growth on two antenatal measurements two to three weeks apart according to French, British, and Canadian recommendations38,39,40. FGR is rarely used as an outcome in environmental epidemiological studies because it is more difficult to diagnose FGR retrospectively than SGA58. However, SGA is defined by birth weight at gestational age and sex and the size and weight of newborns are strongly influenced by those of their parents. Moreover, SGA does not take into account the growth trajectory as opposed to FGR, which is a dynamic measure of growth regardless of the measured weight value. So SGA could be less specifically a disorder of fetal growth than FGR, and seems less appropriate for identifying an association between fetal growth disorders and environmental exposure than FGR. A coding effect cannot be ruled out because FGR implies a dynamic evaluation of fetal growth during pregnancy by obstetricians. However, this potential coding effect was reduced by the multicentric quality of this study.
Specific attention was paid to the collection of data from medical records. The consultation of paper medical records resulted in the collection of potential confounding factors for the adjustment of the analyses. Data was missing for two adjustment factors of the model (malnutrition/obesity and maternal smoking during pregnancy) but involved only 2.4% of pregnancies.
Another limit was the absence of information about a potential move during pregnancy – we used the mother's address at delivery, which was recorded in the hospital information system upon admission, for geocoding.
Individuals spend about 80% of the time in indoor environments (European Commission, 2004) and French women spend 16/24 h (67% of the time) inside their dwelling72, but we did not use indoor air measurements for our study. Due to the retrospective design of this study, NO2 exposure was assessed using modelled outdoor exposure. However, a study in Vancouver, Canada, found a good agreement between indoor air measurements and outdoor values obtained by modeling73,74; a similar approach previously conducted by our team in the study area led to similar conclusions46,47; furthermore the retrospective modeling of exposure allowed for repeatable assessment of exposure. We assessed only residential exposure but time spent at home increases during maternity leave. We cannot however exclude exposure misclassification; but the definition of the air pollution and noise indicators that we used in this study has been explored in precedent works of our team49,50. Particular attention was paid to calculate NO2 and PM10 exposure closest to the home in the immediate neighborhood50. We used the average noise of all façades because the use of the noise of the most (least) exposed façade could induce an over (under) estimation of the exposure. Furthermore, the closer from the main source the building is located, the higher the overestimation could be. So when considering individual exposure, the most exposed façade could lead to differential error measurement. And last, this influence of the proximity of the source on the error assessment is more marked for noise than for air pollution. Moreover, we do not know the floor and the façade of the dwelling of the women included in our study. In one of the two areas, 25% of the buildings are very close to a main road. All these points could lead to a significant overestimation of the noise exposure of the most exposed buildings49, and then to a high risk of differential error assessment between dwelling, and between noise and air pollution exposure.
Conclusions
The study confirms a positive association between fetal growth disorders in single pregnancies and environmental exposure to air pollution at the end of pregnancy, in moderately polluted cities. In these conditions of moderate noise exposure, no argument in favor of a noise effect on the relationship between fetal growth and environmental exposure to air pollution was found in our study. These results need to be confirmed in areas with high levels of noise so as not to overlook the effect of high exposure of noise on the relationship between air pollution and fetal growth disorders.
Data availability
In accordance with the CNIL recommendations and the letter of information sent to each eligible family, we are not authorized to transmit the data.
References
Colella, M., Frérot, A., Novais, A. R. B. & Baud, O. Neonatal and long-term consequences of fetal growth restriction. Curr. Pediatr. Rev. 14(4), 212–218 (2018).
Dall’Asta, A., Brunelli, V., Prefumo, F., Frusca, T. & Lees, C. C. Early onset fetal growth restriction. Matern. Health Neonatol. Perinatol. 18(3), 2 (2017).
Figueras, F. & Gardosi, J. Intrauterine growth restriction: new concepts in antenatal surveillance, diagnosis, and management. Am. J. Obstet. Gynecol. 204(4), 288–300 (2011).
Gardosi, J. & Francis, A. Adverse pregnancy outcome and association with small for gestational age birthweight by customized and population based percentiles. Am. J. Obstet. Gynecol. 201(28), e1-8 (2009).
