Abstract
The prognosis of cutaneous melanoma depends on early detection, and good biomarkers for melanoma risk may provide a valuable tool to detect melanoma development at a pre-clinical stage. By studying the epigenetic profile in pre-diagnostic blood samples of melanoma cases and cancer free controls, we aimed to identify DNA methylation sites conferring melanoma risk. DNA methylation was measured at 775,528 CpG sites using the Illumina EPIC array in whole blood in incident melanoma cases (n = 183) and matched cancer-free controls (n = 183) in the Norwegian Women and Cancer cohort. Phenotypic information and ultraviolet radiation exposure were obtained from questionnaires. Epigenome wide association (EWAS) was analyzed in future melanoma cases and controls with conditional logistic regression, with correction for multiple testing using the false discovery rate (FDR). We extended the analysis by including a public data set on melanoma (GSE120878), and combining these different data sets using a version of covariate modulated FDR (AdaPT). The analysis on future melanoma cases and controls did not identify any genome wide significant CpG sites (0.85 ≤ padj ≤ 0.99). In the restricted AdaPT analysis, 7 CpG sites were suggestive at the FDR level of 0.15. These CpG sites may potentially be used as pre-diagnostic biomarkers of melanoma risk.
Similar content being viewed by others
Introduction
Incidence of cutaneous melanoma, the most aggressive and lethal form of skin cancer, continues to increase in fair-skinned populations1. Ultraviolet radiation (UVR) exposure is the main risk factor of melanoma2. By sequencing lesions in different evolutionary stages, Shain et al.3 found strong signature UVR mutations detectable at all stages, from benign lesions to invasive melanoma3. In some melanoma patients, the tumour suppressor gene TP53 has occasionally shown mutations with a UVR-signature4,5. Still, the most common mutations found in melanomas are the BRAF V600E and V600K mutations6 although these mutations do not carry the typical UVR signature7. The biological pathway from UVR exposure to melanoma onset is not yet fully explained, and could be due to epigenetic changes after UVR exposure.
Epigenetic modifications, such as DNA methylation (DNAm), have been associated with all cancers, including melanoma8. However, to our knowledge, previous studies on the epigenetics of melanoma have mostly been based on post-diagnostic sampling9. Epigenetic biomarkers have been proposed for both survival and response to treatment in melanoma patients10,11. However, the predictive value of these markers for melanoma risk itself has not been evaluated. Since most melanomas appear without association with a precursor lesion12, evaluation of pre-diagnostic markers should be based on samples of skin, blood, or saliva stored in biobanks. The DNA from these mixed tissues such as blood or saliva could be confounded by the composition of the different tissues. Based on epigenetic data, it is possible to estimate the relative contribution from each cell type observed in the tissue mix13.
The population-based Norwegian Women and Cancer (NOWAC) cohort study14 has been used to study the importance of host factors and UVR exposure in melanoma risk15,16,17, and to identify pre-diagnostic epigenetic markers for lung and breast cancer18,19,20. In a nested case–control study within the NOWAC cohort, we aimed to identify biomarkers of melanoma risk in pre-diagnostic blood samples of melanoma cases and cancer free controls, in an epigenome wide association study (EWAS), as well as a subset EWAS on case specific characteristics. To complement our analyses, EWAS was also performed in an open source data set from an independent study on melanoma21 and combined with our results.
Materials and methods
Material (NOWAC)
The NOWAC cohort includes over 172,000 women aged 30–70 years at recruitment in 1991–2006 (response 54%)14. Information on host characteristics and lifestyle factors was collected through baseline questionnaires and up to two follow-up questionnaires. The NOWAC study has high external validity, with no major selection bias14,22. Approximately 50,000 women (46–63 years) constitute the post genome cohort within NOWAC and donated a blood sample at inclusion or at the second follow-up (2003–2006)23. By using the unique identity number of Norwegian citizens, NOWAC is linked to the Cancer Registry of Norway (CRN) for follow-up of cancer incidence and vital status. We included all incident melanoma cases (n = 183) with an isolated DNA and RNA sample in the biobank per December 31, 2013, and matched each case with one cancer free control, based on time since blood sampling and year of birth (1943–1947, 1948–1952, 1953–1957). The Norwegian Malignant Melanoma Registry (NMMR) was established under the CRN in 2008, and information on tumour thickness for incident cases since 2008 was obtained from the NMMR. For melanoma cases diagnosed before 2008, information on tumour thickness was extracted manually by the CRN’s experienced melanoma registrars from histopathological reports in the CRN archive24.
