Abstract
Ischemia–reperfusion injury is a risk factor for poor clinical prognosis in patients with ST-segment elevation myocardial infarction (STEMI). However, due to the inability to predict the risk of its occurrence early, the effect of intervention measures is still being determined. This study intends to construct a nomogram prediction model and evaluate its value in predicting the risk of ischemia–reperfusion injury (IRI) after primary percutaneous coronary intervention (PCI). The clinical admission data of 386 STEMI patients who underwent primary PCI were retrospectively analyzed. According to the degree of ST-segment resolution (STR), the patients were divided into the STR < 70% group (n = 197) and the STR > 70 group (n = 187). The least absolute shrinkage and selection operator (LASSO) regression method was used to screen out IRI's admission-related clinical risk factors. The R language software was used to construct and verify the IRI nomogram prediction model based on the above indicators. The peak troponin level and the incidence of in-hospital death in the STR < 70% group were significantly higher than those in the STR > 70% group (p < 0.01), and the left ventricular ejection fraction was significantly lower than that in the STR > 70% group (p < 0.01). Combined with the results of LASSO regression and receiver operating characteristic curve comparison analysis, we constructed a six-dimensional nomogram predictive model: hypertension, anterior myocardial infarction, culprit vessel, proximal occlusion, C-reactive protein (CRP) > 3.85 mg/L, white blood cell count, neutrophil cell count, and lymphocyte count. The area under the nomogram's receiver operating characteristic (ROC) curve was 0.779. The clinical decision curve found that the nomogram had good clinical applicability when the occurrence probability of IRI was between 0.23 and 0.95. The nomogram prediction model constructed based on six clinical factors at admission has good prediction efficiency and clinical applicability regarding the risk of IRI after primary PCI in patients with acute myocardial infarction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Reperfusion therapy, including primary percutaneous coronary intervention and thrombolytic treatment, can restore the blood supply of ischemic myocardium in time and limit the infarct size1. Paradoxically, however, restoration of blood supply can cause additional heart damage and complications, known as ischemic-reperfusion injury (IRI)2. When coronary occlusion blocks the blood supply to cardiomyocytes, cardiomyocytes eventually become damaged or die. Still, restoring blood supply while the heart muscle is alive may also cause microvascular embolism3, increased calcium overload in cardiomyocytes4, excessive cardiomyocyte contracture 5, massive generation of oxygen free radicals6, leukocyte aggregation causing inflammatory damage7 and platelet activation8, and other harmful changes. The above-mentioned pathophysiological changes can cause abnormalities in the mechanical activity and electrophysiological function of cardiomyocytes, which clinically manifest as adverse clinical events such as arrhythmia9 and heart failure10.
Current clinical treatments for ischemia–reperfusion injury include ischemic preconditioning11, adenosine12, GPIIbIIIa receptor antagonists13, anti-oxidative stress14, and hypothermia therapy15. However, the validity of these methods and the normative approach to their application still need to be determined. Identifying high-risk patients with an ischemia–reperfusion injury early and applying the treatment mentioned above promptly and reasonably is an essential link to achieving effective clinical treatment of IRI.
Currently, the diagnostic methods for IRI include the level of electrocardiogram (ECG) ST-segment resolution16, myocardial blush grade17, the degree of increase and change trend of cardiac biomarkers, cardiac magnetic resonance imaging18, etc. However, the above indicators are obtained after IRI, and there is a lag in timeliness, so it is impossible to identify high-risk patients with IRI early enough to guide the timely adoption of cardioprotective strategies in clinical practice.
This study aims to investigate the early clinical risk factors of ischemia–reperfusion injury in patients with acute ST-segment elevation myocardial infarction (STEMI) after primary PCI and to construct a clinical prediction model to assist in the early risk assessment of IRI high-risk patients and improve clinical treatment strategy.
