Abstract
The transition from controlled to partial support ventilation is a challenge in acute respiratory distress syndrome (ARDS) patients due to the risks of patient-self-inflicted lung injury. The magnitude of tidal volume (VT) and intrapulmonary dyssynchrony (pendelluft) are suggested mechanisms of lung injury. We conducted a prospective, observational, physiological study in a tertiary academic intensive care unit. ARDS patients transitioning from controlled to partial support ventilation were included. On these, we evaluated the association between changes in inflammatory biomarkers and esophageal pressure swing (ΔPes), transpulmonary driving pressure (ΔPL), VT, and pendelluft. Pendelluft was defined as the percentage of the tidal volume that moves from the non-dependent to the dependent lung region during inspiration, and its frequency at different thresholds (− 15, − 20 and − 25%) was also registered. Blood concentrations of inflammatory biomarkers (IL-6, IL-8, TNF-α, ANGPT2, RAGE, IL-18, Caspase-1) were measured before (T0) and after 4-h (T4) of partial support ventilation. Pendelluft, ΔPes, ΔPL and VT were recorded. Nine out of twenty-four patients (37.5%) showed a pendelluft mean ≥ 10%. The mean values of ΔPes, ΔPL, and VT were − 8.4 [− 6.7; − 10.2] cmH2O, 15.2 [12.3–16.5] cmH2O and 8.1 [7.3–8.9] m/kg PBW, respectively. Significant associations were observed between the frequency of high-magnitude pendelluft and IL-8, IL-18, and Caspase-1 changes (T0/T4 ratio). These results suggest that the frequency of high magnitude pendelluft may be a potential determinant of inflammatory response related to inspiratory efforts in ARDS patients transitioning to partial support ventilation. Future studies are needed to confirm these results.
Similar content being viewed by others
Introduction
The transition from controlled to partial support ventilation or spontaneous modes is necessary for withdrawing mechanical ventilation in the acute respiratory distress syndrome (ARDS). Both ventilatory strategies preserve diaphragmatic contraction and allow spontaneous breathing (SB). Likewise, SB favors less sedation as well as improvements in ventilation/perfusion matching, dorsal ventilation, gas exchange, hemodynamics, and attenuate ventilator-induced diaphragmatic dysfunction, among other beneficial effects1,2,3,4. However, under certain conditions, SB has been demonstrated to cause or enhance lung injury and could therefore complicate the weaning5,6,7,8.
Several mechanisms gathered under the name “patient self-inflicted lung injury” (or “P-SILI”) are proposed to explain how spontaneous breathing may damage the lung. They include excessive tidal volume, increased pulmonary transvascular pressure, and high esophageal pressure swings5,6,7,8,9. In addition, the existence of a pendelluft phenomenon, i.e. a shift of air from non-dependent to dependent lung regions during inspiration in supine position, may produce inhomogeneous lung inflation and regional overstretch. This phenomenon has been detected at the bedside through electrical impedance tomography (EIT) and is caused by local negative pleural pressure generated by diaphragmatic contraction not uniformly transmitted through dependent regions of the lungs10.
Nevertheless, there is no definitive evidence that P-SILI mechanisms such as pendelluft may effectively promote lung injury in humans. An additional challenge to advance our understanding of pendelluft is that we lack a validated approach to quantify it in magnitude and frequency, both of which may theoretically influence the impact of pendelluft on lung injury.
In the present study, we aimed to determine the potential contribution of pendelluft, esophageal pressure swing (ΔPes), transpulmonary driving pressure (ΔPL), and tidal volume (VT) to acute lung injury, assessed by changes in inflammatory biomarkers in plasma, during the transition from controlled to partial ventilatory support.
Methods
Study population
We assessed patients with moderate-severe ARDS on protective mechanical ventilation (MV) for more than 48 h, hemodynamically stable, under moderate-light sedation (Richmond Agitation-Sedation Scale (RASS) − 2 to − 3) and without paralytic agents. Patients younger than 18 years old, pregnant, with contraindications to place EIT, central nervous system injury, new sepsis or moderate-severe metabolic acidosis, were excluded.
