Abstract
A converging body of behavioural findings supports the hypothesis that the dispositional use of emotion regulation (ER) strategies depends on trait emotional intelligence (trait EI) levels. Unfortunately, neuroscientific investigations of such relationship are missing. To fill this gap, we analysed trait measures and resting state data from 79 healthy participants to investigate whether trait EI and ER processes are associated to similar neural circuits. An unsupervised machine learning approach (independent component analysis) was used to decompose resting-sate functional networks and to assess whether they predict trait EI and specific ER strategies. Individual differences results showed that high trait EI significantly predicts and negatively correlates with the frequency of use of typical dysfunctional ER strategies. Crucially, we observed that an increased BOLD temporal variability within sensorimotor and salience networks was associated with both high trait EI and the frequency of use of cognitive reappraisal. By contrast, a decreased variability in salience network was associated with the use of suppression. These findings support the tight connection between trait EI and individual tendency to use functional ER strategies, and provide the first evidence that modulations of BOLD temporal variability in specific brain networks may be pivotal in explaining this relationship.
Similar content being viewed by others
Introduction
Emotion regulation (ER) is employed to modify current emotional states and adaptively respond to the environment1,2,3,4. Not surprisingly, difficulties in ER are involved in compromised well-being and mental health5,6. Namely, the selection of specific strategies and the excessive or rigid usage of them has been associated with negative outcomes7,8,9 contributing to a general distinction between dysfunctional and functional strategies. For example, some studies reported that suppression strategy (i.e., the inhibition of the expressive reactions to the emotional events) fails in engendering subjective relief after experiencing negative emotions, and it is associated with costs in terms of physiological, cognitive and social functioning5,10,11, as well as with decreased well-being12 and several psychopathologies (e.g.13,14). Differently, reappraisal strategy (i.e., the ability of cognitively changing the impact of the emotional event) has been generally considered an adaptive strategy, which is associated with healthiness and personal satisfaction5 improving the affective state15 and successfully modulating the emotion-related peripheral physiological indexes16 and the neural activity17.
The simplistic distinction between functional and dysfunctional strategies, however, risks to conceal the role of individual differences in selecting and recurrently using regulation strategies18. What leads someone to use one given strategy or to use it more often than others? Even though individuals’ dispositions may be crucial to comprehend the variability behind the selection and usage of emotion regulation strategies19, investigations in this sense are still scant18,20. Only recently, an interesting line of research suggests a link between ER processes and the construct of emotional intelligence (EI18), with some authors suggesting a theoretical framework to explain such a relationship (e.g.21). In literature, EI can be operationalized both as a set of cognitive abilities, which allows the individual to perceive, reason about, and manage emotional information (ability EI—what individuals are able to do), and as a set of personal dispositions related to emotional events (trait EI—what individuals typically do18). Especially trait EI has been suggested to play a critical role in the ER processes19, since personality traits may shape individual preferences, interpersonal behaviours, and intents, which are all likely to influence the choice of ER strategy (e.g.22). In line with that, behavioural studies reported that individuals with high levels of trait EI tend to use contextually appropriate coping strategies, whereas individuals with low levels of EI adopted non-adaptive ones18,23,24,25. Differently, similar levels of ability EI may result in adaptive as well as deviant responses22,26, and there is evidence that the ability EI fails in predicting specific emotion regulation processes27,28, or when it does, it has to be coupled with trait EI to comprehend the beneficial consequences of the adaptive response29. The measure of traits EI, then, not only represent an individual’s predisposition measure itself, but it also seems promising to explain how ER processes are selected. However, the relationship between trait EI and specific ER strategies, the nature and the extent of their point of connection remain unclear, especially in neural terms. To fill this gap, the present study is aimed to investigate whether the link between trait EI and ER relies on a shared neural substrate, provided more in deep their relationship in terms of self-reported measures.
So far, the few functional30,31 and structural32 neuroimaging studies dealing with trait EI seem to support at least a partial neural overlap with findings from neuroimaging studies of ER33,34,35,36. Indeed, both EI30,31 and ER processes33,34,35,36,37,38 rely on task-related activity in brain areas subserving emotion processing (limbic system) and higher executive functioning (prefrontal regions). Similarly, from a structural neuroimaging perspective, it has been shown that grey matter alterations in fronto-limbic brain areas correlates with specific ER processes39,40 and traits EI32.
Besides structural and task-related functional evidence, some authors relied on resting-state functional connectivity (RS-FC) to understand the neural signature of ER and EI. RS-FC is a valuable source to understand the neural mechanisms behind several psychological states20,41,42, as well as emotional processes17,33. In addition, altered functional connectivity seems to be associated with many psychopathologies43,44 as ER5 and EI also are45. Evidence from resting-state studies showed that higher trait EI scores was associated with the amplitude of low-frequency fluctuations (ALFFs) in networks related to social emotion processing and cognitive control (Ref.46, but see47 for null results). Moreover, Takeuchi et al.48, showed that trait EI was associated with components of social cognition and somatic marker circuitry neural networks. They found the trait EI score positively correlated with resting-state activity between medial prefrontal cortex (mPFC) and the precuneus, the intracalcarine cortex, and between anterior insula (aIC) and the right portion of the dorsolateral prefrontal cortex (dlPFC). In ER context, Pérez et al.49 reported a negative correlation between the connectivity of the right basolateral amygdala, the left insula, and the supplementary motor cortex (SMA) with the frequency of use of cognitive reappraisal, and a positive correlation between emotion suppression strategy and the activity of the right basolateral amygdala, the dorsal anterior cingulate (dACC), the supplementary motor cortex (SMA), and in the left medial portion of the amygdala. In contrast, Uchida et al.50 reported a negative correlation between functional connectivity in the amygdala and medial prefrontal cortex (mPFC) with the success of applying the cognitive reappraisal strategy. It is important to note also that Dörfel et al.51, failed to replicate and extend both the previously mentioned studies. Notably, the inconsistencies in ER and EI studies, may derive from some methodological issues.
Indeed, the majority of studies used massive univariate approaches, and a priori selected regions30,36,46,49,51. Widely distributed processes, such as ER or EI, may be better captured using multivariate approaches and a network perspective52. For this reason, in the present study, we adopted a whole brain data-driven approach (independent component analysis, ICA) that allows us to identify the variations of BOLD signal activity over time within major brain networks rather than considering signals from a priori decided regions of interest. ICA, being a blind source separation method, is an unsupervised machine learning approach able to identify non-overlapping independent neural circuits53. Such independent circuits represent meaningful naturally separated circuits that bypass anatomically and histologically based regions, and have the advantage of reducing brain complexity into low dimensional spaces52. The BOLD temporal variability (SD BOLD) for every resting-state network was used to predict trait EI and ER strategies. BOLD variability is defined as a fluctuation in neural activity over time, and an increment in this measure reflects greater functional network complexity and is associated with more effective information integration54,55,56. According to Moreira et al.57, this feature is particularly relevant for psychological phenomena that develop over time, such as ER58,59, or affective states60 and represents an index of the degree of cognitive flexibility61.