Material and methods
Study population
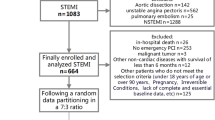
A total of 412 patients with STEMI who were admitted to the department of cardiovascular medicine at Beijing Shijitan Hospital and underwent primary PCI between January 2016 and October 2022 were retrospectively selected as the research subjects. Inclusion criteria: (1) Cardiac biomarkers exceeded 99% of the upper limit of reference values; (2) Sustained chest pain > 30 min or electrocardiogram showed ST-segment elevation of at least 0.1 mV in ≥ 2 adjacent leads; (3) Killip I–III grade on admission; (4) Onset < 12 h and successful primary PCI (5) Only revascularization was performed on the lesion vessel of the myocardial infarction during primary PCI. Exclusion criteria: (1) thrombolytic therapy before direct PCI; (2) cardiogenic shock on admission; (3) chronic total occlusion of non-infarction-related vessels; (4) previous myocardial infarction or coronary artery disease with a history of revascularization; (5) acute and chronic infectious diseases, hematopoietic abnormalities, autoimmune diseases, liver and kidney failure, and malignant tumor diseases. Finally, 386 patients were selected, including 314 males and 72 females, with a median age of 62.
Collection of clinical data
Clinical data of patients were retrospectively collected through our hospital's clinical electronic medical record system and Hospital Information System (HIS), including: 1. demographic and disease-related data: gender, age, smoking history, and comorbidities; 2. admission echocardiogram results within 24 h; 3. Laboratory test results: including pre-intervention hematological parameters, such as white blood cells, lymphocytes, neutrophils, monocyte counts, hemoglobin, etc.; preoperative serum creatinine, preoperative C-reactive protein, peak troponin level (the troponin detection time is immediately after the operation, every 4 h after the procedure, and measured once every 48 h after reaching the peak, respectively); 4. Primary PCI intervention information: culprit vessel, ischemic infarction site, whether the occlusive lesion is located in the proximal end of the culprit’s vessel, postoperative TIMI blood flow classification, whether to use a suction device, use IABP or GPIIbIIIa receptor antagonists, whether to use stent therapy, stent type, and parameters, maximum balloon inflation pressure, etc.; 5. ST-segment resolution ratio (STR): Two cardiologists independently analyzed the ECG before and 60 min after the operation and selected the lead with the highest ST-segment elevation before the procedure to calculate the STR. The calculation formula is STR = (preoperative ST-segment elevation height—ST-segment elevation height 60 min after operation) ÷ ST-segment elevation height before operation. STR ≥ 70% means complete ST-segment regression, and STR < 70% means poor ST-segment regression.
Statistical analysis
Statistical software SPSS 26.0 and R language (R 3.6.1) were used for data analysis, and measurement data were expressed as x ± s or M (P25, P75) according to whether they obeyed a normal distribution. The Mann–Whitney U test was used to compare the non-normally distributed data between the two groups. The independent sample t-test was used to compare the normally distributed data between the two groups. Categorical and graded data were expressed in case numbers, percentages, or rates. Categorical data were compared between the two groups based on the total sample size and the minimum theoretical frequency using the uncorrected Pearson chi-square test or Fisher's exact probability method. The least absolute shrinkage and selection operator (LASSO) regression method was used to screen the characteristic variables affecting STR. The "rms" program package in R language was used to construct the nomogram model and the calibration curve of the nomogram, and the "pROC" program package was used to draw the receiver operating characteristic (ROC) curve. The "caret" package was used to conduct Bootstrap self-sampling 1000 times to evaluate the prediction efficiency after internal validation, and the test level α = 0.05.
Ethical statement
The Beijing Shijitan Hospital Ethics Committee, Capital Medical University approved the study protocol and written informed consent was obtained from all patients. All methods were performed in accordance with the Declaration of Helsinki.
