Abstract
The present study evaluates the non-communicable disease (NCD) patterns and related risk factors among people living with HIV (PLWH) in Iran. This national cross-sectional survey study was conducted on 1173 confirmed PLWHs with a mean age of 35.35 (56.82 Over 50 years old, 33.90 Under 50 years old) admitted from 15 different provinces in the country. Logistic regression was used to analyze the association of factors with having at least one NCD comorbidity. From 1173 PLWH, 225(19.18%) participants experienced at least one NCD (15.20% and 38.69% among under- and over-50-year-old patients, respectively). The prevalence of heart disease, hypertension, diabetes, and sleep apnea among all patients was 1.59%, 2.05%, 1.55%, and 10.26%, respectively. The similar prevalence for each NCD among those over 50 years was 10.11%, 15.71%, 9.01%, 25.44%, and 1.01%, 1.12%, 1.04%, and 9.23% among those under 50 years, respectively. The odds of being at risk of at least one NCD stood higher in patients over 50 years (ORadj = 2.93, 95% CI 1.96–4.37), married (ORadj = 2.48, 95% CI 1.41–4.35), divorced or widowed (ORadj = 2.78, 95% CI 1.48–5.20), and obese (ORadj = 3.82, 95% CI 2.46–5.91). According to our findings regarding the prevalence of NCDs among patients under 50 years of age, we recommend that policymakers give greater consideration to this group in the screening and care programs for NCDs since adults and the elderly are both vulnerable to the risk factors for developing NCDs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
According to the latest reports of the World Health Organization (WHO), there were 38.4 million people living with HIV (PLWH) globally by the end of 2021. Studies have predicted that nearly 75% of PLWH under care and treatment for HIV will be older than 50 at the end of 20301,2. In Iran, model-based projections estimate that 54,850 people were HIV positive and 15,552 were under antiretroviral therapy treatment in 2021. Also, 74% of all diagnosed cases were in the age group of 20–45 years at the time of diagnosis3.
Although PLWH have used a large number of antiretroviral therapies (ART) in recent years, the rapid expansion of access to treatment for elderly HIV-positive patients has some challenges globally, including an increase in the comorbidity of non-communicable diseases (NCDs), such as cardiovascular disease (CVD), type 2 diabetes mellitus (T2DM), hypertension (HTN), chronic kidney disease (CKD), and cancers4. Consequently, this increase in NCDs’ comorbidity with HIV can be defined by improved general health, a decrease in morbidity and mortality in HIV-positive patients, and an increase in life expectancy and survival5.
Many studies have represented the association between aging and NCDs in PLWH 6,7. The prevalence of NCD risk factors among PLWH, including smoking, drug or alcohol use, comorbidity with hepatitis B or C viruses, and lower socio-economic status, is higher8,9,10. Moreover, antiretroviral therapy drugs could increase the risk of hypercholesterolemia, abdominal fat, and metabolic syndrome11,12. Also, in HIV-positive patients, activation in the immune system leads to the secretion of mediators and inflammatory cytokines, which results in a higher risk for atherosclerosis and coronary artery inflammation7. Hence, studying the comorbidity of HIV and aging has become an urgent public health concern because of the increasing number of elderly PLWH13.
Research in this era faced challenges due to the complex nature of NCDs in PLWH. Despite the present challenges, several studies have shown the prevalence of NCDs in PLWH in different parts of the world. However, to our knowledge, there are limited data and screening programs in the context of this emerging problem in Iran. The study of NCDs in PLWH can provide information for policymakers to better manage the overlapped epidemics of HIV and NCDs. So, the present national study aimed to address aging and NCDs, namely diabetes, hypertension, heart disease, obstructive sleep apnea co-morbidity, and related factors in PLWH.
Methods
Sampling method and study population
The prevalence estimation formula was used to estimate the sample size based on non-communicable disease (NCD) prevalence among PLWH14,15,16.
The study’s sample size, considering design effects and attrition, was estimated at about 1200. To allocate the appropriate sample size to each province, three provinces were randomly selected from each part of the North-East, North-West, Central, South-East, and South-West of Iran (a Total of 15 provinces), and the total sample size was divided based on proportion to the size of diagnosed patients. Next, a list of VCTs was prepared separately for each of the 15 provinces; then, two VCTs in each of these provinces were randomly selected for data collection. Finally, in each of the VCTs, convenience sampling was used to choose participants.
