Abstract
Guidelines from the World Health Organization strongly recommend the use of a high fraction of inspired oxygen (FiO2) in adult patients undergoing general anesthesia to reduce surgical site infection (SSI). However, previous meta-analyses reported inconsistent results. We aimed to address this controversy by focusing specifically on abdominal surgery with relatively high risk of SSI. Medline, EMBASE, and Cochrane CENTRAL databases were searched. Randomized trials of abdominal surgery comparing high to low perioperative FiO2 were included, given that the incidence of SSI was reported as an outcome. Meta-analyses of risk ratios (RR) were performed using a fixed effects model. Subgroup analysis and meta-regression were employed to explore sources of heterogeneity. We included 27 trials involving 15977 patients. The use of high FiO2 significantly reduced the incidence of SSI (n = 27, risk ratio (RR): 0.87; 95% confidence interval (CI): 0.79, 0.95; I2 = 49%, Z = 3.05). Trial sequential analysis (TSA) revealed that z-curve crossed the trial sequential boundary and data are sufficient. This finding held true for the subgroup of emergency operations (n = 2, RR: 0.54; 95% CI: 0.35, 0.84; I2 = 0%, Z = 2.75), procedures using air as carrier gas (n = 9, RR: 0.79; 95% CI: 0.69, 0.91; I2 = 60%, Z = 3.26), and when a high level of FiO2 was maintained for a postoperative 6 h or more (n = 9, RR: 0.68; 95% CI: 0.56, 0.83; I2 = 46%, Z = 3.83). Meta-regression revealed no significant interaction between SSI with any covariates including age, sex, body-mass index, diabetes mellitus, duration of surgery, and smoking. Quality of evidence was assessed to be moderate to very low. Our pooled analysis revealed that the application of high FiO2 reduced the incidence of SSI after abdominal operations. Although TSA demonstrated sufficient data and cumulative analysis crossed the TSA boundary, our results should be interpreted cautiously given the low quality of evidence.
Registration: https://www.crd.york.ac.uk/prospero (CRD42022369212) on October 2022.
Similar content being viewed by others
Introduction
Millions of patients undergo surgery under general anesthesia each year1. Anesthesiologists optimize ventilator setting to reduce postoperative morbidity and mortality and the fraction of inspired oxygen (FiO2) is one of the key settings. Surgical site infection (SSI) is a common and serious complication after abdominal surgery2. World Health Organization (WHO) implemented guidelines recommending the use of high fraction of inspired oxygen (FiO2) during the perioperative period to reduce the risk of surgical site infection (SSI)3. Poor quality of evidence and failure to address potential harms4 led to an updated analysis5, limiting the intervention to adult patients undergoing general anesthesia. Currently, the US Centers for Disease Control and Prevention (CDC) and other healthcare organizations have adopted the revised WHO standards6,7. The consensus is that administering high FiO2 and thereby increasing tissue oxygen tension8 could lower SSI by mechanisms such as facilitating neutrophil bacterial killing9. Additional benefits such as the reduction of postoperative nausea and vomiting have been debated10,11.
Nonetheless, concerns still prevail. The current recommendations yet undermine the known adverse effects of high FiO2—among the notable drawbacks lies absorptive atelectasis12, which potentially results in decreased lung compliance and impaired oxygenation, as well as pneumonia13. It is further hypothesized that FiO2 above physiological range imposes systematic oxidative stress; this may lead to respiratory and cardiovascular complications13,14, neurological manifestations15, and death. Acutely ill patients fare better with conservative oxygen therapy16,17, accordingly, high FiO2 is not recommended in emergency operations or for critical care patients18,19.
There have been attempts to tackle this debate. While older studies focused on the prevention of either SSI or postoperative nausea and vomiting20, recent meta-analyses reported diverse clinical effects such as length of hospital stay and mortality21,22,23. Despite the increase in scope, previous reviews have neglected to analyze the substantial differences arising from surgical or patient characteristics. For instance, limiting included trials to those operated under general anesthesia prohibited the exclusion of the effect of general anesthetics on wound infection. General anesthesia corresponded to a higher risk of SSI24. Furthermore, open surgical approach and emergency operations25, male sex, and length of the procedure26 were documented as independent risk factors of SSI.
We believe that surgery type is also a critical factor associated with SSI incidence and that different types should be investigated separately. Previous trials of abdominal surgery indicated a high incidence of 15–25%27,28. SSI remains an unrelenting source of morbidity for colorectal operations with an incidence rate of 9.34%29, or pancreatoduodenectomy with an incidence of 6–17%30. A recent trial of emergency abdominal surgery showed that perioperative administration of 80% FiO2 significantly decreased SSI incidence31. In addition, previous randomized trials reported the significant positive effect of high FiO2 on SSI in abdominal surgeries32,33. However, a meta-analysis focusing solely on abdominal surgery has rarely been conducted to the best of our knowledge. ‘
Therefore, we carried out a meta-analysis on the effect of high FiO2 on SSI and other clinical outcomes focusing only on abdominal surgeries; we aimed to discover causes of heterogeneity unexposed by previous reviews by performing subgroup analysis or meta-regression for the important risk factors previously reported.
Methods
Following the registration on PROSPERO (https://www.crd.york.ac.uk/prospero, registration number: CRD42022369212) on October 2022, the present study was conducted according to the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions. The reporting of this review follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement guidelines.
A search strategy was developed and applied to Medline, Embase, and Cochrane CENTRAL, and is provided in the protocol (Supplemental Text S1). The inclusion and exclusion criteria of our review was reported in Supplemental Text S2. The last search was executed on October 21st, 2022. Only full-text articles in English were considered as candidates.
Two authors (KJH and WHK) independently screened titles and abstracts for relevant trials. The full text of manuscripts that passed the first level of screening was scrutinized to determine eligibility. Studies were qualified for inclusion if they: (1) mentioned surgical site infection (SSI) incidence following abdominal surgery; (2) compared high FiO2 of at least 80% with standard levels of at most 40%; and (3) were randomized controlled trials. The references of previous meta-analyses and related articles were manually inspected to incorporate any studies omitted in the original search. Any disagreements were resolved via discussion.