Results
Characteristics of clinical data
Figure 1 illustrates the study flowchart. Among the 386 subjects, the incidence rate of poor ST-segment elevation recovery (STR < 70%) was 51.0% (197/386). Patients with STR < 70% had no significant difference in age, sex, diabetes mellitus, or smoking status compared with patients with STR ≥ 70%. Compared with patients with ≥ 70% STR, patients with poor STR had a higher proportion of hypertension. In the comparison of interventional treatment data, it was found that there was no significant difference between the two groups in terms of the time from symptom onset to balloon expansion, the application of a Glycoprotein IIb/IIIa antagonist, the application of a suction catheter, and the proportion of implanted stents. However, the proportions of anterior wall myocardial infarction, infarction-related vessel LAD, and proximal vascular occlusion in patients with poor STR were significantly higher than in patients with complete STR. The LVEF of patients with insufficient STR was significantly lower than that of patients with complete STR. Finally, patients with poor ST-segment resolution had significantly higher in-hospital mortality than patients with complete STR (5.1% vs. 0.5%, p = 0.007) (Table 1).
Characteristics of laboratory examination
Compared with patients with > 70% STR, patients with poor STR had significantly higher peak troponin T, C-reactive protein level, total white blood cell, neutrophil, and monocyte counts. The level of lymphocytes in patients with poor STR was significantly lower than that in patients with > 70% STR. Patients with poor STR had higher D-dimer levels than those with > 70% STR, but the difference was insignificant. There were no significant differences in parameters such as hemoglobin level, hematocrit level, red blood cell count, platelet count, mean platelet volume, and platelet distribution width between the two groups. (Table 2).
Determination of the optimal cut-off value of CRP and the neutrophil/lymphocyte ratio for predicting STR < 70%
We used receiver operating characteristic curves to explore the CRP and neutrophil/lymphocyte ratio values in predicting STR < 70%. The results showed that the area under the ROC curve (AUC) of CRP in predicting poor ST-segment regression was 0.702 with a 95% confidence interval (95% CI) of 0.638 to 0.760, and the optimal cut-off value was > 3.85 (Youden index 0.354, sensitivity 61.79%, specificity 73.64%). The AUC of NLR to predict poor ST-segment resolution was 0.652 (95% confidence interval 0.602 to 0.700), and the best cut-off value was > 2.17 (Youden index 0.2346, sensitivity 76.65%, specificity 46.81%) (Fig. 2).
Screening of characteristic variables for the risk of adverse ST-segment regression after emergency PCI in STEMI patients
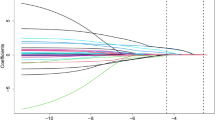
Using LASSO regression, the nine admission and interventional variables with statistical significance (hypertension, anterior myocardial infarction, culprit vessel, proximal occlusion, CRP > 3.85 mg/L, white blood cell count, neutrophil cell count, lymphocyte count, neutrophil/lymphocyte ratio > 2.17, etc.) were subjected to dimensionality reduction processing to screen out further the characteristic variables of STR < 70% risk. Using tenfold cross-validation, the λ value when the cross-validation error value is the smallest is used as the optimal solution of the model. The variable names and variable numbers of the corresponding non-zero regression coefficients are counted at this time. LASSO regression results show that the λ value when the error is the smallest is 0.006, and there are seven variables corresponding to the non-zero regression coefficient, namely, hypertension, anterior wall myocardial infarction, proximal occlusive disease, CRP > 3.85 mg/L, neutrophil count, lymphocyte count, and NLR > 2.17 (Fig. 3). These seven variables are characteristic variables for predicting the occurrence of STR < 70%.
Construction of a nomogram prediction model
According to the influencing factors found in Lasso regression, the "rms" package of R was used to construct the prototype of the nomogram for predicting the occurrence of ST-segment failure, and the scores of the seven indicators in the nomogram were added to obtain the total score. A vertical line is drawn along the corresponding score of the total score. The value on the horizontal axis of "STR < 70% risk" obtained is the risk value of poor ST-segment resolution after emergency PCI.
