Abstract
Genetic recombination is one of the major evolution processes of HIV-1. Despite their great genetic divergence, HIV-1 groups M and O can generate HIV-1/MO intergroup recombinants. The current description of 20 HIV-1/MO unique recombinant forms suggests a possible benefit of the recombination. The aim of this work was to study in vitro the replicative potential of HIV-1/MO recombinant forms. This analysis was based on a simple recombination pattern, [Ogag/pol-Menv], harboring a breakpoint in Vpr. A chimeric infectious molecular clone, pOM-TB-2016 was synthesized from HIV-1/M subtype B and HIV-1/O subgroup T and recombinant viruses were obtained by transfection/co-culture. To compare the replicative potential of these viruses, two markers were monitored in culture supernatants: Reverse Transcriptase (RT) activity and P24 antigen concentration. The results showed a superiority of the group M parental virus compared to group O for both markers. In contrast, for the recombinant virus, RT activity data did not overlap with the concentration of P24 antigen, suggesting a hybrid behavior of the recombinant, in terms of enzyme activity and P24 production. These results highlighted many hypotheses about the impact of recombination on replicative potential and demonstrated again the significant plasticity of HIV genomes and their infinite possibility of evolution.
Similar content being viewed by others
Introduction
HIV-1 is characterized by high genetic diversity, due to its simian origins and replication mode, enhanced by recombination events. Despite the great genetic divergence between HIV-1 group M (HIV-1/M), pandemic, and group O (HIV-1/O), endemic in Cameroon, HIV-1/M + O dual infections can generate HIV-1/MO inter-group recombinants. Over the past 20 years, several case reports and epidemio-molecular studies have shown the ability of HIV-1/M and /O to superinfect a patient already infected with a viral form from another group (1 case)1; to replicate in the context of double infections (20 cases)1,2,3,4,5,6,7,8,9; and to generate intergroup recombinant forms capable of replicating in the absence (13 cases)6,7,10,11 (manuscript submitted) or in the presence of one or both parental forms (12 cases)3,5,6,7,10,12,13. Phylogenetic analysis showed that the 25 recombinant forms corresponded to 20 unique HIV-1/MO recombinant forms (URF_MO)14 (data submitted). The analysis of the genomic profiles and of the breakpoint frequency revealed hotspots in the "central" accessory genes (vif, vpr, vpu), Long Terminal Repeat (LTR) regions, reverse transcriptase (RT) and gp41, and no recombination event in protease, gp120 and nef14 (data submitted). This work also highlighted a variable degree of complexity in profiles, with on average two to three breakpoints per recombinant genome14 (data submitted). The current description of these 20 URF_MOs raises the question of a possible benefit of recombination and the modalities of their emergence. The evolution of the genetic diversity of HIV-1/M, of which the number of recombinant forms, both circulating recombinant forms (CRF) and URFs, has escalated since the beginning of the epidemic, clearly shows the interest of studying URF_MOs. Indeed, studies have suggested a better adaptation and a better fitness of intragroup M recombinants compared to parental forms and have highlighted preferential recombination profiles15,16,17. Therefore, data collected from the URF_MOs currently described in vivo and the presence of HIV-1/MO recombinant forms in patients with neither of the two original parental forms suggest the possible existence of phenotypic properties conferring a selective advantage of these HIV-1/MO recombinant forms, compared to HIV-1/M and HIV-1/O parental forms. Moreover, this emergence of multiple URF_MOs could lead over time to the successful emergence of circulating forms (i.e. a CRF_MO).
Regarding the relative replicative fitness, a pairwise growth competition experiment showed that HIV-1 group O was 10- to 100-fold-less fit than group M forms, when competed in peripheral blood mononuclear cells (PBMC), and depending on the diversity of the forms studied. These results suggested a lesser replicative fitness of group O, contributing to the low prevalence and limited geographical spread of HIV-1/O in the human population although it emerged several years ago18,19. The phenomenon of M/O inter-group recombination could thus rebalance this "pyramid of fitness" and confer a replicative advantage to an HIV-1/MO recombinant form compared to one, the other or both of the parental forms. To date, only one study has investigated the replicative potential of an HIV-1/MO recombinant form and the results indicated a better in vitro adaptive capacity (fitness) of the recombinant virus compared to the HIV-1/M parental virus, suggesting a potential better spread and therefore a selective advantage conferred by recombination, in the absence of selection pressure13.
The aim of this work was to investigate in vitro the viral properties conferred by recombination, on the replicative potential of an HIV-1/MO recombinant virus compared to both of the HIV-1/M and HIV-1/O parental viruses, from which it was derived.
Results
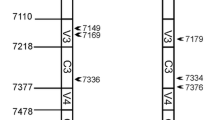
The pattern of the HIV-1/OM recombinant virus (OM-TB-2016) involved RBF206 from the first bases of 5′LTR to the unique but conserved AvrII restriction site, in the vpr gene, and 89.6 from the AvrII site to the last bases of 3′LTR (Fig. 1a). The breakpoint in Vpr was located at 5661–5667, relative to the HIV-1/M HxB2 reference strain (GenBank accession number K03455). The chimeric infectious molecular clone (CIMC) pOM-TB-2016, synthesized by Genecust Society (Luxembourg), corresponded to the OM-TB-2016 full-length sequence inserted into a pET-28b(+) vector and was 15,358 bp in length (Fig. 2).
