Abstract
Computer tomography-derived skeletal muscle index normalized for height in conjunction with muscle density enables single modality-based sarcopenia assessment that accounts for all diagnostic criteria and cutoff recommendations as per the widely accepted European consensus. Yet, the standard approach to quantify skeletal musculature at the third lumbar vertebra is limited for certain patient groups, such as lung cancer patients who receive chest CT for tumor staging that does not encompass this lumbar level. As an alternative, this retrospective study assessed sarcopenia in lung cancer patients treated with curative intent at the tenth thoracic vertebral level using appropriate cutoffs. We showed that skeletal muscle index and radiation attenuation at level T10 correlate well with those at level L3 (Pearson’s R = 0.82 and 0.66, p < 0.001). During a median follow-up period of 55.7 months, sarcopenia was independently associated with worse overall (hazard ratio (HR) = 2.11, 95%-confidence interval (95%-CI) = 1.38–3.23, p < 0.001) and cancer-specific survival (HR = 2.00, 95%-CI = 1.19–3.36, p = 0.009) of lung cancer patients following anatomic resection. This study highlights feasibility to diagnose sarcopenia solely by thoracic CT in accordance with the European consensus recommendations. The straightforward methodology offers easy translation into routine clinical care and potential to improve preoperative risk stratification of lung cancer patients scheduled for surgery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Sarcopenia (Greek sarx 'flesh' and penia 'poverty') is commonly described as a progressive and generalized disorder of skeletal muscle1. Beyond ageing1,2, many factors have been identified to contribute to the sarcopenic phenotype, such as malnutrition, inactivity and chronic diseases like chronic obstructive pulmonary disease, diabetes mellitus and cancer3. Within different cancer types and stages including lung cancer, sarcopenia was shown to be associated with poorer survival4.
To date, no international consensus on sarcopenia has been achieved3. And so far, different approaches have been used to quantify sarcopenia1,5. The most widely used definition and diagnostic criteria for sarcopenia have been established by the European Working Group on Sarcopenia in Older People (EWGSOP)1 and supported by the Asian Working group on Sarcopenia2. They have been endorsed for clinical practice and research by various international scientific societies3. Accordingly, sarcopenia can be determined by muscle strength, quantity, quality, and physical performance1,2. Lumbar third vertebral level (L3) imaging by computed tomography (CT) is considered gold standard for non-invasive muscle assessment1. Muscle quantity can be measured by cross-sectional muscle area (SMA) on axial CT imaging and converted into a height-adjusted index, skeletal muscle index (SMI), similar to body mass index (BMI). CT-derived skeletal muscle radiation attenuation (SMRA) is a measure of muscle quality1,6,7 which is associated with function7,8,9,10, strength7,11,12 and physical performance13. Hence, SMA or SMI in combination with SMRA may enable complete assessment of sarcopenia, solely by CT, if respective cutoff values are available13. The general recommendation is to set the cutoff for sarcopenia-related measurements two standard deviations (SD) below the mean of a young and healthy reference population1. For lumbar level L3 many reference values have been reported14,15. However, as part of the standard of care staging, lung cancer patients usually undergo CT of the chest and upper abdomen that does not include L316. European consensus based CT-cutoff values derived from a healthy cohort of potential kidney donors have been presented for thoracic spine levels13 that, to our knowledge, have not yet been tested for clinical outcomes.
We performed a novel approach using SMI in conjunction with SMRA to provide an assessment of sarcopenia solely by thoracic CT that meets the European consensus definition and diagnostic criteria1 and analyzed its relevance for preoperative risk stratification for long-term survival of lung cancer patients after anatomic resection.