The AUC of the prototype nomogram model was 0.778 (95% CI 0.717–0.840). Considering that there is an interaction among neutrophil count, lymphocyte count, and NLR in the nomogram prediction model, and NLR ≥ 2.17 predicts that the AUC of STR < 70% is less than 0.70, we remove neutrophil count, lymphocyte count, and NLR, respectively, and form three improved models containing six variables. The results showed that the AUCs of the three improved nomogram prediction models were 0.775 (95% CI 0.714–0.829), 0.771 (95% CI 0.709–0.826), and 0.779 (95% CI 0.717–0.832), which were comparable to the 7-variable prototype. There was no significant difference in the AUC of the nomogram prediction model (Fig. 4).
Therefore, we finally selected neutrophil count, lymphocyte count, hypertension, anterior wall myocardial infarction, proximal occlusive lesions, and CRP > 3.85 mg/L to construct a nomogram model (Fig. 5), and the AUC of the model was 0.779 (95% CI 0.717–0.832). The Hosmer–Lemeshow bias test suggested that the deviation between the risk prediction value of the nomogram and the actual value was not statistically significant (χ2 = 4.328, p = 0.826). Bootstrap was used to draw the internal calibration diagram, which showed that the risk-fitting curve of the nomogram was closer to the ideal curve, suggesting that the nomogram had a good calibration (Fig. 6).
Analysis of the clinical applicability of the nomogram model
The predicted probability of the nomogram was used as the test variable, and the occurrence of STR < 70% in patients was used as the state variable to construct the clinical decision curve of the nomogram model. It can be seen from Fig. 7 that when the threshold probability of occurrence of STR < 70% in patients is between 0.23 and 0.95, the net benefit level of the application of the nomogram is higher than that of the "no intervention" and "all intervention" schemes. It is suggested that the nomogram has good clinical applicability.
Discussion
Timely and active restoration of blood supply to the ischemic myocardium (reperfusion) and opening of infarction-related blood vessels are the most effective measures for treating myocardial infarction19. However, reperfusion itself is a double-edged knife. Restoring oxygen and nutrients to myocardial cells and saving the ischemic myocardium can also cause myocardial stun, no-reflow phenomenon, reperfusion arrhythmia, and even irreversible myocardial cell death, that is, "ischemia–reperfusion injury."20 From a pathological point of view, microvascular obstruction (MVO) and intramyocardial hemorrhage (IMH) are the main manifestations of reperfusion injury21. Evaluation methods for ischemia–reperfusion injury include biomarkers of myocardial injury, echocardiography, TIMI myocardial perfusion classification, positron emission tomography (PET/CT), quantitative single photon emission computed tomography (SPECT), cardiac magnetic resonance imaging (MRI), etc.
Studies have found that ST-segment resolution is significantly correlated with myocardial reperfusion at the microvascular level22, and can be used to evaluate reperfusion injury16,23. In this study, the proportion of patients with ST-segment resolution < 70% was 51.0%. These patients had a higher incidence of no-reflow, higher peak troponin levels, and in-hospital mortality compared with patients with ST-segment resolution > 70%. It is suggested that patients with ST-segment resolution < 70% have a clinically significant ischemia–reperfusion injury. This study used poor STR regression as an indirect criterion for judging ischemia–reperfusion injury. It was found that hypertension, anterior wall myocardial infarction, culprit vessel, proximal occlusion, CRP > 3.85 mg/L, white blood cell count, neutrophil count, lymphocyte count, neutrophil/lymphocyte ratio > 2.17, etc., were associated with ischemia–reperfusion injury.
Previous studies have also found that patients with hypertension and ventricular hypertrophy have increased susceptibility to ischemia–reperfusion injury. Hypertension and ventricular hypertrophy are more prone to myocardial cell apoptosis, fibrosis, arrhythmia, systolic dysfunction, and abnormal expression of gene level, thus aggravating the injury effect of ischemia–reperfusion24.
