Abstract
Our study aimed to explore the association between serum C-reactive protein (CRP) and COVID‐19 mortality. This is a retrospective cohort study of all patients admitted to 4 hospitals within the Montefiore Health System between March 1 and April 16, 2020, with SARS-CoV-2 infection. All-cause mortality were collected in 7 May 2020. The mortality risk was estimated using Cox proportional hazards models. Of the 3545 patients with a median age of 63.7 years, 918 (25.9%) died within the time of cohort data collection after admission. When the CRP was < 15.6 mg/L, the mortality rate increased with an adjusted HR of 1.57 (95% CI 1.30–1.91, P < 0.0001) for every 10 mg/L increment in the CRP. When the CRP was ≥ 15.6 mg/L, the mortality rate increased with an adjusted HR of 1.11 (95% CI 0.99–1.24, P = 0.0819) for every 10 mg/L increment in the CRP. For patients with COVID‐19, the association between the CRP and the mortality risk was curve and had a saturation effect. When the CRP was small, the mortality rate increased significantly with the increase of CRP. When CRP > 15.6 mg/L, with the increase of CRP, the mortality rate increases relatively flat.
Similar content being viewed by others
Introduction
At the end of 2019, Global health is being challenged in an unprecedented way by the 2019 coronavirus disease (COVID-19) pandemic. The systemic inflammatory response to the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection is a hallmark of COVID-19, and most COVID-19 patients have abnormal inflammatory biomarkers1. C-reactive protein (CRP), an acute phase protein first described by Tillet and Francis, is synthesized by the liver in response to interleukin 6 (IL-6) and is a widely used biomarker of inflammation2. Elevated CRP concentrations are associated with cardiovascular disease and acute kidney injury3, and with inflammatory rheumatic diseases such as rheumatoid arthritis and gout4. CRP is also related to the severity of H1N1 influenza pneumonia patients5. Besides, most studies in recent years have pointed out that for COVID-19 patients, higher CRP concentrations are closely related to higher mortality risk6,7,8,9,10,11,12. However, some studies have pointed out that CRP may play a protective role in alveolitis, and in patients with acute lung injury, an increase in CRP levels is associated with a decrease in mortality13. Therefore, it is still controversial whether the increase of CRP in the early stage of COVID-19 can be used as a prognostic indicator. Moreover, most studies had small sample sizes and did not consider the curvilinear relationship between CRP and mortality. The aim of this retrospective cohort study was to determine whether elevated levels of CRP measured in the early stages of COVID-19 were associated with higher mortality in COVID-19 patients.
Methods
This is a retrospective cohort study of all patients admitted to 4 hospitals within the Montefiore Health System between March 1 and April 16, 2020, with SARS-CoV-2 infection. Information on demographics, comorbidities, admission laboratory values, discharge, and mortality was identifified through a health care surveillance software package (Clinical Looking Glass; Streamline Health, Atlanta, GA) and review of the primary medical records. The design details in this study have been previously reported14.
Data source
We downloaded the raw data free of charge from the DATADRYAD database provided by Eskandar et al. Source: Altschul, David (2021), Neurologic complications of COVID-19, Dryad, Dataset, <https://doi.org/10.5061/dryad.7d7wm37sz>. Under Dryad's Terms of Service, researchers can use this data for secondary analysis without infringing on the authors' rights.
Study population
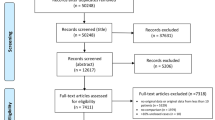
All patients with real-time reverse transcriptase PCR positive assay testing for SARS-CoV-2 RNA were included.For patients with multiple admissions, only the last reported was considered for analysis.Data were collected on May 7, 2020.Exclusion criteria were as follows: (1) patients who were not admitted or died before admission were excluded because they rarely had a complete laboratory study panel and a complete neurological evaluation could not be assessed, (2) CRP = 0 mg/L or Patients with CRP ≥ 100 mg/L. The flow chart of the study is shown in Fig. 1.