Kady, S. & Gardosi, J. Perinatal mortality and fetal growth restriction. Best Pract. Res. Clin. Obstet. Gynaecol. 18(3), 397–410 (2004).
Sharma, D., Farahbakhsh, N., Shastri, S. & Sharma, P. Intrauterine growth restriction: part 2. J. Matern. Fetal Neonatal. Med. 29(24), 4037–4048 (2016).
Barker, D. J. et al. Fetal nutrition and cardiovascular disease in adult life. Lancet 341(8850), 938–941 (1993).
Figueras, F. & Gratacos, E. An integrated approach to fetal growth restriction. Best Pract. Res. Clin. Obstet. Gynaecol. 38, 48–58 (2017).
Jacobsson, B. et al. Cerebral palsy and restricted growth status at birth: population based case-control study. BJOG 115(10), 1250–1255 (2008).
Tenailleau, Q. M. et al. Do outdoor environmental noise and atmospheric NO2 levels spatially overlap in urban areas?. Environ. Pollut. 214, 767–775 (2016).
Biberoglu, E. et al. Circulating and myometrial markers of oxidative stress in pregnant women with fetal growth restriction. J. Obstet. Gynaecol. Res. 42(1), 29–35 (2016).
Browne, V. A. et al. Uterine artery blood flow, fetal hypoxia and fetal growth. Philos. Trans. R Soc. Lond. B Biol. Sci. 370(1663), 20140068 (2015).
Daiber, A. et al. Environmental noise induces the release of stress hormones and inflammatory signaling molecules leading to oxidative stress and vascular dysfunction-Signatures of the internal exposome. BioFactors 45(4), 495–506 (2019).
Duhig, K., Chappell, L. C. & Shennan, A. H. Oxidative stress in pregnancy and reproduction. Obstet Med. 9(3), 113–116 (2016).
Ghio, A. J., Carraway, M. S. & Madden, M. C. Composition of air pollution particles and oxidative stress in cells, tissues, and living systems. J. Toxicol. Environ. Health B Crit. Rev. 15(1), 1–21 (2012).
Ha, S., Zhu, Y., Liu, D., Sherman, S. & Mendola, P. Ambient temperature and air quality in relation to small for gestational age and term low birthweight. Environ. Res. 155, 394–400 (2017).
Møller, P. et al. Oxidative stress and inflammation generated DNA damage by exposure to air pollution particles. Mutat. Res. Rev. Mutat. Res. 762, 133–166 (2014).
Nieuwenhuijsen, M. J., Ristovska, G. & Dadvand, P. WHO environmental noise guidelines for the european region: a systematic review on environmental noise and adverse birth outcomes. Int. J. Environ. Res. Public Health. 14(10), 1252 (2017).
Prada, J. A. & Tsang, R. C. Biological mechanisms of environmentally induced causes of IUGR. Eur. J. Clin. Nutr. 52, S21–S27 (1998) (discussion S27–8).
Slama, R. et al. Meeting report: atmospheric pollution and human reproduction. Environ. Health Perspect. 116(6), 791–798 (2008).
Guo, L. Q. et al. Ambient air pollution and adverse birth outcomes: a systematic review and meta-analysis. J. Zhejiang Univ. Sci. B. 20(3), 238–252 (2019).
Huang, I. et al. A systematic review of associations between maternal exposures during pregnancy other than smoking and antenatal fetal measurements. Environ. Res. 173, 528–538 (2019).
Li, X. et al. Association between ambient fine particulate matter and preterm birth or term low birth weight: an updated systematic review and meta-analysis. Environ. Pollut. 227, 596–605 (2017).
Tsoli, S., Ploubidis, G. B. & Kalantzi, O. I. Particulate air pollution and birth weight: a systematic literature review. Atmosph. Pollut. Res. 10(4), 1084–1122 (2019).
Zheng, T. et al. Effects of environmental exposures on fetal and childhood growth trajectories. Ann. Glob. Health. 82(1), 41–99 (2016).
Arroyo, V., Díaz, J., Carmona, R., Ortiz, C. & Linares, C. Impact of air pollution and temperature on adverse birth outcomes: Madrid, 2001–2009. Environ. Pollut. 218, 1154–1161 (2016).
