Abstract
Triple negative breast cancer (TNBC) is highly immunogenic and high levels of tumor infiltrating lymphocytes (TILs) have been associated with a better prognosis and higher probability to achieve pathological complete response. Here, we explore the potential role of stromal TILs level and composition as a prognostic and predictive biomarker in TNBC. 195 Tumor biospecimens from patients diagnosed with TNBC were included. Stromal TILs (sTILs), positive CD4/CD8 cells were evaluated. Differences in clinic-pathological characteristics according to immune infiltration were assessed. The predictive and prognostic value of immune infiltration was analyzed by multivariate models. Higher immune infiltration was observed in patients with favorable clinical–pathological features. Survival analysis showed that longer overall survival times were observed in patients with a higher infiltration of sTILs (p = 0.00043), CD4 + (p = 0.0074) and CD8 + (p = 0.008). In the multivariate analysis, low levels of sTILs were found to be associated with a higher mortality hazard (HR: 1.59, 95% CI 1.01–2.48). CD4 and CD8 immune infiltration were associated with higher odds for pathological complete response (OR: 1.20, 95% CI 1.00–1.46, OR: 1.28, 1.02–1.65, respectively). Our results suggest that immune infiltration could be used as a prognostic marker for overall survival in TNBC patients.
Similar content being viewed by others
Introduction
Triple-negative breast cancer (TNBC) is characterized by the lack of expression of estrogen receptor (ER), progesterone receptor (PR), and the epidermal growth factor receptor 2 (HER2)1. Its prevalence ranges between 15 and 25%, although it has been reported to occur more frequently among young (< 50 years old) Non-Hispanic Black (NHB) and Latina women2,3,4. It is considered the most aggressive breast cancer subtype characterized by earlier relapse and worse survival compared to other breast cancer subtypes1,5,6.
Unlike hormone receptor (HR) positive and HER2-positive tumors that highly benefit from endocrine and targeted therapies, TNBC has limited therapeutic options and until recently, cytotoxic chemotherapy was the only systemic therapy approved7. The molecular heterogeneity of TNBC has been described by the presence of distinct molecular subtypes, each with different prognosis and possible molecular targets but these data has not changed its clinical management8,9,10,11.
TNBC tumors are known to be the most immunogenic subtype with relatively high levels of TILs when compared to HR-positive subtypes12,13,14. The presence of high TILs, especially in the tumor proximity, has been considered a surrogate for immune response and has emerged as an important immunological biomarker in breast cancer8,15. Recent data have shown TIL density to be both a positive prognostic marker for disease-free and overall survival16,17,18,19 and a predictive marker for pathologic complete response (pCR) to neoadjuvant chemotherapy8,20,21. This beneficial effect might be related to the role of immune cells who are able to identify tumor neoantigens and enhance the adaptive immune response to induce tumoral death22,23. Nonetheless, the physiological impact these cells exert on the tumor vary according to their subclassification17,24. In this study we sought to evaluate the differences in clinical–pathological variables, clinical outcomes and pCR according to TILs composition and levels to better understand its prognostic and predictive value in Colombian patients.
Methods
Patient selection
We conducted a nationwide, multicenter retrospective study series of 195 TNBC patients diagnosed at three health institutions between 2008 and 2016, including the largest reference cancer center in the country, the Colombian National Cancer Institute (NCI) in Bogotá D.C, as well as Fundación Valle de Lili (FVL) in Cali, and Clínica las Américas (CLA) in Medellin. The inclusion criteria were as followed 1) histologically confirmed diagnosis of primary TNBC, 2) availability of formalin-fixed paraffin-embedded (FFPE) tissue blocks from pre-treatment tissue from biopsies and/or surgery (i.e., mastectomies or quadrantectomy for patients that did not receive neoadjuvant chemotherapy) with at least 10% of invasive carcinoma, and 3) availability of clinical–pathological data from the medical records.
TNBC was defined by the lack of ER and PR reactivity (< 1%), and a negative HER2 score (0 + , 1 + , or 2 + with a confirmatory negative result from an in situ hybridization technique), according to the latest American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines25. Hormone receptors and HER2 expression were reviewed from medical records and re-analyzed by a single pathologist to confirm the diagnosis.
