Abstract
Aseptic prosthetic loosening (APL) and prosthetic joint infections (PJI) are frequent complications of hip and knee implants. Polymorphisms of cytokines and nitric oxide (NO), key inflammatory molecules in APL and PJI pathogenesis, could explain individual susceptibility to these complications. Three cytokines (IL-1-a, IL-1-β, TNF-α) and two nitric oxide synthase (NOS2, NOS3) genes polymorphisms were genotyped in 77 APL and 117 PJI patients and 145 controls with aseptic hip or knee implants that were implanted for > 16 years. Plasma cytokines and nitrate-nitrite (NOx) levels also were measured. The TT genotype and T allele of (+3954 C/T, exon 5, rs1143634) IL-1β polymorphism were more frequent in APL patients compared to controls (P = 0.03 and P = 0.02, respectively). No genotypic associations in PJI patients were observed. Plasma IL-6, TNF-α and NOx were significantly different between APL and controls (P < 0.0001). Plasma IL-1β and IL-6 were significantly higher in APL T allele carriers vs. non-carriers (P < 0.03). Knee implant (HR 2.488, 95% CI 1.307–4.739, P = 0.005), male gender (HR 2.252, 95% CI 1.121–4.525, P = 0.023), carriages of the TT genotype of the (+3954 C/T) IL-1β polymorphism (HR 3.704, 95% CI 1.274–10.753, P = 0.016) and AA genotype of the (exon 22) NOS2 polymorphism (HR 3.509, 95% CI 1.266–9.709, P = 0.016) were independently associated with a shorter implant survival by Cox regression. No genotypic associations in PJI patients were observed. Genotyping of IL-1β (+3954 C/T, exon 5, rs1143634) and NOS2 (exon 22) polymorphisms could be useful as predictors of early hip or knee APL.
Similar content being viewed by others
Introduction
Aseptic prosthetic loosening (APL) is a major clinical problem that results in pain and thus the need for revision surgery1,2,3,4,5, but the pathophysiology behind this complication is unclear. However, proinflammatory cytokines play an important role in bone remodelling6,7,8. Interleukin-1 isoforms (IL-1α and β) and tumor necrosis factor-α (TNF- α) are potent stimulators of bone resorption by inducing osteoclastogenesis and studies have shown that cytokines are involved in hip and knee APL. Ex-vivo studies of cultured synovium from patients with failed prostheses produced high levels of IL-1, TNF-α and IL-69. In situ hybridization of the same membranes showed high levels of IL-1β mRNA in macrophages4.
Genes encoding IL-1α, IL-1β, TNF-α and other cytokines are polymorphic, and the various alleles may have different translational efficiency, affecting cytokines production10,11,12,13. Associations between cytokines polymorphisms: (-238 A/G, rs361525) of TNF-α, (-174 G/C, rs1800795), (-597 G/A, rs1800797) and (-572 G/C, rs 1800796) of IL-6, (-330 T/G, rs2069762) of IL-2, and (29 T/C) of the transforming growth factor-β 1 (TGF-β1) have been reported to be risk factors for osteolysis after hip arthroplasty14,15,16,17. Polymorphisms of IL-1α and β, although closely associated with bone inflammatory and infectious diseases11,18,19,20, have not been studied in depth in APL. Late loss of bone after denture implant is associated with genetic polymorphisms of IL-1-α (-889 C/T) rs1800587), IL-1-β (+3954 C/T, exon 5, rs1143634) and VNTR IL1RN, rs223466321.
Nitric oxide (NO), a free radical produced through the metabolism of arginine by the nitric oxide synthase (NOS), is crucial in bone metabolism. The NOS endothelial isoform (eNOS or NOS3) is constitutively expressed in bone, whereas inducible NOS (iNOS or NOS2) is expressed by bone cells in response to IL-1-β and TNF-α. The NO produced stimulates bone loss22,23. NOS3 gene expression is regulated by inflammatory stimuli through the Akt-kinase pathway24. Knock-out mice for NOS3 have marked defects in osteoblast function25,26.
Some NOS3 polymorphisms have been linked to differences in NO blood levels or in protein expression in response to several stimuli, including the 27-bp repeat in intron 4, the (-786 T/C,) in the promoter region (rs2070744), and the missense (E298D) in exon 7 (rs1799983). These NOS3 polymorphisms are associated with bone diseases27,28. Several polymorphisms of NOS2, such as the highly polymorphic CCTTT micro-satellite at the promoter region, the G/A substitution at position 37498, and G/A in exon 22 (iNOS 22), are associated with rheumatoid arthritis29,30. However, the association between NOS3 or NOS2 polymorphisms and APL has not been studied so far.