All participants gave written informed consent and the Medical Ethical Committees of North Norway has approved the NOWAC study, the storage of human biological material, as well as this study (2016/976/REK Nord). All methods in this study were performed in accordance with the relevant ethical guidelines and regulations.
DNA methylation
Details of the DNAm quality control has been described elsewhere25. Briefly, DNA were treated with bi-sulfite and hybridised to the Illumina Infinium MethylationEPIC array according to the manufacturer’s protocol. Background subtraction and control normalization were performed with minfi to reduce background noise and dye bias26. Type I and Type II probes were normalized using the Beta mixture quantile normalization method from the wateRmelon R-package27. After quality control, 775 528 CpG probes remained in the data set. White blood cell composition was estimated using the Houseman algorithm13,28.
To complement our analysis, we included an open source data set from an independent study by Conway et al.21 (hereafter referred to as the GSE120878 study) which compared the epigenetic profiles of melanoma biopsies (n = 89) and nevi biopsies (n = 73), all from suspected melanoma biopsies from different patients using logistic regression. Their DNA methylation data were deposited at the GEO database in April 2019 (accession number GSE120878)21. In this study, DNA methylation was measured on the Illumina Infinium HumanMethylation450 BeadChip array and processed with the minfi R package.
Covariates
Baseline variables include age at recruitment, birth cohort and recruitment year. Age at blood sample and time in freezer were recorded. Participants reported, by questionnaire, education (≤ 10, 11–13, ≥ 14 years), smoking (never, former, current smoker), hair color (dark brown/black, brown, blond/yellow, red), freckling after sunbathing (yes, no), and the number of asymmetric nevi > 5 mm on the legs (0, 1, 2–3, 4–6, 7–12, 13–24, ≥ 25; categorized as 0, 1, ≥ 2). On the basis of average ambient ultraviolet radiation29, region of residence (latitudes 70°–58°) was categorized as low UVR exposure (north Norway), medium–low (central Norway), medium (southwest Norway), and highest UVR exposure (southeast Norway)30. In the baseline and follow-up questionnaires, participants reported history of severe sunburns (never, 1, 2–3, 4–5, ≥ 6 times/year), average number of weeks per year spent on sunbathing vacations (never, 1, 2–3, 4–6, ≥ 7 weeks/year), and average use of an indoor tanning device (never; rarely; 1, 2, or 3–4 times/month; > 1 time/week) in childhood (≤ 9 years), adolescence (10–19 years), and adulthood (> 19 years)30. The reported frequencies of sunburns, sunbathing vacations and indoor tanning were transformed and multiplied by the length of each interval for each questionnaire15. The participants were then classified into five categories; non-exposed and quartiles. To capture the tail of the distribution, the upper quartile was further divided into two equally sized groups (i.e. six categories in total) as described in Page et al.25 Cumulative UVR exposure was constructed by summarizing the categories (i.e. scores 0–5) for indoor tanning and sunbathing vacations15. Reproducibility of melanoma risk factors in the NOWAC questionnaire was good (kappa/intraclass correlation coefficient 0.49–0.77) and independent of age and education31.
Statistical methods
Conditional logistic regression was used to study the association between future status of melanoma as the outcome and white blood cell composition as continuous exposure, accounting for time to diagnosis and potential confounders: hair colour, nevi, and UVR exposure. To minimize technical variation and capture unmeasured confounding, we constructed surrogate variables using the sva package in R32,33,34. The surrogate variables were constructed as orthogonal decompositions of the residuals after projecting melanoma status on the DNA methylation data matrix33. We used conditional logistic regression, with control for the matching variables (age at blood sample and time in freezer) to assess the associations between future melanoma as the outcome and DNA methylation, adjusting for lifetime history of sunburns (as an indicator of severe UVR exposure)15, hair colour (the best measure of skin sensitivity to UVR exposure in the NOWAC cohort)17,35, and surrogate variables as potential confounders.