We piloted a standardized extraction form which one author (JHK) filled out and another (WHK) confirmed. Data was reexamined several times to amend any remaining errors. The following data were extracted: study design, inclusion and exclusion criteria, surgery type, the urgency of surgery, open or laparoscopic methods, definition of SSI, size of groups, demographics (age; sex; body-mass index (BMI); smoking; American Society of Anesthesiologists physical status classification; history of hypertension, diabetes mellitus (DM), coronary artery disease, myocardial injury, chronic pulmonary diseases, cerebrovascular accidents; preoperative hemoglobin; and blood glucose levels), intraoperative and perioperative parameters (percentage of acute operations and transfusions; duration of surgery; and estimated blood loss), and outcomes.
The incidence of SSI was evaluated as the primary outcome, which was defined by either the CDC guidelines6 or the ASEPSIS scoring system34. Secondary outcomes were as follows: short-term mortality, myocardial injury, atelectasis, organ-space SSI, anastomotic leakage, pneumonia, and reoperation.
Two authors (JHK and WHK) independently evaluated the risk of bias of each selected randomized controlled trials (RCT) utilizing the Cochrane tool (RoB assessment tool version 2.0)35. Six domains were addressed: bias arising from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in the measurement of outcomes, and bias in the selection of reported outcomes. Risks were ranked as low, intermediate, or high. Overall risks of bias for each manuscript were reported as the highest risk among the five categories.
The quality of evidence for all study outcomes was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) methodology, which comprises five domains: risk of bias, inconsistency, indirectness, imprecision, and publication bias. Each outcome was rated as having very low, low, moderate, or high certainty of evidence.
Data synthesis and analysis
Review Manager 5.4 software (RevMan, The Cochrane Collaboration, Oxford, United Kingdom) and STATA/SE version 14.0 (StataCorp, College Station, Texas, USA) were utilized for data synthesis and analysis. Binary data were analyzed via the Mantel–Haenszel method with a fixed-effects approach to calculate pooled risk ratios (RR) and 95% confidence intervals (CI), while the Inverse Variance method was applied to measure mean difference (MD) and 95% CI from continuous variables. Fixed effect model assumes one true effect size underlies all the studies in the meta-analysis. However, we also performed random effects approach to allow for the expected heterogeneity across the studies. The results of meta-analysis were depicted by forest plots for the primary outcome and hospital length of stay.
Subgroup analyses were conducted according to demographic data and surgical characteristics based on a priori analysis plan. Moderators were considered for analysis if they were known risk factors of SSI or if their clinical significance was acknowledged by all authors. Only those reported in a sufficient number of trials (at least 18) were ultimately selected, including mean BMI, percentage of patients with DM, urgency of surgery, type of surgery, carrier gas, durationof postoperative oxygenation supplementation, and percentage of current smokers. Continuous data such as mean BMI and duration of oxygen supplementation were converted into dichotomous categories.
Meta-regression was performed to assess the relationship between continuous covariates and our primary outcome. We applied criteria identical as mentioned above, resulting in age, percentage of males, mean BMI, percentage of patients with DM, duration of surgery, and percentage of smokers. Results are reported in bubble plots and corresponding p values. Publication bias was primarily assessed with funnel plots. Egger’s linear regression test was additionally performed for outcomes reported in 10 or more trials.
We performed trial sequential analysis (TSA) with TSA Viewer (Version 0.9.5.10 Beta, Copenhagen Trial Unit, 2016, Copenhagen, Denmark) for SSI, length of hospital stay, short-term mortality, and myocardial injury. It conducts a cumulative meta-analysis, manifested as a Z curve of the pooled observed effect, that reduces the risk of false-positive results from repetitive testing. We used a power of 80% and a 5% alpha error to calculate the required information size, which signifies the threshold for which the effect of the intervention may be confirmed or rejected. A conventional boundary denoting statistical significance (p < 0.05) and the trial sequential boundary (O’Brien-Fleming significance boundary) were also created. Overall, the course of the z-curve in relation to these borders helped us estimate when the effect will be large enough for future studies to be unnecessary. We used a 20% relative risk reduction (RRR) for binary outcomes, except for short-term mortality for which we used 10% RRR considering its clinical importance. For the continuous outcome (length of hospital stay), a mean difference of 0.5 was used.
A sensitivity analysis applying a random-effects approach was carried out to dismiss concerns regarding disparities among operation types and study designs, which may have breached assumptions necessary to undertake a fixed-effects approach. Another sensitivity analysis was done to compare trials with different definitions for SSI, namely per CDC guidelines, per ASEPSIS scoring system, and others. Heterogeneity was represented as the I2 statistic. We examined potential sources of heterogeneity via subgroup analyses and meta-regression.
Results
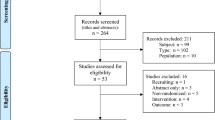
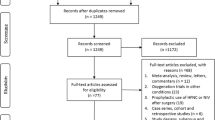
We identified a total of 58,576 manuscripts from the initial search of which 13,279 duplicates were removed. After excluding 44,300 irrelevant studies, full texts of the remaining 997 studies were further inspected for eligibility. A total of 970 articles were removed for the reasons shown in Fig. 1. After evaluating trials additionally identified in references (n = 0), a sum of 27 original records comprising 15,977 patients was included in the final analysis.
The trials represented 25 parallel RCTs31,32,33,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57, one cross-over RCT58, and one case–control study59. All but two39,58 were double-blinded trials. A total of 19 trials were conducted at a single center31,36,37,38,39,41,45,46,47,48,50,51,52,53,54,56,57,58,59, while 8 collected data from multiple centers32,33,40,42,43,44,49,55. Most of the trials compared a perioperative FiO2 of 80% to 30%, except three; one trial used 40% FiO246, another used 35%50, and the other used 33% FiO247 for the control arm (Table 1). Sample size of the trials ranged between 38 to 5749 (median 239), with 9 trials exceeding 500 patients (Table 1). Additional demographic and surgical characteristics are provided in Table 1 and Supplemental Table S1. The distribution of study outcomes across all included studies is shown in Supplemental Table S2.
Most studies showed an overall intermediate risk of bias, except for two determined to be at low risk41,49, and two at high risk39,56 (Supplemental Figure S1). GRADE approach showed that the quality of evidence of our study outcomes is from moderate to very low (Supplemental Table S3).