Study protocol
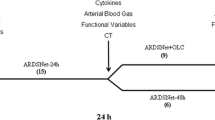
In this physiological study we prospectively monitored ARDS patients transitioning from controlled to partial support ventilation with esophageal manometry and EIT and we applied a systematic approach to measure pendelluft. In parallel, we analyzed the changes in representative biomarkers of acute lung injury to determine whether the development of pendelluft was associated with an increase in any of these biomarkers (Supplementary Figure S1).
Baseline respiratory mechanics and PEEP titration
Before initiating SB, respiratory system compliance was calculated by dividing VT by the difference between plateau pressure and total positive end-expiratory pressure (PEEP), under volume-controlled mode with VT of 6 ml/kg of predicted body weight (PBW) and respiratory rate to keep PaCO2 5.3–6.6 kPa. Optimal PEEP was defined as the PEEP associated with the lowest combination of collapse and overdistension according to EIT11. The patients were maintained at a semi‐recumbent position (30º to 45º) during the study protocol. Further details are provided in the supplementary file.
Mechanical ventilation settings
The ventilator mode was switched from volume-control ventilation (VCV) to biphasic positive airway pressure mode (BiVent, Servo-i ventilator Maquet) after detection of a regular patient’s respiratory rate (RR) (≥ 10 bpm). BiVent was applied during 4-h with a target of 10–50% of spontaneous ventilation relative to total minute ventilation (Supplementary Method S1). Pressure-high was adjusted for VT of 6 ml/kg PBW and Pressure-low for optimal PEEP according to EIT; T-high with 0.8–1.0 s duration and T-low to maintain the same RR as in VCV.
Respiratory mechanics during SB
Airway pressure (Paw), esophageal pressure (Pes), and transpulmonary pressure (PL) were registered synchronously with EIT monitoring using a pneumotachometer (FluxMed MBMED®). PL was calculated as the difference between Paw and Pes. The correct position of the esophageal catheter (Neurovent Research Inc®, Canada) was confirmed12 (Supplementary Method S1).
Ventilatory cycle and pendelluft
Regional ventilation changes were analyzed in four regions-of-interest (ROI) with similar height from non-dependent to dependent regions using EIT (Enlight 1800, Timpel®, Brazil). Because controlled, spontaneous, and mixed cycles coexist in BiVent, an algorithm was implemented to define each ventilatory cycle (Supplementary Figure S2). We analyzed ventilatory cycles for the last 10-min of each monitoring hour. Pendelluft magnitude was defined as the percentage of the normalized VT that moves from non-dependent to dependent regions during inspiration in each ventilatory cycle (expressed in negative values). The mean of pendelluft magnitude was obtained and the pendelluft frequency was estimated as the proportion of cycles presenting pendelluft magnitudes above specific cut-off points (− 15, − 20, − 25%, expressed as “− 0.15”, ” − 0.20″, “− 0.25″ in Fig. 1 and Supplementary Figure S3).
Histograms of inspiratory dyssynchrony (pendelluft) at different magnitudes. X axis corresponds to the magnitude of pendelluft and Y axis, to the percentage of ventilatory cycles with certain magnitude of pendelluft. The negative value of pendelluft represents the lost volume in non-dependent region during inspiration, expressed as fraction. The vertical red dotted line delimits the pendelluft with high magnitude cut-off − 0.2 (i.e. 20% of lost volume in non-dependent region during inspiration). (A) corresponds to Subject #2, a patient with a pendelluft mean of − 0.2 (− 20%), who presented high frequency of high-magnitude pendelluft. (B) corresponds to Subject #23, a patient with pendelluft mean of − 0.02 (− 0.2%), who presented low frequency of high-magnitude pendelluft.
Biomarkers
Pre-specified inflammatory mediators related to acute lung injury and ventilator-induced lung injury (VILI) (pro-inflammatory cytokines [IL-6, IL-8, TNF-α], biomarkers of lung epithelial [The receptor for advanced glycation end products, RAGE] and endothelial [angiopoietin-2, ANGP2] injury, and representative biomarkers of inflammasome activation [IL-18, Caspase-1])13,14,15,16,17,18,19,20,21,22,23 were measured in serum by ELISA (Human magnetic Luminex screening assay and Human Caspase-1/ICE quantikine ELISA kit) at baseline (T0) and after 4 h on BiVent mode (T4) (Supplementary Method S3).