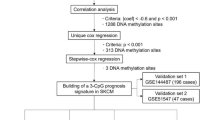
To control for multiple testing, we used the false discovery rate (FDR) procedure of Benjamini and Hochberg36. The genes annotated to the top 2000 CpG sites in our main model were included in an enrichment analysis, using the Enricher web interface37.
An EWAS was run using logistic regression without any covariate adjustments in the GSE120878 dataset, and the log p-values included as covariates in an adaptive multiple testing FDR method called AdaPT38 when correcting our main model for multiple testing. This method is based on the covariate modulated FDR (cmFDR) proposed by Ferkingstad et al.39 which weight the FDR significance by information from the new data set. The combined AdaPT analysis was restricted to CpG sites with a nominal p-value < 10–10 (NCpG = 2176) in the GSE120878 EWAS and the respective log p-values from these sites were included as side information in AdaPT, for each CpG, respectively.
A prediction model was trained on the same CpG sites (p-value < 10–10, NCpG = 2176) from the GSE120878 data, using a regression and decision tree algorithm40 similar to that of Onwuka et al.41. The prediction model was applied on the NOWAC data set of incident melanoma cases and controls.
Lastly, we performed an EWAS including only the melanoma cases in linear regressions with log transformed tumour thickness as the outcome, adjusting for lifetime history of sunburns and hair colour.
Institutional review board statement
The Medical Ethical Committees of North Norway has approved the NOWAC study, the storage of human biological material, as well as this sub-study (2016/976/REK Nord). All methods in this study were performed in accordance with the relevant ethical guidelines and regulations.
Informed consent statement
All participants gave written informed consent.
Results
Baseline characteristics of the cases and controls are presented in Table 1. Having higher education, being a non-smoker, having blond/yellow/red hair, freckling when sunbathing, and large asymmetric nevi on the legs were more common in the melanoma cases than in the controls. Compared to controls, melanoma cases reported more UVR exposure: lower proportion living in the region with low ambient UVR, the lower proportion experiencing no sunburns, the higher proportions in the highest categories of sunbathing vacations, and lower proportions in the two lowest categories of indoor tanning and cumulative UVR exposure (Table 1). Mean age at melanoma diagnosis was 60.2 years (range 49–70) and mean time from blood sampling to diagnosis was 4.4 years (range 0–9.7 years) (Table 1). Estimated cell-type proportions were similar in cases and controls (0.09 ≤ p ≤ 0.94) (Supplementary Table S1). None of the white blood cell fractions were significantly differently distributed between the melanoma cases and controls after adjustment for time to diagnosis (0.14 ≤ padj ≤ 0.56) (Supplementary Table S2).
We did not find any CpGs significantly associated with melanoma risk in the genome-wide analyses (0.85 ≤ padj ≤ 0.99). The top 10 CpG sites are listed in Table 2. The estimated odds ratios (ORs) genome wide were in equal proportions in both directions, indicating no global loss of methylation. The pathway enrichment analysis of the top 2000 CpGs did not identify any pathways previously reported for melanoma (Supplementary Table S3).
In the combined AdaPT analysis, after adjusting our findings with the log p-value from the GSE120878 EWAS, seven CpG sites from the NOWAC study had an FDR adjusted p-value below 0.15 (Table 3). The distribution of the ORs for these seven CpGs were shifted towards higher risk, with 5/7 CpGs having an OR above 1, as compared to the entire set of OR in our main EWAS, where the OR was in equal proportions in both directions.
The prediction model trained on the GSE120878 data set, did not predict melanoma status well: while 48% were predicted true positive, 49% were predicted as false positive, with the true negative only 1% and the false negative 2%. We did not find any significant CpG associations in the EWAS analysis of DNAm and tumour thickness in the melanoma cases (0.86 ≤ padj ≤ 0.99).
Discussion
We compared DNAm profiles of incident melanoma cases to healthy controls, to identify potential biomarkers for melanoma risk. We did not identify any genome wide significant CpGs related to melanoma risk. However, by combining different data sources, weighing the FDR adjustment, we identified seven potentially differentially methylated CpG sites associated to incident melanoma, all previously associated to melanoma in a case–control study21.