As the inclusion criteria mandated the reporting of this outcome, all included 27 trials addressed the incidence of surgical site infection. Of these, 11 trials followed up with the patients for two weeks31,33,36,38,39,41,42,49,50,53,54 and another 11 trials for a month32,37,40,43,44,45,47,48,51,58,59. The remaining studies had various follow-up periods or did not specify the length of follow-up. Overall, the use of high FiO2 significantly reduced SSI (RR: 0.87; 95% CI: 0.79, 0.95; p = 0.002, I2 = 49%) (Fig. 2). The corresponding funnel plot does not suggest publication bias upon visual inspection (Supplemental Figure S2). The Egger test reinforces this assertion as no small-study effect was found (p = 0.773).
Twelve trials reported on the length of hospital stay for a total of 3703 patients33,36,37,39,40,41,42,44,45,46,50,56. There was no significant difference in the length of hospital stay between the two groups (MD: −0.06; 95% CI: −0.19, 0.08; p = 0.43, I2 = 66%) (Supplemental Figure S3). The funnel plot did not reveal any sign of publication bias, nor did the Egger test show a small-study effect (p = 0.645).
Data on mortality were available from thirteen trials involving 12,991 patients32,40,42,43,45,46,49,51,52,53,54,55,58. While short-term mortality of less than or equal to 60 days was reported in twelve trials32,40,42,45,49,51,52,53,54,55,58, two trials provided long-term mortality of six months40,46. The follow-up period for short-term mortality was 30 days for the overwhelming majority, save for two trials that tracked mortality for 60 days55 and 15 days42 each, and two trials that did not disclose the duration of follow-up52,56. No significant difference in short-term mortality was found between the two groups (RR: 1.04; 05% CI: 0.76, 1.43; p = 0.79, I2 = 34%) (Table 2). While the funnel plot was unremarkable, the Egger test warned of a small-study effect (p = 0.020).
Eight studies of a total of 4845 patients reported the incidence of myocardial injury32,37,40,43,45,46,51,53. Two trials43,51 employed a standardized definition based on elevated troponin values, while the remaining six provided data on solely myocardial infarction. No notable difference was shown between the groups (RR: 0.90, 95% CI: 0.71, 1.13; p = 0.36, I2 = 0%) (Table 2). The funnel plot did not indicate any publication bias.
Regarding the incidence of atelectasis, an analysis of seven studies31,32,40,45,46,49,59 consisting of 5254 patients did not indicate a significant difference between groups (RR: 1.11; 95% CI: 0.96, 1.28; p = 0.16) (Table 2). Heterogeneity was very severe (I2 = 88%). The funnel plot was well-balanced.
Regarding organ-space SSI, six trials31,37,44,47,49,58 including a sum of 2095 patients reported data on the subcategories of SSI, namely superficial, deep, and organ-space. Among these, one trial58 documented a combination of deep and organ-space SSI, and another only noted deep SSI31. Following our decision to assess the incidence of organ-space SSI as a secondary outcome, these two trials were not included in the final analysis. There was no remarkable difference between the two groups (RR: 0.85; 95% CI: 0.57, 1.27, p = 0.43, I2 = 0%) (Table 2). The funnel plot did not suggest publication bias.
Six studies of a sum of 7887 patients demonstrated a decrease in anastomotic leakage incidence in the high FiO2 arm (RR: 0.59; 95% CI: 0.43, 0.79; p = 0.0006, I2 = 83%) (Table 2)48,49,52,54,58,59 Six trials documented data on pneumonia for 4859 patients31,32,37,43,46,49. The pooled estimate did not point out a significant difference between groups (RR: 0.89; 95% CI: 0.69, 1.14; p = 0.35, I2 = 46%). The funnel plot was not indicative of publication bias.
Reoperation data were available from six trials for a sum of 2690 patients46,49,50,51,52,54. High versus low FiO2 did not result in a meaningful reduction in reoperation rates (RR: 1.01; 95% CI: 0.81, 1.27; p = 0.92, I2 = 32) (Table 2). No publication bias was discerned from observing the funnel plot.
We further performed subgroup analyses on 9 major demographic and clinical characteristics for the primary outcome (Table 3). High FiO2 resulted in a significant decrease in SSI incidence for trials where the mean BMI was less than 30 (RR: 0.86; 95% CI: 0.75, 0.98; p = 0.03; I2 = 57%), while insignificant in more obese populations. SSI was also reduced in the subgroup where patients with diabetes mellitus constituted less than 20% of the entire population (RR: 0.86; 95% CI: 0.78, 0.95; p = 0.004; I2 = 56%), while otherwise not. The beneficial effect of high FiO2 was apparent in acute surgeries (RR: 0.54; 95% CI: 0.35, 0.84; p = 0.006; I2 = 0%) whereas unclear in trials composed of elective surgeries exclusively. FiO2 levels did not have a significant effect on SSI for Caesarean sections (RR: 1.15; 95% CI: 0.79, 1.67; p = 0.47; I2 = 0%) while a higher level was favorable for colorectal surgeries (RR: 0.78; 95% CI: 0.63, 0.97; p = 0.02; I2 = 51%) and other abdominal procedures (RR: 0.87; 95% CI: 0.78, 0.97; p = 0.01; I2 = 57%). Trials that used air as carrier gas resulted in SSI decrease for higher FiO2 (RR: 0.79; 95% CI: 0.69, 0.91; p = 0.001; I2 = 60) but not trials that used nitrogen or nitrogen oxide. The intervention reduced SSI in trials that supplied oxygen for at least six hours postoperatively (RR: 0.68; 95% CI: 0.56, 0.83; p = 0.0001; I2 = 46%); no such effect was shown in trials that supplied oxygen for shorter durations. A subgroup analysis on the percentage of smokers among patients did not yield significant results for either arm.
Meta-regression revealed no significant interaction between SSI with any covariates, namely age (p = 0.73), sex (p = 0.06), BMI (p = 0.99), DM (p = 0.244), duration of surgery (p = 0.295), and smoking (p = 0.696) (Supplemental Figure S4).