Statistical Analysis
Summary statistics of pendelluft magnitude were estimated. For each patient, the pendelluft frequencies were calculated from the four 10-min monitoring periods. Friedman analysis was performed to compare changes in the pendelluft frequencies, ΔPes, ΔPL, and VT during the four observation periods. The Wilcoxon signed-rank test was used to compare the biomarkers at T0 and at T4.
To evaluate the individual association between ΔPes, ΔPL, VT, and pendelluft frequency with each biomarker ratio [(biomarker at T4)/(biomarker at T0)], simple linear regression models were fitted. To study the independence of associations between pendelluft and ratios while controlling for ΔPes, ΔPL and total and regional VT, multiple linear regressions were fitted. All the analyses were performed using R statistical software (R Foundation for Statistical Computing, Vienna, Austria).
Ethics approval and consent to participate
This study was performed in accordance with the Declaration of Helsinki. The Institutional Review Board reviewed and approved the study (approval number N.027/2016, Comité Ético Científico Hospital Clínico Universidad de Chile). Written informed consent was obtained from all patient’s next of kin. All methods were performed in accordance with the relevant guidelines and regulation.
Results
We included twenty-four ARDS patients of which 14 were males, with a median age of 63 [54–67] years and with body mass index of 29 [23–32] kg m− 2. Before enrollment their worst exchange values were median PaO2/FiO2 of 16.4 [12–20] kPa and worse sequential organ failure assessment (SOFA) score of 12 [9–14] with six patients treated with prone positioning. At the study entry, MV time was 6.5 [4–11] days, gas exchange and lung mechanics were already improving in most patients. Twelve patients presented with patchy, 4 with diffuse, and 8 with lobar computed tomography attenuations (Supplementary Table S1).
The pendelluft magnitude and its frequency for different cut-offs on hourly basis and the overall period are shown in Table 1. Nine patients (37.5%) showed a volume displacement mean ≥ 10% from non-dependent to dependent region during inspiration (i.e. mean pendelluft). Pendelluft frequency was lower at higher cut-off points of magnitude (19 [3.8–32]% at pendelluft-15, 10 [2–23]% at pendelluft-20, and 3 [1–15]% at pendelluft-25). ΔPes, ΔPL, and VT were − 8.5 [− 10.1; − 6.6] cmH2O, 15.1 [12.2–16.7] cmH2O and 8.1 [7.2–8.8] m/kg PBW, respectively. None of these respiratory variables significantly changed during the study period (Table 1). Respiratory rate and total minute ventilation were 24 (19.6—25.9) bpm and 10.2 (8.8—12.0) L/min, respectively; both remained stable through the 4-h period (p-value = 0.809 and p-value = 0.951, respectively).
The overall cohort plasma concentration of the biomarkers did not change between T0 and T4 with the exception of TNF-α that decreased (Supplementary Figure S4). Nevertheless, some individual patients did exhibit an increase in biomarkers concentrations. The main results of single regression models for biomarkers ratios and mean pendelluft magnitude, frequency of different pendelluft magnitudes, or respiratory variables are shown in Fig. 2, Fig. 3 and Additional File 1 Table 2. There was only a trend in correlations between IL-18 and Caspase-1 ratios and the mean pendelluft magnitude (R2 0.147 and p-value 0.064 for both biomarkers), but a significant association was observed between these biomarkers (and IL-8) and the frequency of high-magnitude pendelluft. The R2, estimate (β), and level of significance (p-value) increased as pendelluft magnitude became higher (from 15 to 25%) (Fig. 2). We did not observe association between other cytokines and the mean and frequency of pendelluft at any magnitude (Supplementary Table S2). Likewise, no significant associations were observed between biomarkers and global respiratory variables, with the exception of a positive correlation between RAGE ratio and ΔPL (Fig. 3 and Supplementary Table S2).
Scatter plots and regression analysis between (A) IL-8, (B) IL-18, (C) Caspase-1 ratios and frequencies of pendelluft magnitude − 15, − 20 and − 25%. Biomarker ratio [(biomarker at T4)/(biomarker at T0)] and the mean of pendelluft frequencies at different cut-off points of pendelluft magnitude (− 15, − 20 and − 25%), through the 4 h period of observation, were obtained for each patient. Pendelluft-15: mean of pendelluft frequency at magnitude of − 15%; Pendelluft-20: mean of pendelluft frequency at magnitude of − 20%; Pendelluft-25: mean pendelluft frequency at magnitude of − 25%. ΔPes: mean of negative deflection of esophageal pressure from the onset of inspiratory effort during the ventilatory cycle; ΔPL: mean of tidal change in transpulmonary pressure. VT: tidal volume.