This study suggests that patients with anterior myocardial infarction and the culprit vessel LAD, especially proximal occlusion, are more susceptible to severe ischemia–reperfusion injury than patients with non-anterior myocardial infarction and the culprit vessel LCX or RCA (poor ST-segment regression). There are few studies on the correlation between different infarct locations and the culprit’s vessels and the severity of the ischemia–reperfusion injury. Some studies have found that the degree of ischemia–reperfusion injury in patients with an anterior wall infarction is more severe than that of an inferior wall infarction25. The response of patients with different infarct locations to anti-ischemia–reperfusion injury treatment is different, which is speculated to be related to the anatomical structure characteristics of different regions and the differences in collateral circulation status26. The no-reflow phenomenon is closely related to ischemia–reperfusion injury. This study found that the no-reflow incidence in patients with STR < 70% was significantly higher than in patients with STR ≥ 70% (26.9% vs. 15.3%, p = 0.005). This result is consistent with previous research27,28,29.
CRP level, white blood cell count, neutrophil count, lymphocyte count, neutrophil/lymphocyte ratio, etc., are important indicators to reflect the body’s inflammatory state, and the inflammatory response plays a vital role in ischemia–reperfusion injury30,31. CRP is an acute phase response protein. Animal studies have found that a large amount of CRP is deposited in the myocardial risk area of rat heart ischemia–reperfusion injury32. Excessive neutrophil infiltration can generate ROS and secrete chemokines while exerting phagocytosis on cell debris and matrix degradation33. Boag et al. found that decreased circulating T lymphocytes were associated with myocardial microvascular embolism and a poor prognosis in STEMI patients undergoing primary PCI34. Neutrophils and lymphocytes are markers of inflammatory persistence and regulatory pathways, respectively. As an indicator of systemic inflammatory status, the neutrophil-to-lymphocyte ratio (NLR) (calculated by dividing the neutrophil count by the lymphocyte count) is associated with adverse clinical outcomes in acute myocardial infarction35. Chen et al. found that NLR was positively correlated with myocardial injury in ACS patients undergoing emergency PCI, compared with patients with NLR < 2.76, patients with NLR > 2.76 had significantly increased myocardial injury markers and decreased systolic cardiac function36. Although understanding the pathophysiology of reperfusion injury can help identify potential therapeutic strategies, no treatment for reperfusion injury has been shown to improve clinical outcomes. The reasons for this include the inability to accurately predict the risk of ischemia–reperfusion injury and to give preventive or targeted treatment at the optimal time. Early identification of patients at the highest risk for reperfusion injury may be essential in bringing therapeutic approaches to clinical practice. In diagnosing and treating acute ST-segment elevation myocardial infarction, the average time from the first medical contact to the opening of epicardial culprit vessels to achieve reperfusion at the level of large vessels is 90 min37. During this period, the clinical data obtained by doctors is very limited, including the patient's demographic characteristics, electrocardiogram, blood routine, biochemistry, myocardial enzymes, etc., and coronary lesion characteristics obtained by coronary angiography. How to use these limited clinical data to establish a simple, fast, and accurate prediction model for the risk of ischemia–reperfusion injury to identify high-risk patients early is of great significance to improving the prognosis of STMEI patients.
A nomogram is a tool for transforming statistical models into visual graphics. It is simple, intuitive, and quantifiable. The nomogram has a good predictive effect and application value in predicting the risk of heart failure and death after primary PCI in patients with acute myocardial infarction (AMI). However, there is no nomogram model based on ischemia–reperfusion injury. This study found that indicators such as hypertension, anterior myocardial infarction, culprit vessels, proximal vascular occlusion, CRP > 3.85 mg/L, white blood cell count, neutrophil count, lymphocyte count, and neutrophil/lymphocyte ratio > 2.17 were associated with the risk of ischemia–reperfusion injury in STEMI patients. The clinical decision curve analysis showed that when the threshold probability of STR < 70% in patients was between 0.23 and 0.95, the net benefit of using the nomogram to guide treatment decisions was better than "intervention at all" or "no intervention at all.” The variables included in the prediction model are easy and fast to obtain. The time required for evaluation with model tools is short, which is conducive to clinical use and promotion.