Pathology reports were reviewed to obtain information regarding histopathological diagnosis, lymph node involvement, histological grade, invasion (lymphovascular and/or perineural), and surgical margins. Demographic information, including place of birth, region of origin, as well as clinical data such as age of diagnosis, body mass index (BMI), tumor size, AJCC clinical stage, treatment protocols (neoadjuvant and/or adjuvant treatments), the presence of recurrence and the vital state, were extracted from clinical records at each institution. Pathological complete response (pCR) was evaluated in the cases that had available slides from the surgical procedure following the Chevallier criteria26. The study was conducted in accordance with the tenets of the Declaration of Helsinki and was approved by research ethics committee from Colombian National Cancer Institute (approval number: INTOFI049562018), and each site, and the patients provided written informed consent.
Immunohistochemistry and TILs assessment
TILs evaluation was performed by a single-blinded pathologist. Stromal TILs evaluation was performed on single full-face hematoxylin and eosin (H&E) slide from a pre-treatment tissue (biopsy or treatment free surgery product) following the International TILs Working Group 2014 guidelines27. For each case a single FFPE tissue block with the highest tumoral content was analyzed. sTILs was defined as the percentage of tumoral stromal area that was occupied by mononuclear immunological infiltrate. sTILs was analyzed as a continuous variable and categorized into two groups: high-sTILs (> 10%) and as low-sTILs (≤ 10%). This cut-off point was selected following recommendations from previous studies21,27,28 and were defined before statistical analysis.
TILs subpopulations were assessed on 3 µm thick sections from the same pre-treatment FFPE block selected for sTILs evaluation. Monoclonal antibodies for CD4 (clone SP35, Ventana Medical System) and CD8 (clone SP57, Ventana Medical System) were analyzed in a Roche Benchmark XT automated slide preparation system (Roche Ltd., Switzerland). Positive and negative controls were included and 3,3′ diaminobenzidine (DAB) was used as the chromogen. For each sample, three tumoral areas with the highest immune infiltration were selected and digitalized by a microscopy Olympus EP50 camera at an × 400 magnification (× 40 objective). Using the plugin cell counter from the Image J program, the number of stromal and intratumoral CD4 + T and CD8 + T cells were counted within the three-chosen fields and averaged to obtain the mean score for each sample. TILs subpopulations were analyzed as continuous variables and categorized into two groups using as a cut-off value the median to define groups of high or low infiltration (supplementary Fig. 1).
Statistical analysis
Statistical analyses were performed with R-studio (version 4.2.1). Chi-squared and Fisher’s exact test were used to evaluate differences in clinical–pathological characteristics according to sTILs infiltration (high: > 10% vs. low ≤ 10%), along with TILs subpopulations, CD4 + T (high: > 101.33 vs. low: ≤ 101.33, supplementary Fig. 1a) and CD8 + T (high: > 105 vs. low: ≤ 105, supplementary Fig. 1b). Moreover, differences in sTILs infiltration, CD4 + T and CD8 + T subpopulations, by pCR status (pCR vs. no-pCR) were assessed using the Mann–Whitney and T-student test. All statistical tests were two sided and considered significant when p ≤ 0.05. Univariate and multivariate binary logistic regression models were used to assess the association of pCR and lymphocytic infiltration. Regarding Odds ratio (OR), we analyzed different increments, as a result was chosen, the increment that better fit to prediction model, per every 10% increase in sTILs, and per every 30 cells increase in CD4 + T and CD8 + T.
Two survival endpoints were evaluated: (1) overall survival (OS) defined as the time interval between diagnosis and death from any cause or last follow-up; (2) Disease-free survival (DFS) defined as the time interval between surgery and the date of recurrence of breast cancer (local, regional, or distant) or last follow up. Differences (OS) and DFS were assessed between the high and low sTILs, CD4 + T, and CD8 + T infiltration groups, using the Kaplan–Meier and log-rank test. A multivariate Cox regression analysis was performed including the following variables: pretreatment nodal status (positive vs. negative) and tumor size (T1 (≤ 2 cm) vs. T2 (> 2 cm)). Hazard ratios (HR) and 95% confidence intervals (95% CI) were calculated for high vs low of sTILs, CD4 + T and CD8 + T.
