Abstract
Epigenetic alterations have emerged as fundamental players in development and progression of breast cancer (BC). A hypoxic tumour microenvironment regulates the stemness phenotype in breast cancer stem cells (BCSCs). The aim of this study was to investigate Znhit1 and HIF-2α gene expression in breast cancer tissues as well as their relation to CSCs markers LGR5, ALDH1A1 and β-catenin in tissue and serum of BC patients. The present study included 160 females divided into two groups, group I: 80 healthy females served as control group and group II: 80 breast cancer patients. Gene expression of tissue Znhit1 and HIF-2α was determined by qRT-PCR. Tissue and serum ALDH1A1, LGR5 and β-catenin levels were determined by ELISA. We found that gene expression of Znhit1 was significantly downregulated in BC tissues. Moreover, it was significantly negatively correlated with clinical stage and β-catenin levels in BC patients. Regarding HIF-2α, gene expression of HIF-2α was significantly upregulated in BC tissues. Moreover, it was significantly positively correlated with Her-2/neu expression and β-catenin levels in BC patients. Based upon our results, Znhit1 and HIF-2α may serve as novel therapeutic targets for BC therapy. Additionally, each of serum ALDH1A1, LGR5 and β-catenin may play a crucial role in non-invasive detection of BC with a high specificity and sensitivity.
Similar content being viewed by others
Introduction
Breast cancer accounts for 24.5% of all cancers diagnosed in females and it is the first leading cause of cancer-related mortality in females worldwide1. Epigenetics is the study of heritable changes in gene expression that do not affect primary DNA sequence2. Failure of the proper maintenance of heritable epigenetic marks can lead to abnormal induction or suppression of several signaling pathways and results in many diseases including cancer3. Zinc finger HIT-type containing 1 (Znhit1), is a subunit of sucrose non-fermenting 2 (Snf2)-related cyclic adenosine monophosphate response element binding protein-binding protein (CBP) activator protein (SRCAP) complex, which specifically can alter chromatin structure by promoting the inclusion of H2AZ-H2B dimers into nucleosomes4. Znhit1 is overexpressed in response to genotoxic stresses and activating transcription of numerous p53 target genes5. However, the function of Znhit1 in cancer remains largely unknown.
A hypoxic tumour microenvironment regulates the stemness phenotype in breast cancer stem cells (BCSCs). The activation of Hypoxia-inducible factors (HIFs) is one of the fundamental mechanisms of stemness maintenance triggered by hypoxia6. HIFs are made up of a heterodimer of an O2-regulated α subunit (HIF-1α, HIF-2α and HIF-3α) and a constitutively expressed β subunit7. Despite their great homology, HIF-1α and HIF-2α regulate diverse target genes and have different roles in invasion, proliferation and chemotherapy resistance8.
Accumulating evidence suggests that the presence of a small number of immature cells, known as BCSCs appears to be the most likely cause of tumour progression, metastasis and drug resistance9,10. Aldehyde dehydrogenase 1+ (ALDH1+) and leucine rich repeat containing G protein coupled receptor 5+ (LGR5+) are the most commonly used biomarkers to identify the BCSC phenotype11,12,13.
Aldehyde dehydrogenase 1 is an intracellular enzyme catalyzing the oxidation of intracellular aldehydes. It contributes to differentiation of normal and tumour stem cell14. ALDH1 proteins consist of 3 main isozymes namely ALDH1A1, ALDH1A2 and ALDH1A3 with ALDH1A1 being the most specific which is largely reported to be related to poor prognosis15.
Leucine-rich repeat-containing G protein-coupled receptor 5 is a well-recognized marker for adult SCs as well as CSCs. LGR5 promotes BC progression and CSCs maintenance partially via stimulation of Wnt/β-catenin signaling pathway16,17. BCSCs with activated Wnt/β-catenin signaling pathway are substantially more tumorigenic than those without and blocking Wnt/β-catenin pathway inhibits BC metastasis by suppressing BCSCs phenotype18. β-catenin is the major mediator of Wnt/β-catenin signaling pathway. It is a multifunctional protein that either associates with cadherins at the cell membrane to regulate cellular adhesion or translocates to the nucleus, where it activates transcription of Wnt target genes19.
