Abstract
To analyze the effectiveness of dexamethasone in preventing upper airway obstruction (UAO) symptoms after extubation and the need of reintubation in critically ill children. Multicenter, prospective, double-blind, randomized, phase IV clinical trial involving five pediatric intensive care units. Children between 1 month and 16 years-of-age intubated for more than 48 h were included. Patients were randomized to receive placebo or dexamethasone 0.25 mg/kg every 6 h, 6-to-12 h prior to extubation (four doses). 48 h follow-up was carried out after extubation. Severity of UAO symptoms (Taussig score, stridor) and reintubation requirement were compared. 147 patients were randomized (10 were excluded), 70 patients received dexamethasone and 67 placebo. No global differences were found in the presence of stridor or moderate-to-severe UAO symptoms (Taussig ≥ 5), but Taussig ≥ 5 was less frequent in patients less than 2 years-of-age treated with steroids (p = 0.014). Median Taussig score was lower in the dexamethasone group 1 h after extubation, p < 0.001. 27 patients required reintubation, 9 due to UAO: 3 (4.3%) in the dexamethasone group and 6 (8.9%) in the placebo group, p = 0.319. In those intubated > 5 days, reintubation due to UAO was higher in the placebo group (2.4% vs. 14.3, p = 0.052). Nebulized epinephrine and budesonide were required more frequently in the placebo group in the first 2 h (p = 0.041) and 1 h (p = 0.02) after extubation, respectively. No relevant side effects were observed. Dexamethasone prior to extubation did not significantly reduce moderate-severe UAO symptoms, except for patients under 2-years of age. Dexamethasone could decrease Taussig score and the need of rescue therapies, as well as reintubation rates in those intubated for more than 5 days.
Similar content being viewed by others
Introduction
Post-extubation upper airway obstruction (UAO) in children is a common complication in up to a third of patients, thereby increasing morbidity and mortality rates1,2,3. Laryngeal edema is more frequent and severe in children than in adults due to the small diameter of their airways2,4. Signs of airway obstruction include stridor and respiratory distress, which may require reintubation in 6–13% of patients1,2,3.
Extubation failure and reintubation has significant effects on the outcomes. It is associated with prolonged length of hospital stay, increase of days of mechanical ventilation and higher healthcare-related costs1,2,5,6. For this reason, it is important to prevent this complication, especially in patients at risk. Predisposing risk factors related to post-extubation UAO include underlying respiratory or neurological disease, prolonged intubation (more than 36-48 h), reintubation and young age (≤ 24 months of age)1,2.
Dexamethasone and other steroids have been used in an attempt to reduce post-extubation UAO, although with heterogeneous dosing regimens. However, contradictory results have been obtained in studies assessing its effectiveness in reducing UAO symptoms and reintubation rates5,6,7,8,9,10, although recent meta-analysis suggest that the use of steroids could be favorable11. In light of the lack of consistent evidence about the effectiveness of steroids in preventing UAO in children, high quality studies are needed.
We designed a randomized study to compare the effectiveness of dexamethasone versus placebo in the prevention of post-extubation UAO in patients intubated for more than 48 h.
The main objective of this study is to analyze the effectiveness of dexamethasone in preventing stridor and reducing the frequency of moderate to severe UAO symptoms in critically ill children compared to placebo. Secondary objectives are to analyze if dexamethasone reduces the incidence of reintubation, the need of treatments for UAO, and to evaluate the adverse effects associated with this treatment.
Material and methods
A multicenter, prospective, double-blind, randomized, placebo-controlled, phase IV clinical trial was designed (EudraCT 2009-016596-30, registered on 25/08/2010). It was conducted in five Spanish pediatric intensive care units that care for general and cardiac critically ill patients. The study protocol12 was developed in accordance with the Standard Protocol (SPIRIT). The study was approved by local Ethics Committee of all participating centers (Fundación para la investigación biosanitaria del Hospital Gregorio Marañón Institutional Review Board, FIBHGM-ECNC003-2010; FIBHGM-10-01). All methods were performed in accordance with the local guidelines and regulations.