Results
Clinical–pathological characteristics
Clinical–pathological characteristics of patients included are described in Table 1. Most of the patients were diagnosed over the age of 50 (67.7%) and 20.5% were obese at diagnosis. Additionally, most patients were diagnosed at advanced stages (III: 51.1%), presented poorly differentiated tumors (Scarff-Bloom Richardson III: 85.9%), lymph node involvement (53.8%), and had larger tumors (> 2 cm: 80.9%). Concordantly, more than half of the patients underwent neoadjuvant chemotherapy (59.7%), and among this group, 24.7% achieved pCR. The main surgical treatment approach was the modified radical mastectomy (MRM) (53.0%). At the end of the study, 38.9% of the patients had disease recurrence and 53.8% had died.
Clinical–pathological characteristics by lymphocytic infiltration
For this study, we focused on sTILs as they were predominant in our cases and were highly correlated with the number of intratumoral TILs (iTILs) that were present at a lower density (data not shown). High sTILs levels were observed in 44% of the patients. Regarding TILs subpopulations, a high infiltration of CD4 + and CD8 + cells was observed in 50% of the cases for each marker.
We evaluated TNBC patients’ clinical and pathological characteristics according to sTILs infiltration, and by CD4 + T and CD8 + T subpopulations (Table 2). Patients with clinical stage IV were excluded from these analyses. CD4 and CD8 subpopulations were analyzed in a subset of 178 patients due to FFPE tissue depletion. We observed that patients with less than 10% sTILs presented higher clinical stages (III: 63.5% vs. I/II: 36.5%, p < 0.001), larger tumors (> 2 cm: 89.5% vs. < 2 cm: 10.5%, p = 0.003), and were positive for lymph-node involvement at diagnosis (63.1% vs. negative: 36.9%, p = 0.007), compared to patients with more than 10% of sTILs. Similarly, a higher percentage of patients with low sTILs infiltration received neoadjuvant therapy (67.3% vs. no neoadjuvant therapy: 32.7%, p = 0.025), of whom 15.8% achieved a pCR, compared to the high sTILs group where 42.9% achieved a pCR (p = 0.023). In the same line, more than half of the patients with low sTILs infiltration were already deceased at the end of the study (65.4% vs. alive: 34.6%, p = 0.001). Similar results were observed regarding CD4 + T and CD8 + T cells. Patients with lower CD4 and CD8 counts also presented more advanced clinical stages, larger tumors (> 2 cm), and with a higher frequency lymph-node involvement at diagnosis compared to patients with high CD4 + T and CD8 + T counts (Table 2). Furthermore, patients in the low CD4 + T and CD8 + T infiltration groups were also treated more frequently with neoadjuvant therapy and MRM, compared to high CD4 + T and CD8 + T infiltration groups. Interestingly, only patients with high CD4 + T counts seemed to respond significantly better to neoadjuvant treatment (pCR, high: 43.3% vs. low: 16.3%, p = 0.006). Only variables that showed statistically significant differences between evaluated groups were included in Table 2.
sTILs immune infiltration and pathological complete response
We compared sTILs, CD4 + T and CD8 + T infiltration levels measured as a continuous variable according to neoadjuvant treatment response, using pCR as the defining variable (Fig. 1). Information regarding neoadjuvant chemotherapy administered to these patients are included in supplementary Table 1. We consistently observed that patients that successfully achieved pCR presented higher sTILs levels (p = 0.0076, Fig. 1a), as well as CD4 + T (p = 0.012, Fig. 1b) and CD8 + T (p = 0.019, Fig. 1c) infiltration, compared to patients that did not achieve pCR.
Moreover, univariate models showed a statistically significant association between pCR and sTILs (OR = 1.48, 95% CI 1.14–2.01, p = 0.005) and CD4 and CD8 immune infiltration (CD4 + T: OR = 1.26, 95% CI 1.06–1.53, p = 0.012; CD8 + T: OR = 1.33, 95% CI 1.08–1.69, p = 0.0094). Consistently, after adjusting the model for lymph-node involvement and tumor size, for sTILs and CD4, the association previously observed was attenuated (sTILs: OR = 1.31, 95% CI 0.984–1.806, p = 0.0762; CD4: OR = 1.201, 95% CI 1.003–1.461, p = 0.0568) but remained statistically significant for CD8 immune infiltration (OR = 1.277, 95% CI 1.017–1.647, p = 0.0433) (Table 3).
Prognostic value of immune infiltration in TNBC
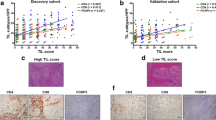
Differences in OS and DFS between sTILs, CD4 + T and CD8 + T infiltration groups were analyzed. We observed that patients with low immune infiltration present significantly lower OS and DFS median times compared to TNBC patients with high immune infiltration (Fig. 2).