Two of the top 10 genes identified in our EWAS (Table 2) have previously been associated to melanoma; RSF142 and NTN443. However, they have been associated with more advanced stages in melanoma from case only studies and cell lines, and not with melanoma risk. We observed an equal number of effect sizes in both directions while the proportion of hypomethylation was larger in the GSE120878 study (50% vs ~ 57.6%), indicating a global loss of methylation in melanoma biopsies, which was not observed in the pre-diagnostic samples. This indicates that the loss of methylation observed in cancers may be a consequence of the disease, and not its cause. The log-odds was consistently over ten times larger in the study including samples from prevalent cases as compared to this pre-diagnostic study, which is also to be expected, given the differences in sample tissues in the two studies.
None of the top genes found in our primary analysis were associated to melanoma risk in the largest GWAS of melanoma to date44, which included almost 37,000 melanoma cases and ten times as many controls. Of the CpGs associated with melanoma risk in Table 3, two are associated to genes MIR196B and SH3RF3, which have been observed differentially expressed in sun exposed skin, as compared to non-exposed skin45. Given the prominent role of UVR exposure in melanoma risk, this is a potentially interesting finding that should be followed up. Further among the genes indicated, the HOXA9-HOXA10 cluster has been observed differentially expressed in multiple cancers46, and upregulation of HOXA9 is related to poor survival in melanoma cases47. Since the analysis was informed using findings from a previous case–control study on melanoma, all findings in this analysis have previously been associated with melanoma.
None of the white blood cell fractions were significantly differently distributed between the melanoma cases and controls, even after adjusting for time to diagnosis. This indicated that the cell type composition would not be a confounder for disease status, and was thus not adjusted for in our analysis.
The discovery of pre-diagnostic biomarkers relies on a large number of samples with biological material stored in biobanks, since the future cancer status of each participant is unknown. Biobanks of the size needed for this type of incident sampling, are almost exclusively storing biological samples derived from blood. The use of blood leukocytes may explain some of the poor performance for the multi-CpG prediction for melanoma, which did not predict case status with high accuracy. Additionally, the tissue differences need to be kept in mind when comparing the results between pre-diagnostic blood samples and tissue specific cancer samples. Using results from cancer tissue to inform the FDR correction could help detect cancer like signals in blood samples early on in the disease. Circulating blood leukocytes are constantly in contact with all organ systems in the body, and exposed to the same environment, thus, weak signals from the environmental exposure can often be detected in blood leukocytes48. Additionally, pre-clinical tumors are likely to shed DNA fragments in the blood stream, which can influence the DNAm signature in the blood sample, and DNAm isolated from whole blood may then contain weak cancer specific signals.
Being nested in the NOWAC cohort, this study benefits from a large population-based cohort with well documented case information and prospective baseline information on major risk factors for melanoma, such as UVR exposure, but the pre-diagnostic biological material was limited to whole blood and with a limited sample size. The distribution of T categories was not even across the cases, as it can be in a selected clinical sample, but reflective of what is found in the general population (i.e. more T1 than T4 melanomas).
NOWAC is a female only cohort, while GSE120878 included both sexes. Previous cancer studies have included only one sex, either only females14 or males49. Studies addressing association between UV exposure and melanoma, found no interaction between sun exposure and sex50. The GSE120878 data set was balanced with respect to sex ratio, and the p-value for any sex differences between the groups not significant21.
The lack of genotype information in the cohort is a limitation. Multiple genetic markers have been found to increase melanoma risk44, most notable variants in the CDKN2A gene51, however, the consent in NOWAC did not open for genotyping of the participants.
We find that the use of covariate modulated FRD methods, like AdaPT, is a good way of combining our results with public data from a different source.
Conclusion
No epigenome-wide significant associations to melanoma risk was found, but 7 CpGs identified by combining data and previous knowledge was suggestive of melanoma risk. Future melanoma status was not well predicted in this study, however, using a more targeted tissue, such as skin biopsies could have resulted in more informative epigenetic markers for melanoma risk.
Data availability
The DNA methylation data generated and/or analysed in the current study can be accessed upon reasonable request to the originating cohort. Access will be conditional on adherence to both local and national ethical and security policy. R codes used for the analyses presented in the paper are available upon request.