Patients
All patients admitted to the participating pediatric intensive care units (PICUs) between 1 month and 16 years of age who required intubation for more than 48 h were eligible. Exclusion criteria were: (1) airway malformations; (2) suspected or confirmed upper airway infection (croup syndrome, tracheitis or epiglottitis); (3) previous surgeries involving upper or lower airway; (4) administration of steroid therapy within the previous seven days; (5) previous extubation failure during PICU stay; (6) parent refusal to participate in the study. Written informed consent was required from parents or guardians before enrollment.
Sample size was established to detect a 50% reduction in the incidence of UAO according to previously published studies (which is estimated to be around 30%). Approximately 110 subjects were expected to be included in each treatment group12.
Screening, randomization and masking
After identification and enrollment candidates were assigned to one of the two therapy groups on a 1:1 ratio by simple randomization. The coordinating center sent a randomization table of patients to each participating center. The Pharmacy Unit of the coordinating center sent treatment-arm assignments labeled and blinded as established in the randomization table for each center. Study medication (dexamethasone or placebo) was also packed and distributed to each participating center by the Pharmacy Unit of the coordinating center. Medication packages were labeled without any information regarding group assignment. Medication ampoules looked the same in both groups.
Study intervention
Patients randomized to the treatment group received intravenous dexamethasone 0.25 mg/kg/dose (to a maximum of 8 mg) every 6 h, for a total of 4 doses. Patients randomized to placebo group received normal saline. The first dose was administered between 6 and 12 h prior to extubation. Dose adjustment, discontinuance or restart of treatment was considered a protocol violation.
On inclusion, demographic and clinical data (diagnosis, intubation characteristics and medical history related with upper airway problems) were collected.
The following variables were recorded at 15 min, 1 h, 2 h, 6 h, 12 h, 24 h and 48 h after extubation: (1) UAO modified Taussig score; (2) need for, and frequency of additional therapies used to treat respiratory distress (nebulized epinephrine, nebulized budesonide, iv steroids; based on the clinical criteria of the attending physician); (3) hemodynamic and respiratory parameters; (4) arterial pCO2 and pO2 and glucose level (blood samples were only drawn if they were needed for clinical purpose); (5) need for reintubation, time point and cause; (6) presence of secondary effects attributable to study treatment: hypertension, hyperglycemia, digestive bleeding and occurrence of infection.
The primary outcome was the reduction in the incidence of moderate to severe UAO symptoms and the reduction of the occurrence of stridor within 48 h after extubation. Moderate UAO symptoms were considered as the presence of stridor or Taussig score ≥ 5. The secondary end points were the need of reintubation and the appearance of adverse effects associated with dexamethasone.
Statistical analysis
Details about sample size determination are specified in the published study protocol12. Continuous variables are presented as mean values and standard deviation (SD), or as median values and interquartile range (IQR), depending on result of Kolmogorov–Smirnov test. Categorical variables are expressed as frequencies and percentages.
Since outcome assessment is not possible if the patient does not undergo extubation, a per-protocol analysis was performed. All patients involved in protocol study violation were excluded from the analysis. The association between categorical variables were assessed by Chi-squared or Fisher test. Continuous variables were compared by Student's t-test or Mann–Whitney U test. A linear mixed model was developed to compare the evolution over time of the different variables, considering each individual as a random effect. Follow-up time, dexamethasone treatment and their interaction over time were considered as fixed effects with compound symmetry covariance structure. Linear mixed models estimate fixed and random effects and are especially useful when the dependent variable is binary and involves repeated measures. In a linear mixed model, we obtain a p value indicating the probability of finding a similar behavior of a continuous variable measured repeatedly in two different groups over time. When the comparison between groups in a linear mixed model achieves statistical significance (p < 0.05), a different evolution for that variable between both groups over time is considered to be present. Secondly, both groups are compared at each timepoint, obtaining specific p values for every timepoint comparison. Linear mixed models can show different evolution of variables over time between groups despite values of variables at each timepoint do not show differences (when variables do not evolve parallel over time). Conversely, linear mixed models can also establish a comparable behavior over time of two variables despite statistically significant differences are present at different timepoints when both variables evolve in parallel form. These models have great flexibility and power to study variables measured repeatedly over time with missing values13,14.