When sTILs levels was analyzed as a categorical variable, we observed that having less than 10% of sTILs, or lower CD4 + T (< 101.3) and CD8 + T (< 105) cell counts was significantly associated with a higher risk of mortality (HR = 2.08, 95% CI 1.35–3.20, p < 0.001; HR = 1.85, 95% CI 1.19–2.89, p = 0.007; HR = 1.79, 95% CI 1.16–2.77, p = 0.008, respectively) and recurrence (HR = 1.78, 95% CI 1.08–2.92, p = 0.023; HR = 1.73, 95% CI 1.05–2.87, p = 0.033; HR = 1.77, 95% CI 1.07–2.92, p = 0.027, respectively). In a model adjusted by tumor size and lymph-node involvement, we found that having a low sTILs infiltration is an independent prognostic factor for mortality (HR = 1.59, 95% CI 1.01–2.48, p = 0.043) (Table 4). Similar results were observed for OS when the model was adjusted by clinical stage (Supplementary Table 2), but there was no association when the model was stratified by clinical (Supplementary Table 3).
Discussion
There is growing interest in analyzing TILs and immune subpopulations in the clinical practice to explore their potential as prognostic and predictive biomarker in a highly aggressive subtype such as TNBC. As has been reported before, tumors with high immune infiltration often present better clinical outcomes and favorable clinical–pathological features20,23,29,30. In this study, we analyzed a Colombian cohort of patients with TNBC with a long-term follow-up to explore the immune infiltration and its association with patient outcome and pCR achievement and found sTILs as a robust and independent prognostic marker for mortality and an association with pCR in TNBCs. We believe this is the first study performed in Colombian women to report this finding. This is important as the prevalence of TNBC in our population is higher than the prevalence reported in European-American women31,32,33, and we highlight the need to better understand TNBC in our patients to improve its prognosis and to better identify patients that could benefit from immune-based treatments.
In accordance with previous studies17,21,34,35, ours showed an association between clinical–pathological features and the prognostic value of immune infiltration. We observed that patients with high levels of sTILs, CD4 + T, and CD8 + T cells are more frequently diagnosed at earlier clinical stages (I/II), with smaller tumor sizes and no lymph node involvement. Additionally high sTILs were associated with longer OS times and this association remained significant with the inclusion of known clinical variables associated with the prognosis. In that sense, we found that sTILs can be an independent prognostic factor for OS, whereas for CD4 + T and CD8 + T subpopulations, although they were found associated with prognosis in the univariate analyses, the statistical significance was attenuated in the multivariate model.
Immune subpopulations, CD4 + T and CD8 + T, represent only a general fraction of the total population of immune cells that integrate TNBC microenvironment17. In that sense, CD4 + T and CD8 + T cell counts are not fully informative and sufficient to establish the effect of immune infiltrate on the disease prognosis, although they might serve as an approximation. A more accurate characterization of the tumor immune infiltration profile should include broader information about other lymphocyte subpopulations with relevant immunological roles36.
Even though our results are consistent with the associations reported in other studies21,35, the findings around the prognostic value of TILs between studies are still controversial. It is important to take into consideration the stage at diagnosis and lymph node status at diagnosis when analyzing results from different studies as this could impact the association with the prognosis. For example, a study conducted on 133 TNBC patients at earlier clinical stages and small tumor burdens did not find TILs associated with survival outcomes37. Presumably, patients at earlier clinical stages and with well-differentiated tumors present lower amounts of tumor antigens and, as result lower immune infiltrate37,38. In our study 46.9% of the patients were at stage I/II and 49% at stage III. Moreover 86.6% of the tumors were poorly differentiated.