TSA for SSI revealed that the z-curve surpassed both the conventional boundary and the O’Brien Fleming significance boundary while also exceeding the required sample size (Fig. 3). This indicates that further trials are unlikely to alter our conclusion that high FiO2 decreases SSI in abdominal surgeries. On the other hand, the z-curve for the length of hospital stay passed the required sample size within the area of futility (Supplemental Figure S5). The curves for short-term mortality (Supplemental Figure S6), myocardial injury (Supplemental Figure S7), and atelectasis (Supplemental Figure S8) remain within the conventional boundary and have yet to reach their respective required sample sizes, pointing out a need for additional data.
A sensitivity analysis applying the random-effects model did not change the effect direction or statistical significance for all outcomes (Supplemental Table S4). A second sensitivity analysis for SSI incidence was performed on the heterogeneous definitions of SSI. Differences between the two groups remained significant for trials defining SSI per ASEPSIS score (RR: 0.43; 95% CI: 0.26, 0.70; p = 0.0007; I2 = 0%) and CDC guidelines (RR: 0.87; 95% CI: 0.79, 0.96; p = 0.006; I2 = 34%) alike. Thirdly, a sensitivity analysis of trials that strictly conformed with WHO recommendations (i.e. trials on patients 18 years or older under general anesthesia)32,33,37,42,43,44,45,46,47,48,49,50,51,52,53,54,55,59 yielded results consistent with the initial analysis (RR: 0.84; 95% CI: 0.75, 0.94; p = 0.002; I2 = 54%). No significant effect was found in trials deviating from the guidelines36,38,39 (RR: 0.94; 95% CI: 0.79, 1.12; p = 0.52, I2 = 35%). Fourthly, the trials by Schietroma et al. should be dealt with cautiously due to retractions among the author’s other manuscripts60. We performed a sensitivity analysis removing the works by Schietroma et al. yielded no significant effect of FiO2 (Supplemental Table S5)52,53,54.
Discussion
Our meta-analysis demonstrated a potential benefit of a FiO2 of 80% compared to 30% or lower in reducing SSI in abdominal surgeries. TSA revealed that the required sample size was reached and the cumulative z-curve crossed the trial sequential boundary of the preference for high FiO2. A sensitivity analysis of the definition of SSI did not alter the results. However, the low quality of evidence still prevents a firm conclusion.
Whilst evidence for WHO guidelines are disputed, the beneficial effect of high FiO2 on SSI reduction continued to be found in recent studies, albeit in limited populations. It has been previously demonstrated for a subgroup of intubated patients23, or trials on a majority of emergency operations21, and when a stringent value of 80% versus 30% oxygen was applied21. This finding was also supported by a previous meta-analysis when inadequately or ambiguously blinded trials were excluded61.
Wound infections are usually established in a “decisive period” of several hours in the postoperative period9, during which host defense vigorously functions to remove pathogens, starting with neutrophils. Their key mechanism is the generation of antimicrobial reactive oxidant species9,42. In vitro studies have shown that neutrophil oxygen consumption and hence its production of oxidants are hampered at low oxygen tension8, which is alleviated by the administration of high levels of oxygen62. Therefore, it has been hypothesized that high FiO2 activates the antimicrobial mechanism of neutrophils and thereby decreases SSI. This theory was true in an animal study which showed that neutrophils in the 80% FiO2 group were more activated than those in the 30% group, although no variance was found in infiltration amounts63. Nonetheless, clinical trials have yielded mixed results due to various confounding factors of the clinical practice. However, the significant result of our subgroup analysis of supplied oxygen for at least postoperative six hours supports the importance of the decisive period.
Our results contrast with the cumulative outcomes of recent meta-analyses that found no significant beneficial or harmful effect of high FiO221,22,23,61,64,65. The different result of our meta-analysis is primarily due to the inclusion of only abdominal surgeries while previous meta-analyses included any types of surgeries. Three previous meta-analyses included randomized trials of non-cardiac or any type of surgeries and reported no significant results for SSI21,61,65. The other meta-analyses analyzed mortality, length of hospital stay, and the incidence of cardiovascular or respiratory complication but reported no significant difference22,23,64, which was consistent with our results. The significant results of our meta-analysis for SSI may be, in part, attributed to certain characteristics specific to abdominal surgery. One possible explanation is a marked variance in pathogens. While Staphylococcus aureus is the most common microorganism causing SSI66, Escherichia coli is the most predominant in SSI following abdominal surgery67. S. aureus is a notorious foe to neutrophils, armed with mechanisms for evasion such as chemotaxis inhibitory protein68 and extracellular fibrinogen binding proteins that block complement activation69. E. coli is armed with other defense mechanisms such as lipopolysaccharides. As such, discrepancies in bacterial characteristics interact with neutrophil activity differently; perhaps those that infest abdominal wounds are more susceptible to neutrophils, which, in turn, may enhance the effect of high FiO2.
The high FiO2 was also associated with a significantly lower incidence of anastomotic leakage. Ischemia in surrounding tissue is essential for anastomotic leakage development, leading to delayed wound healing, necrosis, and dehiscence70. Application of high FiO2 increases tissue oxygen levels and may thereby prevent the formation of anastomotic leakage. No association was found for organ-space SSI; there is yet no evidence for a beneficial effect of oxygen on severe SSI. Previous studies and ours alike showed no association between FiO2 and poor clinical outcomes including mortality, myocardial injury, reoperation, pneumonia, and in length of hospital stay21,22,23.
However, the adverse effect of high FiO2 should be acknowledged. Given that one hundred percent oxygen is known to induce absorption atelectasis even if administered for a short time71 and that another meta-analysis demonstrated detrimental effects of high FiO2 on oxygen parameters and severity of atelectasis72, concerns about pulmonary function cannot be discarded. Atelectasis is not causative of postoperative pneumonia73 and must be dealt with as an independent outcome.
TSA for SSI revealed that we have reached the required information size and the O’Brien-Fleming boundary had been crossed. Nonetheless, the quality of evidence was judged low; robust evidence is still lacking, and our results are subject to change. Secondary adverse outcomes of high FiO2 have yet to reach the required information size.