Patients and Methods
Data acquisition
Data was acquired at two tertiary hospitals, the University Hospital Regensburg and Hospital Barmherzige Brüder Regensburg. For both institutions, the study protocol and ethical clearance were approved by the Institutional Review Board (IRB) of the University Regensburg (Study code 20-2045-101, approved on October 14, 2020). All methods were carried out in accordance with institutional guidelines and regulations. Because pre-existing patient information and CT scans were used retrospectively, the need for informed consent was waived for both hospitals by the IRB of the University Regensburg. At both tertiary institutions, we retrospectively identified 589 patients who underwent anatomic pulmonary resection (segmentectomy, (bi-)lobectomy or pneumonectomy) in the departments of thoracic surgery between 2015 and 2018 (Fig. 1). Inclusion required pathologically confirmed diagnosis of lung cancer and treatment with curative intent. Preoperative contrast-enhanced computed tomography examinations of the thorax and abdomen were included if they were obtained within 60 days before surgery with complete visualization of skeletal muscle at the level of the tenth thoracic vertebral body (T10). Clinical data were extracted from the most recent patient records before surgery. Comorbidities were assessed by the Charlson Comorbidity Index (CCI) based on the information of the preoperative patient report17,18. Since 2017, the staging of lung cancer has been performed according to the eighth edition of the Tumor, Nodes, and Metastasis (TNM) staging system of malignant tumors. Thus, patients who had surgery before 01/01/2017 were classified according to the criteria of the eighth edition based on the seventh edition TNM descriptors19,20. From preoperative pulmonary function testing, forced expiratory volume in one second (FEV1), diffusion capacity for carbon monoxide (DLCO), and vital capacity (VC) were retrieved. Primary outcomes were overall and cancer-specific survival. Cancer-specific decease was determined by death from the index lung cancer. Survival time was calculated from the day of resection. Vital status and date of last follow-up were retrieved from medical records, death certificates, registration offices, and the Clinical Cancer Registry of the Tumor Center–Institute for Quality Management and Health Services Research, University of Regensburg. If no death record was found, survival was censored at date of last visit. The follow-up period ended on 05/31/2022.
Semi-automated CT-based muscle measurement
The most recent preoperative CT image was accessed with free-ware 64-bit DICOM viewer Horos™ (Horos Project, Geneva, Switzerland, version 4.0). Patients underwent routine preoperative CT imaging at a tube voltage of 100 kVp and with contrast injection yielding 3 to 5 mm thick multiplanar reconstructions in the axial plane.
Measurements were performed at the level of the fifth, eighth and tenth thoracic vertebral bodies (T5, 8, 10) and at the third level of the lumbar vertebral body (L3).
Lumbar third vertebra imaging by CT is considered gold standard for non-invasive skeletal muscle and sarcopenia assessment1. T5 serves as an anatomic landmark for the aortic arch and has been previously used to quantify skeletal muscle21. Nevertheless, complete visualization is more frequently available further caudal22,23. T8 was shown to correlate well with T523. More importantly, T10 is the most cranial level for which cutoff values have been established13. The selection of vertebral levels also allowed us to analyze the correlation of skeletal muscle measurements of the upper, middle, and lower thoracic and lumbar spine.
We measured on two consecutive CT slices where both procc. tranversi of the thoracic spine or procc. costales of the lumbar spine were visible and computed the mean of both measurements for further analysis. The distinction between different tissue components is based on Hounsfield units (HU). Using the “Grow Region (2D/3D Segmentation)” application within the Region-of-Interest-tool, a preset threshold range of −30 to 150 HU7 was used to automatically quantify cross-sectional skeletal muscle area and the respective mean skeletal muscle radiation attenuation within the area. If necessary, subsequent manual adjustment was performed by a primary analyst with consensus reading of a board-certified radiologist with more than 10 years of experience.
Sarcopenia assessment
According to the European consensus guidelines, sarcopenia is confirmed by the presence of low muscle quantity or quality in combination with low muscle strength. Poor physical performance indicates severe sarcopenia1. Cross-sectional muscle area (cm2) was normalized for height (m) squared and reported as skeletal muscle index (cm2/m2). We diagnosed sarcopenia by presence of low SMI in combination with low SMRA which were defined as SMI of < 28.8 cm2/m2 for men and < 20.4 cm2/m2 for women and SMRA < 32.4 HU for men and < 26.5 HU for women13, respectively.