Collecting data
For data collection, trained interviewers with experience working on PWLH using standard questionnaires collected data from adult patients aged ≥ 18 diagnosed at least 3 months ago. Verbal informed consent was also obtained from patients all over Iran between April 2021 and March 2022.
Data collection consisted of two parts: a physical examination and a questionnaire. The physical examination consisted of height and weight measurements. A demographic questionnaire and the Berlin Obstructive Sleep Apnea Questionnaire (BQ) were used for data collection. This scale has ten items assessing obstructive sleep apnea risk factors, including snoring, excessive daytime sleepiness and fatigue, obesity, and high blood pressure. A positive score on more than one subscale of the three shows a high risk of developing sleep apnea17. The validity and reliability of this tool have been confirmed with a Cronbach’s alpha coefficient of 0.918 for the original version and 0.90 for the Persian version19.
Self-reported NCD diseases were considered by asking questions about diagnosed diseases by physicians or using related medications in the included questionnaire. Each NCD, heart disease, hypertension, diabetes, was defined as patients who had diseases diagnosed by a physician or were on medications. Medical information and disease-related data—including duration of HIV diagnosis, duration of ART, ways of HIV transmission, hepatitis C virus, hepatitis B virus, and tuberculosis (TB) comorbidity, the last CD4 count, and the last viral load—extracted from the patient's electronic health record available in VCTs.
Ethical consideration
Ethical approval was received for this study from the Tehran University of Medical Sciences (TUMS) that all research was performed in accordance with relevant guidelines and regulations (ethics approval number: IR.TUMS.FNM.REC.1399.066), and all processes were performed in accordance with the Declaration of Helsinki.
Every participant provided verbal informed consent, according to patients with HIV/AIDS research approved by the Tehran University of Medical Sciences Ethics Committee under the code IR.TUMS.FNM.REC.1399.066,
Statistical analysis
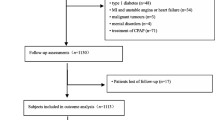
Although data were gathered for 1200 PLWH cases, 27 patients had missing data on at least one of the NCDs that were excluded from the study, and data analysis was performed on 1173 patients with complete NCD data. Other socio-demographic and clinical characteristics with missing values (3.01% of the whole dataset) were imputed by performing a single imputation and regression model.
All analyses were carried out using complex survey methods to regulate cluster sampling and sample weight. Based on the age and sex categories of the 15-year-old national Iranian PLWH population in 2021, the data were weighted. The weighted mean with standard error (SE) and weighted percent were used to first report the characteristics of the individuals. In the complex survey approach, a t-test analysis (p < 0.05) was used to examine differences in age, body mass index (BMI), duration of HIV diagnosis, CD4 count, viral load, and ART duration between the under-50 and over-50-year-old groups and a chi-square test was used for other qualitative variables. The normality of continuous data was checked by using a histogram.
Logistic regression was used to investigate the relationship between baseline variables and having at least one NCD comorbidity with HIV in total patients. For exploring associated factors, a univariable analysis was performed, and variables with a p-value < 0.1 were entered in the multiple regression model as the final model. All analyses were performed using STATA (17 version) for complex survey analysis, and p-values < 0.05 were considered statistically significant.
Results
The present research enrolled 1173 PLWH in the analysis, with a mean age of 35.35 years (SE: 0.06). Of these, 974 (83.03%) and 199 (19.97%) cases were aged under 50 and over 50, respectively. The mean age of participants under 50 and over 50 was 33.90 (SE:0.07) and 56.82 (SE: 0.22) years, respectively. The CD4 count and median of the duration of HIV diagnosis (month) were higher in people under 50, and in terms of NCD prevalence, there is a significant difference between the two age groups (p < 0.001) (Table 1).
As shown in Table 2, the prevalence of HCV, HBV, and TB comorbidity in PLWH under 50 was 13.86%, 2.33%, and 5.38%, and in PLWH over 50, it was 19.69%, 3.38%, and 7.13%, respectively.