References
Whiteman, D. C., Green, A. C. & Olsen, C. M. The growing burden of invasive melanoma: Projections of incidence rates and numbers of new cases in six susceptible populations through 2031. J. Investig. Dermatol. 136, 1161–1171. https://doi.org/10.1016/j.jid.2016.01.035 (2016).
Berwick, M. et al. Melanoma epidemiology and prevention. Cancer Treat. Res. 167, 17–49. https://doi.org/10.1007/978-3-319-22539-5_2 (2016).
Shain, A. H. et al. The genetic evolution of melanoma from precursor lesions. N. Engl. J. Med. 373, 1926–1936. https://doi.org/10.1056/NEJMoa1502583 (2015).
Alexandrov, L. B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421. https://doi.org/10.1038/nature12477 (2013).
Arozarena, I. & Wellbrock, C. Overcoming resistance to BRAF inhibitors. Ann. Transl. Med. 5, 387. https://doi.org/10.21037/atm.2017.06.09 (2017).
Hodis, E. et al. A landscape of driver mutations in melanoma. Cell 150, 251–263. https://doi.org/10.1016/j.cell.2012.06.024 (2012).
Bauer, J. et al. BRAF mutations in cutaneous melanoma are independently associated with age, anatomic site of the primary tumor, and the degree of solar elastosis at the primary tumor site. Pigment Cell Melanoma Res. 24, 345–351. https://doi.org/10.1111/j.1755-148X.2011.00837.x (2011).
Jones, P. A. DNA methylation and cancer. Oncogene 21, 5358–5360. https://doi.org/10.1038/sj.onc.1205597 (2002).
Van Laar, R., Lincoln, M. & Van Laar, B. Development and validation of a plasma-based melanoma biomarker suitable for clinical use. Br. J. Cancer 118, 857–866. https://doi.org/10.1038/bjc.2017.477 (2018).
Guo, Y., Long, J. & Lei, S. Promoter methylation as biomarkers for diagnosis of melanoma: A systematic review and meta-analysis. J. Cell Physiol. 234, 7356–7367. https://doi.org/10.1002/jcp.27495 (2019).
Micevic, G., Theodosakis, N. & Bosenberg, M. Aberrant DNA methylation in melanoma: Biomarker and therapeutic opportunities. Clin. Epigenet. 9, 34. https://doi.org/10.1186/s13148-017-0332-8 (2017).
Shitara, D. et al. Nevus-associated melanomas: Clinicopathologic features. Am. J. Clin. Pathol. 142, 485–491. https://doi.org/10.1309/AJCP4L5CJGKTJVDD (2014).
Houseman, E. A. et al. DNA methylation arrays as surrogate measures of cell mixture distribution. BMC Bioinform. 13, 86. https://doi.org/10.1186/1471-2105-13-86 (2012).
Lund, E. et al. Cohort profile: The Norwegian women and cancer study—NOWAC-Kvinner og kreft. Int. J. Epidemiol. 37, 36–41. https://doi.org/10.1093/ije/dym137 (2008).
Ghiasvand, R. et al. Indoor tanning and melanoma risk: Long-term evidence from a prospective population-based cohort study. Am. J. Epidemiol. 185, 147–156. https://doi.org/10.1093/aje/kww148 (2017).
Ghiasvand, R., Weiderpass, E., Green, A. C., Lund, E. & Veierod, M. B. Sunscreen use and subsequent melanoma risk: A population-based cohort study. J. Clin. Oncol. 34, 3976–3983. https://doi.org/10.1200/JCO.2016.67.5934 (2016).
Veierod, M. B. et al. A prospective study of pigmentation, sun exposure, and risk of cutaneous malignant melanoma in women. J. Natl. Cancer Inst. 95, 1530–1538. https://doi.org/10.1093/jnci/djg075 (2003).
Fasanelli, F. et al. Hypomethylation of smoking-related genes is associated with future lung cancer in four prospective cohorts. Nat. Commun. 6, 10192. https://doi.org/10.1038/ncomms10192 (2015).
van Veldhoven, K. et al. Epigenome-wide association study reveals decreased average methylation levels years before breast cancer diagnosis. Clin. Epigenet. 7, 67. https://doi.org/10.1186/s13148-015-0104-2 (2015).