Statistical methods
Statistical analyses were performed with R version 4.2.024 and the package ggplot225 for data visualization. The significance-level was set at p < 0.05 for hypothesis testing and confidence intervals (CI) unless otherwise indicated. We present categorical variables as count [percentage (%)] and continuous variables as mean [standard deviation (SD)] or median [interquartile range (Q1–Q3)], as appropriate. Differences between groups were analyzed with the chi-square test of independence or Fisher exact test (if the expected frequency was below 5) for categorical variables, the Student’s or Welch’s t test for normally distributed continuous variables, and the Mann-Whitney-U test for not normally distributed continuous variables. We used multivariable logistic regression to analyze the association of sarcopenia with clinical factors, adjusted for age and gender. Pearson’s correlation was used to determine the association between the skeletal muscle areas, indices and radiation attenutation at the different thoracic and lumbar spine levels. Overall and cancer-specific survival was visualized using the Kaplan-Meier-method. Differences in survival curves were assessed with the log-rank test. Multivariable Cox proportional hazards regression determined whether the prevalence of sarcopenia was independently associated with overall and cancer-specific survival. The following covariates were identified and included based on the results of univariable Cox regression, literature26,27, clinical experience and general recommendations to avoid overfitting28: gender, BMI, age-adjusted CCI, number of resected segments, and pathologic tumor stage. We compared multivariable models where sarcopenia as a dichotomous predictor was substituted by continuous crude muscle measures SMA, SMI and SMRA as well as averaged SMI and SMRA percentiles. Using R percentiles package29, the averaged SMI and SMRA percentiles were determined by gender-stratified aggregation of the respective SMI and SMRA percentiles at each available vertebral level in a patient22. Model fit was evaluated using Akaike Information Criterion (AIC) and ANOVA testing.
Results
Cohort summary
The study included 280 patients (35.4% female, Table 1) who underwent chest CT. At level T10, due to limited field of view, measurement of skeletal musculature was not possible for three patients who were excluded from the analysis (Fig. 1). 85 (30.4%) patients additionally received a CT scan of the abdomen. And in another 19 (6.8%) thoracic CT examinations, skeletal muscle was completely visualized at level L3. There were no significant sex differences in median age at diagnosis, mean SMRA, ECOG performance status, and comorbid disease burden as per CCI. Compared to men, females had a lower BMI (p < 0.012), SMA (p < 0.001), SMI (p < 0.001), and were less often smokers (p < 0.001). The median age at diagnosis was 66.1 (58.5–72.7) years. 71 (28.0%) of study participants had ECOG 1. Compared to patients with ECOG = 0, patients with ECOG = 1 were significantly older (median age of diagnosis 70.0 vs 63.6 years, p < 0.001) and had a significantly higher burden of comorbidities (median CCI of 1 (0–2) vs 1 (1–3), p = 0.010). Most patients had pathologic tumor stage I (46.9%). The most frequent histology subtypes were adenocarcinoma (46.4%) and squamous cell carcinoma (33.6%). The median time between CT acquisition and lung cancer resection was 22 (6–36) days. Skeletal muscle tissues at level T5, T8 and L3 were completely visualized on the CT scans of 232 (82.9%), 268 (95.7%) and 103 (36.8%) patients, respectively.
Sarcopenia
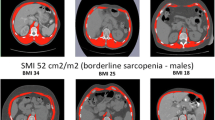
95 (33.9%) patients were classified sarcopenic (Table 1). Of note, the prevalence of sarcopenia was 20.2% in females and 41.4% in males. Figure 2 shows comparison of a male sarcopenic to a male non-sarcopenic patient. Compared to non-sarcopenic patients, sarcopenic patients were older (median age of diagnosis 69.5 vs 65.0 years, p < 0.001), had more comorbidities according to the CCI (median of 2 (1–2) vs 1 (0–2), p = 0.013) and more frequently an impaired ECOG performance status of 1 (frequency of 40.0% vs 21.9%, p = 0.004). After adjusting for age and gender, sarcopenia was significantly associated with lower BMI (OR = 0.88, p < 0.001), higher frequency of positive smoking history (OR = 5.05, p = 0.005) and a lower preoperative DLCO % predicted (OR = 0.97, p < 0.001). Other lung function parameters, FEV1% predicted (OR = 0.99, p = 0.122) and VC % predicted (OR = 0.99, p = 0.113) did not reach the significance level. There were no significant differences between the sarcopenic and non-sarcopenic group with respect to histologic subtypes, pathologic tumor stage, neoadjuvant and adjuvant therapy, procedure, and number of resected segments.