Scatter plots and regression analysis between (A) IL-8, (B) IL-18, (C) Caspase-1 ratios, and respiratory variables. Biomarker ratio [(biomarker at T4)/(biomarker at T0)] and the mean values by patient of ΔPes , ΔPL and VT, through the 4 h period of observation, were obtained for each patient ΔPes: mean of negative deflection of esophageal pressure (Pes) from the onset of inspiratory effort during the ventilatory cycle; ΔPL: mean of tidal change in transpulmonary pressure. Only the association between ΔPes and ΔPL and the pendelluft 25 frequency was significant (R2 0.202 and p-value = 0.047). Several patients presented pendelluft of higher magnitude at lower than − 15 and 20 cmH2O of ΔPes and ΔPL, respectively.
To explore whether pendelluft may be mediator in the pathway between global parameters and inflammatory biomarkers, simple regressions were fitted. The mean pendelluft magnitude and frequencies at different pendelluft magnitudes were associated with VT at the dependent lung regions but not with total VT. We did not observe significant associations between pendelluft and ΔPes nor ΔPL suggesting that pendelluft frequency might be only partially influenced by these variables (Supplementary Table S3). Even more, when ΔPes, ΔPL, and VT were included as covariates in multiple linear regressions for the biomarkers ratios, the observed associations between pendelluft20-25 frequency and IL-8, IL-18, and Caspase-1 were only slightly attenuated, presenting stronger associations than the global parameters as evidenced by the magnitude of the t-statistic (Table 2).
Discussion
In the present physiological study, we found that in ARDS patients transitioning from controlled to partial support ventilation: (1) the overall concentration of inflammatory biomarkers did not change consistently, although a subgroup of patients exhibited an increase in some inflammatory biomarkers; (2) the frequency of high-magnitude pendelluft was the parameter best associated with the increase in specific inflammatory biomarkers (IL-8, IL-18, and Caspase-1), independently of ΔPes, ΔPL, and VT.
The present study describes for the first time in humans the association of high-magnitude pendelluft with the increase of specific inflammatory mediators related to VILI. The absence of a significant correlation between pendelluft and global variables, such as ΔPes, ΔPL and VT provides evidence for the current hypothesis of pendelluft as a regional phenomenon which may be related to a local change in pleural pressure10,24,25.
Not all patients developed high-magnitude pendelluft (Fig. 1) or increased inflammatory biomarkers, but the association between both was positive and significant. Indeed, we observed a progressive increase in R2, in the estimate (β), and in the level of significance (p-value) in relation to the increase in the cut-off points of pendelluft magnitude from 15 to 25% with the IL-8, IL-18 and Caspase-1 ratios. This suggests that the inflammatory response is triggered above a certain threshold of pendelluft in the same way as non-protective MV. High-magnitude pendelluft causes overstretch in lung regions during tidal inflation and it is known that cyclic stretch upregulates IL-8 in a strain-dependent manner15 and thus provides a potential explanation for the association between the frequency of high-magnitude pendelluft and the increase in IL-8. This cytokine is the major chemoattractant for neutrophils16 and the release of IL-8 is considered to play an important role in the inflammatory response and progression of VILI in patients with ARDS17.
A parallel increase in IL-18 and Caspase-1 in association with the frequency of high-magnitude pendelluft suggests a common pathway related to the inflammasome activation. IL-18 and caspase-1 have been shown to play a fundamental role in the spread of lung injury in experimental studies and in critically ill patients18,19,20,21. Even more, an elevated level of serum IL-18 has a suggested association with worse prognosis in patients with ARDS18,19. The use of MV with high VT (for a few hours) has been shown to increase the expression of IL-18 and Caspase-1 in lung tissue and plasma19. The mechanical stress produced by MV on the lung parenchyma is capable of triggering the production of reactive oxygen species in mitochondria from activated alveolar macrophages, which activate the inflammasome leading to the processing and maturation of Pro IL-1β and Pro IL-1820,21.