About 225 (19.18%) participants experienced at least one NCD (15.20% and 38.69% among under- and over-50-year-old patients, respectively). The prevalence of heart disease, hypertension, diabetes, and sleep apnea among all patients was 1.59%, 2.05%, 1.55%, and 10.26%, respectively. The similar prevalence for each NCD among those over 50 years was 10.11%, 15.71%, 9.01%, 25.44%, and 1.01%, 1.12%, 1.04%, and 9.23% among those under 50 years, respectively.
The prevalence of one, two, or three and more non-communicable comorbidities in patients under 50 was 10.63%, 1.32%, and 0.21%, respectively, and patients over 50, it was 22.50%, 11.49%, and 4.97%, respectively.
As Fig. 1 shows, the prevalence of comorbidities increases with age. The peak of one comorbidity occurred in the age group 50–60, and two and three comorbidities occurred in the age group over 60.
Multiple logistic regression was used to address factors associated with having at least one NCD comorbidity. The results of multivariate logistic regression showed that several factors were positively associated with having NCD in PLWH. Age over 50 years (ORadj = 2.93, 95% CI 1.96–4.37), married participants (ORadj = 2.48, 95% CI 1.41–4.35), divorced or widowed (ORadj = 2.78, 95% CI 1.48–5.20), and BMI ≥ 30 (ORadj = 3.82, 95% CI 2.46–5.91) were associated with having at least one NCD comorbidity in PLWH. Nevertheless, patients with an education level diploma had lower odds of at least one non-communicable comorbidity than those under diploma counterparts (ORadj = 0.64, 95% CI 0.41–0.97) (Table 3).
Discussion
Our findings demonstrated that the chance of having at least one noncommunicable disease was higher in patients over 50 years old. Age and obesity were significant risk factors for NCDs in those over 50, while HIV transmission through sexual contact, obesity, level of education, and age were significant risk factors in patients under 50. This study was consistent with other studies20. For those who have HIV infection, the comorbidity of NCDs is becoming a greater burden. However, information on the extent of HIV-NCD comorbidity and the factors that contribute to it in Iran is scarce. With the improvement in HIV-infected individuals’ survival, the probability of other diseases is increasing, like in the general population21,22. According to reports from the World Health Organization, ART may affect and progress NCDs10. By increasing NCD risk factors in low-resource settings, the prevalence of NCDs increases and shifts from the affluent to the less affluent population23. One of the main causes of NCDs among PLWH is aging, which is called accelerated aging and leads to NCDs affecting PLWH decades earlier than people without HIV24. The objective of this study was to determine the effect of aging and non-communicable comorbidities in PLWH.
Obesity is known as a risk factor for NCDs and diabetes and is a behavioral risk factor and consequence of lifestyle25. The increase in overweight and obesity in low-resource-setting countries is greater than in high-resource-setting countries26. Also, obesity in men and women is associated with higher socioeconomic status, whereas in high-income countries it is related to poverty27. Studies show that as individual’s age, their risk of NCDs considerably increases. In the current study, age and obesity were found to be predictors of HIV-NCD comorbidity. Numerous studies have shown that the incidence of NCDs increases with age. Investigations, for instance, showed that impairments in glucose tolerance were more prevalent in people who were older28.
Some investigators agree with the finding that PLWH on ART have a lower BMI and a lower prevalence of obesity and overweight than their counterparts29. It could be attributed to the later initiation of ART30. Weight loss and wasting are common symptoms of HIV-related illness. However, it is well-established that antiretroviral therapy (ART) can reverse HIV-related weight loss and wasting31. The results of this comprehensive study on NCD comorbidities in PLWH showed that the prevalence of obesity in patients over 50 years was 19.60%.
PLWH on ART are at higher risk of NCDs at older ages in the present study. Older age increases the odds of NCD comorbidity by 5% and 8% in patients under 50 and over 50, respectively. The patients younger than 30 years faced one NCD. As patients get older, they encounter more than one NCD. The peak of multi-morbidity in patients occurred at ages 60 and older. Although communicable and lung disease comorbidities were not significant, the prevalence of all non-communicable comorbidities was higher in patients over 50 years. The results of the Oni et al. study show a higher prevalence of multi-morbidity in younger ages (18–35 years) for women and, by contrast, in older ages (46–55 years) for men, whereas after adjusting for age, comorbidity in younger ages was more prevalent32. The suggested reason for this was previously mentioned as being premature aging33. In contrast, the study conducted by Zhang et al. found that the elderly population in China has a high prevalence of multi-morbidity34. Antiretroviral therapy (ART) has decreased virological failure, but the results of a cohort study on the initiation of ART show those patients who start ART at 50 years of age or older are at higher risk for death than those who start ART at younger ages35.