T10-SMA correlated highly with SMA at level T5, T8 and L3 (Pearson’s Rho ≥ 0.891 for all, p < 0.001 for all, Fig. 3). Similarly, Pearson’s correlation coefficients for the correlation of T10-SMI with SMI at level T5, T8 and L3 were ≥ 0.821 (p < 0.001 for all), respectively. SMRA at level T10 correlated well with SMRA at level T5, T8 and L3 (Pearson’s Rho ≥ 0.659 for all, p < 0.001 for all).
Correlation analysis of skeletal muscle areas (SMA; a–c), indices (SMI; d–f), and radiation attenuation (SMRA; g–i). SMA, SMI and SMRA at the level of the tenth (T10) thoracic vertebral body were compared to SMAs, SMIs and SMRAs at the level of the fifth (T5) and eighth (T8) thoracic and third (L3) lumbar vertebral body, respectively. Pearson’s Rho ranges from 0.659 to 0.908. P < 0.001 for all.
Survival
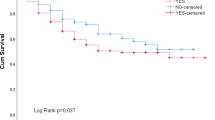
During a median follow-up period of 55.7 (46.8–71.3) months, death of 100 (35.7%) patients was registered. 53 (28.6%) of non-sarcopenic patients died, whereas 47 (49.5%) of sarcopenic patients died. In the Kaplan-Meier analysis, the sarcopenic group had worse overall (log-rank-p < 0.001) and cancer-specific survival (log-rank-p = 0.005, Fig. 4). There was no prognostic difference for overall and cancer-specific survival between the two most common histologic subtypes adenocarcinoma and squamous cell carcinoma (Table 2 and 3). Therefore, we did not add histology as a covariate to the multivariable model.
Kaplan-Meier analysis of overall (a) and cancer-specific survival (b) of patients with lung cancer following anatomic resection. Survival and number at risk of the sarcopenic (red) and non-sarcopenic group (blue) are plotted in monthly intervals. Red and blue areas indicate 95%-confidence intervals. Log-rank-p < 0.001 for overall and 0.005 for cancer-specific survival.
After multivariable adjustment, sarcopenia was independently associated with worse overall (hazard ratio (HR) 2.11, 95%-confidence interval (95%-CI) 1.38–3.23, p < 0.001, Table 2) and cancer-specific survival (HR 2.00, 95%-CI 1.19–3.36, p = 0.009, Table 3). In addition, pathologic tumor stage was found to be an unfavorable prognostic factor for overall survival as well as for cancer-specific survival. This multivariable Cox regression model using presence of sarcopenia as a binary variable provided a significantly better model fit as per AIC compared to models comprising continuous SMA, SMI and SMRA and averaged SMI percentiles as predictors for overall survival (p < 0.001, Table 4). Only the model that included averaged SMRA percentiles showed a lower AIC value in comparison to the use of dichotomous sarcopenia predictor.
Discussion
While CT imaging at the third lumbar vertebra is considered gold standard to quantify skeletal musculature, it is, however, not readily available in some patient groups, including lung cancer patients. This study evaluated a method to diagnose sarcopenia on chest CT in accordance with the widely accepted European consensus diagnostic criteria and cutoff recommendations1,13. At the tenth thoracic vertebral level, sarcopenia was indicated by low skeletal mass index in conjunction with low skeletal muscle radiation attenuation to account for presence of all diagnostic criteria—muscle quantity, quality, strength, and physical performance1—with reference to previously published cutoff values1,13. Sarcopenia was an independent prognostic factor for poorer long-term overall and cancer-specific survival of lung cancer patients following anatomic resection with curative intent. In addition, T10-SMI and SMRA correlated well with muscle indices and densities at other thoracic and lumbar spine levels, respectively. Our study emphasizes that assessment of sarcopenia solely by chest CT is feasible, easily translatable into routine clinical practice, and helpful to improve preoperative risk stratification of lung cancer patients undergoing surgery.