Variables
The variable for this study was C-reactive protein, and the covariate was chosen based on our clinical experience, original studies, and other studies investigating risk factors for progression in COVID-19 patients. Based on the above principles, the following variables are therefore used as covariates: (1) Continuous variables: age, D-Dimer, temperature, oxygen saturation, mean arterial pressure, platelets, INR, BUN, creatinine, sodium, glucose, AST, WBC, ALT, lymphocytes, interleukin-6, ferritin, procalcitonin, troponin; (2) Categorical variables: ethnicity, myocardial infarction, peripheral vascular disease, congestive heart failure, cerebrovascular disease, dementia, chronic obstructive pulmonary disease, diabetes mellitus simple, renal disease, stroke.
Outcomes
Te outcome of the study was all-cause mortality.
Statistical analysis
Continuous variables were described as the means ± SD or median and interquartile ranges (IQR). Categorical data were presented as numbers and percentages. The diference according to the tertiles of the CRP was compared using one-way analysis of variance (ANOVA) for continuous data and chi-squared tests for categorical variables.
Calculations for Kaplan Meier survival were performed using Kaplan Meier analysis, and a Kaplan Meier curve was developed as a result.
We used a generalized additive model (GAM) to investigate the dose–response relationship between the CRP and mortality (Fig. 2). Adjusted for age, D-Dimer, temperature, oxygen saturation, mean arterial pressure, platelets, INR, BUN, creatinine, sodium, glucose, AST, WBC, ALT, lymphocytes, interleukin-6, ferritin, procalcitonin, troponin, ethnicity, myocardial infarction, peripheral vascular disease, congestive heart failure, cerebrovascular disease, dementia, chronic obstructive pulmonary disease, diabetes mellitus simple, renal disease, stroke. Estimating the hazard ratio (HR) of death was done using Cox proportional hazards modeling .
We then used a two-piece-wise linear regression model to examine the threshold effect of the CRP on mortality (Table 3). The turning point for the CRP was determined using “exploratory” analyses, which is to move the trial turning point along the predefned interval and pick up the one which gave maximum model likelihood. We also performed a log-likelihood ratio test and compared the one-line linear regression model with the two-piece-wise linear model. All the statistical analyses were performed using the EmpowerStats (www.empowerstats.com, X&Y solutions, Inc. Boston MA) and R software version 3.6.1 (http://www.r-project.org)15.
Ethics approval and consent to participate
The study received approval from Albert Einstein College of Medicine, Montefiore Medical Center ethical standards committee on human experimentation. Written informed consent was waived by the Albert Einstein College of Medicine, Montefiore Medical Center ethical standards committee given the retrospective design of the study. In addition to this, Methods of the study were performed in accordance with the relevant guidelines and regulations.
Results
Baseline characteristics
Data from 3545 patients were analyzed. The median age is 63.7 years (IQR54-76 years). Table 1 compares patient’s demographics, vital signs, laboratory results, and complications by tertile of CRP. Subjects in the highest tertile of CRP were older and had lower Oxygensaturation on admission than those in the lowest tertile of CRP (Table 1).
Associations between the CRP and mortality in all patients with Covid-19. A threshold, nonlinear association between the CRP and mortality was found in a generalized additive model (GAM). Red rad line represents the smooth curve fit between variables. Blue bands represent the 95% of confdence interval from the fit. Adjusted for age, D-Dimer, temperature, oxygen saturation, mean arterial pressure, platelets, INR, BUN, creatinine, sodium, glucose, AST, WBC, ALT, lymphocytes, interleukin-6, ferritin, procalcitonin, troponin, ethnicity, myocardial infarction, peripheral vascular disease, congestive heart failure, cerebrovascular disease, dementia, chronic obstructive pulmonary disease, diabetes mellitus simple, renal disease, stroke.
Mortality
The mortality rate was 918/3545 = 25.90% in our cohort. The mortality rate from the lowest tertile (0.50–5.50) to the highest (15.00–68.40) CRP was 141 (11.94%), 286 (24.32%), and 491 (41.33%) (Table 1).
Unadjusted association between baseline variables and mortality
In the univariate COX regression analysis model, CRP, age, D-Dimer, oxygen saturation, mean arterial pressure, BUN, creatinine, sodium, glucose, AST, WBC, lymphocytes, interleukin-6 and Procalcitonin are all correlated with mortality, and have statistical significance, as shown in Table 2.