A subgroup analysis in children aged younger than one year and two years old and patients with length of intubation of more than five days were additionally performed. Statistical analysis was performed using SPSS version 20 (SPSS Inc, Chicago, USA).
Results
Study population
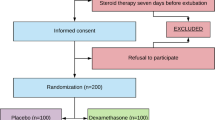
From February of 2013 to February of 2020, 2746 were assessed for eligibility. 293 met the inclusion criteria and 147 patients were randomized (Fig. 1 shows CONSORT flow diagram). There was balance between centers and treatment groups. Ten patients were excluded due to study protocol violations: in 6 children extubation was delayed, in four patients the data collection was inadequate. One hundred and thirty-seven patients (137) were managed per protocol. Seventy (51.1%) received dexamethasone and 67 (48.9%) placebo. Baseline characteristics before extubation are shown in Table 1. There were no statistical differences between groups except for heart rate and blood glucose level prior to extubation.
Primary and secondary outcomes
No differences were found in the presence of stridor or moderate-to-severe UAO symptoms (Taussig ≥ 5) between treatment groups (see Table 2). No differences were found in the maximum Taussig score (registered at any evaluation point) between dexamethasone and placebo group (Table 2). Evolution of Taussig score over time in both treatment groups was compared using a linear mixed model. Global evolution was different between groups (p = 0.023, Fig. 2). Additionally, comparison of Taussig scores at the different evaluation timepoints is shown in Table 3. Taussig score 1 h after extubation was lower in the dexamethasone group compared to the placebo group, p < 0.001. Evolution over time of the other variables (heart rate, mean arterial pressure, glycemia, pCO2, and respiratory rate) introduced in the linear mixed model was comparable between groups except for glycemia (p < 0.001, Fig. 2). Individual comparisons at the different timepoints for the different variables are shown in Additional file 3: Table S1.
Linear mixed model comparing evolution over time of estimated means and 95% CI after extubation of respiratory (Taussig score, arterial CO2 paCO2 and respiratory rate (RR)), hemodynamic and metabolic parameters (heart rate, mean arterial pressure (MAP) and glycemia). Significant differences in different moments of follow-up (p < 0.05) were marked as *.
Regarding the need of reintubation, 27 patients required reintubation for various reasons, without differences between groups. Nine (33.3%) of these 27 patients were reintubated specifically due to UAO obstruction: 3 (4.3%) in patients treated with dexamethasone and 6 (8.9%) in the placebo group (Table 2).
A mixed model approach was used to analyze the evolution over time after extubation of several respiratory and hemodynamic parameters. No differences were found in the evolution of respiratory rate and paCO2 (p = 0.402 and p = 0.087 respectively), Fig. 2. Regarding hemodynamic parameters (Fig. 2), the evolution of heart rate (p = 0.373) and mean arterial pressure (p = 0.077) throughout the follow-up was not different between groups. Nevertheless, mean arterial pressure was higher in the dexamethasone group from 2 to 12 h after extubation (p < 0.05) and heart rate was higher in the placebo group from extubation to 6 h of follow-up (p < 0.05).
The need for rescue therapies for UAO within 48 h from extubation is shown in Table 4. The use of nebulized epinephrine in the first 2 h after extubation was needed in 16.9% of placebo patients versus 5.8% in the dexamethasone group (p = 0.041). Nebulized budesonide was used more frequently in the placebo group, 7.5% versus 0% in the first hour after extubation (p = 0.02) and 9.4% versus 1.5% at 12 h after extubation (p = 0.05). Intravenous dexamethasone (additional to study medication) in the first hour after extubation was required in 7 patients (10.4%) in the placebo group and in one patient (1.4%) in the dexamethasone group (p = 0.03).