In our study, presence of sarcopenia evaluated by SMI and SMRA on thoracic CT was independently associated with worse overall and cancer-specific survival of lung cancer patients scheduled for anatomic resection. This observation held true after adjusting for variables known to be associated with mortality following lung cancer surgery26,27. Additionally, pathologic tumor stage was found to be an independent predictor of survival. In otherwise identical models, including sarcopenia as a dichotomous predictor for overall survival resulted in a model with significant higher goodness of fit compared to the use of singular continuous muscle measures SMA, SMI and SMRA as well as averaged SMI percentiles that aggregated skeletal muscle indices from all available measurement levels in a single patient. Still, averaged multi-vertebral level SMRA percentiles yielded a better fitted model, underlining the additional value of muscle quality assessment to determine sarcopenia.
SMRA relates to muscle quality1,6,7 and is associated with function7,8,9,10, strength7,11,12 and physical performance13. Hence, it can bypass unavailability of functional testing13. All diagnostic criteria for sarcopenia recommended by European and Asian consensus can be met by SMRA in conjunction with a parameter for muscle quantity, such as SMA or SMI, and, enable diagnosis of sarcopenia solely by chest CT if cutoff values are specified. The European consensus recommended to set cutoffs two standard deviations below the mean of a young, healthy reference population1. In accordance with this guideline, reference values for SMI and SMRA derived from kidney donors aged 18–40 years have been published for levels T10 to L313. Hence, our study highlights feasibility of sarcopenia assessment based on a single modality that is readily available in lung cancer patients. The preoperative staging of lung cancer includes a contrast-enhanced CT scan of the chest and upper abdomen that usually does not encompass L316, the gold standard vertebral level for sarcopenia assessment1. Consistently, only 36.8% of patients underwent additional CT of the abdomen and 6.8% of chest CT scans also included level L3. Nevertheless, our results showed that measures of skeletal muscle quantity, SMA and SMI, as well as quality, SMRA, at level L3 correlate well with measures at level T10, the most cranial thoracic spine level for which reference values from a young, healthy reference population have previously been published13. In addition, T10-SMA, SMI and SMRA correlated well with SMAs, SMIs and SMRAs of the upper and middle thoracic spine. Compared to level T5 and T8, skeletal musculature was more frequently completely visualized at level T10. This finding is consistent with other study groups22,23,30.
Consistent with the majority of studies, a higher percentage of men were affected by sarcopenia (41.4% vs 20.2%)31,32,33. Compared to non-sarcopenic patients, sarcopenic patients were significantly older, had a higher ECOG and burden of comorbid disease as per CCI. After adjustment for age and gender, sarcopenia was significantly associated with a lower BMI and lower preoperative DLCO % predicted. Hence, our findings are consistent with the pathophysiology of sarcopenia3. With a median age at diagnosis of 69.5 years, the sarcopenic patients were at a stage in life with ongoing, physiologic decline in skeletal muscle mass and maximization of body fat12,34. The western lifestyle, especially excessive energy intake, sedentary behavior, and physical inactivity, contribute to fat deposition in muscle and muscle loss35,36. Chronic diseases such as chronic obstructive pulmonary disease, type 2 diabetes mellitus and inflammatory processes promote muscle mass reduction3. Furthermore, malignancies can lead to cancer cachexia including muscle wasting and physical impairment37 that may become apparent as a higher ECOG and poorer preoperative lung function. Yet, sarcopenia can be therapeutically targeted by aerobic and resistance training38,39, protein supplement40 and Mediterranean diet41. Thus, intervention may improve sarcopenia and possibly clinical outcomes.