Higher immune infiltration before neoadjuvant chemotherapy has been significantly associated with higher pCR rates20,39,40,41. In our study population, 23.3% of the patients achieved pCR. This percentage is concordant with the rates reported in other studies where 16.6–48% of TNBC patients achieved pCR. The variability in the percentages could be related with differences in chemotherapy schemes between studies42,43,44. Regarding groups of high or low immune infiltration, we observed that patients with high immune infiltrate received less neoadjuvant chemotherapy and were mostly treated with conservative surgeries but on the other hand a higher percentage of patients with high levels of sTILs, CD4 + T, and CD8 + T achieved pCR. The degree of the sTILs antitumor immune response against cancer cells acts synergistically with natural immunity induced by chemotherapy to restore the cytotoxic response19,45 Moreover, chemotherapy can promote an antitumor immune response through the induction of danger associated molecular patterns (DAMP) signals during cell death, in addition to other molecules like the calreticulin (CALR) and the high mobility group release box 1 (HMGB1), which can increase levels of TILs in the TME as in the residual disease after treatment30,46,47. sTILs were not clearly shown to have an association with pCR, whereas CD4 + and specially CD8 + cells were found to be associated with this outcome. A possible explanation for that is that different immune populations are included when sTILs are evaluated in H&E slides. For example, Tumor associated macrophages (TAMs) within the TME contribute to evasion and suppression of the immune response and likewise this has been associated with resistance to chemotherapy48,49. It should be noted that none of the patients in this study receive immunotherapy. Further investigations are needed to keep exploring the interactions between specific immune cell populations, the tumor phenotype and treatment regimen including immunotherapy, in order to have a better understanding of their role in TNBC.
There is lack of an established standard methodology for immune infiltration assessment. Studies in the field have used different approaches for TILs evaluation to test its association with breast cancer prognosis, managing this variable either as categorical or continuous, and with undefined cut-off values27 contributing to the highly heterogenous reports around TILs in breast cancer. In 2014, the International TILs Working Group27 published a series of recommendations for TILs assessment, where they came to the consensus that TILs evaluation may provide more accurate information when scored as a continuous variable, given that it would allow a more standard categorization around different thresholds. In the present study, we analyzed TILs both ways, as continuous and categorical variable. Either way, consistent associations were observed, where better clinical–pathological features and longer OS and DFS were found for patients with higher immune infiltration.
The study has some limitations, including the heterogeneity of the specimens and the difference in sample sizes between the three health institutions involved. FFPE blocks were taken from each institution’s pathology archive, therefore, it is possible that sample quality and their general management might have varied considerably from center to center, affecting downstream analyses like the IHC. On the same line, given that we worked with biopsies which are tissue-limited specimens, it was not possible to test additional IHC immune biomarkers to assess a broader spectrum of TILs subpopulations. New methodologies based on tissue microarrays, flow cytometry, and the use of gene expression data are being developed to quantify different immune cells subpopulations50. These approaches can be implemented in future studies to improve immune infiltrate assessment and gain a better understanding of TILs effect on TNBC.
Conclusions
This is the first study in Colombian women to assess immune infiltration as prognostic and potential predictive biomarker in breast cancer patients with TNBC. The results obtained in this study suggest that patients with TNBC, high infiltration of sTILs, and CD4 + T and CD8 + T immune populations, present clinical–pathological characteristics of favorable prognosis. In addition, high levels of immune infiltration were found as an independent factor for overall survival, and a potential biomarker for pCR. However, it is still necessary to continue exploring the relationship between infiltrate immune and prognosis in a higher sample size also including patients from different Colombian regions. Moreover, it is important to evaluate more specific immune marker by different methodologies.
We want to highlight the interdisciplinary work conducted by pathologists, oncologists, molecular biologist, and other scientific professionals, which have enriched this work, contributing this way to the progress of science in Colombia.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Lin, N. U. et al. Clinicopathologic features, patterns of recurrence, and survival among women with triple-negative breast cancer in the National Comprehensive Cancer Network. Cancer 118(22), 5463–5472. https://doi.org/10.1002/CNCR.27581 (2012).
Abramson, V. G., Lehmann, B. D., Ballinger, T. J. & Pietenpol, J. A. Subtyping of triple-negative breast cancer: Implications for therapy. Cancer 121(1), 8–16. https://doi.org/10.1002/CNCR.28914 (2015).
Scott, L. C., Mobley, L. R., Kuo, T. M. & Il’yasova, D. Update on triple-negative breast cancer disparities for the United States: A population-based study from the United States Cancer Statistics database, 2010 through 2014. Cancer 125(19), 3412–3417. https://doi.org/10.1002/CNCR.32207 (2019).
Almansour, N. M. Triple-negative breast cancer: A brief review about epidemiology, risk factors, signaling pathways, treatment and role of artificial intelligence. Front. Mol. Biosci. 9, 32. https://doi.org/10.3389/FMOLB.2022.836417/BIBTEX (2022).