In our study, there was no significant association between men and women in disease comorbidities with HIV. In a study in South Africa, women were at higher risk for obesity than men36. Oni et al. confirm the significant difference between men and women in multi-morbidity patterns32. In a systematic review, results confirm the prior finding about higher odds of comorbidities in females versus37. This finding was in line with the 2018 Academy of Medical Science report about multi-morbidity38. A higher rate of comorbidity in females could be explained by more self-reporting39, higher life expectancy40 and adherence to healthcare providers’ advice, and frequency of visits41.
A higher prevalence of NCDs in PLWH than in the past could be due to a combination of several factors, like aging, more affection for PLWH by traditional NCD risk factors, and the effects of human viruses and ART side effects42. ART effects on lifespans led PLWH to be exposed to NCD risk factors as much as normal people7,16. The rapid change in demographic characteristics of this population and the epidemiological transition enhanced by unhealthy behaviors such as an unhealthy diet and lower physical activity could increase the chance of NCDs43. Hypertension is one of the main cardiovascular disease (CVD) risk factors44. Our results confirm the previous finding that hypertension is one of the most common comorbidities among PLWH. In this study, 16.08%, 9.05%, and 6.53% of PLWH aged 50 and older were impressed by hypertension, diabetes, and TB, respectively. A study that examined the comorbidity pattern of HIV, TB, and NCDs reported that the prevalence of hypertension in individuals 50 years of age and older was 77%, and it was the most common comorbidity in HIV patients on ATR14. Also, Kaluvu et al., in a systematic review, cited hypertension as the most frequent (23.3%) comorbidity with HIV. Diabetes, TB, and HIV (26.6%) and hypertension, diabetes, and HIV were the most frequent triples (63%)37. The results of a meta-analysis conducted in low- and high-income countries showed that the prevalence of HIV comorbidity due to hypertension and obesity was 21.2% and 7.8%, respectively16.
In the present study, the prevalence of HCV and HBV in PLWH older than 50 was higher than in their counterparts younger than 50, but the difference was not statistically significant. In a study in India, the prevalence of HBV and HCV among HIV-positive participants who injected drugs was 9.7% and 53.7%, respectively45. The prevalence of HCV and HIV comorbidity in homeless men was 5.76% in a study in Iran46. The comorbidity of HIV with viral hepatitis because of some complications such as liver problems, renal disorders, immunological and hematological problems, and CVD must be considered47.
To the best of our knowledge, this study is the first such effort to investigate the current health service landscape for populations in Iran that are co-morbid with HIV and NCDs.
The finding of the present study demonstrates that obstructive sleep apnea affects 25.63% of PLWH aged 50 and older, which could be due to the higher prevalence of obesity in older ages. In a study by Njoh et al., the prevalence of HIV infection in patients with sleep apnea was higher than in those without sleep apnea syndrome. This study reveals a strong association between HIV and obstructive sleep apnea48, which is the opposite of our study’s finding. It may be due to different study designs (case–control study vs. cross-sectional study).
Although there is little information on integrated HIV care and NCD services, shreds of evidence show it could be beneficial for PLWH49. So that most of the risk factors are behavioral, and the majority of them potentially require intervention50. The NCDs as emerging problems among PLWH must be confronted and leveraged through HIV health care investments to enhance well-being and reduce mortality among PLWH51. Thereby, early detection of risk factors of NCDs as preventive behaviors would reduce the costs imposed, morbidity, and mortality.
This study was conducted on the national data of PLWH. These data were used to determine the effect of aging on the comorbidity of HIV with NCDs for the first time in the country, which is the most important strength of the study. Also, we examined obstructive sleep apnea in addition to NCDs.
Despite the promising results of this study, there are some limitations that should be acknowledged. Due to the coincidence of the COVID-19 era, we had to use convenience sampling based on the regular visits of HIV patients to VCTs to get their own antiretroviral regimens. We try to use weighted analysis to manage this issue. On the other hand, the measurement of non-communicable diseases here was stated through self-report and was not measured accurately for each patient, which led to an underestimation of the number of samples.