To date, the European consensus criteria for sarcopenia which closely align with the Asian consensus are most widely recognized for clinical practice and research1,2,3. We have outlined that all criteria of the European consensus can be met radiologically by CT-derived SMI and SMRA1. To facilitate implementation in daily clinical practice, additional tests may be omitted13. So far, lumbar assessment of skeletal muscle is considered the gold standard to quantify sarcopenia1. Recent availability of cutoff values from a young, healthy reference population, allows the diagnosis of sarcopenia on chest CT which—unlike CT imaging of the abdomen—is readily available in lung cancer patients13. This is the first time these thoracic reference values have been tested against clinical outcomes. Our retrospective analysis showed that, in a first step, the skeletal muscle parameters SMA, SMI and SMRA at the third lumbar vertebra level correlated well with those at the tenth thoracic vertebra level. In a second step, preoperative sarcopenia as determined by SMI and SMRA at level T10 was shown to be an independent prognostic factor for poor overall and cancer-specific long-term survival of lung cancer patients following anatomic resection. Thus, our study implements single modality-based evaluation of sarcopenia on chest CT in accordance with European and Asian consensus guidelines, demonstrating its feasibility for straightforward integration into routine clinical practice and its value in enhancing preoperative risk stratification of lung cancer patients scheduled for surgery.
There are several limitations to our study. First, the retrospective design resulted in a 47%-exclusion rate due to unavailable imaging or clinical data elements. Furthermore, we had to retrospectively convert some TNM stages from the seventh to the eighth edition. Third, no minimum was set for the follow-up interval. However, the median follow-up period was 55.7 months. Fourth, the standard of care staging protocol for lung cancer patients includes IV contrast-enhanced CT imaging of the chest and upper abdomen. Consequently, most of the CT scans were acquired with contrast-enhancement and, for standardization purposes, patients who underwent plain CT imaging were excluded from the study. The cutoff-values used for sarcopenia diagnosis, however, originated from non-contrast-enhanced CT imaging at 120kVp13. It was shown that contrast injection can increase radiation attenuation by up to 5.99%42. Conversely, increase in slice thickness may decrease radiation attenuation42. Since this study included CT scans with a slice thickness of 3 to 5 mm, this may affect threshold-based segmentation. Overall, IV contrast, slice thickness, tube voltage and current may cause slight variations of skeletal muscle measures42. Nevertheless, sarcopenia was defined by both low SMRA and SMA normalized by patients’ height, and influence of IV contrast on SMA was shown to be minimal (up to 1.88%42). While, we used level T10 since it was the most cranial level for which cutoff-values from a young, healthy reference population were available, other thoracic levels may also be utilized. Finally, only patients that deemed fit enough for anatomic resection, were included in this study. Therefore, results may not be generalizable to patients who were assigned to other treatment modalities.
Future longitudinal observational studies that repeatedly assess sarcopenia-related measures in the pre- and postoperative course may yield important insights into the dynamics of muscle wasting and guide implementation of targeted interventions. Furthermore, conversion of the semi-automated sarcopenia quantification into a fully automated pipeline may allow immediate detection of sarcopenia when CT scans for diagnosis and staging purposes are performed as well as direct inclusion in preoperative risk stratification models.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Cruz-Jentoft, A. J. et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 48, 16–31. https://doi.org/10.1093/ageing/afy169 (2019).
Chen, L.-K. et al. Asian Working Group for Sarcopenia: 2019 Consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 21, 300-307.e2. https://doi.org/10.1016/j.jamda.2019.12.012 (2020).
Cruz-Jentoft, A. J. & Sayer, A. A. Sarcopenia. Lancet 393, 2636–2646. https://doi.org/10.1016/S0140-6736(19)31138-9 (2019).
Shachar, S. S., Williams, G. R., Muss, H. B. & Nishijima, T. F. Prognostic value of sarcopenia in adults with solid tumours: A meta-analysis and systematic review. Eur. J. Cancer (Oxford, England : 1990) 57, 58–67. https://doi.org/10.1016/j.ejca.2015.12.030 (2016).
Lim, W. H. & Park, C. M. Validation for measurements of skeletal muscle areas using low-dose chest computed tomography. Sci. Rep. 12, 463. https://doi.org/10.1038/s41598-021-04492-1 (2022).
Goodpaster, B. H., Kelley, D. E., Thaete, F. L., He, J. & Ross, R. Skeletal muscle attenuation determined by computed tomography is associated with skeletal muscle lipid content. J. Appl. Physiol. (Bethesda, Md. : 1985) 89, 104–110. https://doi.org/10.1152/jappl.2000.89.1.104 (2000).