Dadvand, P. et al. Residential proximity to major roads and term low birth weight: the roles of air pollution, heat, noise, and road-adjacent trees. Epidemiology 25(4), 518–525 (2014).
Díaz, J., Arroyo, V., Ortiz, C., Carmona, R. & Linares, C. Effect of environmental factors on low weight in non-premature births: a time series analysis. PLoS ONE 11(10), e0164741 (2016).
Gehring, U., Tamburic, L., Sbihi, H., Davies, H. W. & Brauer, M. Impact of noise and air pollution on pregnancy outcomes. Epidemiology 25(3), 351–358 (2014).
Hjortebjerg, D. et al. Associations between maternal exposure to air pollution and traffic noise and newborn’s size at birth: a cohort study. Environ. Int. 95, 1–7 (2016).
Nieuwenhuijsen, M. J. et al. Influence of the urban exposome on birth weight. Environ. Health Perspect. 127(4), 47007 (2019).
Smith, R. B. et al. Impact of London’s road traffic air and noise pollution on birth weight: retrospective population based cohort study. BMJ 5(359), j5299 (2017).
Barba-Vasseur, M. et al. Do low to moderate environmental exposure to noise and air pollution influence preterm delivery in medium-sized cities?. Int. J. Epidemiol. 46(6), 2017–2027 (2017).
Brembilla, A. et al. Pregnancy vulnerability in urban areas: a pragmatic approach combining behavioral, medico-obstetrical, socio-economic and environmental factors. Sci. Rep. 9(1), 18878 (2019).
Mariet, A. S. et al. Multiple pregnancies and air pollution in moderately polluted cities: Is there an association between air pollution and fetal growth?. Environ. Int. 121(Pt 1), 890–897 (2018).
Association of users of computerized records in pediatrics, obstetrics and gynecology (Audipog). Presentation of the sentinel network, accessed 1st December 2020. https://www.audipog.net/Reseau-Sentinelle.
Ego, A. et al. Customized and non-customized French intrauterine growth curves: I: methodology. J. Gynecol. Obstet. Biol. Reprod. (Paris). 45(2), 155–164 (2016).
French national college of gynecologists and obstetricians (CNGOF). Recommendations for clinical practice. Intrauterine growth retardation. 37th national days. Paris, 2013, accessed 1st December 2020 http://www.cngof.asso.fr/data/RCP/CNGOF_2013_FINAL_RPC_rciu.pdf.
Lausman, A. & Kingdom, J. Maternal fetal medicine committee: Intrauterine growth restriction: screening, diagnosis, and management. J. Obstet. Gynaecol. Can. 35(8), 741–748 (2013).
Royal College of Obstetricians and Gynaecologists (RCOG). The investigation and management of the small-for-gestational age fetus. London (UK): Royal College of Obstetricians and Gynaecologists (RCOG); 2013 [Green-top guideline; No. 31].
EUROCAT. EUROCAT Guide 1.3: Instruction for the registration and surveillance of congenital anomalies. EUROCAT Central Registry, University of Ulster; 2005.
INSEE. National Institute of Statistic and Economics Studies. Definition of IRIS. 2016 Oct, accessed 1st December 2020 https://www.insee.fr/en/metadonnees/definition/c1523.
Lalloué, B. et al. A statistical procedure to create a neighborhood socioeconomic index for health inequalities analysis. Int. J. Equity Health. 28(12), 21 (2013).
Messer, L. C. et al. The development of a standardized neighborhood deprivation index. J. Urban Health Bull. New York Acad. Med. 83(6), 1041–1062 (2006).
Pornet, C. et al. Construction of an adaptable European transnational ecological deprivation index: the French version. J. Epidemiol. Commun. Health 66(11), 982–989 (2012).
Pujol, S. et al. Urban ambient outdoor and indoor noise exposure at home: a population-based study on schoolchildren. Appl. Acoust. 73, 741–750 (2012).
Pujol, S. et al. Indoor noise exposure at home: a field study in the family of urban schoolchildren. Indoor Air 24(5), 511–520 (2014).