A subgroup analysis showed that high FiO2 is beneficial only when the mean BMI was below 30. This result lacks sufficient power as revealed by TSA and is contrary to our expectation; obesity is a known risk factor for surgical site infection74, especially for colorectal surgery75. Hypoperfusion of adipose tissue in obese patients delays wound healing and forms dead space, predisposing patients to SSI76. Additionally, poor tissue oxygenation makes it difficult for prophylactic antibiotics to reach sufficient concentrations.
We found high FiO2 to be beneficial in emergency operations, consistent with the subgroup analysis of a previous meta-analysis21 yet contrasting with another study on acutely ill adults16 and a third inconclusive study77. These discrepancies may be due to insufficient power and heterogeneity in included surgery types. The previous meta-analysis21 was based on three trials of 509 patients and our analysis was on only two trials with 388 patients. Studies on acute appendicitis took up the majority with a short operation time duration and a predominantly laparoscopic approach.
Subgroup analysis comparing nitrous oxide to air showed that FiO2 decreased SSI in only the latter. While a previous meta-analysis claimed that usage of nitrous oxide does not significantly alter SSI rates78, its assertion may lack power as it was based on six trials with high heterogeneity. A randomized trial published afterward also found no association between N2O and SSI79. We believe that N2O may potentially serve as a confounding factor; it is known to inhibit methionine production, which leads to a reduction in protein expression which in turn deters the healing process80. It is also known to depress chemotactic migration81 and inhibit methionine synthase82.
Subgroup analysis revealed that the beneficial effect of high FiO2 on SSI was consistent to a subgroup of colorectal surgeries and other abdominal surgeries, but not to trials of Cesarean sections. Caesarean sections are commonly operated under regional anesthesia, which was the case for all trials included in our analysis. It is also a relatively safe procedure with a low SSI rate, hence the effect of FiO2 may have been statistically trivial even if favorable.
High FiO2 appeared to be useful only when the duration of exposure was 6 h or longer. This starkly contrasts with the current WHO guidelines, which advise oxygen administration for 2–6 h postoperatively5. The suggested duration is not based on physiological evidence and is rather attributable to additional factors such as resource use. As far as we know, the optimal duration of exposure has not been meticulously studied to date. The decisive period for oxygen to benefit a patient is unknown; and though a need for a direct comparison between exposure duration has been suggested33, we found no trials delving into this issue. Further trials comparing the effect of oxygenation for different durations are required to better understand the effect of oxygen on SSI reduction.
Our meta-analysis provided timely analysis, including recently published studies31,43 and a trial previously left out for unknown causes55, as well as re-incorporating studies that have been omitted from conservative meta-analyses due to concerns of authors with retracted articles52,53,54. Despite several previous meta-analyses on broader classes of surgery, an inspection of abdominal surgery has not been conducted. It has been analyzed as a subgroup analysis with insignificant results21. Including recently published trials and trials with regional anesthesia as well have resulted in a contrasting conclusion. We also performed a subgroup analysis on diabetes mellitus and BMI, well-known risk factors of SSI. Our findings that diabetes and obesity may hinder or counterbalance the positive effects of oxygen may contribute to stratifying the intervention to relevant patients in future operations. While concerns have been raised on the vast range of postoperative oxygenation duration16, no analysis has been done on the topic; we found a significant benefit of oxygenation administered for at least 6 h. Heterogeneous definitions of the endpoint were also brought up as a source of potential imprecision, yet our sensitivity analysis showed that trials on CDC or ASEPSIS definitions benefited from the intervention alike. A final sensitivity analysis including only the population under general anesthesia with tracheal intubation as indicated by WHO guidelines revealed that high FiO2 does indeed reduce SSI. This finding effectively diminishes the risks of imprecision arising from our attempt to include more studies than previous reviews. Meta-regression found that age, mean BMI, percentage of patients with DM, and percentage of smokers did not significantly alter our results. Male sex is a disputed risk factor; a recently published multicenter study found its effect non-significant83.
There are several important limitations in our study. Firstly, the quality of evidence is low. Only two41,49 included trials were deemed at low risk of bias, while two trials judged to be at high risk39,56 impose concerns on the possible bias. We rated high-risk trials due to the concerns with deviations from intended interventions39 and outcome measurement56. Regarding ethical consideration, the studies by Schietroma et al. should be dealt with caution due to questionable methodology based on some retractions60. Some of other studies by Schietroma et al. were retracted, although the studies included in our analyses were not retracted or are under investigation60. Our sensitivity analysis removing the works of Schietroma yielded no significant effect of FiO252,53,54.
Secondly, heterogeneity among data is also a downfall. The I2 value for the main analysis and notable subgroup analyses was high. We attempted to explore major potential sources of heterogeneity with subgroup analyses and meta-regression; unresolved factors may include variations in anesthetic regimes and protocols of prophylactic antibiotics. Aspects of the study population such as comorbidities or selectioncriteria may also have played a role. Study settings and the baseline quality of performance of each research center were also discrepant. As surgical and anesthetic techniques have evolved, older trials may be outdated; the year of inclusion showed indeed a significant influence21. Variance in sample size is another potential source of heterogeneity, as was found in the aforementioned study.
In conclusion, this meta-analysis found that high FiO2 reduced the incidence of SSI and anastomotic leakage after abdominal surgery, a viewpoint distinct from the current consensus that perioperative high FiO2 does not benefit patients. This difference may be attributable to our focus on solely abdominal operations. Taking together the variance in subgroups, we suggest that the administration of high FiO2 should be individualized based on patient and surgery characteristics rather than being standardized. Based on our significant subgroup analyses for colorectal surgery and other abdominal surgeries but not for Caesarean section, further studies for these specific types of surgeries are required. The TSA for SSI revealed that the cumulative analysis crossed the trial sequential boundary while the required information size was reached. However, our results of the meta-analysis and TSA should be cautiously interpreted as the quality of evidence was judged moderate to very low for all outcomes. Further large trials focusing on high-risk surgeries, and a direct comparison of oxygenation duration are required to explore the true effects of this intervention which underlies countless operations.
Data availability
All other data is available in the Supplementary Information files. Any further information including all data used in our analyses are available upon request from the corresponding author.