Concerning adverse effects, blood glucose level was higher in the dexamethasone group at the baseline measurement and during the first 6 h of follow-up, p < 0.05 (Fig. 2), but it decreased and fell to a similar level as in the placebo group at 12 h after extubation. Only one patient, receiving steroids, required insulin therapy due to hyperglycemia. No bleeding, infection, hypertension or other adverse events were observed in the treatment group. PICU length of stay was similar in both groups (23.8 days with dexamethasone vs. 23.4 days with placebo, p = 0.906). None of the patients died.
Subgroups analysis
Analysis of subgroups of higher risk of UAO (patients younger than two years old) and those patients intubated for more than 5 days is shown in Table 2. Moderate-to-severe UAO symptoms were less frequent in patients less 2 years of age treated with steroids (Table 2). There were no global differences in the presence of stridor between treatments in the different subgroups. Nevertheless, the presence of stridor was significantly different at some time-points: twelve hours after extubation it was present in the 20.9% in the control group versus 4.8% in the treatment group in children younger than two years old (p = 0.05). In patients with a length of intubation of more than five days, stridor was more frequent in the placebo group one hour after intubation (34.1% vs. 15.6%, p = 0.045).
In patients with an intubation of more than five days, reintubation due to UAO obstruction was higher in placebo group than dexamethasone group, with almost significant differences (Table 2).
Discussion
Intubation and invasive mechanical ventilation are common practices in pediatric intensive care units. The patients requiring intubation are at high risk of presenting complications of the upper airway. UAO symptoms include stridor and respiratory distress, and the most severe consequence is the need of reintubation. Extubation failure has an important impact on prognosis of patients admitted to PICU2,4, increasing mortality, length of hospital stay, duration of mechanical ventilation and hospitalization costs1,2,5,6.
Corticosteroids, due to their anti-inflammatory effects, have been previously used to prevent UAO, but the results of different studies has been contradictories5,6,7,8,9,10. Most of studies were performed in adults. Steroids reduce incidence of upper airway complications and reintubations in high risk patients in a meta-analysis of adult patients7. Studies in pediatric patients are heterogeneous, with small sample sizes, involving different ages, diagnoses, doses and timings of steroids therapy4,7.
Our study showed that dexamethasone decreased the UAO severity evaluated using Taussig score over time and the need of rescue treatments for UAO. Moreover, stridor was less frequent in children younger than two years old receiving dexamethasone and in those with prolonged intubation (more than 5 days). Statistically significant decrease of reintubation rates was not reached, although the incidence was the half in patients treated with dexamethasone, but we found a significant reduction in reintubation due to UAO in those intubated for more than 5 days.
Some authors reported a decrease in the incidence of stridor5,8, whereas two studies showed a reduction in the incidence of reintubation8,9,15. A study in neonates found that dexamethasone reduces the risk for reintubation10. In contrast, this benefit has not been proven in other studies in children16 and neonates17.
There are few randomized double-blind studies performed in children3,5,8,9,18,19 (one include adults and children5), and our study is one of the largest (see Table 5). Two meta-analysis were published in 20094,20. The review by Khemani et al. concluded that dexamethasone prior to extubation in children reduces the incidence of stridor, but the evidence is insufficient to conclude that rates of re-intubation are reduced4. The meta-analysis by McCaffrey et al.20 concluded that corticosteroids reduce laryngeal edema and importantly reduce the incidence of extubation failure in critically ill patients of all ages. This study included one more randomized, double-blind study with prednisolone in patients with croup21. A posterior study by Baranwal et al.3 compared two regimens in high-risk pediatric patients versus placebo: starting dexamethasone therapy 6 h versus 24 h before extubation. The authors found that the 24-h regimen significantly reduced the incidence of UAO, as compared to the 6-h regimen.
Our results do not provide enough evidence to recommend systematic administration of dexamethasone prior to extubation in critically ill children, except for intubations of more than 5 days. Our results suggest that dexamethasone reduces UAO symptoms, but the number of patients included in the study was not sufficient to find significant differences in the primary outcomes. Expected sample size (110 patients per treatment arm) could not finally be achieved due to difficulties during recruitment of patients and expiration of study period. Beside this, incidence of UAO in our study population was found to be much lower of previously published studies. This fact implies that the number of patients needed to identify differences between treatment groups would be much higher than initially expected (more than 450 patients per treatment arm). Thus, only a much larger multicenter (and probably international) randomized study could reach this number of patients.