Aubrey, J. et al. Measurement of skeletal muscle radiation attenuation and basis of its biological variation. Acta Physiol. (Oxford, England) https://doi.org/10.1111/apha.12224 (2014).
Bye, A. et al. Muscle mass and association to quality of life in non-small cell lung cancer patients. J. Cachexia Sarcopenia Muscle 8, 759–767. https://doi.org/10.1002/jcsm.12206 (2017).
Williams, G. R. et al. Skeletal muscle measures and physical function in older adults with cancer: sarcopenia or myopenia?. Oncotarget 8, 33658–33665. https://doi.org/10.18632/oncotarget.16866 (2017).
West, M. A. et al. Myosteatosis is associated with poor physical fitness in patients undergoing hepatopancreatobiliary surgery. J. Cachexia Sarcopenia Muscle 10, 860–871. https://doi.org/10.1002/jcsm.12433 (2019).
Goodpaster, B. H. et al. Attenuation of skeletal muscle and strength in the elderly: The Health ABC Study. J. Appl. Physiol. (Bethesda, Md. : 1985) 90, 2157–2165. https://doi.org/10.1152/jappl.2001.90.6.2157 (2001).
Goodpaster, B. H. et al. The loss of skeletal muscle strength, mass, and quality in older adults: The health, aging and body composition study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 61, 1059–1064. https://doi.org/10.1093/gerona/61.10.1059 (2006).
Derstine, B. A. et al. Skeletal muscle cutoff values for sarcopenia diagnosis using T10 to L5 measurements in a healthy US population. Sci. Rep. 8, 11369. https://doi.org/10.1038/s41598-018-29825-5 (2018).
Martin, L. et al. Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J. Clin. Oncol. https://doi.org/10.1200/JCO.2012.45.2722 (2013).
Prado, C. M. M. et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol. 9, 629–635. https://doi.org/10.1016/S1470-2045(08)70153-0 (2008).
Peters, S. et al. Metastatic non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 23(Suppl 7), vii56-64. https://doi.org/10.1093/annonc/mds226 (2012).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 40, 373–383. https://doi.org/10.1016/0021-9681(87)90171-8 (1987).
Charlson, M., Szatrowski, T. P., Peterson, J. & Gold, J. Validation of a combined comorbidity index. J. Clin. Epidemiol. https://doi.org/10.1016/0895-4356(94)90129-5 (1994).
Wittekind, C. TNM-Klassifikation Maligner Tumoren. Mit Allen Ergänzungen der UICC aus den Jahren 2017 bis 2019. 8th Ed. (2020).
Robold, T. et al. Ergeben sich durch die Einführung der 8. Auflage der TNM-Klassifikation Änderungen für eine leitliniengerechte, chirurgische Therapiestrategie des Lungenkarzinoms?. Zentralblatt Chir. 145, 589–596. https://doi.org/10.1055/a-1164-7058 (2020).
McDonald, M.-L.N. et al. Quantitative computed tomography measures of pectoralis muscle area and disease severity in chronic obstructive pulmonary disease. A cross-sectional study. Ann. Am. Thoracic Soc. 11, 326–334. https://doi.org/10.1513/AnnalsATS.201307-229OC (2014).
Marquardt, J. P. et al. Percentile-based averaging and skeletal muscle gauge improve body composition analysis: Validation at multiple vertebral levels. J. Cachexia Sarcopenia Muscle 13, 190–202. https://doi.org/10.1002/jcsm.12848 (2022).
Troschel, F. M. et al. Preoperative thoracic muscle area on computed tomography predicts long-term survival following pneumonectomy for lung cancer. Interactive Cardiovasc. Thoracic Surg. 28, 542–549. https://doi.org/10.1093/icvts/ivy280 (2019).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2022).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, 2016).
Fernandez, F. G. et al. The Society of Thoracic Surgeons lung cancer resection risk model: Higher quality data and superior outcomes. Ann. Thorac. Surg. 102, 370–377. https://doi.org/10.1016/j.athoracsur.2016.02.098 (2016).
Onaitis, M. W. et al. Prediction of long-term survival after lung cancer surgery for elderly patients in the Society of Thoracic Surgeons General Thoracic Surgery Database. Ann. Thorac. Surg. 105, 309–316. https://doi.org/10.1016/j.athoracsur.2017.06.071 (2018).