Abbreviations
- BMI:
-
Body-mass index
- CDC:
-
Centers for Disease Control and Prevention
- CI:
-
Confidence interval
- DM:
-
Diabetes mellitus
- FiO2 :
-
Fraction of inspired oxygen
- GRADE:
-
Grading of Recommendations Assessment, Development, and Evaluation
- MD:
-
Mean difference
- RCT:
-
Randomized controlled trials
- RR:
-
Relative risk
- RRR:
-
Relative risk reduction
- SSI:
-
Surgical site infection
- TSA:
-
Trial sequential analysis
- WHO:
-
World Health Organization
References
Weiser, T. G. et al. Size and distribution of the global volume of surgery in 2012. Bull. World Health Organ. 94, 201–209f (2016).
Coello, R. et al. Adverse impact of surgical site infections in English hospitals. J. Hosp. Infect. 60, 93–103 (2005).
World Health Organization. Global Guidelines for the Prevention of Surgical Site Infection, 2nd edition https://www.who.int/publications/i/item/global-guidelines-for-the-prevention-of-surgical-site-infection-2nd-ed (2018).
Akca, O. et al. WHO needs high FIO2?. Turk. J. Anaesthesiol. Reanim 45, 181–192 (2017).
Solomkin, J. et al. World Health Organization responds to concerns about surgical site infection prevention recommendations. Anesthesiology 128, 221–222 (2018).
Berrios-Torres, S. I. et al. Centers for disease control and prevention guideline for the prevention of surgical site infection. JAMA Surg. 152, 784–791 (2017).
Ban, K. A. et al. American college of surgeons and surgical infection society: Surgical site infection guidelines, 2016 update. J. Am. Coll. Surg. 224, 59–74 (2017).
Govinda, R. et al. Early postoperative subcutaneous tissue oxygen predicts surgical site infection. Anesth. Analg. 111, 946–952 (2010).
Allen, D. B. et al. Wound hypoxia and acidosis limit neutrophil bacterial killing mechanisms. Arch. Surg. 132, 991–996 (1997).
Greif, R., Laciny, S., Rapf, B., Hickle, R. S. & Sessler, D. I. Supplemental oxygen reduces the incidence of postoperative nausea and vomiting. Anesthesiology 91, 1246–1252 (1999).
Goll, V. et al. Ondansetron is no more effective than supplemental intraoperative oxygen for prevention of postoperative nausea and vomiting. Anesth. Analg. 92, 112–117 (2001).
Rothen, H. U. et al. Influence of gas composition on recurrence of atelectasis after a reexpansion maneuver during general anesthesia. Anesthesiology 82, 832–842 (1995).
van Kaam, A. H. et al. Reducing atelectasis attenuates bacterial growth and translocation in experimental pneumonia. Am. J. Respir. Crit. Care Med. 169, 1046–1053 (2004).
McNulty, P. H. et al. Effects of supplemental oxygen administration on coronary blood flow in patients undergoing cardiac catheterization. Am. J. Physiol. Heart Circ. Physiol. 288, H1057-1062 (2005).
Hirunpattarasilp, C., Shiina, H., Na-Ek, N. & Attwell, D. The effect of hyperoxemia on neurological outcomes of adult patients: A systematic review and meta-analysis. Neurocrit. Care 36, 1027–1043 (2022).
Chu, D. K. et al. Mortality and morbidity in acutely ill adults treated with liberal versus conservative oxygen therapy (IOTA): A systematic review and meta- analysis. Lancet 391, 1693–1705 (2018).
Mackle, D. et al. Conservative oxygen therapy during mechanical ventilation in the ICU. N. Engl. J. Med. 382, 989–998 (2020).
O’Driscoll, B. R., Howard, L. S., Earis, J. & Mak, V. British Thoracic Society Guideline for oxygen use in adults in healthcare and emergency settings. BMJ Open Respir. Res. 4, e000170 (2017).
Barbateskovic, M. et al. Higher versus lower fraction of inspired oxygen or targets of arterial oxygenation for adults admitted to the intensive care unit. Emergencias 33, 309–311 (2021).
Orhan-Sungur, M., Kranke, P., Sessler, D. & Apfel, C. C. Does supplemental oxygen reduce postoperative nausea and vomiting? A meta-analysis of randomized controlled trials. Anesth. Analg. 106, 1733–1738 (2008).
Hoybye, M. et al. Fraction of inspired oxygen during general anesthesia for non-cardiac surgery: Systematic review and meta-analysis. Acta Anaesthesiol. Scand. 66, 923–933 (2022).
Lim, C. H. et al. Effects of high versus low inspiratory oxygen fraction on postoperative clinical outcomes in patients undergoing surgery under general anesthesia: A systematic review and meta-analysis of randomized controlled trials. J. Clin. Anesth. 75, 110461 (2021).
Mattishent, K. et al. Safety of 80% vs 30–35% fraction of inspired oxygen in patients undergoing surgery: A systematic review and meta-analysis. Br. J. Anaesth. 122, 311–324 (2019).
Ban, K. A. et al. Executive Summary of the American College of Surgeons/surgical infection society surgical site infection guidelines-2016 update. Surg. Infect. (Larchmt) 18, 379–382 (2017).
Imai, E. et al. Surgical site infection risk factors identified by multivariate analysis for patient undergoing laparoscopic, open colon, and gastric surgery. Am. J. Infect. Control 36, 727–731 (2008).
Alkaaki, A. et al. Surgical site infection following abdominal surgery: A prospective cohort study. Can. J. Surg. 62, 111–117 (2019).
Aga, E. et al. Surgical site infections after abdominal surgery: Incidence and risk factors. A prospective cohort study. Infect. Dis. (Lond.) 47, 761–767 (2015).
Legesse Laloto, T., Hiko Gemeda, D. & Abdella, S. H. Incidence and predictors of surgical site infection in Ethiopia: Prospective cohort. BMC Infect. Dis. 17, 119 (2017).
Hedrick, T. L., Sawyer, R. G., Friel, C. M. & Stukenborg, G. J. A method for estimating the risk of surgical site infection in patients with abdominal colorectal procedures. Dis. Colon Rectum 56, 627–637 (2013).
Sugiura, T., Uesaka, K., Ohmagari, N., Kanemoto, H. & Mizuno, T. Risk factor of surgical site infection after pancreaticoduodenectomy. World J. Surg. 36, 2888–2894 (2012).