The aim of this study was to investigate Znhit1 and HIF-2α gene expression in breast cancer tissues as well as their relation to CSCs markers LGR5, ALDH1A1 and β-catenin in tissue and serum of BC patients. Therefore, this study can help to find new target against BC progression and reveal a possible molecular mechanism for the Znhit1and HIF-2α associated regulation in BC, providing innovate therapeutic strategies for the treatment of BC.
Results
Znhit1 gene expression in BC tissues
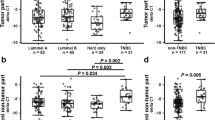
The range and mean ± SE fold change of Znhit1 gene expression in BC tissues and adjacent normal breast tissues were shown in Table 1 and illustrated in Fig. 1. The statistical analyses of these results were also shown in Table 1. As presented in Table 1 the mean fold change of Znhit1 gene expression in BC tissues was statistically significantly lower than that in adjacent normal breast tissues (P = 0.009).
HIF-2α gene expression in BC tissues
The range and mean ± SE fold change of HIF-2α gene expression in BC tissues and adjacent normal breast tissues were shown in Table 1 and illustrated in Fig. 2. The statistical analyses of these results were also shown in Table 1. As presented in Table 1 the mean fold change of HIF-2α gene expression in BC tissues was statistically significantly higher than that in adjacent normal breast tissue (P = 0.02).
ALDH1A1 levels (pg/ml) in BC tissues
The range and mean ± SE of ALDH1A1 (pg/ml) in BC tissues and adjacent normal breast tissues were shown in Table 2 and illustrated in Fig. 3a. The statistical analyses of these results were also shown in Table 2. As presented in Table 2 the mean value of ALDH1A1 in BC tissues was statistically significantly higher than that in adjacent normal breast tissues (P < 0.001).
ALDH1A1 levels (pg/ml) in serum of BC patients
The range and mean ± SE of serum ALDH1A1 (pg/ml) in control group and BC patients group were shown in Table 3 and illustrated in Fig. 3b. The statistical analyses of these results were also shown in Table 3. As presented in Table 3 the mean value of serum ALDH1A1 in BC patients group was statistically significantly higher than that in control group (P < 0.001).
LGR5 levels (pg/ml) in BC tissues
The range and mean ± SE of LGR5 (pg/ml) in BC tissues and adjacent normal breast tissues were shown in Table 2 and illustrated in Fig. 4a. The statistical analyses of these results were also shown in Table 2. As presented in Table 2 the mean value of LGR5 in BC tissues was statistically significantly higher than that in adjacent normal breast tissues (P < 0.001).
LGR5 levels (pg/ml) in serum of BC patients
The range and mean ± SE of serum LGR5 (pg/ml) in control group and BC patients group were shown in Table 3 and illustrated in Fig. 4b. The statistical analyses of these results were also shown in Table 3. As presented in Table 3 the mean value of serum LGR5 in BC patients group was statistically significantly higher than that in control group (P < 0.001).
β-catenin levels (pg/ml) in BC tissues
The range and mean ± SE of β-catenin (pg/ml) in BC tissues and adjacent normal breast tissues were shown in Table 2 and illustrated in Fig. 5a. The statistical analyses of these results were also shown in Table 2. As presented in Table 2 the mean value of β-catenin in BC tissues was statistically significantly higher than that in adjacent normal breast tissues (P < 0.001).
β-catenin levels (pg/ml) in serum of BC patients
The range and mean ± SE of serum β-catenin (pg/ml) in control group and BC patients group were shown in Table 3 and illustrated in Fig. 5b. The statistical analyses of these results were also shown in Table 3. As presented in Table 3 the mean value of serum β-catenin in breast cancer patients group was statistically significantly higher than that in control group (P < 0.001).
Correlation between different biochemical studied parameters and clinicopathological parameters in BC patients
Znhit1 gene expression was statistically significantly negatively correlated with clinical stage (r = − 0.336, P = 0.037), HIF-2α gene expression was statistically significantly positively correlated with Her-2/neu expression (r = 0.601, P < 0.001) and LGR5 either in tissue or serum was statistically significantly negatively correlated with PR status (r = − 0.300, P = 0.044 and r = − 0.344, p = 0.043; respectively), while there was insignificant correlation between these parameters and the other clinicopathological characters.