Similar to previous studies4 we did not find significant side effects related to the use of corticosteroids. Blood glucose level was slightly higher in the dexamethasone group during the first hours after extubation, but without clinical relevancy. Blood glucose levels were comparable in both groups 12 h after discontinuation of dexamethasone administration. Similarly, baseline heart rate was higher in the dexamethasone-treated group from baseline to the first six hours of follow-up. No other clinically relevant cardiovascular differences were observed. Therefore, corticosteroids would be safe for this indication at the tested dose.
Limitations
This study has several limitations. Although it was a multicenter, randomized, double-blind study, the number of patients included was not sufficient to reach statistical significance. Some of the inclusion criteria such as previous administration of steroids and anticipation of the first dose 6–12 h before extubation may have hindered the inclusion of a higher number of patients. Furthermore, some patients could not be recruited due to research collaborator availability issues. Besides, the authorized period for the study expired before the estimated sample size was reached. The study excluded high risk patients with croup and upper airway surgery which might affect the generalizability of this study to those populations.
The primary outcome as a clinical score of UAO symptoms could be subjective, in contrast to the objective secondary outcome of extubation failure.
Participants were not homogeneous in age, diagnosis, duration of mechanical ventilation, although both groups were comparable at baseline. However, baseline data were obtained prior to extubation and not prior starting the treatment. We cannot exclude the influence of other factors, as neurologic, cardiac, or respiratory disturbances, on the post-extubation respiratory failure. Although the stratified subgroup analysis by age was planned, the analysis in patients intubated for more than 5 days was post-hoc.
Finally, rescue therapies used to treat UAO symptoms were not protocolized and there could be differences between hospitals and physicians' practices.
Conclusions
Dexamethasone prior to extubation did not significantly reduce moderate-severe UAO symptoms, except for patients under 2-years of age. There were no differences in stridor. Dexamethasone could decrease Taussig score and the need of rescue therapies. It could also reduce reintubation rates in those intubated for more than 5 days. Further large multicenter studies are needed to assess if corticosteroid could reduce UAO severity and reintubation incidence in critically ill children.
References
Kurachek, S. C. et al. Extubation failure in pediatric intensive care: A multiple-center study of risk factors and outcomes. Crit. Care Med. 31, 2657–2664 (2003).
Khemani, R. G. et al. Evaluating risk factors for pediatric post-extubation upper airway obstruction using a physiology-based tool. Am. J. Respir. Crit. Care Med. 193, 198–209 (2016).
Baranwal, A. K., Meena, J. P., Singhi, S. C. & Muralidharan, J. Dexamethasone pretreatment for 24 h versus 6 h for prevention of postextubation airway obstruction in children: A randomized double-blind trial. Intensive Care Med. 40, 1285–1294 (2014).
Khemani, R. G., Randolph, A. & Markovitz, B. Corticosteroids for the prevention and treatment of post-extubation stridor in neonates, children and adults. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD001000.pub3 (2009).
Malhotra, D., Gurcoo, S., Qazi, S. & Gupta, S. Randomized comparative efficacy of dexamethasone to prevent postextubation upper airway complications in children and adults in ICU. Indian J. Anaesth. 53, 442–449 (2009).
Fan, T. et al. Prophylactic administration of parenteral steroids for preventing airway complications after extubation in adults: Meta-analysis of randomised placebo controlled trials. BMJ 337, 1088–1091 (2008).
Kuriyama, A., Umakoshi, N. & Sun, R. Prophylactic corticosteroids for prevention of post-extubation stridor and reintubation in adults: A systematic review and meta-analysis. Chest 151, 1002–1010 (2017).
Anene, O., Meert, K. L., Uy, H., Simpson, P. & Sarnaik, A. P. Dexamethasone for the prevention of postextubation airway obstruction: a prospective, randomized, double-blind, placebo-controlled trial. Crit. Care Med. 24, 1666–1669 (1996).