Guida, F. et al. Dynamics of smoking-induced genome-wide methylation changes with time since smoking cessation. Hum. Mol. Genet. 24, 2349–2359. https://doi.org/10.1093/hmg/ddu751 (2015).
Conway, K. et al. Identification of a robust methylation classifier for cutaneous melanoma diagnosis. J. Investig. Dermatol. 139, 1349–1361. https://doi.org/10.1016/j.jid.2018.11.024 (2019).
Lund, E. et al. External validity in a population-based national prospective study—The Norwegian Women and Cancer Study (NOWAC). Cancer Causes Control 14, 1001–1008. https://doi.org/10.1023/b:caco.0000007982.18311.2e (2003).
Dumeaux, V. et al. Deciphering normal blood gene expression variation—The NOWAC postgenome study. PLoS Genet. 6, e1000873. https://doi.org/10.1371/journal.pgen.1000873 (2010).
Ghiasvand, R. et al. Patient age and risk of recurrence of primary melanoma at high risk of spread. Br. J. Dermatol. 184, 566–568. https://doi.org/10.1111/bjd.19601 (2021).
Page, C. M. et al. Lifetime ultraviolet radiation exposure and DNA methylation in blood leukocytes: The Norwegian women and cancer study. Sci. Rep. 10, 4521. https://doi.org/10.1038/s41598-020-61430-3 (2020).
Aryee, M. J. et al. Minfi: A flexible and comprehensive Bioconductor package for the analysis of Infinium DNA methylation microarrays. Bioinformatics 30, 1363–1369. https://doi.org/10.1093/bioinformatics/btu049 (2014).
Pidsley, R. et al. A data-driven approach to preprocessing Illumina 450K methylation array data. BMC Genom. 14, 293. https://doi.org/10.1186/1471-2164-14-293 (2013).
Reinius, L. E. et al. Differential DNA methylation in purified human blood cells: Implications for cell lineage and studies on disease susceptibility. PLoS One 7, e41361. https://doi.org/10.1371/journal.pone.0041361 (2012).
Edvardsen, K. et al. Vitamin D-effective solar UV radiation, dietary vitamin D and breast cancer risk. Int. J. Cancer 128, 1425–1433. https://doi.org/10.1002/ijc.25463 (2011).
Ghiasvand, R., Lund, E., Edvardsen, K., Weiderpass, E. & Veierod, M. B. Prevalence and trends of sunscreen use and sunburn among Norwegian women. Br. J. Dermatol. 172, 475–483. https://doi.org/10.1111/bjd.13434 (2015).
Veierod, M. B., Parr, C. L., Lund, E. & Hjartaker, A. Reproducibility of self-reported melanoma risk factors in a large cohort study of Norwegian women. Melanoma Res. 18, 1–9. https://doi.org/10.1097/CMR.0b013e3282f120d2 (2008).
Leek, J. T., Johnson, W. E., Parker, H. S., Jaffe, A. E. & Storey, J. D. The sva package for removing batch effects and other unwanted variation in high-throughput experiments. Bioinformatics 28, 882–883. https://doi.org/10.1093/bioinformatics/bts034 (2012).
Leek, J. T. & Storey, J. D. Capturing heterogeneity in gene expression studies by surrogate variable analysis. PLoS Genet. 3, 1724–1735. https://doi.org/10.1371/journal.pgen.0030161 (2007).
R Core Team. R: A language and environment for statistical computing. (2013).
Veierod, M. B., Adami, H. O., Lund, E., Armstrong, B. K. & Weiderpass, E. Sun and solarium exposure and melanoma risk: Effects of age, pigmentary characteristics, and nevi. Cancer Epidemiol. Biomark. Prev. 19, 111–120. https://doi.org/10.1158/1055-9965.EPI-09-0567 (2010).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate—A practical and powerful approach to multiple testing. J. R. Stat. Soc. B. 57, 289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x (1995).
Kuleshov, M. V. et al. Enrichr: A comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90-97. https://doi.org/10.1093/nar/gkw377 (2016).