Correlation between different biochemical studied parameters in BC patients
As presented in Fig. 6a,b, Znhit1 gene expression was statistically significantly negatively correlated with tissue and serum β-catenin (r = − 0.348, P = 0.04 and r = − 0.352, P = 0.038; respectively), while it was insignificantly negatively correlated with ALDH1A1 and LGR5 levels in tissue and serum of BC patients. As presented in Fig. 7, HIF-2α gene expression was statistically significantly positively correlated with tissue β-catenin (r = 0.328, P = 0.04), while it was insignificantly correlated with serum β-catenin, tissue and serum ALDH1A1 and LGR5. Tissue ALDH1A1 was statistically significantly positively correlated with serum ALDH1A1, tissue LGR5 and tissue β-catenin (r = 0.489, P = 0.003, r = 0.338, P = 0.04 and r = 0.391, P = 0.02; respectively). Serum ALDH1A1 was statistically significantly positively correlated with serum LGR5 and serum β-catenin (r = 0.628, P < 0.001 and r = 0.550, P = 0.001; respectively). Tissue LGR5 was statistically significantly positively correlated with serum LGR5 and tissue β-catenin (r = 0.466, P = 0.005 and r = 0.419, P = 0.012; respectively). Serum LGR5 was statistically significantly positively correlated with serum β-catenin (r = 0.606, P < 0.001). Tissue β-catenin was statistically significantly positively correlated with serum β-catenin (r = 0.397, P = 0.018).
Diagnostic performance of ALDH1A1, LGR5 and β-catenin in serum of breast cancer patients
The ROC curve analysis was used to compare the diagnostic values of the studied parameters. The higher area under the ROC curve (AUC) corresponds to a better diagnostic test. As presented in Fig. 8, serum ALDH1A1 had significant AUC (0.986, P < 0.001) with 100% specificity and 94% sensitivity at cut off value ≥ 21.5 pg/ml, serum LGR5 had significant AUC (0.99, P < 0.001) with 90% specificity and 97% sensitivity at cut off value ≥ 28.75 pg/ml and serum β-catenin had significant AUC (0.991, P < 0.001) with 90% specificity and 94% sensitivity at cut off value ≥ 166 pg/ml.
Discussion
The results of our study indicated that the gene expression of Znhit1 in BC tissues was statistically significantly lower than that in adjacent normal breast tissues. Moreover, our results revealed statistically significant negative correlation between gene expression of Znhit1 in BC tissues and clinical stage of patients, suggesting the role of Znhit1 repression in tumor progression. Similarly, Sun et al. demonstrated that expression of Znhit1 gene was significantly decreased in acute myeloid leukemia (AML) patients compared with healthy blood cell samples20. Znhit1 has been shown to trigger cell cycle arrest as well as play an important role in controlling cell proliferation21. According to Cuadrado et al. study which demonstrated that under normal growing conditions, cyclin G1 stimulates degradation of Znhit1 and can possibly regulate its expression levels5, we suggest that cyclin G1 is overexpressed in cancer cells leading to downregulaion of Znhit1 expression.
Moreover, the current study revealed that Znhit1 gene expression in BC tissues was statistically significantly negatively correlated with β-catenin levels in tissue and serum of BC patients. This finding suggests that downregulation of Znhit1 may contribute to induction and maintenance of stemness in BC. To the best of our knowledge, the present study is the first study revealed this correlation. Our explanation of this negative correlation that the downregulation of Znhit1 expression in BC tissues may downregulate Phosphatase and tensin homolog (PTEN) expression along with activation of Phosphoinositide 3-kinase (PI3K)/A serine threonine protein kinase (Akt) signaling pathway20, PI3K/Akt activation can stimulate the Wnt/β-catenin signaling pathway via the Glycogen synthase kinase 3 beta phosphorylation by the phosphorylated Akt1/2, preventing β-catenin destruction complex formation22.