The decision to assess inflammatory changes through the ratio of biomarker levels at T0 and T4 was based in the large inter-individual variability reported in previous studies13,14,15,16,17,18,19,20,21,22,23,24, as well as, the expectation that only a subgroup of patients would have intense inspiratory efforts. In addition, we believe that the biomarker ratio, by representing a relationship is sensitive to changes, discriminates the most relevant interactions and allows a more personalized analysis26.
Remarkably, there was a low frequency of high-magnitude pendelluft at the SB onset. Three potential causes may explain these findings: (1) In this study, we used a personalized titration of “optimal” PEEP according to EIT to reduce lung collapse11. The titrated PEEP may reduce the neuromuscular efficiency as suggested by recent physiologic studies27,28; (2) The ventilatory mode used (BiVent) usually generates lower tidal volumes and transpulmonary pressure than fully synchronized or partially synchronized pressure-targeted modes despite similar settings on the ventilator and patient’s effort29 and has shown to be a safe and potentially beneficial ventilatory strategy30,31,32,33. We applied a similar ventilatory strategy to that used in a randomized controlled trial with BiVent (BiRDS study, ClinicalTrials.gov Identifier: NCT01862016). (3) Finally, the sedation was titrated to maintain RASS − 2 to − 3 and SB up to 50% of the total minute ventilation, which was successfully obtained and remained between 35 and 50% in all patients. In this scenario, which may be considered partially inhibitory of strong inspiratory efforts, no associations were found between the percentage of SB over total minute ventilation and pendelluft magnitude or inflammatory biomarkers.
P-SILI may be more likely when intense inspiratory efforts occur in severe ARDS ("solid-like" lungs), compared to mild ARDS ("fluid-like" lungs)34,35. In the present study, all patients had moderate to severe ARDS upon admission to the ICU. However, at the time of inclusion, patients had remained on MV for over a week on average and thus: gas exchange, lung disease, and lung mechanics were already improving in most patients. Nevertheless, high-magnitude pendelluft was observed in some patients despite protective values of ΔPes, ΔPL, and VT. Both the global parameters and pendelluft may share some common mechanisms and thus the observed associations are attenuated when including both variables in the multivariate model, but the signal from pendelluft was a stronger predictor of inflammatory response and it might be mediated by specific pathways unrelated to global parameters.
Study Limitations
Our findings must be interpreted with caution due to several limitations such as: (1) the small sample size; (2) the use of BiVent mode with optimized PEEP and analgosedation enough to maintain spontaneous ventilation up to 50% of total minute ventilation, which may have avoided high-magnitude pendelluft and a stronger inflammatory response; (3) the short period of spontaneous ventilation; (4) the potential effect of simultaneous phenomena acting as confounders on biomarkers changes; (5) the study does not allow to infer whether the changes in IL-18, Caspase 1 and IL-8 plasma concentrations observed in patients with high-magnitude pendelluft were indeed produced in the lung; and, (7) the lack of validation of the results of pendelluft in the absence of a unified definition of the phenomenon.
For all the above, the present study should be considered as a pilot exploratory and hypothesis generating study. Further research is needed to assess the role of pendelluft in clinical practice. However, to the best of our knowledge, this is the first attempt to explore the association between pendelluft and inflammation in subjects with ARDS during the first hours of spontaneous ventilation.
Conclusions
In conclusion, in ARDS patients transitioning from controlled to partial support ventilation high-magnitude pendelluft was independently associated with an increase in specific inflammatory biomarkers related to VILI (IL-8, IL-18 and Caspase-1). The development of pendelluft may be a potential determinant of P-SILI at the spontaneous ventilation onset. Future studies are needed to confirm this conclusion.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information file.
References
Vimlati, L., Kawati, R., Hedenstierna, G., Larsson, A. & Lichtwarck-Aschoff, M. Spontaneous breathing improves shunt fraction and oxygenation in comparison with controlled ventilation at a similar amount of lung collapse. Anesth. Analg. 113, 1089–1095 (2011).
Neumann, P. et al. Spontaneous breathing affects the spatial ventilation and perfusion distribution during mechanical ventilatory support. Crit. Care Med. 33, 1090–1095 (2005).