Conclusion
In general, the study found that patients over 50 had a considerable amount of comorbidity. Being older and having a BMI over 25 increased the chance of developing NCDs in people living with HIV. Targeted NCD prevalence screening and caring, addressing modifiable risk factors, and providing integrated care are all advised in order to improve the quality of life for comorbid patients.
It is essential to make investments in improved NCD management. Making sure individuals who require it have access to palliative care is a part of the detection, screening, and treatment of NCDs. We focused on the comorbidity of NCDs in PLWH because it is a growing problem in the context of HIV-positive patients. Policymakers should take into account these issues, adopt coordinated management between HIV and chronic disease care, and promote NCD screening programs for PLWH due to the impact of aging on the growing NCDs and the rising population of PLWH aged under and beyond 50.
This national study, which was conducted in Iran, revealed that there is typically no screening policy for non-communicable diseases in PLWH. However, we also emphasized the significance of NCD prevalence in patients under the age of 50 compared to the general population; this group should not be ignored. In order to get medical professionals and health policymakers to pay more attention to the spread of non-communicable diseases in PLWH, we recommended that HIV patients be checked for comorbidity with non-communicable diseases.
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to[REASON WHY DATA ARE NOT PUBLIC] but are available from the corresponding author on reasonable request.
References
Organization WH. in HIV, Estimated number of people (all ages) living with HIV 2023 [Available from: https://www.who.int/data/gho/data/indicators/indicator-details/GHO/estimated-number-of-people--living-with-hiv.
Smit, M. et al. Projections of non-communicable disease and health care costs among HIV-positive persons in Italy and the U.S.A.: A modelling study. PLOS ONE 12(10), e0186638. https://doi.org/10.1371/journal.pone.0186638 (2017).
UNAIDS. in HIV/AIDS 2021 [Available from: https://www.unaids.org/en//resources/fact-sheet.
Coetzee, L. et al. HIV, antiretroviral therapy and non-communicable diseases in sub-Saharan Africa: empirical evidence from 44 countries over the period 2000 to 2016. J. Int. AIDS Soc. 22(7), e25364 (2019).
Harris, T. G. et al. HIV and aging among adults aged 50 years and older on antiretroviral therapy in Eswatini. Afr. J. AIDS Res. 20(1), 107–115 (2021).
Achwoka, D. et al. High prevalence of non-communicable diseases among key populations enrolled at a large HIV prevention & treatment program in Kenya. PLoS ONE 15(7), e0235606 (2020).
Feinstein, M. J. et al. Characteristics, prevention, and management of cardiovascular disease in people living with HIV: A scientific statement from the American Heart Association. Circulation 140(2), e98–e124 (2019).
Deeks, S. G., Lewin, S. R. & Havlir, D. V. The end of AIDS: HIV infection as a chronic disease. Lancet 382(9903), 1525–1533 (2013).
Seang, K. et al. Differences in prevalence and risk factors of non-communicable diseases between young people living with HIV (YLWH) and young general population in Cambodia. PLoS ONE 17(6), e0269989 (2022).
Kansiime, S., Mwesigire, D. & Mugerwa, H. Prevalence of non-communicable diseases among HIV positive patients on antiretroviral therapy at joint clinical research centre, Lubowa, Uganda. PLoS ONE 14(8), e0221022 (2019).
Thet, D. & Siritientong, T. Antiretroviral therapy-associated metabolic complications: Review of the recent studies. HIV/AIDS – Res. Palliative Care 12, 507–524. https://doi.org/10.2147/HIV.S275314 (2020).
Gausi, B., Otiku, P., Jacob, N. & Oni, T. Patient outcomes in integrated HIV and non-communicable disease models of care: A scoping review. J. Global Health Rep. 5, e2021084 (2021).
Brown, M. J. & Adeagbo, O. HIV and aging: Double stigma. Curr. Epidemiol. Rep. 8, 72–78 (2021).
Peltzer, K. & Phaswana-Mafuya, N. Hypertension and associated factors in older adults in South Africa: Cardiovascular topics. Cardiovasc. J. Afr. 24(3), 66–71 (2013).
Kaluvu, L. et al. Multimorbidity of communicable and non-communicable diseases in low-and middle-income countries: A systematic review. J. Multimorb. Comorb. 12, 26335565221112590 (2022).