The IRI prediction model constructed in this study has limitations, such as the lack of large samples and multi-center external verification. It still needs to be further verified and improved in future research. In this study, only STR was used as the criterion for judging IRI, which may have deviations from the actual changes of myocardial ischemia–reperfusion injury. Future analysis and exploration combined with myocardial imaging technology may further improve the accuracy of this nomogram in predicting the risk of IRI. Finally, since this study is retrospective, we did not routinely detect and collect indicators such as B-type natriuretic peptide (BNP)38, low-density lipoprotein cholesterol level39, and intravascular ultrasound (IVUS)-detected attenuated plaque40 that may be associated with ischemia–reperfusion injury in STEMI patients. In future studies, we will prospectively incorporate the above index parameters to explore the possibility of further improving the accuracy of the prediction model.
In conclusion, this study screened the clinical risk factors for admission of ischemia–reperfusion injury and constructed a nomogram prediction model. This model has specific predictive efficiency and clinical applicability and may help to identify STEMI patients with a high risk of ischemia–reperfusion injury early and provide corresponding prevention and treatment.
Data availability
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
References
Markman Filho, B. & Lima, S. G. Coronary reperfusion in acute myocardial infarction: trying the optimal. Executing the possible. Arq Bras Cardiol. 117(1), 130–131 (2021).
Heusch, G. Myocardial ischaemia-reperfusion injury and cardioprotection in perspective. Nat Rev Cardiol. 17(12), 773–789 (2020).
Konijnenberg, L. S. F. et al. Pathophysiology and diagnosis of coronary microvascular dysfunction in ST-elevation myocardial infarction. Cardiovasc Res. 116(4), 787–805 (2020).
Ye, J. et al. Hydroxysafflor yellow A ameliorates myocardial ischemia/reperfusion injury by suppressing calcium overload and apoptosis. Oxid Med Cell Longev. 2021, 6643615 (2021).
Seara, F. A. C. et al. Cardiac ischemia/reperfusion injury is inversely affected by thyroid hormones excess or deficiency in male Wistar rats. PLoS ONE 13(1), e0190355 (2018).
Cadenas, S. ROS and redox signaling in myocardial ischemia-reperfusion injury and cardioprotection. Free Radic Biol Med. 117, 76–89 (2018).
Starz, C. et al. Elevated platelet-leukocyte complexes are associated with, but dispensable for myocardial ischemia-reperfusion injury. Basic Res Cardiol. 117(1), 61 (2022).
Ziegler, M., Wang, X. & Peter, K. Platelets in cardiac ischaemia/reperfusion injury: a promising therapeutic target. Cardiovasc Res. 115(7), 1178–1188 (2019).
van der Weg, K., Prinzen, F. W. & Gorgels, A. P. Editor’s Choice-reperfusion cardiac arrhythmias and their relation to reperfusion-induced cell death. Eur Heart J Acute Cardiovasc Care. 8(2), 142–152 (2019).
Wu, M. Y. et al. Current mechanistic concepts in ischemia and reperfusion injury. Cell Physiol Biochem. 46(4), 1650–1667 (2018).
Hausenloy, D. J. & Yellon, D. M. Ischaemic conditioning and reperfusion injury. Nat Rev Cardiol. 13(4), 193–209 (2016).
Headrick, J. P. & Lasley, R. D. Adenosine receptors and reperfusion injury of the heart. Handb Exp Pharmacol. 193, 189–214 (2009).