Building on the above considerations, the present study is aimed to support and extend previous findings on the role of trait EI in determining ER strategies, and to disclose which overlapping resting-state networks may subserve this relationship. We hypothesize that the level of trait EI (measured by the TeiQue-SF questionnaire62) is associated with the use of different ER processes, assessed using two different scales, ERQ12 and CERQ63. These scales are largely used together in literature since they allow to cover a wide range of emotion regulation strategies. Namely, we expect that the higher the trait EI and/or subscales scores, the higher the usage of adaptive ER strategies, and/or the lesser the usage of non-adaptive ER strategies. Then, we delineate the networks underlying EI using an ICA approach on the resting-state functional connectivity, and we assess whether these networks may be associated with the use of different ER strategies. We expect to find that the activity in regions responsible for the processing and control of socio-emotional information (e.g., insula, frontal and parietal regions64) underly both the individual differences in EI (as measured by TeiQueSF), and the ER usage (measured by ERQ and CERQ).
Results
Trait measures
The total EI trait index is negatively associated (negative β values) with the frequency of use of the following strategies: Suppression (ERQ) (F(1,78) = 9.705, R2 = 0.11, r = − 0.335; p = 0.003; β = − 0.335; pFDR = 0.022) and Self-blame (CERQ) (F(1,78) = 7.867, R2 = 0.093, r = − 0.304 p = 0.006; β = − 0.304, pFDR = 0.022). For what concerns the four TEIQue-SF factors, we firstly confirmed that there was no multicollinearity between these predictors, as the Pearson’s correlation coefficient (less than 0.965) and the Variance Inflation Factor (VIF, less than 1066) showed (see Table 1). Then, regression results showed that Emotionality (β = − 0.305, pFDR = 0.04), Well-being (β = − 0.255, pFDR = 0.01) and female gender (β = − 0.255, pFDR = 0.04) were associated with Suppression (ERQ) (F(4,75) = 4.908 R2 = 0.21; p = 0.001); Self-control (β = -0.366, pFDR = 0.01), Emotionality (β = 0.323, pFDR = 0.01), and Well-being (β = − 0.254, pFDR = 0.03) were associated with Rumination (CERQ) (F(4,75) = 5.332, R2 = 0.28, p < 0.005); Finally, Self-control (β = − 0.285, pFDR = 0.05), and Well-being (β = − 0.336, pFDR = 0.02) were associated with Catastrophizing (CERQ) (F(4,75) = 4.258, R2 = 0.21, p < 0.001). To conclude, as can be noted from the negative β values of the predictors, the lower is the EI, the higher is the individual tendency to use non-adaptive ER regulation strategies such as Rumination, Self-blame, Catastrophizing, Suppression.
Functional connectivity results
Multiple Regression analyses (stepwise) showed that the BOLD variability of IC20 (β = 0.302, pFDR = 0.008) significantly predicts total EI index (F(21,76) = 0.07, R2 = 0.08, p = 0.008); IC18 (β = 0.360, pFDR = 0.04) and IC16 (β = 0.239, pFDR = 0.05) predict Sociability (F(4,74) = 6.02, R2 = 0.250, p < 0.001); Finally, IC20 (β = 0.380, pFDR = 0.03), IC1 (β = − 0.309, pFDR = 0.01), and IC7 (β = 0.234, pFDR = 0.03) predict Well-being (F(3,75) = 5.734, R2 = 0.18, p = 0.001). To summarise, the ICs associated with the total index and the subscale of the TEIQue-SF questionnaire were the IC1, IC7, IC16, IC18, and IC20. These IC’s include clusters of cortical and subcortical regions at a cluster significance level p < 0.05 (pFDR corrected) and voxel significance p < 0.001 (pFDR corrected). Based on CONN’s correlational spatial match to template approach, the identified ICs are attributable to known resting state networks: IC20 (r = 0.46) and IC16 (r = 0.36) = sensorimotor network; IC1 (r = 0.49) = cerebellar network; IC18 (r = 0.58) = visual network, and IC7 (r = 0.10) = salience network.
Among the IC’s associated with trait EI, the BOLD variability of IC20 (β = 0.492, pFDR = 0.005) was also associated with cognitive reappraisal (ERQ) (F(6,72) = 3.151, R2 = 0.21, p = 0.008). The BOLD variability of IC20 (β = 0.345, pFDR = 0.01), IC16 (β = − 0.275, pFDR = 0.02) and IC7 (β = 0.299, pFDR = 0.01), was also associated with Positive Reappraisal (CERQ) (F(7,71) = 3.544, R2 = 0.22, p = 0.004). Finally, the BOLD variability of IC7 (β = − 0.335, pFDR = 0.006) and female gender (β = − 0.360, pFDR = 0.006) was associated with Suppression (ERQ) (F(6,72) = 4.271, R2 = 0.26, p = 0.001) (see Table 2).
To sum up, the higher is the BOLD variability the higher is the frequency of use of cognitive reappraisal and positive reappraisal (with the exception of IC16). Whereas, the lower is the BOLD variability the higher is the frequency of use of Suppression.
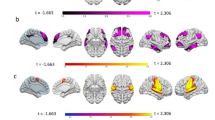
The brain networks identified by the IC7, IC20, and IC16 include clusters of cortical and subcortical regions (see Table 3 for details) at a cluster significance level p < 0.05 (pFDR corrected) and voxel significance p < 0.001 (pFDR corrected) (see Fig. 1).
Representation of the ICs shared by trait EI and ER. (a) IC7; (b) IC20 and (c) IC16. Left of the panel: 2D (above) and 3D inflated (below) brain model for the three ICs (cluster significance pFDR < 0.05 and voxel significance pFDR < 0.001). Colour bar represents positive t-values in orange and negative t-values in blue. Right of the panel: Relation between the ICs and the subscales of trait EI and ER questionnaires. In red, the positive relation; In blue, the negative relation. C. Reappraisal cognitive reappraisal, P. Reappraisal positive reappraisal.
Discussion
ER processes are daily employed by individuals to modify the emotional states they are experiencing. However, the individuals’ variability to use one or other strategy is still poorly understood. How can such variability be explained? In the present study, we investigated the relationship between trait EI and ER abilities, on the basis of previous evidence that trait EI may represent a suitable measure for the individual predisposition to the choice of specific ER strategy (e.g.18,25). Namely, we were interested in finding the neural bases associated with trait EI by analysing the functional connectivity of naturally grouping circuits decomposed by an unsupervised machine learning approach (ICA). One intriguing question was whether EI and ER share at least some neural bases. This would be an additional proof of their intimate relationship.