Regarding HIF-2α, the results of our study indicated that gene expression of HIF-2α in BC tissues was statistically significantly higher than that in adjacent normal breast tissues. Moreover, gene expression of HIF-2α was statistically significantly positively correlated with Her-2/neu expression in human BC tissues. These results were in accordance with in vitro study conducted by Jarman et al. who revealed that Her-2 overexpression in MCF-7 cells leads to upregulation of HIF-2α in normoxia and in response to hypoxia. In addition, they showed that upregulation of Her-2 in this model promotes the expression of many hypoxic response genes both in normoxia and in response to acute (24 h) and chronic (10 weeks) hypoxia, indicating that growth factor signaling may increase the cellular response to hypoxia through HIF-2α23. Additionally, our results revealed statistically significant positive correlation between HIF-2α gene expression and β-catenin levels in human BC tissues, suggesting the role of HIF-2α in driving stemness in BC. This finding is consistent with the study of Yan et al. who demonstrated that upregulation of HIF-2α in MCF-7 and MDA-MB-231 BC cells induced stemness and promoted paclitaxel resistance by stimulating the Notch and Wnt signaling pathways. Wnt pathway activation was related to overexpression of β-catenin24. To the best of our knowledge, this is the first study showed that HIF-2α expression is related to Her-2/neu expression and the expression of β-catenin in BC patients.
To elucidate the biological significance of ALDH1A1 and its regulation in BC, we examined the ALDH1A1 levels in tissue and serum of BC patients. The results of this study revealed that the ALDH1A1 levels in tissue and serum of BC patients were statistically significantly higher than that in adjacent normal breast tissue and serum of control subjects respectively. Furthermore, our results revealed statistically significant positive correlation between tissue and serum ALDH1A1 levels in BC patients. These findings suggest that serum ALDH1A1 can be used as biomarker for non-invasive detection of cancer stem cells in BC patients. Our results support study conducted by Gyan et al. who examined the ALDH1A1 protein expression in African BC patients by Immunohistochemistry assay. The results of their study demonstrated that protein expression of ALDH1A1 was high in African BC patients25. Consistent with our results, Rossi et al. showed that serum ALDH1A1 protein levels are statistically higher in patients with non-small cell lung cancer (NSCLC) compared to control group26.
Moreover, our results showed that ALDH1A1 levels were statistically significantly positively correlated with LGR5 and β-catenin levels in tissue and serum of BC patients. These findings suggest that ALDH1A1 expression may be regulated by LGR5 and β-catenin in BC. In line with our findings, Gao et al. demonstrated that the protein expression of ALDH1A1 and LGR5 were significantly related in NSCLC and were correlated with the NSCLC progression and indicated poor prognosis27. Cui et al. indicated that Notch and Wnt/β-catenin signaling pathways that are important in drug resistance were aberrantly induced in stem-like ALDHhigh/CD44+ cells compared with non-stem-like ALDHlow/CD44+ cells in BC28. Furthermore, Sun et al. suggested that ALDH1A1 expression may be regulated by β-catenin in BC29. Collectively, these findings suggest that ALDH1A1 levels may help to identify high-risk BC patients and thus can be considered as potential therapeutic and/or diagnostic target.
The results of our study revealed that LGR5 levels in tissue and serum of BC patients were statistically significantly higher than that in adjacent normal breast tissue and serum of control subjects respectively. Moreover, there was statistically significant positive correlation between tissue and serum LGR5 levels in BC patients. These findings suggest that serum LGR5 will be one of the potential candidate biomarkers for non-invasive detection of cancer stem cells in BC patients. Our results were consistent with previous studies evaluated LGR5 protein expression in BC tissues by Immunohistochemistry assay and observed that LGR5 protein expression was lower in normal breast tissue compared to BC tissue17,30.
Nevertheless, results of our study showed that LGR5 levels in tissue and serum of BC patients were statistically significantly positively correlated with β-catenin levels. This finding supports the notion that LGR5 activates Wnt/β-catenin pathway in BC patients17. This correlation may be explained by recent in vitro study conducted by Chen et al. who demonstrated that a protein kinase A (PKA) activity was increased by LGR5 overexpression and was reduced by suppression of LGR5 in BC cells. Also, expression level of LGR5 and activity of PKA were positively related to activation of β-catenin while negatively related to activation of Glycogen synthase kinase 3 beta in BC cells. These mean that LGR5 stimulates the Wnt/β-catenin pathway in BC cells by activation of PKA31. These findings suggest that LGR5 is more of a molecular marker for CSCs but may also promote cancer cell growth and tumor development in BC. Thus, targeting of LGR5 could thus be a possible therapeutic strategy for BC treatment.