Despite careful management of preventive measures, prosthetic joint infections (PJI) can develop in up to 1.7% of primary hip and 2.5% of knee arthroplasties31. A functional variant of of IL-1β (-511 C/T, rs16944) is associated with increased susceptibility to PJI as are polymorphisms of the mannose-binding lectin (MBL) (-550 C/G, rs11003125, codon 54 A/G, rs1800450), toll-like receptor 9 (TLR9) (-1486 T/C, rs 187084), and the vitamin D receptor (VDR) (T/C, rs1544410)32,33.
The aim of this study was to analyze a potential association between hip and knee PJI and APL and some polymorphisms of cytokines and NOS that were previously associated with inflammatory bone diseases, ([-889 C/T] IL-1α, [+ 3954 C/T, exon 5] IL-1β, [-308 G/A] TNF-α]), ([-786 T/C] NOS3 and [exon 22, NOS2]). We compared a group of APL and PJI patients with hip or knee implants to control patients who had neither complication after ≥ 16 years. A secondary aim was to study the expression of cytokines and NOS in the different genotypes by measuring IL-1β, IL-6, TNF-α and nitrate-nitrite (NOx) plasma levels in APL and PJI patients and controls.
Material and methods
Patients
We recruited patients who were admitted with APL of hip or knee for a second arthroplasty or with PJI for antimicrobial treatment at the Hospital Universitario Central de Asturias (HUCA) between January 2003 and April 2021. Surgical confirmation of the APL and a negative bacteriological culture of surgical samples were required for inclusion as APL. PJI patients were included if they fulfilled the 2013 Infectious Diseases Society of America PJI diagnostic criteria34. Cultures of surgical and sinus tract samples from PJI patients were collected.
The main indication for the primary arthroplasty was degenerative osteoarthritis in all the APL and PJI patients, and controls. Other diseases which could affect the bone metabolism, such as hyperparathyroidism, chronic kidney insufficiency, rheumatoid arthritis and other inflammatory arthropathies, steroids use, autoimmune diseases, and avascular necrosis of the femoral head, were excluded from the study.
Controls were individuals admitted to the HUCA to undergo a primary hip or knee arthroplasty since January 2003 and were followed ≥ 16 years until April 2022 or implant–unrelated death without developing APL or PJI.
Patients and controls were members of a homogeneous population; all were Europeans and residents of the same region (Asturias, Northern Spain). Each participant gave written informed consent for the study, which was approved by the Ethics Committee of the HUCA with the number 2020.113.
Genotypic analysis
Genomic DNA was extracted from peripheral leukocytes following a salting-out method35. Polymorphisms of cytokines ([-889 C/T] IL-1α, rs1800587, [+3954 C/T] exon 5 IL-1β, rs1143634, and [–308 G/A]), TNF-α, rs1800629], NOS3 [-786 T/C], rs2070744, and NOS2 [exon 22]) were determined by PCR (Table 1)18,27. The PCR results were confirmed by sequencing representative samples for each genotype of each polymorphism. PCR products were electrophoresed on a 2% low-melting agarose gel and the fragments were excised from the gel, purified with spin columns (DNA gel extraction Kit, Millipore, Billerica, MA, USA) and sequenced on an ABI Prism 310 Genetic Analyser (Applied Biosystems, Foster City, CA, USA).
Cytokines levels
Plasma cytokine levels (IL-1β, IL-6, TNF-α) were measured by ELISA kits from R&D Systems (R&D Systems Inc., 614 McKinley Place, MN, USA). Cytokine concentrations were calculated by comparing sample absorbance with the absorbance of pooled plasma enriched with increasing amounts of recombinant human cytokine.
Plasma nitrate and nitrite levels
NO is a short-lived free radical gas that rapidly reacts with oxygen to generate the stable metabolites nitrate and nitrite. Thus, NO levels were assessed indirectly by measuring the accumulation of nitrates and nitrites. Plasma nitrate and nitrite (NOx) determinations were performed using the Griess reaction27,36. Results were expressed as μM of NOx/sample.