Kingma, J. G. Jr., Plante, S. & Bogaty, P. Platelet GPIIb/IIIa receptor blockade reduces infarct size in a canine model of ischemia-reperfusion. J Am Coll Cardiol. 36(7), 2317–2324 (2000).
González-Montero, J., Brito, R., Gajardo, A. I. & Rodrigo, R. Myocardial reperfusion injury and oxidative stress: therapeutic opportunities. World J Cardiol. 10(9), 74–86 (2018).
El Farissi M, Keulards DCJ, Zelis JM, van 't Veer M, Zimmermann FM, Pijls NHJ, Otterspoor LC. Hypothermia for reduction of myocardial reperfusion injury in acute myocardial infarction: closing the translational gap. Circ Cardiovasc Interv. 2021;14(8):e010326.
Reinstadler, S. J. et al. ST-segment depression resolution predicts infarct size and reperfusion injury in ST-elevation myocardial infarction. Heart 101(22), 1819–1825 (2015).
Majidi, M. et al. Implications of ventricular arrhythmia “bursts” with normal epicardial flow, myocardial blush, and ST-segment recovery in anterior ST-elevation myocardial infarction reperfusion: a biosignature of direct myocellular injury “downstream of downstream”. Eur Heart J Acute Cardiovasc Care. 4(1), 51–59 (2015).
Liu, T. et al. Intramyocardial hemorrhage and the “Wave Front” of reperfusion injury compromising myocardial salvage. J Am Coll Cardiol. 79(1), 35–48 (2022).
Saito, Y., Oyama, K., Tsujita, K., Yasuda, S. & Kobayashi, Y. Treatment strategies of acute myocardial infarction: updates on revascularization, pharmacological therapy, and beyond. J Cardiol. S0914–5087(22), 00165–00174 (2022).
Vytrykhovskyi, A. I. & Fedorchenko, M. V. Reperfusion injury in acute period of myocardial infarction-ways of prevention and correction. Wiad Lek. 75(10), 2514–2518 (2022).
Ibanez, B. et al. Cardiac MRI endpoints in myocardial infarction experimental and clinical trials: JACC scientific expert panel. J Am Coll Cardiol 74(2), 238–256 (2019).
de Lemos, J. A. & Braunwald, E. ST segment resolution as a tool for assessing the efficacy of reperfusion therapy. J Am Coll Cardiol. 38(5), 1283–1294 (2001).
Campo, G. et al. Fo ATP synthase C subunit serum levels in patients with ST-segment elevation myocardial infarction: preliminary findings. Int J Cardiol. 221, 993–997 (2016).
Pagliaro, P. & Penna, C. Hypertension, hypertrophy, and reperfusion injury. J Cardiovasc Med (Hagerstown). 18(3), 131–135 (2017).
Feldmann, K. J., Goldstein, J. A., Marinescu, V., Dixon, S. R. & Raff, G. L. Disparate impact of ischemic injury on regional wall dysfunction in acute anterior vs inferior myocardial infarction. Cardiovasc Revasc Med. 20(11), 965–972 (2019).
Bär FW, Tzivoni D, Dirksen MT, Fernández-Ortiz A, Heyndrickx GR, Brachmann J, Reiber JH, Avasthy N, Tatsuno J, Davies M, Hibberd MG, Krucoff MW; CASTEMI Study Group. Results of the first clinical study of adjunctive CAldaret (MCC-135) in patients undergoing primary percutaneous coronary intervention for ST-Elevation Myocardial Infarction: the randomized multicentre CASTEMI study. Eur Heart J. 2006;27(21):2516–23.
Sorajja, P. et al. Combined prognostic utility of ST-segment recovery and myocardial blush after primary percutaneous coronary intervention in acute myocardial infarction. Eur Heart J. 26(7), 667–674 (2005).
Schröder, R. Prognostic impact of early ST-segment resolution in acute ST-elevation myocardial infarction. Circulation 110(21), e506–e510 (2004).