Li, X. et al. Triple-negative breast cancer has worse overall survival and cause-specific survival than non-triple-negative breast cancer. Breast Cancer Res. Treat. 161(2), 279–287. https://doi.org/10.1007/S10549-016-4059-6 (2016).
Ignatov, A., Eggemann, H., Burger, E. & Ignatov, T. Patterns of breast cancer relapse in accordance to biological subtype. J. Cancer Res. Clin. Oncol. 144(7), 1347–1355. https://doi.org/10.1007/S00432-018-2644-2 (2018).
Cardoso, F. et al. 4th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 4). Ann. Oncol. 29(8), 1634–1657. https://doi.org/10.1093/annonc/mdy192 (2018).
Leon-Ferre, R. A. et al. Impact of histopathology, tumor-infiltrating lymphocytes, and adjuvant chemotherapy on prognosis of triple-negative breast cancer. Breast Cancer Res. Treat. 167(1), 89–99. https://doi.org/10.1007/s10549-017-4499-7 (2018).
Burstein, M. D. et al. Comprehensive genomic analysis identifies novel subtypes and targets of triple-negative breast cancer. Clin. Cancer Res. 21(7), 1688–1698. https://doi.org/10.1158/1078-0432.CCR-14-0432 (2015).
Lehmann, B. D. et al. Refinement of triple-negative breast cancer molecular subtypes: Implications for neoadjuvant chemotherapy selection. PLoS One 11(6), e0157368. https://doi.org/10.1371/journal.pone.0157368 (2016).
Lehmann, B. D. et al. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J. Clin. Investig. 121(7), 2750–2767. https://doi.org/10.1172/JCI45014 (2011).
He, T. F. et al. Multi-panel immunofluorescence analysis of tumor infiltrating lymphocytes in triple negative breast cancer: Evolution of tumor immune profiles and patient prognosis. PLoS One 15(3), e0229955. https://doi.org/10.1371/JOURNAL.PONE.0229955 (2020).
Kim, G. et al. The contribution of race to breast tumor microenvironment composition and disease progression. Front. Oncol. 10, 1022. https://doi.org/10.3389/FONC.2020.01022/BIBTEX (2020).
Denkert, C. et al. Tumour-infiltrating lymphocytes and prognosis in different subtypes of breast cancer: A pooled analysis of 3771 patients treated with neoadjuvant therapy. Lancet Oncol. https://doi.org/10.1016/S1470-2045(17)30904-X (2018).
García-Teijido, P., Cabal, M. L., Fernández, I. P. & Pérez, Y. F. Tumor-infiltrating lymphocytes in triple negative breast cancer: The future of immune targeting. Clin. Med. Insights Oncol. 10s1, CMO.S34540. https://doi.org/10.4137/CMO.S34540 (2016).
Pruneri, G. et al. Tumor-infiltrating lymphocytes (TILs) are a powerful prognostic marker in patients with triple-negative breast cancer enrolled in the IBCSG phase III randomized clinical trial 22–00. Breast Cancer Res. Treat. https://doi.org/10.1007/s10549-016-3863-3 (2016).
Ibrahim, E. M., Al-Foheidi, M. E., Al-Mansour, M. M. & Kazkaz, G. A. The prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancer: A meta-analysis. Breast Cancer Res. Treat. 148(3), 467–476. https://doi.org/10.1007/S10549-014-3185-2 (2014).
Loi, S. et al. Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: Results from the FinHER trial. Ann. Oncol. 25(8), 1544–1550. https://doi.org/10.1093/annonc/mdu112 (2014).
Loi, S. et al. Prognostic and predictive value of tumor-infiltrating lymphocytes in a phase III randomized adjuvant breast cancer trial in node-positive breast cancer comparing the addition of docetaxel to doxorubicin with doxorubicin-based chemotherapy: BIG 02–98. J. Clin. Oncol. https://doi.org/10.1200/JCO.2011.41.0902 (2013).
Denkert, C. et al. Tumor-infiltrating lymphocytes and response to neoadjuvant chemotherapy with or without carboplatin in human epidermal growth factor receptor 2-positive and triple-negative primary breast cancers. J. Clin. Oncol. https://doi.org/10.1200/JCO.2014.58.1967 (2015).
Jang, N., Kwon, H. J., Park, M. H., Kang, S. H. & Bae, Y. K. Prognostic value of tumor-infiltrating lymphocyte density assessed using a standardized method based on molecular subtypes and adjuvant chemotherapy in invasive breast cancer. Ann. Surg. Oncol. https://doi.org/10.1245/s10434-017-6332-2 (2018).