Yerra, P. et al. Effect of peri-operative hyperoxygenation on surgical site infection in patients undergoing emergency abdominal surgery: A randomized controlled trial. Surg. Infect. (Larchmt) 22, 1052–1058 (2021).
Myles, P. S. et al. Avoidance of nitrous oxide for patients undergoing major surgery: A randomized controlled trial. Anesthesiology 107, 221–231 (2007).
Belda, F. J. et al. Supplemental perioperative oxygen and the risk of surgical wound infection: A randomized controlled trial. JAMA 294, 2035–2042 (2005).
Wilson, A. P., Treasure, T., Sturridge, M. F. & Gruneberg, R. N. A scoring method (ASEPSIS) for postoperative wound infections for use in clinical trials of antibiotic prophylaxis. Lancet 1, 311–313 (1986).
Sterne, J. A. C. et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 366, l4898 (2019).
Bickel, A., Gurevits, M., Vamos, R., Ivry, S. & Eitan, A. Perioperative hyperoxygenation and wound site infection following surgery for acute appendicitis: A randomized, prospective, controlled trial. Arch. Surg. 146, 464–470 (2011).
Chen, Y. et al. Leukocyte DNA damage and wound infection after nitrous oxide administration: A randomized controlled trial. Anesthesiology 118, 1322–1331 (2013).
Duggal, N., Poddatorri, V., Noroozkhani, S., Siddik-Ahmad, R. I. & Caughey, A. B. Perioperative oxygen supplementation and surgical site infection after cesarean delivery: A randomized trial. Obstet. Gynecol. 122, 79–84 (2013).
Fariba, F., Loghman, G., Daem, R., Dina, S. & Jamal, S. Effect of supplemental oxygen on the incidence and severity of Wound Infection after cesarean surgery. J. Chem. Pharma. Sci. 9, 3320–3325 (2016).
Ferrando, C. et al. Effects of oxygen on post-surgical infections during an individualised perioperative open-lung ventilatory strategy: A randomised controlled trial. Br. J. Anaesth. 124, 110–120 (2020).
Gardella, C. et al. High-concentration supplemental perioperative oxygen to reduce the incidence of postcesarean surgical site infection: A randomized controlled trial. Obstet. Gynecol. 112, 545–552 (2008).
Greif, R., Akca, O., Horn, E. P., Kurz, A. & Sessler, D. I. Supplemental perioperative oxygen to reduce the incidence of surgical- wound infection. N. Engl. J. Med. 342, 161–167 (2000).
Holse, C. et al. Hyperoxia and antioxidants for myocardial injury in noncardiac surgery: A 2 × 2 factorial, Blinded, randomized clinical trial. Anesthesiology 136, 408–419 (2022).
Kurz, A. et al. Effects of supplemental oxygen and dexamethasone on surgical site infection: A factorial randomized trialdouble dagger. Br. J. Anaesth. 115, 434–443 (2015).
Li, X. F. et al. Comparison of low and high inspiratory oxygen fraction added to lung- protective ventilation on postoperative pulmonary complications after abdominal surgery: A randomized controlled trial. J. Clin. Anesth. 67, 110009 (2020).
Lin, X. et al. Intraoperative oxygen concentration and postoperative delirium after laparoscopic gastric and colorectal malignancies surgery: A randomized, double-blind, controlled trial. Clin. Interv. Aging 16, 1085–1093 (2021).
Mayank, M., Mohsina, S., Sureshkumar, S., Kundra, P. & Kate, V. Effect of perioperative high oxygen concentration on postoperative SSI in elective colorectal surgery—A randomized controlled trial. J. Gastrointest. Surg. 23, 145–152 (2019).
Mayzler, O. et al. Does supplemental perioperative oxygen administration reduce the incidence of wound infection in elective colorectal surgery?. Miner. Anestesiol. 71, 21–25 (2005).
Meyhoff, C. S. et al. Effect of high perioperative oxygen fraction on surgical site infection and pulmonary complications after abdominal surgery: The PROXI randomized clinical trial. JAMA 302, 1543–1550 (2009).
Pryor, K. O., Fahey, T. J. 3rd., Lien, C. A. & Goldstein, P. A. Surgical site infection and the routine use of perioperative hyperoxia in a general surgical population: A randomized controlled trial. JAMA 291, 79–87 (2004).
Reiterer, C. et al. Perioperative supplemental oxygen and NT-proBNP concentrations after major abdominal surgery—A prospective randomized clinical trial. J. Clin. Anesth. 73, 110379 (2021).
Schietroma, M. et al. Prevention of anastomotic leakage after total gastrectomy with perioperative supplemental oxygen administration: A prospective randomized, double-blind, controlled, single-center trial. Ann. Surg. Oncol. 20, 1584–1590 (2013).
Schietroma, M. et al. Supplemental peri-operative oxygen and incision site infection after surgery for perforated peptic ulcer: A randomized, double-blind monocentric trial. Surg. Infect. (Larchmt) 17, 106–113 (2016).
Schietroma, M. et al. Effect of high perioperative oxygen fraction on surgical site infection following surgery for acute sigmoid diverticulitis. A prospective, randomized, double blind, controlled, monocentric trial. Chirurgia (Bucur) 111, 242–250 (2016).
Wadhwa, A., Kabon, B., Fleischmann, E., Kurz, A. & Sessler, D. I. Supplemental postoperative oxygen does not reduce surgical site infection and major healing-related complications from bariatric surgery in morbidly obese patients: A randomized, blinded trial. Anesth. Analg. 119, 357–365 (2014).
Wasnik, N., Agrawal, V. P., Yede, J., Gupta, A. & Soitkar, S. Role of supplemental oxygen in reducing surgical site infection in acute appendicities: Our experience of sixty four cases. Int. J. Biomed. Adv. Res. 6, 124–127 (2015).
Williams, N. L., Glover, M. M., Crisp, C., Acton, A. L. & McKenna, D. S. Randomized controlled trial of the effect of 30% versus 80% fraction of inspired oxygen on cesarean delivery surgical site infection. Am. J. Perinatol. 30, 781–786 (2013).