In terms of trait measures, we found that individuals with low levels of trait EI are associated with the use of non-adaptive ER processes (i.e., suppression, and self-blame). This result is complementary to past evidence18,25,67 showing a relationship between high trait EI and adaptive coping strategies. The different, still complementary result can be explained by the fact that the previous studies (e.g.25) focused mostly on coping abilities and not on typical emotion regulation strategies. Instead, by the wide range of strategies considered, we found that low scores in the trait EI subscale of self-control are associated with blaming others and rumination, whereas low scores in the well-being subscale are associated with the use of suppression, rumination, and catastrophizing. Finally, low scores in emotionality are associated to the use of suppression and rumination.
In literature, rumination and suppression strategies have been usually related to negative outcomes and psychopathologies5. Likewise, low level of traits EI are also associated with psychopathological disorders68,69, making reasonable the strong link we found between low trait EI and maladaptive strategies. The specific association between rumination and self-control, which is associated with difficulties in managing stressful situations and impulsive behaviours, is supported by recent evidence suggesting that impulsivity plays a critical role in rumination70. Low self-esteem—a relevant aspect in the trait EI subscale of well-being, already emerged as an important predictor of rumination71, and it has been indirectly linked to suppression since it is involved in shame emotion, which has been already coupled with this regulation strategy56,72. In addition, low emotionality and the related difficulty in emotions recognition and expression62, may lead individuals to use suppression that occurs late in the process of emotion generation73. In our study, that trait EI subscale of sociability did not predict any emotion regulation strategy, can be explained by evidence showing that low sociability is not associated with indexes of emotion dysregulation (e.g., high psychological reactivity, negative emotional intensity, dispositional negative affect, and personal distress). Low sociability is rather associated with low social support seeking74, strategy which may have escaped the taxonomy used in the ERQ and CERQ. In support of that, trait EI predicts social sharing when this latter is included in the set of regulation strategies28. Our result also showed that women are more likely to use suppression with respect to men. Although also previous studies reported gender difference in the use of suppression75,76,77), this result is not consistent in literature (see6,78), suggesting that gender difference possibly depend on other factors such as the situation itself79 Further researches are needed to better understand the role of gender in the trait EI-ER relationship.
Besides trait measures, functional connectivity results showed that modulations of BOLD temporal variability in sensorimotor (IC20 and IC16), visual (IC18), salience (IC7) and cerebellar (IC1) networks is associated with the total traits EI index and the four subscales. Most importantly for the present study, we found that increased BOLD variability especially in sensorimotor (when identified by the IC20 but not IC16) and salience networks also predicted the use of reappraisal strategy (in ERQ and CERQ), whereas decreased BOLD variability in salience network predicted the use of emotion suppression. These results suggest that BOLD temporal variability in sensorimotor and salience networks underlies trait EI and at the same time explains differences in ER strategies. Namely, this finding enriches our understanding of the relationship between trait EI and ER, by showing for the first time that different networks involved in the former, are also involved in the latter.
The brain areas belonging to IC20 have been already related to the sensorimotor network80. Among these areas, the inferior frontal gyrus also emerged in previous resting-state study on trait EI46 and it was interpreted as a part of a circuit related to social and emotional processing. Similarly, a positive correlation between r-state connectivity involving frontal regions and trait EI was reported by Takeuchi et al.48. On the other hand, the supplementary motor cortex (SMA) and the cerebellum positively correlated with trait EI46, playing a critical role in cognitive control mechanisms. In particular, the somatosensory cortex, and its portion of the supramarginal gyrus (SMG) are involved in the recognition of emotions, the understanding of the emotional states of others81, and are more generally part of the mirror neuron system82,83. In addition, other studies provided evidence that the somatosensory cortex is associated with emotion generation84 and interoceptive awareness85. The insula, instead, is deemed to facilitate social interaction, and decision making by integrating sensory, affective, and bodily information, and it has been traditionally reported as neural correlate of trait EI. The implications of these areas in socio-emotional processing and cognitive control46,86 make intuitive their involvement also in reappraisal strategy, which involve cognitive abilities (e.g., attention and memory) to control emotional responses87. Consistently, there is evidence of a correlation between the frequency of use of reappraisal and the functional connectivity of the left insula, supplementary motor cortex (SMA)49, and inferior frontal gyrus88. These areas, more generally, represent a well-established network underlying cognitive control of emotions by reappraisal34,38. Our result concerning the negative correlation between the BOLD variability in the IC16-related sensorimotor network and the positive reappraisal seems apparently incongruent, then. However, this incongruency may be better interpreted in line with our trait measures results, which show no association between the trait EI-sociability scale and ER strategies. Similarly, indeed, that the IC16 (but not the IC20) is associated with the sociability scale may explain why this component yield a negative correlation with reappraisal. In addition, it is worth mentioning that the spatial match to template approach reports the IC20 (vs the IC16), as the best match for the sensorimotor network (see correlations in the Results section), and, yet, the two sensorimotor-related components involve overlapping, but still different brain regions, such as the middle temporal gyrus. Since this is the first attempt showing common neural substrates between trait EI and ER, further researchers are needed to better identify the specific involved brain areas.
Along the sensorimotor network, our study showed the involvement of the salience network, the BOLD variability of which positively correlates and predicts adaptive (reappraisal) strategy, and negatively correlates with maladaptive (suppression) strategy. In line with our results, previous studies reported the link between a similar component and the salience network89, as well as the relationship between the key nodes of this network and trait EI and ER90. Especially the amygdala and the basal ganglia (i.e., putamen), allowing the organism to adaptively respond to the emotional context, are strictly related to the emotion processing91 and as a such to the two constructs48,49. In addition, there is evidence that emotion suppression is associated with activation of anterior cingulate, ventrolateral prefrontal cortex92, inferior frontal gyrus, putamen, pre-supplementary motor area and supramarginal gyrus93.
The relation between BOLD variability, trait EI and adaptive vs maladaptive emotion regulation strategies, however, can be better understood coming back to what the BOLD variability means. Several studies point out that greater BOLD variability positively influences cognitive performance94, fluid intelligence95 and most importantly for our results, interoceptive awareness96, adaptability, flexibility and efficiency of neural system in response to the multiplicity and uncertainty of environmental stimuli54,56,97,98. As a such, it is reasonable that cognitive and affective mechanisms related to the functionally connected regions are better implemented by individuals showing increased temporal variability in the network96. Accordingly, our findings suggest that the increased BOLD variability in the sensorimotor and salience networks play a critical role in predicting both high level of trait EI and adaptive emotion regulation strategy, in terms of a better social and emotional information integration, self-awareness along with a more efficient cognitive control. That temporal variability in these networks significantly predicts high traits EI and the frequency of use of Cognitive Reappraisal strategy is explained as an adaptive feature of the neural response, allowing the brain to easily access different “states”, required to complete cognitive tasks55,99. By the same token, we could also infer that less variability in salience network could imply a difficulty of the subjects to process emotional information resulting in the maladaptive emotion suppression strategy. Then, greater temporal variability may represent a neural predisposition marker which facilitates individuals in the stages underlying the dynamic process of emotional regulation, identification, selection, and implementation100. This finding provides a context for and corroborates the hypothesis that regulation strategies and their outcome may depend on factors such as the individual differences. Neural flexibility and adaptability increase perception and control of the emotional event determining at the same time the success of an emotion regulation process7. Importantly, resting-state functional connectivity may be helpful to investigate task-independent constructs101 such as those related to personal traits.