Bahrami, A. et al. The therapeutic potential of targeting tumor microenvironment in breast cancer: Rational strategies and recent progress. J. Cell Biochem. https://doi.org/10.1002/jcb.26183 (2018).
Huertas-Caro, C. A., Ramirez, M. A., Gonzalez-Torres, H. J., Sanabria-Salas, M. C. & Serrano-Gómez, S. J. Immune lymphocyte infiltrate and its prognostic value in triple-negative breast cancer. Front. Oncol. 12, 3462. https://doi.org/10.3389/FONC.2022.910976/BIBTEX (2022).
Huang, Y. et al. CD4+ and CD8+ T cells have opposing roles in breast cancer progression and outcome. Oncotarget 6(19), 17462–17478. https://doi.org/10.18632/ONCOTARGET.3958 (2015).
Wolff, A. C. et al. Recommendations for human epidermal growth factor receptor 2 testing in breast. J. Clin. Oncol. 31(31), 3997–4013. https://doi.org/10.1200/JCO.2013.50.9984 (2013).
Chevallier, B., Roche, H., Olivier, J. P., Chollet, P. & Hurteloup, P. Inflammatory breast cancer. Pilot study of intensive induction chemotherapy (FEC-HD) results in a high histologic response rate. Am. J. Clin. Oncol. 16(3), 223–228 (1993).
Salgado, R. et al. The evaluation of tumor-infiltrating lymphocytes (TILS) in breast cancer: Recommendations by an International TILS Working Group 2014. Ann. Oncol. https://doi.org/10.1093/annonc/mdu450 (2015).
Hida, A. I. et al. Prognostic and predictive impacts of tumor-infiltrating lymphocytes differ between Triple-negative and HER2-positive breast cancers treated with standard systemic therapies. Breast Cancer Res. Treat. https://doi.org/10.1007/s10549-016-3848-2 (2016).
Krishnamurti, U., Wetherilt, C. S., Yang, J., Peng, L. & Li, X. Tumor-infiltrating lymphocytes are significantly associated with better overall survival and disease-free survival in triple-negative but not estrogen receptor–positive breast cancers. Hum. Pathol. https://doi.org/10.1016/j.humpath.2017.01.004 (2017).
Dieci, M. V. et al. Prognostic value of tumor-infiltrating lymphocytes on residual disease after primary chemotherapy for triple-negative breast cancer: A retrospective multicenter study. Ann. Oncol. https://doi.org/10.1093/annonc/mdt556 (2014).
Serrano-Gomez, S. J. et al. High prevalence of luminal B breast cancer intrinsic subtype in Colombian women. Carcinogenesis 37(7), 669–676. https://doi.org/10.1093/carcin/bgw043 (2016).
Zevallos, A. et al. The hispanic landscape of triple negative breast cancer. Crit. Rev. Oncol. Hematol. 155, 103094. https://doi.org/10.1016/J.CRITREVONC.2020.103094 (2020).
BONILLA-SEPÚLVEDA, O. A., MATUTE-TURÍZO, G. & SEVERICHE, C. Classification of intrinsic subtypes of breast carcinomas analyzed in a pathology center of Medellin in 2011. CES Med. 29(1), 36–45 (2015). Accessed: Feb. 20, 2023. [Online]. Available: http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S0120-87052015000100004&lng=en&nrm=iso&tlng=es
Castaneda, C. A. et al. Tumor infiltrating lymphocytes in triple negative breast cancer receiving neoadjuvant chemotherapy. World J. Clin. Oncol. https://doi.org/10.5306/wjco.v7.i5.387 (2016).
Gu-Trantien, C. et al. CD4+ follicular helper T cell infiltration predicts breast cancer survival. J. Clin. Invest. 123(7), 2873. https://doi.org/10.1172/JCI67428 (2013).
Zander, R. et al. CD4+ T cell help is required for the formation of a cytolytic CD8+ T cell subset that protects against chronic infection and cancer. Immunity https://doi.org/10.1016/j.immuni.2019.10.009 (2019).
Park, H. S. et al. No effect of tumor-infiltrating lymphocytes (TILs) on prognosis in patients with early triple-negative breast cancer: Validation of recommendations by the International TILs Working Group 2014. J. Surg. Oncol. 114(1), 17–21. https://doi.org/10.1002/JSO.24275 (2016).