The specific role of β-catenin in breast carcinoma remains unclear, results of our study demonstrated that β-catenin levels in tissue and serum of BC patients were statistically significantly higher than that in adjacent normal breast tissue and serum of control subjects respectively. Moreover, there was statistically significant positive correlation between tissue and serum β-catenin levels in BC patients. These suggest that serum β-catenin levels may be used as non-invasive marker to distinguish BC patients from normal controls. These results were consistent with Xu et al. study which demonstrated that the aberrant membranous expression of β-catenin and the positive nuclear/cytoplasmic expression of β-catenin were detected in 43.5% and 78.1% of the BC tissues, respectively32. Recent study on BC proposed that deregulation of Wnt/β-catenin pathway may be due to downregulation of its negative regulators. This was supported by the finding that the extracellular inhibitor of Wnt/β-catenin pathway expression named secreted frizzled-related protein 1 (SFRP1) is significantly downregulated in many breast tumors and this is related to poor prognosis as well as therapy resistance33.
Our results showed that each of serum β-catenin, LGR5 and ALDH1A1 has high diagnostic efficacy in BC. Similarly, Li et al. study demonstrated that β-catenin levels in serum had high effectiveness in colorectal carcinoma diagnosis and showed that serum β-catenin levels play a role in early detection of colorectal carcinoma34. To the best of our knowledge, the diagnostic efficacy of serum β-catenin, LGR5 and ALDH1A1 in BC not yet reported in any literature.
Conclusion
From our study we can conclude that Znhit1 repression may play a role in tumour progression as well as induction and maintenance of stemness in BC. HIF-2α may play a role in aggressiveness of Her-2-positive BC and driving stemness in BC patients. The elevated levels of β-catenin in BC patients may be due to downregulation of Znhit1 and upregulation of HIF-2α and LGR5 which activate Wnt/β-catenin signaling pathway. Moreover, the elevated levels of β-catenin may lead to upregulation of Wnt target gene ALDH1A1. β-catenin may be a marker of advanced BC and thus may represent a new diagnostic and therapeutic target. Each of serum ALDH1A1, LGR5 and β-catenin may play a crucial role in non-invasive detection of BC with a high specificity and sensitivity.
Recommendations
Further studies are required to explain the mechanism of Znhit1 downregulation in BC to explore new therapeutic agents that upregulate Znhit1 expression in BC. Targeting of HIF-2α in Her-2-positive BC may be an effective therapeutic intervention. Follow up of BC patients involved in our study is required to investigate the role of studied parameters in BC prognosis.
Subjects and methods
This study was approved by ethical committee of Medical Research Institute (MRI)-Alexandria University and was conducted on one hundred sixty females. They were divided into two groups, Group I which included 80 apparently normal healthy female volunteers served as control group and group II which included 80 females of matched age and menstrual state as the previous group having breast carcinoma of clinical stage II–III.
All Patients were chosen from those admitted to Experimental and Clinical Surgery Department and Department of Cancer Management and Research, MRI, Alexandria University. A written informed consents were taken from all contributors in this study, in accordance with the Declaration of Helsinki. Patients who were metastatic at the time of diagnosis and those who were receiving neoadjuvant therapy were excluded from this study.
Patients were subjected to preoperative evaluation by fine needle aspiration cytology to detect the presence of malignancy and clinical examination to detect the site of the tumour and the presence of axillary lymph node infiltration. Patients were subjected to surgery (mastectomy or conservative) and a written informed consent was taken from all patients before surgery. Pathological grading of the malignant tumour, tumour size, vascular invasion, ER and PR status and Her-2/neu expression were confirmed. The clinico-pathological characteristics of BC patients are represented in Table 4.
Tissue sampling
Human BC tissues and adjacent normal breast tissues were taken from patients who underwent surgical resection and were stored at − 80 °C until used for determination of the Znhit1 and HIF-2α gene expression by quantitative reverse transcription polymerase chain reaction (qRT-PCR) as well as ALDH1A1, LGR5 and β-catenin levels in tissue homogenate by enzyme-linked Immunosorbent assay (ELISA).