Statistical analysis
Non-parametric tests were used to assess the comparison of continuous variables because of their non-Gaussian distribution. Values are reported as median and interquartile range (IQR) or percentage as appropriate. Mann–Whitney U and Kruskal–Wallis tests were used to compare two or more groups, respectively, and the-chi-square test to compare proportions. Multivariate Cox proportional hazards models were constructed to assess the relationship of prosthesis loosening and infection with the diverse genotypes and the factors independently associated with such clinical outcomes. All the reported p values are two-sided. A P value < 0.05 was considered as statistically significant. SPSS v. 25 software (IBM Corp., Armonk, NY, USA) was used for statistical calculations. Hardy–Weinberg equilibrium was calculated for each genotype and allele.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Hospital Universitario Central de Asturias (HUCA).
Consent to participate
Each participant gave written informed consent for inclusion in the study.
Results
A total of 339 patients were included in the study: 77 with APL, 117 with PJI, and 145 controls. The median age at the time of the first surgery was 68.8 years (IQR 61.2–74.9), and 60.5% were women. Controls had a median follow-up of their hip or knee implants without developing APL or PJI of 16.46 years (IQR 16.16–16.90). As compared with controls, APL patients were younger and had more hip implants than controls, whereas PJI patients were more commonly males (Table 2).
The most frequently isolated microorganism was Staphylococcus epidermidis, which was recovered alone or in combination with other bacteria in 46 (39.3%) of the PJI patients. Other pure or mixed isolates were Staphylococcus aureus in 35 patients (29.9%, 13 of them, 37.1%, methicillin-resistant), other Gram-positive bacteria in 25 (21.4%) and Gram-negative bacteria in 23 (19.7%) patients.
There were no differences in IL-1β levels among the groups, but controls had substantially higher levels of IL-6 than the APL and PJI groups, whereas APL patients had higher TNF-α serum levels than the other two groups. Plasma NOx levels were significantly higher in the controls compared to APL patients (Table 3).
All genotypes and alleles analyzed were in Hardy Weinberg equilibrium except for the IL-1α (-889 C/T) polymorphism in the controls. Table 4 describes the relationships among the diverse polymorphisms analyzed and the three groups of patients. Overall, there were no statistically significant associations when the three groups were considered altogether. However, when compared individually, the genotypes and alleles of the IL-1β (+3954 C/T, exon 5, rs1143634) polymorphisms were differently distributed (P = 0.03 and P = 0.02, respectively) between APL patients and controls, mainly due to a higher representation of the variant T allele and TT genotype in the APL group (these individual comparisons data are not shown in Table 4).
Plasma IL-1β and IL-6 were significantly higher in APL IL-1β (+3954C/T, exon 5) T allele carriers compared to non-carriers (IL-1 β: 11.79 [IQR 3.41–21.14] vs. 2.11 [IQR 1.61–5.5], P < 0.03); IL-6: 43.59 [IQR 16.21–146.47 vs. 38.12 (IQR 15.0–128.7, P = 0.006]). Plasma levels of NOx did not differ in carriers of the different NOS3 and NOS2 genotypes.
Multivariate Cox proportional hazard models were elaborated for each polymorphism to evaluate the associations between the diverse genotypes with prosthesis loosening and infection over time. No polymorphisms genotypes were significantly associated with PJI, but genotypes of the IL-1β (+3954 C/T, exon 5) and NOS2 (exon 22) SNPs showed different behavior in the APL group. Table 5 shows the results of these two multivariate regressions. IL-1β (+ 3954 C/T, exon 5) SNP independently associated with knee prosthesis while the NOS2 (exon 22) SNP also associated with knee prosthesis and male gender.
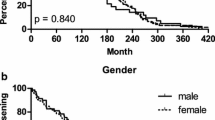
Figure 1 depicts the Cox regression curves survival of the joint replacement according to the IL-1β (+3954 C/T) and NOS2 (exon 22) genotypes. Patients carrying the respective variant homozygous genotypes experienced earlier prosthesis loosening than those carrying the heterozygous and homozygous wild genotypes, an effect that was mainly noted during the first 5 years after surgery.
Discussion
Total hip and knee replacements are some of the most successful and cost-effective surgeries performed. Although the success rate at 10 years exceeds 90% in hip37,38 and knee arthroplasties39,40,41,42,43, prosthesis, failure remains a problem necessitating revision surgery, which is costly both in monetary terms and morbidity42,43. It is difficult to predict who will require an early replacement.