In addition, we can intuitively see from that the higher the CRP, the lower the survival rate. See Fig. 3 for details.
Identifcation of nonlinear relationship
We observed a nonlinear dose–response relationship between the CRP and mortality (Fig. 2 and Table 3). When the CRP was < 15.6 mg/L, the mortality rate increased with an adjusted HR of 1.57 (95% CI 1.30–1.91, P < 0.0001) for every 10 mg/L increment in the CRP. When the CRP was ≥ 15.6 mg/L, the mortality rate increased with an adjusted HR of 1.11 (95% CI 0.99–1.24, P = 0.0819) for every 10 mg/L increment in the CRP (Table 3).
Using the generalized additive model, an inflection point between CRP and mortality was detected (Table 3). The linear regression model and a two-piece-wise linear regression model were compared, and the P value of the log-likelihood ratio test was 0.007. This result indicates that the two-piece-wise linear regression model should be used to fit the model.
Discussion
This retrospective cohort study found a curved relationship and saturation effect between CRP and the mortality risk in 3545 patients infected with SARS-CoV-2 admitted to four hospitals within the Montefiore health system between March 1 and April 16, 2020. When the CRP was small, the mortality rate increased significantly with the increase of CRP. When CRP > 15.6 mg/L, with the increase of CRP, the mortality rate increases relatively flat. Few studies have shown a curvy relationship between CRP and mortality in patients with COVID-19.
C-reactive protein is recognized as a marker of systemic inflammation and severe infection. As an acute phase reactant, CRP binds to phosphorylcholine in pathogen and host cell membranes and acts as an opsonin to enhance phagocytosis and facilitate clearance. Ligand-bound C-reactive protein also efficiently activates the classical pathway of the complement system, an essential component of natural host defense16. Prior to the COVID-19 global pandemic, up to 90% of significantly elevated C-reactive protein concentrations were attributable to infectious pathogens, most commonly bacterial pathogens17. Elevated C-reactive protein concentrations have also been reported in severe viral infections, including H1N1 influenza pneumonia and now SARS-CoV-2 infection5,18.In a previous study of 95 COVID-19 patients, CRP For every 10 mg/L increase, the patient's risk of death increased by 11%, and C-reactive protein concentration was associated with mortality9. Smilowitz et al. reported that patients with COVID-19 who had high levels of C-reactive protein on admission were associated with higher mortality, and that patients with both high D-dimer and CRP had a greater risk of death8. Similarly, among 281 hypertensive COVID-19 patients hospitalized with COVID-19, those who died had higher levels of C-reactive protein compared with survivors19. Although the studies collected patients over different time periods, the trends were similar. In addition, the high mortality rate of COVID-19 is mainly related to the poor control of the inflammatory cascade and the subsequent acute respiratory distress syndrome (ARDS)20. However, some studies have pointed out that elevated plasma CRP levels in patients with acute respiratory distress syndrome are associated with reduced mortality, which seems to be contrary to the established views of previous scholars13. Therefore, the correlation between CRP and the prognosis of COVID-19 infection is controversial, and most of the previous similar studies had small sample sizes, and rarely analyzed the curve fitting relationship between CRP and the mortality rate of COVID-19 patients. This is exactly the problem that this study strives to solve.
C-reactive protein concentration reflects the degree of acute inflammatory response. The role of systemic inflammation in the pathogenesis of COVID-19 is still not fully understood. A causal relationship between the inflammatory response measured by CRP and poor prognosis remains speculative. However, the deleterious inflammatory response observed in some patients with COVID-19 is similar to macrophage activation syndrome and may independently lead to multiple organ damage in COVID-1921,22. Studies have shown that the application of glucocorticoids Decreases C-reactive protein concentrations in patients with ARDS23. A recent randomized trial of immunosuppression noted that hormone therapy reduced mortality in critically ill COVID-19 patients. The clinical benefit of immunosuppression further supports the hypothesis that inflammation from viral infection contributes to adverse outcomes in COVID-1924. In addition, thrombotic inflammation has been implicated as a mediator of adverse events in COVID-19. In response to inflammatory stress, thrombus inflammation dysregulates the normal antithrombotic function of the endothelium, leading to leukocyte recruitment, complement and platelet activation, and enhanced microvascular coagulation25,26. This is supported by autopsy studies of COVID-19 patients. In these studies, platelets with fibrin microthrombi were found in the microcirculation of the lung, kidney, liver, and heart27. Among them, pulmonary embolism has a high mortality rate and is very common in new coronary pneumonia. Therefore, the risk of poor prognosis associated with SARS-CoV-2 infection increases when patients experience concomitant elevated CRP concentrations. However, what we did not expect is that there is a saturation effect between CRP and mortality. So far, the author has not found relevant literature to explain this phenomenon.