Blood sampling
5 ml of venous blood was withdrawn from all BC patients (before surgery) and from the control group. Blood samples were allowed to clot for thirty minutes then centrifuged for 10 min at about 3000×g for separating serum. Serum was removed and stored at – 80 ℃ until used for assaying ALDH1A1, LGR5 and β-catenin levels by ELISA.
Gene expression analysis of Znhit1 and HIF-2α
Total RNA was extracted from Human BC tissues and adjacent normal breast tissues using RNeasy Mini Kit (Qiagen, Germany). Using a NanoDrop 2000 Spectrophotometer, the concentration of extracted RNA was measured by measuring the absorbance at 260 nm (A260) and RNA purity was determined by using the ratio of absorbance values at 260 nm and 280 nm (A260/A280), A260/A280 ratio of pure RNA is 1.9–2.1. The expression of both Znhit1 and HIF-2α genes was determined by qRT-PCR using RealMOD™ Green qRT-PCR mix Kit according to manufacturer’s protocols. The Real time PCR results were analyzed using the comparative cycle threshold (ΔΔCt) method for calculating fold change of gene expression as the following:
where, GOI: Genes of Interest (Znhit1 and HIF-2α), Norm Normalizer (GAPDH).
Determination of tissue and serum ALDH1A1 levels
ALDH1A1 levels in serum of all studied individuals and tissue homogenate of BC tissues and adjacent normal tissues were determined by ELISA assay kit according to the manufacturer’s operating instructions (Cloud-Clone, USA). The assay is based on sandwich enzyme-linked immunosorbent technique. The microwells were coated with an anti-ALDH1A1 antibody (primary antibody). Primary antibody adsorbed to the microwells binds to human ALDH1A1 found in the sample or standard. Anti-human ALDH1A1 conjugated to horseradish peroxidase (HRP) was applied to human ALDH1A1 captured by the primary antibody. After incubation, unbound HRP-conjugated anti-human ALDH1A1 antibody was eliminated by washing and substrate solution was added. The formation of colored product was proportional to ALDH1A1 quantity in the sample or standard. By adding acid, the process was stopped and the absorbance (Abs) was read at 450 nm. The standard curve was constructed by graphing the Abs at 450 nm obtained for each of the six standard concentrations on the vertical (Y) axis against the appropriate concentration on the horizontal (X) axis, then the concentration of ALDH1A1 in samples was determined.
Determination of tissue and serum LGR5 levels
LGR5 levels in serum of all studied individuals and tissue homogenate of BC tissues and adjacent normal tissues were determined by ELISA assay kit according to the manufacturer’s operating instructions (Cloud-Clone, USA). The assay is based on sandwich enzyme-linked immunosorbent technique. The microwells were coated with an anti-LGR5 antibody (primary antibody). Primary antibody adsorbed to the microwells binds to human LGR5 found in the sample or standard. Anti-human LGR5 conjugated to HRP was applied to human LGR5 captured by the primary antibody. After incubation, unbound HRP-conjugated anti-human LGR5 antibody was eliminated by washing and substrate solution was added. The formation of colored product was proportional to LGR5 quantity in the sample or standard. By adding acid, the process was stopped and Abs was read at 450 nm. The standard curve was constructed by graphing the Abs at 450 nm obtained for each of the six standard concentrations on the vertical (Y) axis against the appropriate concentration on the horizontal (X) axis, then the concentration of LGR5 in samples was determined.
Determination of tissue and serum β-catenin levels
β-catenin levels in serum of all studied individuals and tissue homogenate of BC tissues and adjacent normal tissues were determined by ELISA assay kit according to the manufacturer’s operating instructions (Cloud-Clone, USA). The assay is based on sandwich enzyme-linked immunosorbent technique. The microwells were coated with an anti-β-catenin antibody (primary antibody). Primary antibody adsorbed to the microwells binds to human β-catenin found in the sample or standard. Anti-human β-catenin conjugated to HRP was applied to human β-catenin captured by the primary antibody. After incubation, unbound HRP-conjugated anti-human β-catenin antibody was eliminated by washing and substrate solution was added. The formation of colored product was proportional to β-catenin quantity in the sample or standard. By adding acid, the process was stopped and Abs was read at 450 nm. The standard curve was constructed by graphing the Abs at 450 nm obtained for each of the six standard concentrations on the vertical (Y) axis against the appropriate concentration on the horizontal (X) axis, then the concentration of β-catenin in samples was determined.