We found a significant association between a polymorphism within exon 5 of IL-1β (+3954 C/T, rs1143634) and early APL. The TT genotype of this IL-1β polymorphism showed a 3.7-fold higher probability of requiring a joint replacement because of loosening within the first 5 years after surgery, compared to controls with implants lasting ≥ 16 years. This association between IL-1β polymorphism and early loosening association was especially strong with knee APL (Table 5).
IL-1 and TNF-α genes are transcriptionally activated in patients and in experimental models of APL44,45,46,47. IL-1 and TNF-α activate bone-resorbing giant cells that can increase bone loss around implants, which is characteristic of APL48. Osteoporotic fractures due to a reduction in the bone mineral density are associated with an 86-base pair repeat polymorphism in the IL-1RN gene49. There is a linkage disequilibrium between IL-1α, IL-1β and IL-1RN genes, all of which are encoded very close to each other on the long arm of chromosome 250. Thus, it is difficult to be sure whether these associations are specific for a particular gene in the IL-1 family, or even depend on other unknown gene from the same chromosome. Some haplotypes of the IL1R1-IL1A-IL1B-IL1RN gene cluster associated with enhancement to (IL1A-IL1B-IL1RN haplotype) or protection against knee osteoarthritis (IL1B-IL1RN haplotype)20.
Associations between polymorphisms in the IL-1 gene and aseptic loosening have been studied in maxillofacial surgery. An association between carriage of the IL-1β (+3954 C/T, exon 5) T allele and other IL-1 polymorphisms and unsuccessful retaining overdentures and periodontitis in smokers and non-smokers was reported21,51,52,53,54,55,56. An association between a (-889) polymorphism of the promoter region of IL-1α and susceptibility to juvenile rheumatoid arthritis and to osteomyelitis was also reported11,18.
The IL-1 gene variations were previously examined for associations with various chronic inflammatory diseases and with altered levels of inflammatory mediators56,57. We observed that APL patients with the TT genotype of the IL-1β polymorphism had the highest plasma levels of IL-1β and IL-6 compared to carriers of other genotypes of the APL group. Although immunohistochemistry studies of the removed synovial membranes or measure of the cytokine levels in synovial fluid were not performed in our study, based on the work of others58 we would expect that patients with the TT genotype of the IL-1β polymorphism might have proinflammatory cytokines (IL-1β and IL-6) overexpressed in synovial fluids around the implants, facilitating the aseptic loosening of the prosthesis.
We also found an association of the NOS2 (exon 22) polymorphisms with early APL. Carriers of the AA genotype and A allele of this NOS2 polymorphism had a 3.5 fold higher probability of early loss of their implants, mainly during the first 5 years after surgery. The association of NOS2 (exon 22 polymorphism with APL was especially strong with knee prosthesis loosening and with male gender.
Although plasma NOx levels were significantly decreased in APL patients compared to controls, we could find no differences in plasma NOx levels among carriers of the different genotypes of the NOS2 and NOS3 polymorphisms. However we have measured plasma NOx only at one point in time, rather than a 24 h urine nitrate assay that would give a better picture of how much NOx is produced. A relationship of the NOS3 (27-bp, intron 4) polymorphism and osteomyelitis27 and of the NOS3(Glu298Asp) polymorphism and hip osteoporotic fractures28 was previously reported emphasizing the key role of NO in bone metabolism.Thus, the mechanism underneath the association of this NOS2 (exon 22) polymorphism and APL remain elusive.
Regarding PJI, no association of cytokines or NOS polymorphism were observed in our study. An association of the other IL-1 β genetic variant, the IL-1β (-511C/T, rs 16944) polymorphism, not genotyped here, and PJI was reported32,33. However the TNF-α (-308 G/A rs1800629) polymorphism negative association with PJI we observed fully agree with others59.
The main limitations of this study is the relatively small number of APL patients included and the limited number of cytokines and NOS2 and NOS3 polymorphisms genotyped due to budget limitations. The strong points of the work are the novelty of genotyping NOS2 and NOS3 polymorphisms, the inclusion of a good number of PJI patients and the use of patients with orthopaedic prosthesis lasting > 16 years as controls.
To our knowledge this the first study describing an association of IL-1β gene (+3954 C/T, exon 5, rs1143634) and NOS2 (exon 22) polymorphisms and early APL. However, further research in larger European and non-European populations is needed to clarify these genetic associations with early APL.
Data availability
All data generated or analysed during this study are included in this published article.