Besides these new findings, the study has some limitations to point out. While the data-driven approach allowed us to consider the activity of the whole brain and the role of naturally grouping circuits, theory driven analyses (i.e., dynamic causal model) that may facilitate inferences with respect to specific brain regions, or to identify causal relationships between them, may be a valuable and complementary alternative. Moreover, we acknowledge that a larger sample size (including thousands of participants) would have been ideal for increasing reproducibility and statistical power, as a recent paper suggested102. For what concerns the discussed networks as identified by the spatial match, they also included portions of executive networks. Future studies are needed to explore the contributions of such networks to both EI and ER. Finally, building on the existing literature, we focused on trait EI. However, it would be worthy not only to investigate other aspects of EI18, but also to extend the knowledge on the interaction between affective mechanisms and personality, considering the variability this latter may show across different situations103.
To conclude, the present findings reveal that the role of trait EI in predicting adaptive ER strategies relies on a shared and more efficient functional connectivity network involved in social and emotional information processing to understand self and others’ affective states, and in higher cognitive mechanisms which contribute to the control of emotions. Consequently, our study not only further strengthened the association between low traits EI and maladaptive ER strategies but is also represents a first step to understand the neural mechanisms able to explain this relationship. Increased variability of the BOLD signal within a sensorimotor and salience network is a mainstay for the neural structure of high traits EI and at the same time predisposes to the use of adaptive emotional regulation strategy. By contrast, a decreased variability in salience network predisposed to the use of a maladaptive emotion regulation strategy.
Methods
Participants
The data analysed in this study were selected from the open-source dataset “Max Planck Institute Leipzig Mind-Brain-Body Dataset LEMON”104. All subjects were recruited by researchers at the University of Leipzig, in Germany, between 2013 and 2015, and data were collected in accordance with the Declaration of Helsinki for a study which protocol was approved by the ethics committee at the medical faculty of the University of Leipzig. For the present study, we extracted a subset of participants representing a young adult healthy population. Based on the information provided by the authors, indeed, we defined the sample by the following inclusion criteria: no substance use or abuse (negative at the Multi 8/2 Drogen-Tauchtest)104, and no past or present psychopathologies diagnosis, screened by the SCID-I, and by the Hamilton Depression Scale (HAM-D105). The final subset was composed of 79 subjects (23 females; age range: 20–35 years; mean education: 12.39 years). For each participant, we extracted raw data from structural MRI scans (T1 Weighted-MP2RAGE) and functional MRI scans (rs-fMRI). With regards to trait measures, the scores of the following self-administered questionnaires were selected: TeiQUE-SF (Trait EI Questionnaire-Short Form), ERQ (ER Questionnaire) and CERQ (Cognitive ER Questionnaire).
Trait measures
The trait EI questionnaire short-form (TEIQue—SF) is used to measure EI as a personality trait62. The questionnaire was administered in the German validated version106 and assessed four factors: well-being (α = 0.94), self-control (α = 0.86), emotionality (α = 0.90), sociability (α = 0.88), plus a total index of trait EI, which consists of the average of the above factors (α = 0.96). As reported by the authors, reliability of the model-fit of the German validated version was acceptable to good: χ2 (54, N = 352) = 147.78, CFI = 0.95, SRMR = 0.049 and RMSEA = 0.07) and the test validity was reliable (0.88 ≤ α ≤ 0.96)106.
The emotion regulation questionnaire (ERQ) is adopted to measure the interindividual differences in the frequency of use of ER strategies12. The ERQ questionnaire was administered in the German validated version107, and consists of 10 items evaluating the tendency to use cognitive reappraisal (6 items, α = 0.73) and suppression of emotions (4 items, α = 0.76). The German version showed a good internal validity (0.73 ≤ α ≤ 0.76) and a good reliability (RMSEA = 0.068, CFI = 0.94, SRMR = 0.056)107.
The cognitive emotion regulation questionnaire (CERQ108) was administered in German validated version63 and consists of 36 items divided into 9 scales that measure nine strategies defined as adaptive: acceptance (α = 0.75), positive refocusing (α = 0.86), refocus on planning (α = 0.77), focus on positivity (α = 0.78), putting into perspective (α = 0.77); and four non-adaptive strategies: self-blame (α = 0.73), blaming others (α = 0.67), rumination (α = 0.66), catastrophizing (α = 0.73) . The German version of the scale showed good reliability (0.66 ≤ α ≤ 0.84) and a good model-fit factorial validity (χ2 = 482,267, df = 288, p < 0.01; RMSEA = 0.066; CFI = 0.922; TLI = 0.905)63 (see Table 4 for a summary of sample’s descriptive statistics for each scale and subscale).
MRI data acquisition
Structural and functional MRI data in the LEMON dataset were acquired with a 3 Tesla MRI scanner (Verio, Siemens Healthcare GmbH). During the acquisition, subjects were asked to remain awake with open eyes while looking at a low-contrast fixation cross. For our analyses we considered a BOLD rs-fMRI scan, using T2-weighted multiband EPI* sequence (TR = 1400 ms, TE = 30 ms, flip angle = 69°, echo spacing = 0.67 ms number of volumes = 657, voxel size = 2.3 mm, total acquisition time was 15 min 30 s) and T1-weighted structural volumes acquired using MP2RAGE sequence (TR = 5000 ms, TE = 2.92 ms, TI1 = 700 ms, TI2 = 2500 ms, FOV = 256 mm, voxel size = 1 mm isotropic) The structural volumes were acquired with 176 slices interspersed during 8 min 22 s of scanning104.
Trait measures analyses
To test whether trait EI predicts ER processes, we implemented two different analyses using SPSS Statistics for Windows, version 25.0 (SPSS Inc., Chicago, Ill., USA). In the first analysis we implemented a multivariate linear regression (MLR) with ERQ and CERQ questionnaires as dependent variables, while the total trait EI was included as a predictor. Moreover, to assess the effect of every subscale of trait EI, we next implemented a multivariate multiple linear regression (MMLR) with each subscale of the ERQ and CERQ questionnaires as dependent variables, and the four factors of the TeiQue-SF as predictors. Variance inflation factor (VIF) and Pearson’s correlation among the TEIQue-SF subscales were used in order to examine multi-collinearity and relative association in the regression model. Gender was used as a categorical fixed factor to test its effect in the regression model. The type I error was controlled by applying false discovery rate (FDR) correction to p-values.