Statistical analysis
The IBM SPSS software program version 20.0 was used to analyze the data. The mean and standard error (SE) were used to describe quantitative data. The Kolmogorov–Smirnov test was used to check the normality distribution of the quantitative variables. The parametric tests were used since the variables have a normal distribution. The independent t-test was used to compare two independent groups, while the paired t-test was used to compare two dependent groups. Pearson coefficient was used to assess correlations between two quantitative variables. The significance of our results was assessed at a 5% level. The receiver operating characteristic (ROC) curve was used to assess the diagnostic performance of the investigated parameters.
Ethics approval and consent to participate
This study was approved by ethical committee of Medical Research Institute (MRI)-Alexandria University. A written informed consents were taken from all contributors in this study, in accordance with the Declaration of Helsinki.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 71(3), 209–249 (2021).
Tollefsbol, T. O. Chapter 1—An overview of epigenetics. In Handbook of Epigenetics 2nd edn (ed. Tollefsbol, T. O.) 1–6 (Academic Press, 2017).
Shamsi, M. B., Firoz, A. S., Imam, S. N., Alzaman, N. & Samman, M. A. Epigenetics of human diseases and scope in future therapeutics. J. Taibah Univ. Med. Sci. 12(3), 205–211 (2017).
Wong, M. M., Cox, L. K. & Chrivia, J. C. The chromatin remodeling protein, SRCAP, is critical for deposition of the histone variant H2A.Z at promoters. J. Biol. Chem. 282(36), 26132–26139 (2007).
Cuadrado, A. et al. A new p38 MAP kinase-regulated transcriptional coactivator that stimulates p53-dependent apoptosis. EMBO J. 26(8), 2115–2126 (2007).
Zhang, C. et al. Hypoxia induces the breast cancer stem cell phenotype by HIF-dependent and ALKBH5-mediated m6A-demethylation of NANOG mRNA. Proc. Natl. Acad. Sci. U.S.A. 113(14), E2047–E2056 (2016).
Vadde, R. et al. Role of hypoxia-inducible factors (HIF) in the maintenance of stemness and malignancy of colorectal cancer. Crit. Rev. Oncol. Hematol. 113, 22–27 (2017).
Keith, B., Johnson, R. & Simon, M. HIF1 alpha and HIF2 alpha: Sibling rivalry in hypoxic tumour growth and progression. Nat. Rev. Cancer 12, 9–22 (2011).
De Angelis, M. L., Francescangeli, F. & Zeuner, A. Breast cancer stem cells as drivers of tumor chemoresistance, dormancy and relapse: New challenges and therapeutic opportunities. Cancers 11(10), 1569 (2019).
Zou, W. et al. Association of CD44 and CD24 phenotype with lymph node metastasis and survival in triple-negative breast cancer. Int. J. Clin. Exp. Pathol. 13(5), 1008–1016 (2020).
Rabinovich, I. et al. Cancer stem cell markers ALDH1 and CD44+/CD24- phenotype and their prognosis impact in invasive ductal carcinoma. Eur. J. Histochem. 62(3), 2943 (2018).
Yang, L. et al. LGR5 promotes breast cancer progression and maintains stem-like cells through activation of Wnt/β-catenin signaling. Stem Cells 33(10), 2913–2924 (2015).
Leung, C., Tan, S. H. & Barker, N. Recent advances in Lgr5(+) stem cell research. Trends Cell Biol. 28(5), 380–391 (2018).
Vassalli, G. Aldehyde dehydrogenases: Not just markers, but functional regulators of stem cells. Stem Cells Int. 2019, 3904645 (2019).
Yang, W., Wang, Y., Wang, W., Chen, Z. & Bai, G. Expression of aldehyde dehydrogenase 1A1 (ALDH1A1) as a prognostic biomarker in colorectal cancer using immunohistochemistry. Med. Sci. Monit. 24, 2864–2872 (2018).
Lin, W. et al. Lgr5-overexpressing mesenchymal stem cells augment fracture healing through regulation of Wnt/ERK signaling pathways and mitochondrial dynamics. FASEB J. 33(7), 8565–8577 (2019).