Kaplan, L. J., Bailey, H. & Formosa, V. Airway pressure release ventilation increases cardiac performance in patients with acute lung injury/adult respiratory distress syndrome. Crit. Care 5, 221–226 (2001).
Sassoon, C. S., Zhu, E. & Caiozzo, V. J. Assist-control mechanical ventilation attenuates ventilator-induced diaphragmatic dysfunction. Am. J. Respir. Crit. Care Med. 170, 626–632 (2004).
Sassoon, C. S., Zhu, E. & Caiozzo, V. J. Assist-control mechanical ventilation attenuates ventilator-induced diaphragmatic dysfunction. Am. J. Respir. Crit. Care Med. 170, 626–632 (2004).
Tonelli, R. et al. Early inspiratory effort assessment by esophageal manometry predicts noninvasive ventilation outcome in de novo respiratory failure: A pilot study. Am. J. Respir. Crit. Care Med. 202, 558–567 (2020).
Kallet, R. H., Alonso, J. A., Luce, J. M. & Matthay, M. A. Exacerbation of acute pulmonary edema during assisted mechanical ventilation using a low-tidal volume lung-protective ventilator strategy. Chest 116, 1826–1832 (1999).
Yoshida, T., Uchiyama, A., Matsuura, N., Mashimo, T. & Fujino, Y. Spontaneous breathing during lung-protective ventilation in an experimental acute lung injury model: High transpulmonary pressure associated with strong spontaneous breathing effort may worsen lung injury. Crit. Care Med. 40, 1578–1585 (2012).
Yoshida, T., Fujino, Y., Amato, M. B. & Kavanagh, B. P. Fifty years of research in ARDS. Spontaneous breathing during mechanical ventilation. Risks, mechanisms and management. Am. J. Respir. Crit. Care Med. 195, 985–92 (2017).
Brochard, L., Slutsky, A. & Pesenti, A. Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am. J. Respir. Crit. Care Med. 195, 438–442 (2017).
Yoshida, T. et al. Spontaneous effort causes occult pendelluft during mechanical ventilation. Am. J. Respir. Crit. Care Med. 188, 1420–1427 (2013).
Franchineau, G. et al. Contribution of electrical impedance tomography to setting positive end-expiratory pressure for extracorporeal membrane oxygenation-treated patients with severe acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 196, 447–457 (2017).
Baydur, A., Behrakis, P. K., Zin, W. A., Jaeger, M. & Milic-Emili, J. A simple method for assessing the validity of the esophageal balloon technique. Am. Rev. Respir. Dis. 126, 788–791 (1982).
Stüber, F. et al. Kinetic and reversibility of mechanical ventilation-associated pulmonary and systemic inflammatory response in patients with acute lung injury. Intensive Care Med. 28, 834–841 (2002).
Ranieri, V. M. et al. Effect of mechanical ventilation on inflammatory mediators in patients with acute respiratory distress syndrome: A randomized controlled trial. JAMA 282, 54–61 (1999).
Iwaki, M. et al. Mechanical stretch enhances IL-8 production in pulmonary microvascular endothelial cells. Biochem. Biophys. Res. Commun. 389, 531–536 (2009).
Donnelly, S. C. et al. Interleukin-8 and development of adult respiratory distress syndrome in at-risk patient groups. Lancet 341, 643–647 (1993).
Oudin, S. & Pugin, J. Role of MAP kinase activation in interleukin-8 production by human BEAS-2B bronchial epithelial cells submitted to cyclic stretch. Am. J. Respir. Cell. Mol. Biol. 27, 107–114 (2002).
Dong, G. et al. Serum interleukin-18: A novel prognostic indicator for acute respiratory distress syndrome. Medicine 98, e15529 (2019).
Dolinay, T. et al. Inflammasome-regulated cytokines are critical mediators of acute lung injury. Am. J. Respir. Crit. Care Med. 185, 1225–1234 (2012).
Wu, J. et al. Activation of NLRP3 inflammasome in alveolar macrophages contributes to mechanical stretch-induced lung inflammation and injury. J. Immunol. 190, 3590–3599 (2013).
Zhang, Y., Liu, G., Dull, R., Schwartz, D. E. & Hu, G. Autophagy in pulmonary macrophages mediates lung inflammatory injury via NLRP3 inflammasome activation during mechanical ventilation. Am. J. Physiol. Lung Cell Mol. Physiol. 307, L173–L185 (2014).