Neuroimaging analyses
Pre-processing and functional connectivity analysis were conducted using CONN MATLAB Toolbox (version 18b)109. Firstly, we implemented CONN’s default pre-processing pipeline using SMP12 default parameters which includes the following steps: functional realignment and unwarping, translation and centering, functional outlier detection (conservative settings), functional direct segmentation and normalization (1 mm resolution), structural translation, and centering, structural segmentation and normalization (2.4 mm resolution), functional and structural smoothing (spatial convolution with Gaussian kernel 8 mm). Next, the denoising phase was implemented. The objective of this phase is the identification and elimination of confounding variables and artefacts from the estimated BOLD signal. Briefly, these factors are derived from three different sources (BOLD signal coming from white matter or cerebrospinal fluid masks, parameters and outliers defined in the pre-processing step, and an estimate of the pre-processing the subjects’ motion parameters)110. Once identified, the factors are entered into a regression model (Ordinary Least Squares) as covariates. Finally, a 0.0008–0.09 Hz temporal band-pass filter standard for resting-state connectivity analyses was applied to the time series. Next, the functional connectivity analysis has been implemented. For this study, we chose to use a data-driven approach by implementing a group-independent component analysis (group-ICA). The group-ICA implemented by CONN includes the following steps: pre-conditioning variance normalization, concatenation of the BOLD signal along the temporal dimension, dimensionality reduction at the group level, fast-ICA for spatial component estimation, and the back-projection for spatial estimation on the individual subject110. The number of independent components to be identified was set to 20 as software CONN suggests as default, and in line with previous studies using low model order analysis111,112,113. In order to separate noise components from the underlying resting-state networks, every identified IC were visually inspected and compared with CONN’s networks atlas using spatial match-to-template function. This feature measures the overlap between eight brain networks (Default Mode Network, Sensorimotor, Visual, Salience, Dorsal Attention, Frontoparietal, Language, Cerebellar; defined from CONN’s ICA analyses of HCP dataset/497 subjects) and the IC’s spatial map associated with each individual network component. One out of 20 ICs (IC17) did not allow for the delineation of specific areas due to its extent and it was discarded from the following analyses. We then extracted the temporal variability of each remaining IC’s, calculated in CONN as SD of each BOLD time-series110. Type I error was controlled using cluster-size-based false discovery rate (FDR) correction (p < 0.05, voxel thresholded at p < 0.001114 tab, within each analysis). Next, to assess the relationship between IC’s temporal variability and both trait EI and ER, we implemented 2 different analysis by using SPSS Statistics for Windows, version 25.0 (SPSS Inc., Chicago, Ill., USA). Firstly, to address which of the 20 identified IC’s predicted the trait EI, we tested the individual explanatory variables effect (IC’s BOLD variability values) on the TEIQue-SF factors and total index by using a Multiple Linear Regression model (Ordinary Least Squares) with a stepwise method (forward) for each dependent variable, and gender as a categorical fixed factor in order to test its effect in the regression model. Since we do not expect that all the identified components were related to the investigated construct, we chose a method of fitting regression models in which the choice of predictor variables is made by an automatic procedure. This methodology consists of testing the incremental predictivity of the model: starting from a model with no predictor, each explanatory variable is added to the model and compared to the inclusion or exclusion threshold criterion (in our case predictor’s p-value ≤ 0.05 for inclusion) until the model reaches its maximum predictivity. Finally, the BOLD temporal variability of IC’s that resulted to be significant predictors of trait EI in the previous analysis were entered a Multivariate Multiple Regression (MMR) as independent variables to predict ER scores (ERQ and CERQ subscales) and gender as a categorical fixed factor in order to test its effect in the regression model. To avoid multiple comparisons issues, type I error was controlled applying false discovery rate correction (FDR) within each analysis.
Data availability
The complete LEMON Data can be accessed via Gesellschaft für wissenschaftliche Datenverarbeitung mbH Göttingen (GWDG) https://www.gwdg.de/. Raw and preprocessed data at this location is accessible through web browser https://ftp.gwdg.de/pub/misc/MPI-Leipzig_Mind-Brain-Body-LEMON/ and a fast FTP connection (ftp://ftp.gwdg.de/pub/misc/MPI-Leipzig_Mind-Brain-Body-LEMON/). In the case the location of the data changes in the future, the location of the dataset can be resolved with PID 21.11101/0000-0007-C379-5 (e.g. http://hdl.handle.net/21.11101/0000-0007-C379-5).
References
Campbell-Sills, L. & Barlow, D. H. Incorporating emotion regulation into conceptualizations and treatments of anxiety and mood disorders. In Handbook of Emotion Regulation (ed. Gross, J.) 542–559 (The Guilford Press, 2007).
Cole, P. M., Martin, S. E. & Dennis, T. A. Emotion regulation as a scientific construct: Methodological challenges and directions for child development research. Child Dev. 75, 317–333 (2004).
Grecucci, A., Frederickson, J. & Job, R. Editorial: Advances in emotion regulation: From neuroscience to psychotherapy. Front. Psychol. 8, 985 (2017).
Gross, J. J. & Thompson, R. A. Emotion regulation: Conceptual foundations. In Handbook of emotion regulation (ed. Gross, J.) 3–24 (The Guilford Press, 2007).
Aldao, A., Nolen-Hoeksema, S. & Schweizer, S. Emotion-regulation strategies across psychopathology: A meta-analytic review. Clin. Psychol. Rev. 30, 217–237 (2010).
Nolen-Hoeksema, S., Wisco, B. E. & Lyubomirsky, S. Rethinking rumination. Perspect. Psychol. Sci. 3, 400–424 (2008).
Bonanno, G. A. & Burton, C. L. Regulatory flexibility: An individual differences perspective on coping and emotion regulation. Perspect. Psychol. Sci. 8, 591–612 (2013).
Eftekhari, A., Zoellner, L. A. & Vigil, S. A. Patterns of emotion regulation and psychopathology. Anxiety Stress Coping 22, 571–586 (2009).
Sheppes, G., Suri, G. & Gross, J. J. Emotion regulation and psychopathology. Annu. Rev. Clin. Psychol. 11, 379–405 (2015).
Brans, K., Koval, P., Verduyn, P., Lim, Y. L. & Kuppens, P. The regulation of negative and positive affect in daily life. Emotion 13, 926–939 (2013).
Nezlek, J. B. & Kuppens, P. Regulating positive and negative emotions in daily life. J. Pers. 76, 561–580 (2008).
Gross, J. J. & John, O. P. Individual differences in two emotion regulation processes: Implications for affect, relationships, and well-being. J. Pers. Soc. Psychol. 85, 348–362 (2003).