Pujol, S. et al. Association between ambient noise exposure and school performance of children living in an urban area: a cross-sectional population-based study. J. Urban Health. 91(2), 256–271 (2014).
Tenailleau, Q. M. et al. Assessing residential exposure to urban noise using environmental models: Does the size of the local living neighborhood matter?. J. Expo. Sci. Environ. Epidemiol. 25(1), 89–96 (2015).
Tenailleau, Q. M., Mauny, F., Joly, D., François, S. & Bernard, N. Air pollution in moderately polluted urban areas: How does the definition of “neighborhood” impact exposure assessment?. Environ. Pollut. 206, 437–448 (2015).
EEA (2011). Air Quality in Europe. European Environment Agency, Luxemburg (Luxemburg) (Technical report).
World Health Organization Europe (2003). Health Aspects of Air Pollution with Particulate Matter, Ozone and Nitrogen Dioxide. World Health Organization – Regional Office for Europe. Copenhagen (Denmark).
EEA (2011). The Application of Models under the European Union's Air Quality Directive. European Environmental Agency, Luxembourg (Luxembourg).
Commission, E. Directive 2002/49/EC of the European Parliament and of the Council of 25 June 2002 (European Parliament, 2002).
Ballester, F. et al. Air pollution exposure during pregnancy and reduced birth size: a prospective birth cohort study in Valencia, Spain. Environ. Health. 29(9), 6 (2010).
Liu, S., Krewski, D., Shi, Y., Chen, Y. & Burnett, R. T. Association between gaseous ambient air pollutants and adverse pregnancy outcomes in Vancouver, Canada. Environ. Health Perspect. 111(14), 1773–1778 (2003).
Liu, S., Krewski, D., Shi, Y., Chen, Y. & Burnett, R. T. Association between maternal exposure to ambient air pollutants during pregnancy and fetal growth restriction. J. Expo. Sci. Environ. Epidemiol. 17(5), 426–432 (2007).
Nobles, C. J. et al. Ambient air pollution and fetal growth restriction: Physician diagnosis of fetal growth restriction versus population-based small-for-gestational age. Sci. Total Environ. 650(Pt 2), 2641–2647 (2019).
Wang, Q. et al. Seasonal analyses of the association between prenatal ambient air pollution exposure and birth weight for gestational age in Guangzhou, China. Sci. Total Environ. 1(649), 526–534 (2019).
Wang, Q. et al. Interaction of Air Pollutants and Meteorological Factors on Birth Weight in Shenzhen, China. Epidemiology 30(Suppl 1), S57–S66 (2019).
Stieb, D. M. et al. A national study of the association between traffic-related air pollution and adverse pregnancy outcomes in Canada, 1999–2008. Environ. Res. 148, 513–526 (2016).
Capobussi, M. et al. Air pollution impact on pregnancy outcomes in Como, Italy. J. Occup. Environ. Med. 58(1), 47–52 (2016).
Gehring, U. et al. Traffic-related air pollution and pregnancy outcomes in the Dutch ABCD birth cohort study. Occup. Environ. Med. 68(1), 36–43 (2011).
Hannam, K., McNamee, R., Baker, P., Sibley, C. & Agius, R. Air pollution exposure and adverse pregnancy outcomes in a large UK birth cohort: use of a novel spatio-temporal modelling technique. Scand. J. Work Environ. Health. 40(5), 518–530 (2014).
Poirier, A. et al. Maternal exposure to air pollution and adverse birth outcomes in Halifax, Nova Scotia. J. Occup. Environ. Med. 57(12), 1291–1298 (2015).
Dzhambov, A. M. & Lercher, P. Road traffic noise exposure and birth outcomes: an updated systematic review and meta-analysis. Int. J. Environ. Res. Public Health. 16(14), 2522 (2019).
Dzhambov, A. M., Markevych, I. & Lercher, P. Associations of residential greenness, traffic noise, and air pollution with birth outcomes across Alpine areas. Sci. Total Environ. 15(678), 399–408 (2019).
Hao, J. et al. Association between ambient air pollution exposure and infants small for gestational age in Huangshi, China: a cross-sectional study. Environ. Sci. Pollut. Res. Int. 26(31), 32029–32039 (2019).