Among COVID-19 patients, higher C-reactive protein levels were associated with higher mortality risk with a saturation effect. This finding has interesting potential clinical and research implications. To the best of our knowledge, there are few clear references to the curvilinear relationship between CRP and mortality risk in the early stages of COVID-19. Compared with the linear regression relationship, the curve relationship in this study can more scientifically and accurately reflect the judgment value of CRP level on the mortality risk of patients with COVID-19.
Study limitations
A common problem in observational studies is unmeasured confounders. As shown in Table 1, subjects in the highest tertile of CRP were older and had lower Oxygensaturation compared with subjects in the lowest tertile of CRP. These differences may be indicative of unmeasured confounders, such as income and health insurance, which may affect the mortality risk. Although race, length of hospital stay, and age were included in the data set, the effect of unmeasured confounders on HR could not be estimated. In addition, the first measured CRP concentration on admission was used for the primary analysis. The admission physician's knowledge of the initial CRP concentration may have altered subsequent patient care and may have confounded the results. Although there are no data on steroid use, few patients may have received steroids prior to the initial CRP measurement during hospitalization. In some cases, steroids or the IL-6 inhibitor tocilizumab are used in critically ill patients. However, the timing of administration, duration of use, and dosage of these drugs were not recorded in this dataset.
In addition, the lack of information on interventions during the initial stabilization period may have influenced the relationship between CRP levels and survival. It is noteworthy that the potential of the intervention would be biased towards reducing mortality, leading to an underestimation of the association between CRP levels and mortality, but this reinforces the positive findings of this study.
Conclusions
The study identified a nonlinear dose–response relationship between CRP and mortality. For patients with COVID‐19, the association between the CRP and the mortality risk was curve and had a saturation effect. When the CRP was small, the mortality rate increased significantly with the increase of CRP. When CRP > 15.6 mg/L, with the increase of CRP, the mortality rate increases relatively flat.
Data availability
The datasets generated and/or analysed during the current study are available in the DATADRYAD repository, https://doi.org/10.5061/dryad.7d7wm37sz.
References
Petrilli, C. M. et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: Prospective cohort study. BMJ 369, m1966 (2020).
Tillett, W. S. & Francis, T. Serological reactions in pneumonia with a non-protein somatic fraction of pneumococcus. J. Exp. Med. 52, 561–571 (1930).
Murashima, M. et al. Inflammation as a predictor of acute kidney injury and mediator of higher mortality after acute kidney injury in non-cardiac surgery. Sci Rep. 9, 20260 (2019).
Folsom, A. R., Lutsey, P. L., Astor, B. C. & Cushman, M. C-Reactive protein and venous thromboembolism: A prospective investigation in the ARIC Cohort. Thromb Haemost. 102, 615–619 (2009).
Vasileva, D. & Badawi, A. C-reactive protein as a biomarker of severe H1N1 influenza. Inflamm. Res. 68, 39–46 (2019).
Bouayed, M. Z. et al. C-reactive protein (CRP): A poor prognostic biomarker in COVID-19. Front Immunol. 13, 1040024 (2022).
Ali, A. et al. Myoglobin and C-reactive protein are efficient and reliable early predictors of COVID-19 associated mortality. Sci Rep. 11, 5975 (2021).
Smilowitz, N. R. et al. C-reactive protein and clinical outcomes in patients with COVID-19. Eur. Heart J. 42, 2270–2279 (2021).