Change history
13 December 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41598-022-26126-w
References
Santavirta, S. et al. Aggressive granulomatous lesions associated with hip arthroplasty: Inmunopathological studies. J. Bone Joint Surg. [Am.] 72A, 252–258. https://doi.org/10.3109/17453678909150108 (1990).
Schmalzried, T. P. et al. The mechanism of loosening of cemented acetabular components in total hip arthroplasty: Analysis of specimens retrieved at autopsy. Clin. Orthop. Relat. Res. 274, 60–78 (1992).
Amstutz, H. C., Campbell, P., Kossovsky, N. & Clarke, I. C. Mechanism and clinical significance of wear debris induced osteolysis. Clin. Orthop. Relat. Res. 276, 7–18 (1992).
Jiranek, W. A. et al. Production of cytokines around loosened cemented acetabular components: Analysis with inmunohistochemical techniques and in situ hibridization. J. Bone Joint Surg. [Am.] 75A, 863–879. https://doi.org/10.2106/00004623-199306000-00007 (1993).
Wooley, P. H. & Schwarz, E. M. Aseptic loosening. Gene Ther. 11, 402–407. https://doi.org/10.1038/sj.gt.3302202 (2004).
Norredin, S. & Masri, B. Periprosthetic osteolysis: Genetics, mechanisms and potential therapeutic interventions. J. Can. Chir. 55, 408–417. https://doi.org/10.1503/cjs.003711 (2012).
Wallach, S., Avioli, L. V., Feinblatt, J. D. & Carstens, J. H. Jr. Cytokines and bone metabolism. Calcif. Tissue Int. 53, 293–296. https://doi.org/10.1007/BF01351830 (1993).
Kurori, T., Shingu, M., Koshihara, Y. & Nobunaga, M. Effects of cytokines on alkaline phosphatase and osteocalcin production, calcification and calcium release by human osteoblastic cells. Br. J. Rheumatol. 33, 224–230. https://doi.org/10.1093/rheumatology/33.3.224 (1994).
Chiba, J., Rubash, H. E., Kim, K. J. & Iwaki, Y. The characterization of cytokines in the interface tissue obtained from failed cementless total hip arthroplasty with and without femoral osteolysis. Clin. Orthop. Relat. Res. 300, 304–312 (1994).
Pociot, F., Molvig, J., Wogensen, L., Worsaae, H. & Nerup, J. A. TaqI polymorphism in the human interleukin-1β (IL-1β) gene correlates with IL-1β secretion in vitro. Eur. J. Clin. Invest. 22, 396–402. https://doi.org/10.1111/j.1365-2362.1992.tb01480.x (1992).
McDowell, T. L., Symons, J. A., Ploski, R., Forre, O. & Duff, G. W. Genetic association between juvenile rheumatoid arthritis and a novel interleukin-1 alpha polymorphism. Arthritis Rheum. 38, 221–228. https://doi.org/10.1002/art.1780380210 (1995).
D’Alfonso, S., Colombo, G., Della-Bella, S., Scorza, R. & Momigliano-Richiardi, P. P. Association between polymorphism in the TNF region and systemic lupus erythematosus in the Italian population. Tissue Antigens 47, 551–555. https://doi.org/10.1111/j.1399-0039.1996.tb02598.x (1996).
Stuber, F., Petersen, M., Bokelmann, F. & Schade, U. A genomic polymorphism within the tumor necrosis factor locus influences plasma tumor necrosis factor-alpha concentrations and outcome of patients with severe sepsis. Crit. Care Med. 24, 381–384. https://doi.org/10.1097/00003246-199603000-00004 (1996).
Wilkinson, J. M. et al. Variation in the TNF gene promoter and risk of osteolysis after total hip arthroplasty. J. Bone Miner. Res. 18, 1995–2001. https://doi.org/10.1359/jbmr.2003.18.11.1995 (2003).
Kolundzic, R., Orlic, D., Trkulja, V., Pavelic, K. & Troselj, K. G. Single nucleotide polymorphisms in the interleukin-6 gene promoter, tumor necrosis factor-alpha gene promoter, and transforming growth factor-beta1 gene signal sequence as predictors of time to onset of aseptic loosening after total hip arthroplasty: Preliminary study. J. Orthop. Sci. 11, 592–600. https://doi.org/10.1007/s00776-006-1069-y (2006).
Gallo, J., Mrazek, F. & Petrek, M. Variation in cytokine genes can contribute to severity of acetabular osteolysis and risk for revision in patients with ABDG I total hip arthroplasty: A genetic association study. BMC Med. Genet. 10, 109. https://doi.org/10.1186/1471-2350/10/109 (2009).