Peduzzi, P., Concato, J., Kemper, E., Holford, T. R. & Feinstein, A. R. A simulation study of the number of events per variable in logistic regression analysis. J. Clin. Epidemiol. 49, 1373–1379. https://doi.org/10.1016/S0895-4356(96)00236-3 (1996).
Marquardt, J.P. Percentiles: Calculate (Stratified) Percentiles (2021).
Troschel, F. M. et al. Sarcopenia on preoperative chest computed tomography predicts cancer-specific and all-cause mortality following pneumonectomy for lung cancer: A multicenter analysis. Cancer Med. 10, 6677–6686. https://doi.org/10.1002/cam4.4207 (2021).
Yang, M., Shen, Y., Tan, L. & Li, W. Prognostic value of sarcopenia in lung cancer: A systematic review and meta-analysis. Chest https://doi.org/10.1016/j.chest.2019.04.115 (2019).
Collins, J., Noble, S., Chester, J., Coles, B. & Byrne, A. The assessment and impact of sarcopenia in lung cancer: A systematic literature review. BMJ Open 4, e003697. https://doi.org/10.1136/bmjopen-2013-003697 (2014).
Baracos, V. E., Reiman, T., Mourtzakis, M., Gioulbasanis, I. & Antoun, S. Body composition in patients with non-small cell lung cancer: A contemporary view of cancer cachexia with the use of computed tomography image analysis. Am. J. Clin. Nutr. 91, 1133S-1137S. https://doi.org/10.3945/ajcn.2010.28608C (2010).
Mitchell, W. K. et al. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; A quantitative review. Front. Physiol. 3, 260. https://doi.org/10.3389/fphys.2012.00260 (2012).
Blüher, M. Obesity: Global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 15, 288–298. https://doi.org/10.1038/s41574-019-0176-8 (2019).
Fielding, R. A. et al. Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 12, 249–256. https://doi.org/10.1016/j.jamda.2011.01.003 (2011).
Muscaritoli, M. et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: Joint document elaborated by Special Interest Groups (SIG) “cachexia–anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin. Nutr. (Edinburgh, Scotland) 29, 154–159. https://doi.org/10.1016/j.clnu.2009.12.004 (2010).
Edvardsen, E. et al. High-intensity training following lung cancer surgery: A randomised controlled trial. Thorax 70, 244–250. https://doi.org/10.1136/thoraxjnl-2014-205944 (2015).
Dieli-Conwright, C. M. et al. Effects of aerobic and resistance exercise on metabolic syndrome, sarcopenic obesity, and circulating biomarkers in overweight or obese survivors of breast cancer: A randomized controlled trial. J. Clin. Oncol. 36, 875–883. https://doi.org/10.1200/JCO.2017.75.7526 (2018).
Rondanelli, M. et al. Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am. J. Clin. Nutr. 103, 830–840. https://doi.org/10.3945/ajcn.115.113357 (2016).
Silva, R. et al. Mediterranean diet and musculoskeletal-functional outcomes in community-dwelling older people: A systematic review and meta-analysis. J. Nutr. Health Aging 22, 655–663. https://doi.org/10.1007/s12603-017-0993-1 (2018).
Fuchs, G. et al. Quantifying the effect of slice thickness, intravenous contrast and tube current on muscle segmentation: Implications for body composition analysis. Eur. Radiol. 28, 2455–2463. https://doi.org/10.1007/s00330-017-5191-3 (2018).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
T.R. and C.N. designed the research. S.K., C.S. and N.Z. ascertained and curated the data. S.K. performed data analysis and interpreted results. F.Z. provided statistical support. S.K. and C.N. wrote the original draft. J.G. and C.G. edited the manuscript. H.S.H. and C.S. supervised the project. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kaltenhauser, S., Niessen, C., Zeman, F. et al. Diagnosis of sarcopenia on thoracic computed tomography and its association with postoperative survival after anatomic lung cancer resection. Sci Rep 13, 18450 (2023). https://doi.org/10.1038/s41598-023-45583-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-45583-5
- Springer Nature Limited