Villoteau, A. et al. Elevated C-reactive protein in early COVID-19 predicts worse survival among hospitalized geriatric patients. PLoS One. 16, e256931 (2021).
Sharifpour, M. et al. C-reactive protein as a prognostic indicator in hospitalized patients with COVID-19. PLoS One. 15, e242400 (2020).
Acar, E., Demir, A., Yildirim, B., Kaya, M. G. & Gokcek, K. The role of hemogram parameters and C-reactive protein in predicting mortality in COVID-19 infection. Int. J. Clin. Pract. 75, e14256 (2021).
Ozdemir, I. H. et al. Prognostic value of c-reactive protein/albumin ratio in hypertensive COVID-19 patients. Clin. Exp. Hypertens. 43, 683–689 (2021).
Bajwa, E. K. et al. Plasma C-reactive protein levels are associated with improved outcome in ARDS. Chest. 136, 471–480 (2009).
Eskandar, E. N. Neurologic syndromes predict higher in-hospital mortality in COVID-19. Neurology. 96, 551 (2021).
Chang, L., Chen, X. & Lian, C. The association between the non-HDL-cholesterol to HDL-cholesterol ratio and 28-day mortality in sepsis patients: A cohort study. Sci Rep. 12, 3476 (2022).
Volanakis, J. E. Human C-reactive protein: Expression, structure, and function. Mol. Immunol. 38, 189–197 (2001).
Landry, A., Docherty, P., Ouellette, S. & Cartier, L. J. Causes and outcomes of markedly elevated C-reactive protein levels. Can. Fam. Physician. 63, e316–e323 (2017).
Gao, Y. et al. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19. J. Med. Virol. 92, 791–796 (2020).
Özdemir, İH. et al. Prognostic value of c-reactive protein/albumin ratio in hypertensive COVID-19 patients. Clin. Exp. Hypertens. 43, 683–689 (2021).
Wu, C. et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan. China. JAMA Intern. Med. 180, 934–943 (2020).
Merad, M. & Martin, J. C. Pathological inflammation in patients with COVID-19: A key role for monocytes and macrophages. Nat. Rev. Immunol. 20, 355–362 (2020).
Mehta, P. et al. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet. 395, 1033–1034 (2020).
Meduri, G. U. et al. Methylprednisolone Infusion in Early Severe ARDS: Results of a Randomized Controlled Trial. Chest. 131, 954–963 (2007).
Horby, P. et al. Dexamethasone in hospitalized patients with Covid-19. Massachusetts Medical Society. (2020).
Connors, J. M. & Levy, J. H. Thromboinflammation and the Hypercoagulability of COVID-19. J. Thromb. Haemost. 18, 1559–1561 (2020).
Jackson, S. P., Darbousset, R. & Schoenwaelder, S. M. Thromboinflammation: Challenges of therapeutically targeting coagulation and other host defense mechanisms. Blood. 133, 906–918 (2019).
Rapkiewicz, A. V. et al. Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: A case series. EClinicalMedicine. 24, 100434 (2020).
Acknowledgements
The author is very grateful to the data providers of the study.
Funding
This project was funded by National Natural Science Foundation of China (No. 82202789), The Science, Technology and Innovation Commission of Shenzhen (JCYJ20220530165211026), The Clinical Research Project of Shenzhen Dapeng New District Medicine and Health Group (No.2022JTLCYJ06) and The Yu Miao Project of Shenzhen Dapeng New District Medicine and Health Group (No.2021JTYM04).
Author information
Authors and Affiliations
Contributions
M.C.Z. and M.J.H performed statistical analysis. Y.J.Z. Z.J.L. and Y.Y.L. cleaned the data. Y.W. and F.L. conceived and designed the research. F.L. drafed the manuscript. Y.L.W. and Y.W. made critical revision of the manuscript for key intellectual content.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, F., He, M., Zhou, M. et al. Association of C-reactive protein with mortality in Covid-19 patients: a secondary analysis of a cohort study. Sci Rep 13, 20361 (2023). https://doi.org/10.1038/s41598-023-47680-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-47680-x
- Springer Nature Limited