Del Buono, A., Denaro, V. & Maffulli, N. Genetic susceptibility to aseptic loosening following total hip arthroplasty: A systematic review. Br. Med. Bull. 101, 39–55. https://doi.org/10.1093/bmb/ldr011 (2012).
Asensi, V. et al. IL-1α (-889) promoter polymorphism is a risk factor for osteomyelitis. Am. J. Med. Gen. 119A, 132–136. https://doi.org/10.1128/IAI.72.7.3823-3828.2004 (2003).
Knight, J. Polymorphisms in tumor necrosis factor and other cytokines as risks for infectious diseases and the septic syndrome. Curr. Infect. Dis. Rep. 3, 427–439. https://doi.org/10.1007/s11908-007-1010-3 (2001).
Smith, A. J. et al. Extended haplotypes and linkage disequilibrium in the IL1R1-IL1A-IL1B-IL1RN gene cluster: Association with knee osteoarthritis. Genes Immunity 5, 451–460. https://doi.org/10.1038/sj.gene.6364107 (2004).
Sampaio Fernandes, M., Vaz, P., Braga, A. C., Sampaio Fernandes, J. C. & Figueiral, M. H. The role of IL-1 gene polymorphisms (IL-1A, IL1B, and IL1RN) as a risk factor in unsuccesful implant retaining overdentures. J. Prosthodont. Res. 61, 439–449. https://doi.org/10.1016/j.jpor.2017.01.004 (2017).
Ralston, S. H. et al. Nitric oxide: A cytokine-induced regulator of bone resorption. J. Bone Miner. Res. 10, 1040–1049. https://doi.org/10.1002/jbmr.5650100708 (1995).
Van’tHof, R. J. & Ralston, S. Nitric oxide and bone. Immunology 2001(103), 255–261. https://doi.org/10.1046/j.1365-2567.2001.01261.x (2001).
Michell, B. J. et al. The Akt kinase signals directly to endothelial nitric oxide synthase. Curr. Biol. 9, 845–848. https://doi.org/10.1016/s0960-9822(99)80371-6 (1999).
Armour, K. E. et al. Defective bone formation and anabolic response to exogenous estrogen in mice with targeted disruption of endothelial nitric oxide synthase. Endocrinology 142, 760–766. https://doi.org/10.1210/endo.142.2.7977 (2001).
Aguirre, J. et al. Endothelial nitric oxide synthase gene-deficient mice demonstrate marked retardation in postnatal bone formation, reduced bone volume, and defects in osteoblast maduration and activity. Am. J. Pathol. 158, 247–257. https://doi.org/10.1016/S0002-9440(10)63963-6 (2001).
Asensi, V. et al. The NOS3 (27-bp repeat, intron 4) polymorphism is associated with susceptibility to osteomyelitis. Nitric Oxide 16, 44–53 (2007).
Taylor, B. C. et al. Association of endothelial nitric oxide synthase genotypes with bone mineral density, bone loss, hip structure, and risk of fracture in older women. The SOF study. Bone 39, 174–180. https://doi.org/10.1016/j.niox.2006.06.00 (2006).
Pascual, M. et al. Genetic determinants of rheumathoid arthritis: the inducible nitric oxide synthase (NOS2) gene promoter polymorphism. Genes Immun. 3, 299–301. https://doi.org/10.1038/sj.gene.6363856 (2002).
Gonzalez-Gay, M. A. et al. Inducible but not endothelial nitric oxide synthase polymorphism is associated with susceptibility to rheumatoid arthritis in Northwest Spain. Rheumatology 43, 1182–1185. https://doi.org/10.1093/rheumatology/keh283 (2004).
Cataldo, M. A., Petrosillo, N., Cipriani, M., Cauda, R. & Tacconelli, E. Prosthetic joint infection.recent developments in diagnosis and management. J. Infect. 61, 443–448. https://doi.org/10.1016/j.inf.2010.09.033 (2010).
Stahelova, A., Mrazek, F., Smizansky, M., Petrek, M. & Gallo, J. Variation in the IL1B, TNF and IL6 genes and individual susceptibility to prosthetic joint infection. BMC Immunol. 13, 25. https://www.biomedcentral.com/1471-21-2172/13/25 (2012).