Yang, L. et al. LGR5 promotes breast cancer progression and maintains stem-like cells through activation of Wnt/beta-catenin signaling. Stem Cells 33(10), 2913–2924 (2015).
Jang, G.-B. et al. Blockade of Wnt/β-catenin signaling suppresses breast cancer metastasis by inhibiting CSC-like phenotype. Sci. Rep. 5, 12465 (2015).
Bhavanasi, D. & Klein, P. S. Wnt signaling in normal and malignant stem cells. Curr. Stem Cell Rep. 2(4), 379–387 (2016).
Sun, S. et al. Chromatin remodeler Znhit1 preserves hematopoietic stem cell quiescence by determining the accessibility of distal enhancers. Leukemia 34(12), 3348–3358 (2020).
Yang, Z. et al. Znhit1 causes cell cycle arrest and down-regulates CDK6 expression. Biochem. Biophys. Res. Commun. 386(1), 146–152 (2009).
Robertson, B. W. & Chellaiah, M. A. Osteopontin induces beta-catenin signaling through activation of Akt in prostate cancer cells. Exp. Cell Res. 316(1), 1–11 (2010).
Jarman, E. J. et al. HER2 regulates HIF-2α and drives an increased hypoxic response in breast cancer. BCR 21(1), 10 (2019).
Yan, Y. et al. HIF-2α promotes conversion to a stem cell phenotype and induces chemoresistance in breast cancer cells by activating Wnt and Notch pathways. J. Exp. Clin. Cancer Res. 37(1), 256 (2018).
Gyan, E. et al. The role of ALDH1A1 in contributing to breast tumour aggressiveness: A study conducted in an African population. Ann. Diagn. Pathol. 51, 151696 (2021).
Rossi, A. et al. High aldehyde dehydrogenase levels are detectable in the serum of patients with lung cancer and may be exploited as screening biomarkers. J. Oncol. 2019, 8970645 (2019).
Gao, F. et al. The role of LGR5 and ALDH1A1 in non-small cell lung cancer: Cancer progression and prognosis. Biochem. Biophys. Res. Commun. 462(2), 91–98 (2015).
Cui, J., Li, P., Liu, X., Hu, H. & Wei, W. Abnormal expression of the Notch and Wnt/beta-catenin signaling pathways in stem-like ALDH(hi)CD44(+) cells correlates highly with Ki-67 expression in breast cancer. Oncol. Lett. 9(4), 1600–1606 (2015).
Sun, M. et al. Combined expression of aldehyde dehydrogenase 1A1 and β-catenin is associated with lymph node metastasis and poor survival in breast cancer patients following cyclophosphamide treatment. Oncol. Rep. 34(6), 3163–3173 (2015).
Hou, M. F., Chen, P. M. & Chu, P. Y. LGR5 overexpression confers poor relapse-free survival in breast cancer patients. BMC Cancer 18(1), 219 (2018).
Chen, Z. & Xue, C. G-protein-coupled receptor 5 (LGR5) overexpression activates β-catenin signaling in breast cancer cells via protein kinase A. Med. Sci. Monit. Basic Res. 25, 15–25 (2019).
Xu, C. et al. β-catenin nuclear localization positively feeds back on EGF/EGFR-attenuated AJAP1 expression in breast cancer. J. Exp. Clin. Cancer Res. 38(1), 238 (2019).
Clemenceau, A., Diorio, C. & Durocher, F. Role of secreted frizzled-related protein 1 in early mammary gland tumorigenesis and its regulation in breast microenvironment. Cells 9(1), 208 (2020).
Li, S. et al. Serum expression of β-catenin is a potential detection marker in patients with colorectal cancer. Dis. Mark. 2019, 5070524 (2019).
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
S.A.E., S.A.E. and S.E.D. designed research proposal idea, analyzed and interpreted results of this study. S.A.E. and S.E.D. performed the practical part of the research and wrote the main manuscript text. N.A.A. provided data of BC patients. R.R. contributed to sample collection from BC patients. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ebeid, S.A., Abd El Moneim, N.A., El-Benhawy, S.A. et al. Znhit1 and HIF-2α are correlated with cancer stem cell markers in breast cancer patients. Sci Rep 12, 13918 (2022). https://doi.org/10.1038/s41598-022-18133-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-18133-8
- Springer Nature Limited