Jabaudon, M. et al. Association between intraoperative ventilator settings and plasma levels of soluble receptor for advanced glycation end-products in patients without pre-existing lung injury. Respirology 20, 1131–1138 (2015).
Agrawal, A. et al. Plasma angiopoietin-2 predicts the onset of acute lung injury in critically ill patients. Am. J. Respir. Crit. Care Med. 187, 736–742 (2013).
Yoshida, T. et al. Volume-controlled ventilation does not prevent injurious inflation during spontaneous effort. Am. J. Respir. Crit. Care Med. 196, 590–601 (2017).
Huang, X. et al. A computational method of defining potential biomarkers based on differential sub-networks. Sci. Rep. 7, 14339 (2017).
Yoshida, T. et al. Spontaneous effort during mechanical ventilation: Maximal injury with less positive end-expiratory pressure. Crit. Care Med. 44, e678–e688 (2016).
Morais, C. C. A. et al. High positive end-expiratory pressure renders spontaneous effort noninjurious. Am. J. Respir. Crit. Care Med. 197, 1285–1296 (2018).
Richard, J. C. et al. Potentially harmful effects of inspiratory synchronization during pressure preset ventilation. Intensive Care Med. 39, 2003–2010 (2013).
Lim, J. & Litton, E. Airway pressure release ventilation in adult patients with acute hypoxemic respiratory failure: A systematic review and meta-analysis. Crit. Care Med. 47, 1794–1799 (2019).
Jain, S. V. et al. The 30-year evolution of airway pressure release ventilation (APRV). Intensive Care Med. Exp. 4, 11 (2016).
Carvalho, N. C. et al. Higher levels of spontaneous breathing reduce lung injury in experimental moderate acute respiratory distress syndrome. Crit. Care Med. 42, e702–e715 (2014).
Xia, J. et al. Spontaneous breathing with biphasic positive airway pressure attenuates lung injury in hydrochloric acid-induced acute respiratory distress syndrome. Anesthesiology 120, 1441–1449 (2014).
Yoshida, T., Uchiyama, A., Matsuura, N., Mashimo, T. & Fujino, Y. The comparison of spontaneous breathing and muscle paralysis in two different severities of experimental lung injury. Crit. Care Med. 41, 536–545 (2013).
Yoshida, T., Amato, M. B. P., Kavanagh, B. P. & Fujino, Y. Impact of spontaneous breathing during mechanical ventilation in acute respiratory distress syndrome. Curr. Opin. Crit. Care 25, 192–198 (2019).
Acknowledgements
The authors thank Dr. Diego Ugalde for his assistance in figure design, and the nurses, respiratory therapists, medical staff, and medical technologists from Hospital Clínico Universidad de Chile for their support during the execution of the study.
Funding
This work was supported by FONDECYT [grant No 1161510 and grant No 1221829] awarded to Rodrigo Cornejo. The funding bodies had no role in the design of the study, or the collection, analysis, or interpretation of data or the manuscript preparation.
Author information
Authors and Affiliations
Contributions
R.C., D.A., P.R., D.G., C.M., A.G., M.L., R.B., M.C., S.G., V.R., G.D., L.L., J.N., D.S., A.B., M.A., N.E.: All authors contributed to the study design, data interpretation, and manuscript revisions. R.B. and D.S. performed biochemical analyses. Data analysis was performed by R.C., P.R and A.G. The first draft of the manuscript was written by R.C., and all authors commented on drafts of the manuscript. All authors agree to be accountable for all aspects of the submitted work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cornejo, R.A., Arellano, D.H., Ruiz-Rudolph, P. et al. Inflammatory biomarkers and pendelluft magnitude in ards patients transitioning from controlled to partial support ventilation. Sci Rep 12, 20233 (2022). https://doi.org/10.1038/s41598-022-24412-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-24412-1
- Springer Nature Limited
This article is cited by
-
Fluid management strategies and their interaction with mechanical ventilation: from experimental studies to clinical practice
Intensive Care Medicine Experimental (2023)
-
Pendelluft in patients with acute respiratory distress syndrome during trigger and reverse triggering breaths
Scientific Reports (2023)