Giugliano RP, Sabatine MS, Gibson CM, Roe MT, Harrington RA, Murphy SA, Morrow DA, Antman EM, Braunwald E. Combined assessment of thrombolysis in myocardial infarction flow grade, myocardial perfusion grade, and ST-segment resolution to evaluate epicardial and myocardial reperfusion. Am J Cardiol. 2004;93(11):1362–7, A5–6.
Liu, J., Wang, H. & Li, J. Inflammation and inflammatory cells in myocardial infarction and reperfusion injury: a double-edged sword. Clin Med Insights Cardiol. 10, 79–84 (2016).
Marshall, K. et al. Natural immunoglobulin M initiates an inflammatory response important for both hepatic ischemia reperfusion injury and regeneration in mice. Hepatology 67(2), 721–735 (2018).
Oh, S. J., Na Kim, E., Jai Kim, C., Choi, J. S. & Kim, K. B. The effect of C-reactive protein deposition on myocardium with ischaemia-reperfusion injury in rats. Interact Cardiovasc Thorac Surg. 25(2), 260–267 (2017).
Ong, S. B. et al. Inflammation following acute myocardial infarction: multiple players, dynamic roles, and novel therapeutic opportunities. Pharmacol Ther. 186, 73–87 (2018).
Boag, S. E. et al. T lymphocytes and fractalkine contribute to myocardial ischemia/reperfusion injury in patients. J Clin Invest. 125(8), 3063–3076 (2015).
Haybar, H., Pezeshki, S. M. S. & Saki, N. Evaluation of complete blood count parameters in cardiovascular diseases: an early indicator of prognosis?. Exp Mol Pathol. 110, 104267 (2019).
Chen, C. et al. Neutrophil to lymphocyte ratio as a predictor of myocardial damage and cardiac dysfunction in acute coronary syndrome patients. Integr Med Res. 7(2), 192–199 (2018).
Chacón-Diaz M, Custodio-Sánchez P, Rojas De la Cuba P, Yábar-Galindo G, Rodríguez-Olivares R, Miranda-Noé D, López-Rojas LM, Hernández-Vásquez A. Outcomes in ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention or pharmacoinvasive strategy in a Latin American country. BMC Cardiovasc Disord. 2022;22(1):296.
Arakawa, K. et al. B-type natriuretic peptide as a predictor of ischemia/reperfusion injury immediately after myocardial reperfusion in patients with ST-segment elevation acute myocardial infarction. Eur Heart J Acute Cardiovasc Care. 5(1), 62–70 (2016).
Reindl, M. et al. Relation of low-density lipoprotein cholesterol with microvascular injury and clinical outcome in revascularized ST-elevation myocardial infarction. J Am Heart Assoc. 6(10), e006957 (2017).
Shiono, Y. et al. Impact of attenuated plaque as detected by intravascular ultrasound on the occurrence of microvascular obstruction after percutaneous coronary intervention in patients with ST-segment elevation myocardial infarction. JACC Cardiovasc Interv. 6(8), 847–853 (2013).
Author information
Authors and Affiliations
Contributions
Z. W.: Conception and design of the research, acquisition of data, analysis, and interpretation of the data, statistical analysis, and manuscript writing. J. P.: Supervision, review & editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Z., Peng, J. The predictive value of the nomogram model of clinical risk factors for ischemia–reperfusion injury after primary percutaneous coronary intervention. Sci Rep 13, 5084 (2023). https://doi.org/10.1038/s41598-023-32222-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-32222-2
- Springer Nature Limited
This article is cited by
-
Ischemic preconditioning modulates the DNA methylation process of the rat heart to provide tolerance to withstand ischemia reperfusion injury and its associated mitochondrial dysfunction
3 Biotech (2024)
-
Optimization strategy for the early timing of bronchoalveolar lavage treatment for children with severe mycoplasma pneumoniae pneumonia
BMC Infectious Diseases (2023)