Dryman, M. T. & Heimberg, R. G. Emotion regulation in social anxiety and depression: A systematic review of expressive suppression and cognitive reappraisal. Clin. Psychol. Rev. 65, 17–42 (2018).
Ehring, T., Tuschen-Caffier, B., Schnülle, J., Fischer, S. & Gross, J. J. Emotion regulation and vulnerability to depression: Spontaneous versus instructed use of emotion suppression and reappraisal. Emotion 10, 563–572 (2010).
Webb, T. L., Miles, E. & Sheeran, P. Dealing with feeling: A meta-analysis of the effectiveness of strategies derived from the process model of emotion regulation. Psychol. Bull. 138, 775–808 (2012).
Schönfelder, S., Kanske, P., Heissler, J. & Wessa, M. Time course of emotion-related responding during distraction and reappraisal. Soc. Cogn. Affect. Neurosci. 9, 1310–1319 (2014).
Morawetz, C., Bode, S., Baudewig, J. & Heekeren, H. R. Effective amygdala-prefrontal connectivity predicts individual differences in successful emotion regulation. Soc. Cogn. Affect. Neurosci. 12, 569–585 (2017).
Peña-Sarrionandia, A., Mikolajczak, M. & Gross, J. J. Integrating emotion regulation and emotional intelligence traditions: A meta-analysis. Front. Psychol. 6, 160 (2015).
Hughes, D. J. & Evans, T. R. Comment: Trait EI moderates the relationship between ability EI and emotion regulation. Emot. Rev. 8, 331–332 (2016).
Smith, S. M. et al. Correspondence of the brain’s functional architecture during activation and rest. Proc. Natl. Acad. Sci. 106, 13040–13045 (2009).
Hughes, D. J. & Evans, T. R. Putting ‘emotional intelligences’ in their place: Introducing the integrated model of affect-related individual differences. Front. Psychol. 9, 2155 (2018).
Côté, S. & Hideg, I. The ability to influence others via emotion displays: A new dimension of emotional intelligence. Organ. Psychol. Rev. 1, 53–71 (2011).
Goldenberg, I., Matheson, K. & Mantler, J. The assessment of emotional intelligence: A comparison of performance-based and self-report methodologies. J. Pers. Assess. 86, 33–45 (2006).
MacCann, C., Fogarty, G. J., Zeidner, M. & Roberts, R. D. Coping mediates the relationship between emotional intelligence (EI) and academic achievement. Contemp. Educ. Psychol. 36, 60–70 (2011).
Mikolajczak, M., Nelis, D., Hansenne, M. & Quoidbach, J. If you can regulate sadness, you can probably regulate shame: Associations between trait emotional intelligence, emotion regulation and coping efficiency across discrete emotions. Personal. Individ. Differ. 44, 1356–1368 (2008).
Mayer, J. D., Caruso, D. R. & Salovey, P. The ability model of emotional intelligence: Principles and updates. Emot. Rev. 8, 290–300 (2016).
Bastian, V. A., Burns, N. R. & Nettelbeck, T. Emotional intelligence predicts life skills, but not as well as personality and cognitive abilities. Personal. Individ. Differ. 39, 1135–1145 (2005).
Bucich, M. & MacCann, C. Emotional intelligence and day-to-day emotion regulation processes: Examining motives for social sharing. Personal. Individ. Differ. 137, 22–26 (2019).
Davis, S. K. & Humphrey, N. Ability versus trait emotional intelligence: Dual influences on adolescent psychological adaptation. J. Individ. Differ. 35, 54–62 (2014).
Killgore, W. D. S. & Yurgelun-Todd, D. A. Neural correlates of emotional intelligence in adolescent children. Cogn. Affect. Behav. Neurosci. 7, 140–151 (2007).
Kreifelts, B., Ethofer, T., Huberle, E., Grodd, W. & Wildgruber, D. Association of trait emotional intelligence and individual fMRI-activation patterns during the perception of social signals from voice and face. Hum. Brain Mapp. 31, 979–991 (2009).
Mestre, J. M., MacCann, C., Guil, R. & Roberts, R. D. Models of cognitive ability and emotion can better inform contemporary emotional intelligence frameworks. Emot. Rev. 8, 322–330 (2016).
Banks, S. J., Eddy, K. T., Angstadt, M., Nathan, P. J. & Phan, K. L. Amygdala–frontal connectivity during emotion regulation. Soc. Cogn. Affect. Neurosci. 2, 303–312 (2007).
Buhle, J. T. et al. Cognitive reappraisal of emotion: A meta-analysis of human neuroimaging studies. Cereb. Cortex 24, 2981–2990 (2014).
Carlson, J. M. & Mujica-Parodi, L. R. A disposition to reappraise decreases anterior insula reactivity during anxious anticipation. Biol. Psychol. 85, 383–385 (2010).
Drabant, E. M., McRae, K., Manuck, S. B., Hariri, A. R. & Gross, J. J. Individual differences in typical reappraisal use predict amygdala and prefrontal responses. Biol. Psychiatry 65, 367–373 (2009).
Grecucci, A., Giorgetta, C., van’t Wout, M., Bonini, N. & Sanfey, A. G. Reappraising the ultimatum: An fMRI study of emotion regulation and decision making. Cereb. Cortex 23, 399–410 (2013).
Ochsner, K. N., Silvers, J. A. & Buhle, J. T. Functional imaging studies of emotion regulation: A synthetic review and evolving model of the cognitive control of emotion: Functional imaging studies of emotion regulation. Ann. N. Y. Acad. Sci. 1251, E1–E24 (2012).
Giuliani, N. R., Drabant, E. M. & Gross, J. J. Anterior cingulate cortex volume and emotion regulation: Is bigger better? Biol. Psychol. 86, 379–382 (2011).
Hermann, A., Bieber, A., Keck, T., Vaitl, D. & Stark, R. Brain structural basis of cognitive reappraisal and expressive suppression. Soc. Cogn. Affect. Neurosci. 9, 1435–1442 (2014).
Sala-Llonch, R. et al. Brain connectivity during resting state and subsequent working memory task predicts behavioural performance. Cortex 48, 1187–1196 (2012).
Tavor, I. et al. Task-free MRI predicts individual differences in brain activity during task performance. Science 352, 216–220 (2016).
Kaiser, R. H., Andrews-Hanna, J. R., Wager, T. D. & Pizzagalli, D. A. Large-scale network dysfunction in major depressive disorder: A meta-analysis of resting-state functional connectivity. JAMA Psychiatry 72, 603 (2015).
Veer, I. M. Whole brain resting-state analysis reveals decreased functional connectivity in major depression. Front. Syst. Neurosci. 4, 41 (2010).
Davis, S. K., Nowland, R. & Qualter, P. The role of emotional intelligence in the maintenance of depression symptoms and loneliness among children. Front. Psychol. 10, 1672 (2019).