Tellez, D. W. et al. Dexamethasone in the prevention of postextubation stridor in children. J. Pediatr. 118, 289–294 (1991).
Couser, R. J. et al. Effectiveness of dexamethasone in preventing extubation failure in preterm infants at increased risk for airway edema. J. Pediatr. 121, 591–596 (1992).
Kimura, S., Ahn, J. Y. B., Takahashi, M., Kwon, S. & Papatheodorou, S. Effectiveness of corticosteroids for post-extubation stridor and extubation failure in pediatric patients: A systematic review and meta-analysis. Ann Intensive Care 10, 1–9 (2020).
Manrique, G. et al. Effectiveness of steroids versus placebo in preventing upper airway obstruction after extubation in critically ill children: Rationale and design of a multicentric, double-blind, randomized study. Trials 21, 4–11 (2020).
Krueger, C. & Tian, L. A comparison of the general linear mixed model and repeated measures ANOVA using a dataset with multiple missing data points. Biol. Res. Nurs. 6, 151–157 (2004).
Burton, P., Gurrin, L. & Sly, P. Extending the simple linear regression model to account for correlated responses: An introduction to generalized estimating equations and multi-level mixed modelling. Stat. Med. 17, 1261–1291 (1998).
Veldhoen, E. S. et al. Post-extubation stridor in respiratory syncytial virus bronchiolitis: Is there a role for prophylactic dexamethasone?. PLOS ONE 12, 1–10 (2017).
Saleem, A. F., Bano, S. & Haque, A. Does prophylactic use of dexamethasone have a role in reducing post extubation stridor and reintubation in children?. Indian J. Pediatr. 76, 555–557 (2009).
Ferrara, T. B., Georgieff, M. K., Ebert, J. & Fisher, J. B. Routine use of dexamethasone for the prevention of postextubation respiratory distress. J. Perinatol. 9, 287–290 (1989).
Harel, Y. et al. Extubation failure due to post-extubation stridor is better correlated with neurologic impairment than with upper airway lesions in critically ill pediatric patients. Int. J. Pediatr. Otorhinolaryngol. 39, 147–158 (1997).
de Carvalho, H. T. et al. Use of dexamethasone to prevent extubation failure in pediatric intensive care unit: A randomized controlled clinical trial. J. Pediatr. Intensive Care https://doi.org/10.1055/s-0040-1719044 (2020).
McCaffrey, J. et al. Corticosteroids to prevent extubation failure: A systematic review and meta-analysis. Intensive Care Med. 35, 977–986 (2009).
Tibballs, J., Shann, F. A. & Landau, L. I. Placebo-controlled trial of prednisolone in children intubated for croup. Lancet 340, 745–748 (1992).
Acknowledgements
We thank the healthcare professionals of the Pediatric Intensive Care Units and Pharmacy Units of the participating centers.
Funding
RETICS funded by the PN I+D+I 2013-2016 (Spain), ISCIII- Sub-Directorate General for Research Assessment and Promotion and the European Regional Development Fund (ERDF), ref. RD16/0022.
Author information
Authors and Affiliations
Contributions
J.L.-H. conceived the study, participated in the study design, and revised the manuscript. R.G. participated in the study design and revised the manuscript and is responsible for data collection. G.M. and L.B.-L. drafted the manuscript and are responsible for data collection. C.R., Z.M.-C., J.G., and A.R.-N. are responsible for data collection. C.M. and S.M. participated in the study design and are responsible for the labeling and blinding of medication kits. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Butragueño-Laiseca, L., Manrique Martín, G., González Cortés, R. et al. Multicenter randomized clinical trial comparing dexamethasone versus placebo in preventing upper airway obstruction after extubation in critically ill children. Sci Rep 12, 4336 (2022). https://doi.org/10.1038/s41598-022-08178-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-08178-0
- Springer Nature Limited
This article is cited by
-
What’s new in paediatric ventilator liberation?
Intensive Care Medicine (2022)