Savas, P. et al. Clinical relevance of host immunity in breast cancer: From TILs to the clinic. Nat. Rev. Clin. Oncol. https://doi.org/10.1038/nrclinonc.2015.215 (2016).
Goto, W. et al. Predictive value of improvement in the immune tumour microenvironment in patients with breast cancer treated with neoadjuvant chemotherapy. ESMO Open https://doi.org/10.1136/esmoopen-2017-000305 (2018).
Asano, Y. et al. Prediction of treatment response to neoadjuvant chemotherapy in breast cancer by subtype using tumor-infiltrating lymphocytes. Anticancer Res. 38(4), 2311–2321. https://doi.org/10.21873/anticanres.12476 (2018).
Ruan, M. et al. Predictive value of tumor-infiltrating lymphocytes to pathological complete response in neoadjuvant treated triple-negative breast cancers. Diagn. Pathol. https://doi.org/10.1186/s13000-018-0743-7 (2018).
Krishnan, Y., Alawadhi, S. A., Sreedharan, P. S., Gopal, M. & Thuruthel, S. Pathological responses and long-term outcome analysis after neoadjuvant chemotheraphy in breast cancer patients from Kuwait over a period of 15 years. Ann. Saudi Med. 33(5), 443. https://doi.org/10.5144/0256-4947.2013.443 (2013).
Kawajiri, H. et al. Prognostic significance of pathological complete response following neoadjuvant chemotherapy for operable breast cancer. Oncol. Lett. 7(3), 663. https://doi.org/10.3892/OL.2014.1792 (2014).
Huang, M. et al. Association of pathologic complete response with long-term survival outcomes in triple-negative breast cancer: A meta-analysis. Cancer Res. 80(24), 5427–5434. https://doi.org/10.1158/0008-5472.CAN-20-1792/662474/AM/ASSOCIATION-OF-PATHOLOGICAL-COMPLETE-RESPONSE-WITH (2020).
Adams, S. et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J. Clin. Oncol. https://doi.org/10.1200/JCO.2013.55.0491 (2014).
Galluzzi, L., Buqué, A., Kepp, O., Zitvogel, L. & Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 17(2), 97–111. https://doi.org/10.1038/nri.2016.107 (2016).
Ladoire, S. et al. In situ immune response after neoadjuvant chemotherapy for breast cancer predicts survival. J. Pathol. 224(3), 389–400. https://doi.org/10.1002/PATH.2866 (2011).
Deepak, K. G. K. et al. Tumor microenvironment: Challenges and opportunities in targeting metastasis of triple negative breast cancer. Pharmacol. Res. https://doi.org/10.1016/J.PHRS.2020.104683 (2020).
Santoni, M. et al. Triple negative breast cancer: Key role of Tumor-Associated Macrophages in regulating the activity of anti-PD-1/PD-L1 agents. Biochim. Biophys. Acta (BBA) Rev. Cancer 1869(1), 78–84. https://doi.org/10.1016/J.BBCAN.2017.10.007 (2018).
Shen, Y., Peng, X. & Shen, C. Identification and validation of immune-related lncRNA prognostic signature for breast cancer. Genomics 112(3), 2640–2646. https://doi.org/10.1016/J.YGENO.2020.02.015 (2020).
Acknowledgements
This study was supported by Minciencias (Contrato 838-2018 to SJSG) and the Colombian national Cancer Institute (C-19010300431 to SJSG). The publication costs of this article were defrayed by the Colombian National Cancer Institute.
Author information
Authors and Affiliations
Contributions
Conception and design of the study C.H., M.S., and S.S. Methodology C.H., L.R., L.B., D.B., J.Z., and L.F. Acquisition of data C.H., M.R., J.M., L.S., and A.C.. Analysis and interpretation of data C.H., M.R., and M.N. Drafting the work C.H., M.R., and S.S. Writing–review and editing L.R., L.B., A.C., L.F., and M.S. Supervision, funding acquisition M.S., and S.S. Project administration C.H., S.S. All authors contributed to manuscript revision, read, and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huertas-Caro, C.A., Ramírez, M.A., Rey-Vargas, L. et al. Tumor infiltrating lymphocytes (TILs) are a prognosis biomarker in Colombian patients with triple negative breast cancer. Sci Rep 13, 21324 (2023). https://doi.org/10.1038/s41598-023-48300-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48300-4
- Springer Nature Limited