Pan, W. et al. Identifying the core components of emotional intelligence: Evidence from amplitude of low-frequency fluctuations during resting state. PLoS ONE 9, e111435 (2014).
Killgore, W. D. S. et al. Emotional intelligence is associated with connectivity within and between resting state networks. Soc. Cogn. Affect. Neurosci. 12, 1624–1636 (2017).
Takeuchi, H. et al. Resting state functional connectivity associated with trait emotional intelligence. Neuroimage 83, 318–328 (2013).
Picó-Pérez, M. et al. Dispositional use of emotion regulation strategies and resting-state cortico-limbic functional connectivity. Brain Imaging Behav. 12, 1022–1031 (2018).
Uchida, M. et al. Emotion regulation ability varies in relation to intrinsic functional brain architecture. Soc. Cogn. Affect. Neurosci. 10, 1738–1748 (2015).
Dörfel, D., Gärtner, A. & Scheffel, C. Resting state cortico-limbic functional connectivity and dispositional use of emotion regulation strategies: A replication and extension study. Front. Behav. Neurosci. 14, 128 (2020).
Grecucci, A., Sorella, S. & Consolini, J. Decoding Individual Differences in Controlling, Expressing and Inhibiting Anger from Structural Brain Networks: A Supervised Machine Learning Approach.
Bijsterbosch, J. Introduction to Resting State fMRI Functional Connectivity (Oxford University Press, 2017).
Garrett, D. D., Kovacevic, N., McIntosh, A. R. & Grady, C. L. Blood oxygen level-dependent signal variability is more than just noise. J. Neurosci. 30, 4914–4921 (2010).
Garrett, D. D., Kovacevic, N., McIntosh, A. R. & Grady, C. L. The importance of being variable. J. Neurosci. 31, 4496–4503 (2011).
Vakorin, V. A., Lippe, S. & McIntosh, A. R. Variability of brain signals processed locally transforms into higher connectivity with brain development. J. Neurosci. 31, 6405–6413 (2011).
Guassi Moreira, J. F., McLaughlin, K. A. & Silvers, J. A. Spatial and temporal cortical variability track with age and affective experience during emotion regulation in youth. Dev. Psychol. 55, 1921–1937 (2019).
Aldao, A., Sheppes, G. & Gross, J. J. Emotion regulation flexibility. Cogn. Ther. Res. 39, 263–278 (2015).
Heller, A. S. & Casey, B. J. The neurodynamics of emotion: Delineating typical and atypical emotional processes during adolescence. Dev. Sci. 19, 3–18 (2016).
Sorella, S., Vellani, V., Siugzdaite, R., Feraco, P. & Grecucci, A. Structural and functional brain networks of individual differences in trait anger and anger control: An unsupervised machine learning study. Eur. J. Neurosci. https://doi.org/10.1111/ejn.15537 (2021).
Armbruster-Genç, D. J. N., Ueltzhöffer, K. & Fiebach, C. J. Brain signal variability differentially affects cognitive flexibility and cognitive stability. J. Neurosci. 36, 3978–3987 (2016).
Petrides, K. V. Psychometric properties of the trait emotional intelligence questionnaire (TEIQue). In Assessing Emotional Intelligence (eds Parker, J. D. A. et al.) 85–101 (Springer, 2009).
Loch, N., Hiller, W. & Witthöft, M. D. cognitive emotion regulation questionnaire (CERQ): Erste teststatistische Überprüfung einer deutschen Adaption. Z. Für Klin. Psychol. Psychother. 40, 94–106 (2011).
Etkin, A., Büchel, C. & Gross, J. J. The neural bases of emotion regulation. Nat. Rev. Neurosci. 16, 693–700 (2015).
Pallant, J. SPSS Survival Manual: A Step by Step Guide to Data Analysis Using IBM SPSS (McGraw Hill, 2013).
Multivariate Data Analysis. (Prentice Hall, 2010).
Petrides, K. V., Pita, R. & Kokkinaki, F. The location of trait emotional intelligence in personality factor space. Br. J. Psychol. 98, 273–289 (2007).
Fernández-Berrocal, P. & Extremera, N. Ability emotional intelligence, depression, and well-being. Emot. Rev. 8, 311–315 (2016).
Hertel, J., Schütz, A. & Lammers, C.-H. Emotional intelligence and mental disorder. J. Clin. Psychol. 65, 942–954 (2009).
Hasegawa, A., Somatori, K., Nishimura, H., Hattori, Y. & Kunisato, Y. Depression, rumination, and impulsive action: A latent variable approach to behavioral impulsivity. J. Psychol. 155, 717–737 (2021).
Bugay-Sökmez, A., Manuoğlu, E., Coşkun, M. & Sümer, N. Predictors of rumination and co-rumination: The role of attachment dimensions, self-compassion and self-esteem. Curr. Psychol. https://doi.org/10.1007/s12144-021-01799-0 (2021).
Lanteigne, D. M., Flynn, J. J., Eastabrook, J. M. & Hollenstein, T. Discordant patterns among emotional experience, arousal, and expression in adolescence: Relations with emotion regulation and internalizing problems. Can. J. Behav. Sci. Rev. Can. Sci. Comport. 46, 29–39 (2014).
Gross, J. J. The emerging field of emotion regulation: An integrative review. Rev. Gen. Psychol. 2, 271–299 (1998).
Eisenberg, N. & Fabes, R. A. The relation of young children’s vicarious emotional responding to social competence, regulation, and emotionality. Cogn. Emot. 9, 203–228 (1995).
Spaapen, D. L., Waters, F., Brummer, L., Stopa, L. & Bucks, R. S. The emotion regulation questionnaire: Validation of the ERQ-9 in two community samples. Psychol. Assess. 26, 46–54 (2014).
Zhao, Y. & Zhao, G. Emotion regulation and depressive symptoms: Examining the mediation effects of school connectedness in Chinese late adolescents. J. Adolesc. 40, 14–23 (2015).
Zimmermann, P. & Iwanski, A. Emotion regulation from early adolescence to emerging adulthood and middle adulthood: Age differences, gender differences, and emotion-specific developmental variations. Int. J. Behav. Dev. 38, 182–194 (2014).
Yeh, K.-H., Bedford, O., Wu, C.-W., Wang, S.-Y. & Yen, N.-S. Suppression benefits boys in Taiwan: The relation between gender, emotional regulation strategy, and mental health. Front. Psychol. 8, 315 (2017).
Jiang, X., Moreno, J. & Ng, Z. Examining the interplay of emotion regulation strategies, social stress, and gender in predicting life satisfaction of emerging adults. Personal. Individ. Differ. 185, 111255 (2022).
Pamilo, S. et al. Functional subdivision of group-ICA results of fMRI data collected during cinema viewing. PLoS ONE 7, e42000 (2012).