Zhou, X. et al. Genetic susceptibility to prosthetic joint infection following total joint arthroplasty: A systematic review. Gene 563, 76–82. https://doi.org/10.1016/j.gene.2015.03.005 (2015).
Osmon, D. R. et al. Diagnosis and management of prosthetic joint infection: Clinical practice guidelines by the Infectious Diseases Society of America. Clin. Infect. Dis. 56, e1-125. https://doi.org/10.1093/cid/cis803 (2013).
Miller, S. A., Dykes, D. D. & Polesky, H. F. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 16, 1215–1222. https://doi.org/10.1093/nar/16.3.1215 (1988).
Giovannoni, G., Land, J. M., Keir, G., Thompson, E. J. & Heales, S. J. Adaptation of the nitrate reductase and Griess reaction methods for the measurement of serum nitrate plus nitrite levels. Ann. Clin. Biochem. 34, 193–198. https://doi.org/10.1177/000456329703400212 (1997).
Malchau, H., Herberts, P. & Ahnfelt, L. Prognosis of total hip replacement in Sweden: Follow-up of 92.675 operations performed 1978–1990. Acta Ortop. Scand. 64, 497–506. https://doi.org/10.3109/17453679308993679 (1993).
Eskelinen, A. et al. Total hip arthroplasty for primary osteoarthrosis in younger patients in the Finnish arthroplasty register: 4,661 primary replacements followed for 0–22 years. Acta Orthop. 76, 28–41. https://doi.org/10.1080/00016470510030292 (2005).
Ranawat, C. S., Flynn, W. F., Saddler, S., Hansraj, K. K. & Maynard, M. J. Long-term results of the total condylar knee arthroplasty. A 15-year survivorship study. Clin. Orthop. Relat. Res. 286, 94–102 (1993).
Maloney, W. J. Aseptic failure in total knee arthroplasty. In Revision Total Knee Arthroplasty (eds Engh, G. A. & Rorabek, C. H.) 46–60 (Williams & Wilkins, 1997).
Mangaleshkar, S. R., Bajaj, S. K. & Thomas, A. P. Denham total knee arthroplasty: A 10-year follow-up study. J Arthroplasty 17, 550–555. https://doi.org/10.1054/arth.2002.31976 (2002).
Crowe, J. F., Sculco, T. P. & Kahn, B. Revision total hip arthroplasty: Hospital cost and reimbursement analysis. Clin. Orthop. Relat. Res. 413, 175–182. https://doi.org/10.1097/01.blo.0000072469.32680.b6 (2003).
Paprosky, W. G., Weeden, S. H. & Bowling, J. W. Jr. Component removal in revision total hip artrhoplasty. Clin. Orthop. Relat. Res. 393, 181–193. https://doi.org/10.1097/00003086-200112000-00021 (2001).
Granchi, D. et al. Bone-resorbing cytokines in serum of patients with aseptic loosening of hip prosthesis. J. Bone Joint Surg. Br. 80B, 912–917. https://doi.org/10.1302/0301-620x.80b5.8513 (1998).
Haynes, D. R., Rogers, S. D., Hay, S., Pearcy, M. J. & Howie, D. W. The differences in toxicity and release of bone-resorbing mediators induced by titanium and cobalt-chromium-alloy wear particles. J. Bone Joint Surg. Am. 75A, 825–834. https://doi.org/10.2106/00004623-199306000-00004 (1993).
Glant, T. T. et al. Particulate-induced prostaglandin- and cytokine mediated bone resorption in an experimental system and in failed joint replacements. Am. J. Therapeut. 3, 27–41. https://doi.org/10.1097/00045391-199601000-00006 (1996).
Goodman, S. B. et al. A clinical-pathologic-biochemical study of the membrane surrounding loosened and non-loosened total hip arthroplasties. Clin. Orthop. Relat. Res. 244, 182–187 (1989).
Kobayashi, K. et al. Tumor necrosis factor alpha stimulates osteoclast differentiation by a mechanism independent of the OD/RANKL-RANK interaction. J. Exp. Med. 191, 275–286. https://doi.org/10.1084/jem.191.2.275 (2000).
Langhdahl, B. L., Lokke, E., Carstens, M., Stenkjaer, L. L. & Eriksen, E. F. Osteoporotic fractures are associated with an 86-base pair repeat polymorphism in the interleukin-1 receptor antagonist gene, but not with polymorphisms in the interleukin-1β gene. J. Bone Miner. Res. 15, 402–414. https://doi.org/10.1359/jbmr.2000.15.3.402 (2000).