Raz, R., Kioumourtzoglou, M. A. & Weisskopf, M. G. Live-birth bias and observed associations between air pollution and autism. Am. J. Epidemiol. 187(11), 2292–2296 (2018).
Blondel B, Kermarrec M. Perinatal survey 2010. Births in 2010 and their evolution since 2003. 2011, accessed 1st December 2020. http://www.epopé-inserm.fr/wp-content/uploads/2015/01/Rapport-Naisances-ENP2010.pdf.
Clifton, V. L. Review: Sex and the human placenta: mediating differential strategies of fetal growth and survival. Placenta 31(Suppl), S33–S39 (2010).
Zeghnoun, A. & Dor, F. Description du budget espace-temps et estimation de l’exposition de la population française dans son logement. Saint-Maurice (Fra): Institut de veille sanitaire; 2010, accessed 1st December 2020. https://www.santepubliquefrance.fr/determinants-de-sante/exposition-a-des-substances-chimiques/monoxyde-de-carbone/documents/rapport-synthese/description-du-budget-espace-temps-et-estimation-de-l-exposition-de-la-population-francaise-dans-son-logement.
Nethery, E., Leckie, S. E., Teschke, K. & Brauer, M. From measures to models: an evaluation of air pollution exposure assessment for epidemiological studies of pregnant women. Occup. Environ. Med 65(9), 579–586 (2008).
Nethery, E., Teschke, K. & Brauer, M. Predicting personal exposure of pregnant women to traffic-related air pollutants. Sci. Total. Environ. 395(1), 11–22 (2008).
Acknowledgements
The authors acknowledge the local associations for air quality monitoring: Atmo Franche-Comté and Atmosf’air Bourgogne; O. Allemandet, A. Bourtembourg, A. Dussaucy, A. Eckmann, M. Puyraveau and M. Vasseur-Barba from the Centre Hospitalier Régional Universitaire (CHRU) of Besançon; J. Besson and S. Belhadj from the CHRU of Dijon; S. Rankin for thorough proofreading of this article; and the Maison des Sciences de l’Homme et de l’Environnement (MSHE USR3124) for assistance in this project. Computations were performed on the supercomputer facilities at the Mésocentre de Calcul de Franche-Comté.
Funding
This work was supported by the Agence De l’Environnement et de la Maitrise de l’Energie (ADEME) [1217C0065] as part of the National Environmental Research and Occupational Health Program 2012 from the French Agency for Food, Environmental and Occupational Health and Safety (ANSES), and the “Fondation de France” [00069262]. This paper presents the results from the second part of the PrémaBQuA project: Preterm Birth and Environmental Multiple Exposure: Noise and Air Urban Areas.
Author information
Authors and Affiliations
Contributions
A.S.M., N.B., F.M. and C.Q. conceived the design of this present study; P.S., G.T. and D.R. provided its implementation and data collection; S.P., M.B., J.D. and H.H. conducted the environmental data collection and analysis; A.S.M., A.L.P. and E.B. conducted statistical analysis; All authors contributed to the interpretation of the results; A.S.M. drafted the manuscript; All authors critically revised the manuscript, read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
Authors ASM, NB, SP, GT, DR, MB, JD, HH, ALP, EB, FM and CQ do not declare any conflict of interest. Author PS received funding from the following commercial companies: Merck Serono, Finox Biotech, Ferring, MSD France SAS, Teva Santé SAS, Allergan France, Gedeon Richter France, Effik SA, Karl Storz Endoscopie France, GE Medical Systems SCS, Laboratoires Genevrier, HAC Pharma, and Ipsen. The authors confirm that none of this funding was used to support the research in this study. There are no patents, products in development or marketed products to declare. This does not alter the authors' adherence to all the ICMJE policies on sharing data and materials.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mariet, AS., Bernard, N., Pujol, S. et al. Association between moderated level of air pollution and fetal growth: the potential role of noise exposure. Sci Rep 11, 11238 (2021). https://doi.org/10.1038/s41598-021-90788-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-90788-1
- Springer Nature Limited
This article is cited by
-
To promote healthy aging, focus on the environment
Nature Aging (2023)