Kurz, A. et al. Supplemental oxygen and surgical-site infections: An alternating intervention controlled trial. Br. J. Anaesth. 120, 117–126 (2018).
Alvandipour, M., Mokhtari-Esbuie, F., Baradari, A. G., Firouzian, A. & Rezaie, M. Effect of hyperoxygenation during surgery on surgical site infection in colorectal surgery. Ann. Coloproctol. 35, 9–14 (2019).
Myles, P. S., Carlisle, J. B. & Scarr, B. Evidence for compromised data integrity in studies of liberal peri-operative inspired oxygen. Anaesthesia 74, 573–584 (2019).
Wetterslev, J. et al. The effects of high perioperative inspiratory oxygen fraction for adult surgical patients. Cochrane Database Syst. Rev. 2015, CD008884 (2015).
Hopf, H. W. et al. Wound tissue oxygen tension predicts the risk of wound infection in surgical patients. Arch. Surg. 132, 997–1004 (1997).
Kroin, J. S. et al. Perioperative high inspired oxygen fraction therapy reduces surgical site infection with Pseudomonas aeruginosa in rats. J. Med. Microbiol. 65, 738–744 (2016).
Markwei, M. T. et al. Perioperative supplemental oxygen and postoperative nausea and vomiting: Subanalysis of a trial, systematic review, and meta-analysis. Anesthesiology 138, 56–70 (2023).
de Jonge, S. et al. Effectiveness of 80% vs 30–35% fraction of inspired oxygen in patients undergoing surgery: An updated systematic review and meta-analysis. Br. J. Anaesth. 122, 325–334 (2019).
El-Saed, A. et al. High contribution and impact of resistant gram negative pathogens causing surgical site infections at a multi-hospital healthcare system in Saudi Arabia, 2007–2016. BMC Infect. Dis. 20, 275 (2020).
Alexiou, K. et al. A prospective randomised trial of isolated pathogens of surgical site infections (SSI). Ann. Med. Surg. (Lond.) 21, 25–29 (2017).
de Haas, C. J. et al. Chemotaxis inhibitory protein of Staphylococcus aureus, a bacterial antiinflammatory agent. J. Exp. Med. 199, 687–695 (2004).
Lee, L. Y. et al. Inhibition of complement activation by a secreted Staphylococcus aureus protein. J. Infect. Dis. 190, 571–579 (2004).
Marland, J. R. K. et al. Post-operative monitoring of intestinal tissue oxygenation using an implantable microfabricated oxygen sensor. Micromachines (Basel) 12, 810 (2021).
Lundquist, H., Hedenstierna, G., Strandberg, A., Tokics, L. & Brismar, B. CT-assessment of dependent lung densities in man during general anaesthesia. Acta Radiol. 36, 626–632 (1995).
Koo, C. H., Park, E. Y., Lee, S. Y. & Ryu, J. H. The effects of intraoperative inspired oxygen fraction on postoperative pulmonary parameters in patients with general anesthesia: A systemic review and meta-analysis. J. Clin. Med. 8, 583 (2019).
Crompton, J. G., Crompton, P. D. & Matzinger, P. Does atelectasis cause fever after surgery? Putting a damper on dogma. JAMA Surg. 154, 375–376 (2019).
Meijs, A. P. et al. The effect of body mass index on the risk of surgical site infection. Infect. Control Hosp. Epidemiol. 40, 991–996 (2019).
Hourigan, J. S. Impact of obesity on surgical site infection in colon and rectal surgery. Clin. Colon Rectal. Surg. 24, 283–290 (2011).
Anaya, D. A. & Dellinger, E. P. The obese surgical patient: A susceptible host for infection. Surg. Infect. (Larchmt) 7, 473–480 (2006).
Barbateskovic, M. et al. Higher vs lower oxygenation strategies in acutely Ill adults: A systematic review with meta-analysis and trial sequential analysis. Chest 159, 154–173 (2021).
Sun, R. et al. Nitrous oxide-based techniques versus nitrous oxide-free techniques for general anaesthesia. Cochrane Database Syst. Rev. 11, CD008984 (2015).
Fleischmann, E. et al. Nitrous oxide and risk of surgical wound infection: A randomised trial. Lancet 366, 1101–1107 (2005).
Hassanain, H. H. et al. Smooth muscle cell expression of a constitutive active form of human Rac 1 accelerates cutaneous wound repair. Surgery 137, 92–101 (2005).
Kripke, B. J., Kupferman, A. & Luu, K. C. Suppression of chemotaxis to corneal inflammation by nitrous oxide. Zhonghua Min Guo Wei Sheng Wu Ji Mian Yi Xue Za Zhi 20, 302–310 (1987).
Perry, J., Chanarin, I., Deacon, R. & Lumb, M. Chronic cobalamin inactivation impairs folate polyglutamate synthesis in the rat. J. Clin. Invest. 71, 1183–1190 (1983).
Zwicky, S. N. et al. No impact of sex on surgical site infections in abdominal surgery: A multi-center study. Langenbecks Arch. Surg. 407, 3763–3769 (2022).
Acknowledgements
This work was supported by the New Faculty Startup Fund from Seoul National University (800-20230296).
Author information
Authors and Affiliations
Contributions
J.H.K.: This author helped with the literature search, data collection and writing the primary draft of the manuscript; W.-S.J.: This author helped with the literature search, data collection, and reviewing and editing the manuscript; L.L.: This author helped with reviewing and editing the manuscript; H.K.Y.: This author helped with reviewing and editing the manuscript; J.-W.J.: This author helped with data analysis and reviewing and editing the manuscript; H.-J.L.: This author helped with data analysis and reviewing and editing the manuscript; W.H.K.: This author helped with study conceptualization, literature search, data collection, data analysis, data interpretation, supervision, writing the original draft, and reviewing and editing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuh, J.H., Jung, WS., Lim, L. et al. The effect of high perioperative inspiratory oxygen fraction for abdominal surgery on surgical site infection: a systematic review and meta-analysis. Sci Rep 13, 15599 (2023). https://doi.org/10.1038/s41598-023-41300-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-41300-4
- Springer Nature Limited
This article is cited by
-
Impact of hyperoxia on the gut during critical illnesses
Critical Care (2024)