Lei, L. H. & Fithian, W. AdaPT: An interactive procedure for multiple testing with side information. J. R. Stat. Soc. B 80, 649–679. https://doi.org/10.1111/rssb.12274 (2018).
Ferkingstad, E., Frigessi, A., Rue, H., Thorleifsson, G. & Kong, A. Unsupervised Empirical Bayesian Multiple Testing with External Covariates. Ann Appl Stat 2, 714–735. https://doi.org/10.1214/08-Aoas158 (2008).
Breiman, L., Friedman, J., Stone, C. J. & Olshen, R. A. Classification and Regression Trees. (CRC Press, 1984).
Onwuka, J. U. et al. A panel of DNA methylation signature from peripheral blood may predict colorectal cancer susceptibility. BMC Cancer 20, 692. https://doi.org/10.1186/s12885-020-07194-5 (2020).
He, J., Fu, L. & Li, Q. Rsf1 regulates malignant melanoma cell viability and chemoresistance via NFkappaB/Bcl2 signaling. Mol. Med. Rep. 20, 3487–3498. https://doi.org/10.3892/mmr.2019.10610 (2019).
Jayachandran, A. et al. Identifying and targeting determinants of melanoma cellular invasion. Oncotarget 7, 41186–41202. https://doi.org/10.18632/oncotarget.9227 (2016).
Landi, M. T. et al. Genome-wide association meta-analyses combining multiple risk phenotypes provide insights into the genetic architecture of cutaneous melanoma susceptibility. Nat. Genet. 52, 494–504. https://doi.org/10.1038/s41588-020-0611-8 (2020).
Uhlen, M. et al. Proteomics. Tissue-based map of the human proteome. Science 347, 1260419. https://doi.org/10.1126/science.1260419 (2015).
Paco, A., de Bessa Garcia, S. A. & Freitas, R. Methylation in HOX clusters and its applications in cancer therapy. Cells https://doi.org/10.3390/cells9071613 (2020).
Uhlen, M. et al. A pathology atlas of the human cancer transcriptome. Science https://doi.org/10.1126/science.aan2507 (2017).
Terry, M. B., Delgado-Cruzata, L., Vin-Raviv, N., Wu, H. C. & Santella, R. M. DNA methylation in white blood cells: Association with risk factors in epidemiologic studies. Epigenetics 6, 828–837. https://doi.org/10.4161/epi.6.7.16500 (2011).
Rimm, E. B. et al. A prospective-study of alcohol-consumption and the risk of coronary-disease in men. Am. J. Epidemiol. 134, 785–786 (1991).
Liu, F. C. et al. Ultraviolet radiation and risk of cutaneous melanoma and squamous cell carcinoma in males and females in the Norwegian Offshore Petroleum Workers cohort. Am. J. Ind. Med. 64, 496–510. https://doi.org/10.1002/ajim.23240 (2021).
Begg, C. B. et al. Lifetime risk of melanoma in CDKN2A mutation carriers in a population-based sample. J. Natl. Cancer Inst. 97, 1507–1515. https://doi.org/10.1093/jnci/dji312 (2005).
Acknowledgements
We thank all the participants in the NOWAC cohort, who donated blood and answered the questionnaires.
Funding
CMP was funded by a grant from the South-East Regional Health Authority (#2076086). The DNA methylation arrays were funded by grants from the Norwegian Research Council (#248804) and UNIFOR Frimed. The NOWAC post-genome cohort study was funded by the ERC advanced grant Transcriptomic in Cancer Epidemiology (ERC-2008-AdG-232997).
Author information
Authors and Affiliations
Contributions
Conception and design: T.M.S., A.F., M.T., M.B.V. Acquisition of data; T.M.S., T.H.N., M.T., M.B.V. Analysis of data: C.M.P., V.D. Interpretation of data: All authors. Drafted the work: C.M.P., M.B.V. Funding: A.F., M.T., M.B.V. Revising the work: All authors. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Page, C.M., Nøst, T.H., Djordjilović, V. et al. Pre-diagnostic DNA methylation in blood leucocytes in cutaneous melanoma; a nested case–control study within the Norwegian Women and Cancer cohort. Sci Rep 12, 14200 (2022). https://doi.org/10.1038/s41598-022-18585-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-18585-y
- Springer Nature Limited