Grecucci, A., Rubicondo, D., Siugzdaite, R., Surian, L. & Job, R. Uncovering the social deficits in the autistic brain. A source-based morphometric study. Front. Neurosci. 10, 388 (2016).
Gallese, V. & Keysers, C. Mirror neurons: A sensorimotor representation system. Behav. Brain Sci. 24, 983–984 (2001).
Pineda, J. A. Sensorimotor cortex as a critical component of an ‘extended’ mirror neuron system: Does it solve the development, correspondence, and control problems in mirroring? Behav. Brain Funct. 4, 47 (2008).
Damasio, A. R. et al. Subcortical and cortical brain activity during the feeling of self-generated emotions. Nat. Neurosci. 3, 1049–1056 (2000).
Critchley, H. D., Wiens, S., Rotshtein, P., Öhman, A. & Dolan, R. J. Neural systems supporting interoceptive awareness. Nat. Neurosci. 7, 189–195 (2004).
Lisowska, A. et al. Joint pairing and structured mapping of convolutional brain morphological multiplexes for early dementia diagnosis. Brain Connect. 9, 22–36 (2019).
Ochsner, K. N. & Gross, J. J. Cognitive emotion regulation: Insights from social cognitive and affective neuroscience. Curr. Dir. Psychol. Sci. 17, 153–158 (2008).
Papousek, I. et al. The capacity for generating cognitive reappraisals is reflected in asymmetric activation of frontal brain regions. Brain Imaging Behav. 11, 577–590 (2017).
Huang, H. et al. Aberrant resting-state functional connectivity of salience network in first-episode schizophrenia. Brain Imaging Behav. 14, 1350–1360 (2020).
Seeley, W. W. The salience network: A neural system for perceiving and responding to homeostatic demands. J. Neurosci. 39, 9878–9882 (2019).
Pierce, J. E. & Péron, J. The basal ganglia and the cerebellum in human emotion. Soc. Cogn. Affect. Neurosci. 15, 599–613 (2020).
Phan, K. L. et al. Neural substrates for voluntary suppression of negative affect: A functional magnetic resonance imaging study. Biol. Psychiatry 57, 210–219 (2005).
Vanderhasselt, M.-A., Kühn, S. & De Raedt, R. ‘Put on your poker face’: Neural systems supporting the anticipation for expressive suppression and cognitive reappraisal. Soc. Cogn. Affect. Neurosci. 8, 903–910 (2013).
Garrett, D. D., Kovacevic, N., McIntosh, A. R. & Grady, C. L. The modulation of BOLD variability between cognitive states varies by age and processing speed. Cereb. Cortex 23, 684–693 (2013).
Yang, S. et al. Temporal variability of cortical gyral-sulcal resting state functional activity correlates with fluid intelligence. Front. Neural Circuits 13, 36 (2019).
Stoica, T. & Depue, B. Shared characteristics of intrinsic connectivity networks underlying interoceptive awareness and empathy. Front. Hum. Neurosci. 14, 571070 (2020).
Garrett, D. D. et al. Moment-to-moment brain signal variability: A next frontier in human brain mapping? Neurosci. Biobehav. Rev. 37, 610–624 (2013).
Hudetz, A. G., Liu, X. & Pillay, S. Dynamic repertoire of intrinsic brain states is reduced in propofol-induced unconsciousness. Brain Connect. 5, 10–22 (2015).
Petroni, A. et al. The variability of neural responses to naturalistic videos change with age and sex. eNeuro. https://doi.org/10.1523/ENEURO.0244-17.2017 (2018).
Gross, J. J. The extended process model of emotion regulation: Elaborations, applications, and future directions. Psychol. Inq. 26, 130–137 (2015).
Elliott, M. L. et al. What is the test-retest reliability of common task-functional MRI measures? New empirical evidence and a meta-analysis. Psychol. Sci. 31, 792–806 (2020).
Marek, S. et al. Reproducible brain-wide association studies require thousands of individuals. Nature 603, 654–660 (2022).
Mischel, W. & Shoda, Y. A cognitive-affective system theory of personality: Reconceptualizing situations, dispositions, dynamics, and invariance in personality structure. Psychol. Rev. 102, 246–268 (1995).
Babayan, A. et al. A mind-brain-body dataset of MRI, EEG, cognition, emotion, and peripheral physiology in young and old adults. Sci. Data 6, 180308 (2019).
Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 23, 56–62 (1960).
Freudenthaler, H. H., Neubauer, A. C., Gabler, P., Scherl, W. G. & Rindermann, H. Testing and validating the trait emotional intelligence questionnaire (TEIQue) in a German-speaking sample. Personal. Individ. Differ. 45, 673–678 (2008).
Abler, B. & Kessler, H. Emotion regulation questionnaire—Eine deutschsprachige Fassung des ERQ von Gross und John. Diagnostica 55, 144–152 (2009).
Garnefski, N., Kraaij, V. & Spinhoven, P. Negative life events, cognitive emotion regulation and emotional problems. Personal. Individ. Differ. 30, 1311–1327 (2001).
Whitfield-Gabrieli, S. & Nieto-Castanon, A. Conn: A functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2, 125–141 (2012).
Nieto-Castanon, A. Handbook of Functional Connectivity Magnetic Resonance Imaging Methods in CONN (Hilbert Press, 2020).
Abou Elseoud, A. et al. Group-ICA model order highlights patterns of functional brain connectivity. Front. Syst. Neurosci. 5, 37 (2011).
Saviola, F. et al. Trait and state anxiety are mapped differently in the human brain. Sci. Rep. 10, 11112 (2020).
Sorella, S., Vellani, V., Siugzdaite, R., Feraco, P. & Grecucci, A. Structural and functional brain networks of individual differences in trait anger and anger control: An unsupervised machine learning study. Eur. J. Neurosci. 55, 510–527 (2022).
Worsley, K. J. et al. A unified statistical approach for determining significant signals in images of cerebral activation. Hum. Brain Mapp. 4, 58–73 (1996).
Author information
Authors and Affiliations
Contributions
F.Z. conceived the work, analysed data, wrote the manuscript. B.M. wrote and reviewed the manuscript. A.G. conceived and supervised the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zanella, F., Monachesi, B. & Grecucci, A. Resting-state BOLD temporal variability in sensorimotor and salience networks underlies trait emotional intelligence and explains differences in emotion regulation strategies. Sci Rep 12, 15163 (2022). https://doi.org/10.1038/s41598-022-19477-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-19477-x
- Springer Nature Limited
This article is cited by
-
Decoding acceptance and reappraisal strategies from resting state macro networks
Scientific Reports (2024)
-
Resting-state functional connectivity and structural differences between smokers and healthy non-smokers
Scientific Reports (2024)
-
Neural variability in three major psychiatric disorders
Molecular Psychiatry (2023)