Nguyen, K. A., Peer, N., Mills, E. J. & Kengne, A. P. A Meta-analysis of the metabolic syndrome prevalence in the global HIV-infected population. PLoS ONE 11(3), e0150970 (2016).
Tan, A. et al. Using the Berlin questionnaire to predict obstructive sleep apnea in the general population. J. Clin. Sleep Med. 13(3), 427–432 (2017).
Sharma, S. K. et al. Validation of the modified Berlin questionnaire to identify patients at risk for the obstructive sleep apnoea syndrome. Indian J. Med. Res. 124(3), 281–290 (2006).
Ghanei Gheshlagh, R., Hemmati Moslek, M. & Baghi, V. A study on the relation between body mass index and sleep apnea in patients suffering diabetes type2. J. Diabetes Nursing. 1(2), 12–20 (2014).
Getahun, Z., Azage, M., Abuhay, T. & Abebe, F. Comorbidity of HIV, hypertension, and diabetes and associated factors among people receiving antiretroviral therapy in Bahir Dar city Ethiopia. J. Comorbid. 10, 2235042X19899319 (2020).
Jaggers, J. R. et al. Associations between physical activity and sedentary time on components of metabolic syndrome among adults with HIV. AIDS Care 26(11), 1387–1392 (2014).
Lerner, A. M., Eisinger, R. W. & Fauci, A. S. Comorbidities in persons with HIV: The lingering challenge. JAMA 323(1), 19–20 (2020).
Gowshall, M. & Taylor-Robinson, S. The increasing prevalence of non-communicable diseases in low-middle income countries: The view from Malawi. Int. J. General Med. 11, 255–264. https://doi.org/10.2147/IJGM.S157987 (2018).
Gonah, L., Moodley, I. & Hlongwana, K. Effects of HIV and non-communicable disease comorbidity on healthcare costs and health experiences in people living with HIV in Zimbabwe. S. Afr. J. HIV Med. 21(1), 1–8 (2020).
Hanson, S. K. et al. Stunting at 24 months is not related to incidence of overweight through young adulthood in an urban South African Birth cohort. J Nutr. 148(6), 967–973 (2018).
Hopkins, K. L. et al. The substantial burden of non-communicable diseases and HIV-comorbidity amongst adults: Screening results from an integrated HIV testing services clinic for adults in Soweto South Africa. EClinicalMedicine. 38, 101015 (2021).
Hampl, S. E. et al. Clinical practice guideline for the evaluation and treatment of children and adolescents with obesity. Pediatrics 151(2), e2022060640 (2023).
Rasmussen, L. D. et al. Risk of diabetes mellitus in persons with and without HIV: A Danish nationwide population-based cohort study. PLoS ONE 7(9), e44575. https://doi.org/10.1371/journal.pone.0044575 (2012).
Asiimwe, S. B. et al. HIV status and antiretroviral therapy as predictors of disability among older South Africans: Overall association and moderation by body mass index. J. Aging Health 32(10), 1335–1344 (2020).
Houlihan, C. F. et al. Cohort profile: Hlabisa HIV treatment and care programme. Int. J. Epidemiol. 40(2), 318–326 (2011).
Bailin, S. S., Gabriel, C. L., Wanjalla, C. N. & Koethe, J. R. Obesity and weight gain in persons with HIV. Curr. HIV/AIDS Rep. 17, 138–150 (2020).
Oni, T. et al. Patterns of HIV, TB, and non-communicable disease multi-morbidity in peri-urban South Africa- a cross sectional study. BMC Infect Dis. 15, 20 (2015).
McGettrick, P., Barco, E. A. & Mallon, P. W. G. Ageing with HIV. Healthcare 6(1), 17. https://doi.org/10.3390/healthcare6010017 (2018).
Zhang, R., Lu, Y., Shi, L., Zhang, S. & Chang, F. Prevalence and patterns of multimorbidity among the elderly in China: A cross-sectional study using national survey data. BMJ Open 9(8), e024268 (2019).
Carriquiry, G. et al. Virologic failure and mortality in older ART initiators in a multisite Latin American and caribbean cohort. J. Int. AIDS Soc. 21(3), e25088 (2018).