Nicklin, M. J., Weith, A. & Duff, G. W. A physical map of the region encompassing the human interleukin-1 alpha, interleukin-1 beta, and interleukin-1 receptor antagonist genes. Genomics 19, 382–384. https://doi.org/10.1006/geno.1994.1076 (1994).
Lopez, N. J., Jara, L. & Valenzuela, C. Y. Association of interleukin-1 polymorphisms with periodontal disease. J. Periodontol. 76, 234–243. https://doi.org/10.1902/jop.2005.76.2.234 (2005).
Feloutzis, A. et al. IL-1 gene polymorphism and smoking as risk factors for peri-implant bone loss in a well-maintained population. Clin. Oral Implants Res. 14, 10–17. https://doi.org/10.1034/j.1600-0501.2003.140102.x (2003).
Gruica, B., Wang, H. Y., Lang, N. P. & Buser, D. Impact of IL-1 genotype and smoking status on the prognosis of osseointegrated implants. Clin. Oral Implants Res. 15, 393–400. https://doi.org/10.1111/j.1600-0501.2004.01026.x (2004).
Jansson, H., Hamberg, K., De Bruyn, H. & Bratthall, G. Clinical consequences of IL-1 genotype on early implant failures in patients under periodontal maintenance. Clin. Implant Dent. Relat. Res. 7, 51–59. https://doi.org/10.1111/j.1708-8208.2005.tb00047.x (2005).
Kornman, K. S. & Duff, G. W. Candidate genes as potential links between periodontal and cardiovascular diseases. Ann. Periodontol. 6, 48–57. https://doi.org/10.1902/annals.2001.6.1.48 (2001).
Buchs, N. et al. IL-1B and IL-1Ra gene polymorphisms and disease severity in rheumatoid arthritis: Interaction with their plasma levels. Genes Immun. 2, 222–228. https://doi.org/10.1038/sj.gene.6363766 (2001).
McDevitt, M. J., Russell, C. M., Schmid, M. J. & Reinhardt, R. A. Impact of increased occlusal contact, interleukin-1 genotype, and periodontitis severity on gingival crevicular fluid IL-1 beta levels. J. Periodontol. 74, 1302–1307. https://doi.org/10.1902/jop.2003.74.9.1302 (2003).
Wang, C. T., Lin, Y. T., Chiang, B. L., Lee, S. S. & Hou, S. M. Over-expression of receptor activator of nuclear factor-kappa B ligand (RANKL), inflammatory cytokines, and chemokines in periprosthetic osteolysis of loosened total hip arthroplasty. Biomaterials 31, 77–82. https://doi.org/10.1016/j.biomaterials.2009.09.017 (2010).
Salles, A. G., Antunes, L. A. A., Carvalho, P. A., Küchler, E. C. & Antunes, L. S. Association between apical periodontitis and TNF-α -308 G>A gene polymorphism: A systematic review and meta-analysis. Braz. Dent. J. 28, 535–542. https://doi.org/10.1590/0103-6440201701491 (2017).
Acknowledgements
This work was presented in part at the 42nd Meeting of the Spanish Society of Orthopaedic Surgery and Traumatology (SECOT), October 2005 and at the 2022 Infectious Diseases (ID) Week, October 2022, Washington DC, USA, abstract 205. We want to thank Professor Joshua Fierer from the Infectious Diseases Section. Veterans Administration Medical Center, University of California, San Diego (UCSD), USA for critical review and English editing of the manuscript.
Funding
Partial financial support for this study was received from the Instituto de Salud Carlos III (ISCIII) through the project PI 19/00876 and co-funded by the European Union and a research grant from VIIV Healthcare Spain given to Víctor Asensi. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by E.L.A., J.C., A.H.M., L.P.-I., I.P.-H., S.-J.-T., T.S.Z., V.A., E.V.-G. and V.A.. The first draft of the manuscript was written by E.L.A., J.C. and V.A. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained errors in reference citations in the Discussion section. Full information regarding the corrections made can be found in the correction for this Article.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
López-Anglada, E., Collazos, J., Montes, A.H. et al. IL-1 β gene (+3954 C/T, exon 5, rs1143634) and NOS2 (exon 22) polymorphisms associate with early aseptic loosening of arthroplasties. Sci Rep 12, 18382 (2022). https://doi.org/10.1038/s41598-022-22693-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-22693-0
- Springer Nature Limited