Manafe, M., Chelule, P. K. & Madiba, S. The perception of overweight and obesity among South African adults: Implications for Intervention strategies. Int. J. Environ. Res. Public Health 19(19), 12335 (2022).
Kaluvu, L. et al. Multimorbidity of communicable and non-communicable diseases in low- and middle-income countries: A systematic review. J. Multimorb. Comorb. 12, 26335565221112590 (2022).
AOMS. Multimorbidity: A Priority for Global Health Research (Academy of Medical Sciences, 2018).
Afshar, S., Roderick, P. J., Kowal, P., Dimitrov, B. D. & Hill, A. G. Multimorbidity and the inequalities of global ageing: A cross-sectional study of 28 countries using the World Health Surveys. BMC Public Health 15(1), 1–10 (2015).
Narayan, K. V. et al. HIV and noncommunicable disease comorbidities in the era of antiretroviral therapy: A vital agenda for research in low-and middle-income country settings. JAIDS J. Acquir. Immune Defic. Syndr. 67, S2–S7 (2014).
Kalgotra, P., Sharda, R. & Croff, J. M. Examining health disparities by gender: A multimorbidity network analysis of electronic medical record. Int. J. Med. Informat. 108, 22–28 (2017).
van Heerden, A. et al. High prevalence of HIV and non-communicable disease (NCD) risk factors in rural KwaZulu-Natal, South Africa. J. Int. AIDS Soc. 20(2), e25012 (2017).
Chang, D. et al. Non-communicable diseases by age strata in people living with and without HIV in four African countries. J. Int. AIDS Soc. 25, e25985 (2022).
Kjeldsen, S. E. Hypertension and cardiovascular risk: General aspects. Pharmacol. Res. 129, 95–99 (2018).
Ray Saraswati, L. et al. HIV, Hepatitis B and C among people who inject drugs: High prevalence of HIV and hepatitis C RNA positive infections observed in Delhi India. BMC Public Health. 15(1), 1–11 (2015).
Doosti-Irani, A. et al. Prevalence of HIV, HBV, and HCV and related risk factors amongst male homeless people in Lorestan province, the West of Iran. J Res Health Sci. 17(1), e00373 (2017).
(CDC) CfDCaP. in People Coinfected with HIV and Viral Hepatitis 2023 [updated September 21,2020]. Available from: https://www.cdc.gov/hepatitis/populations/hiv.htm.
Njoh, A. A. et al. Likelihood of obstructive sleep apnea in people living with HIV in Cameroon–preliminary findings. Sleep Sci. Pract. 1(1), 1–9 (2017).
Adeyemi, O. et al. Integration of non-communicable disease and HIV/AIDS management: A review of healthcare policies and plans in East Africa. BMJ Glob. Health 6(5), e004669 (2021).
Magnusson, R. Developing a global framework to address non-communicable diseases. Diabetes Voice 53, 9–12 (2008).
Patel, P. et al. Noncommunicable diseases among HIV-infected persons in low-income and middle-income countries: A systematic review and meta-analysis. Aids 32(Suppl 1), S5–S20 (2018).
Acknowledgements
For helping to advance this national study, we would like to thank the Iranian Ministry of Health as well as the staff members who work the voluntary counselling and testing centres for HIV in various Iranian provinces.
Author information
Authors and Affiliations
Contributions
1-Conceived and designed the experiments; S.A., M.S. 2-Performed the experiments; F.H., M.S., S.M.N., S.N., S.A. 3-Analyzed and interpreted the data; M.S., S.M.N., S.A. 4-Contributed reagents, materials, analysis tools, or data; F.H., M.S., S.M.N., M.E.M., A.N., M.M., S.N., S.A. 5-Wrote the paper; F.H., M.S., S.M.N., M.E.M., A.N., M.M., S.N., S.A. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hadavandsiri, F., Shafaati, M., Mohammad Nejad, S. et al. Non-communicable disease comorbidities in HIV patients: diabetes, hypertension, heart disease, and obstructive sleep apnea as a neglected issue. Sci Rep 13, 12730 (2023). https://doi.org/10.1038/s41598-023-39828-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-39828-6
- Springer Nature Limited
This article is cited by
-
Association between psychological discomforts and sleep quality among people living with HIV/AIDS
AIDS Research and Therapy (2023)