HYPERLINK "sps:id::fig1||locator::gr1||MediaObject::0"Representation of the HIV-1 intergroup M and O recombinant genomes. Two recombination patterns were designed, with a breakpoint in the vpr gene: [Mgag/pol-Oenv] pattern, in (a) and [Ogag/pol-Menv] pattern, in (b). The location of the breakpoints is indicated relative to the HIV-1/M HxB2 reference strain (GenBank accession number K03455).
Profile of the chimeric infectious molecular clone pOM-TB-2016. The genome portions from the parental infectious molecular clones pRBF206 (HIV-1/O) and p89.6 (HIV-1/M) are shown in blue and red, respectively. The vector pET-28b(+) is shown in green. The breakpoint, in purple, corresponds to the restriction site of the AvrII enzyme.
A pool of each virus (HIV-1/M, HIV-1/O and HIV-1/OM) was constituted and characterized. P24 antigen concentrations were between 4.5 and 5.9 Log10pg/mL, viral load values were between 8.8 and 9.3 Log10cp/mL and TCID50 values were between 91 376 and 267 184 IU/mL. (Table 1). These results showed very high viral production and reflected good infectivity for the three viruses considered.
The RT-PCRs targeting the Vpr region confirmed the absence of contamination. Indeed, only the [M-M], [O-O] and [O-M] patterns were found for the parental (HIV-1/M and HIV-1/O) and recombinant (HIV-1/OM) viruses, respectively (Table 1). The [M-O] pattern, used as a negative control, was never amplified, as expected.
The triplicates of each virus showed good reproducibility, with only small differences calculated between the maximum and minimum values, both for the time to appearance of the peak and for viral production (Table 2). The recombinant virus showed a greater heterogeneity between triplicates than the parental viruses, with a difference of 5 days for the time to appearance of the peaks, and of 0.3Log10 and 0.8Log10 for the intensity of the peaks for RT activity and P24 antigen concentration, respectively. Finally, the kinetics of the two markers monitored, carried out independently in triplicate for each virus, proved to be reproducible and superimposable.
The mean kinetics of RT activity and P24 antigen concentration of the three viruses are shown in Figure 3a and b, respectively. The HIV-1/M parental virus grew faster and more intensively than the HIV-1/O parental virus, but also than the HIV-1/OM recombinant virus. Therefore, the supernatant samplings were stopped earlier for HIV-1/M than for the two other viruses (Fig. 3). The replication kinetics of the recombinant virus appeared closer to its HIV-1/O parental virus than to its HIV-1/M parental virus (Fig. 3). In order to quantitatively evaluate the differences between the three strains, we calculated the means of the data at the peaks of RT activity and of P24 antigen concentration of the triplicates and the growth rates as well as the differences for the three viruses (Table 3).
Mean kinetics of RT activity and P24 antigen concentration of parental and recombinant strains. (a) Representation of the kinetics of the mean of RT activity, in Log10 pg/mL of the three triplicates, over time in days, for the parental strains HIV-1/M (in red) and HIV-1/O (in blue) and the recombinant strain HIV-1/OM (in orange). (b) Representation of the kinetics of the mean of P24 Ag concentration, in Log10 pg/mL of the three triplicates, over time in days, for the parental strains HIV-1/M (in red) and HIV-1/O (in blue) and the recombinant strain HIV-1/OM (in orange).
This comparative analysis highlighted the hybrid behavior of the recombinant virus. Indeed, the peak of RT activity appeared earlier than for the parental virus HIV-1/O, which could be linked to the first steps of the viral replication cycle with an entry into the target cell favored by an envelope derived from HIV-1/M. On the other hand, concerning the intensity of RT activity and of P24 antigen production, the HIV-1/OM recombinant virus showed a behavior closer to that of the HIV-1/O parental virus, which could be similarly related to the recombination profile [Ogag/pol-Menv] we designed for our chimera (Table 3).
Discussion
A previous study suggested that an inter-group M and O recombinant form could be fitter than its parental forms13. Moreover, the abundant epidemic intragroup M recombinant forms, having emerged over time, are also in favor of this hypothesis of a better adaptation than the parental forms. In this context, the objective of this study was to evaluate, for the first time, the in vitro replication of an inter-group M and O recombinant virus compared to its HIV-1/M and HIV-1/O parental viruses, to assess an increased potential for emergence in the human population.
Infectious molecular clones (IMCs) correspond to a full-length HIV genome sequence inserted into a vector and can produce virions, after transfection into susceptible cells. These newly formed virions all derive from a unique and known genetic sequence, as opposed to the high genetic diversity observed among quasispecies. The use of parental infectious molecular clones (PIMCs) to generate CIMCs, leading to the production of HIV-1/OM recombinant viruses by reverse genetics, allowed us to overcome any bias related to the use of heterologous strains, since the recombinant virus was compared to both the HIV-1/M and HIV-1/O parental strains, used for its generation.
Given that the HIV-1/M parental and HIV-1/OM recombinant viruses presented an X4 tropism in the V3 loop while the HIV-1/O parental virus presented an R5 tropism, the production of the viruses was carried out with the Jurkat CD4-CCR5 cell line. This cell line expresses the two co-receptors at its surface and can thus replicate viruses of different tropism. However, the proportions of expression of each co-receptor at the cell surface are unknown and an overexpression of one or the other could lead to a sensitivity of these cells to an infection depending on the tropism of the virus. In this context, we measured these expression percentages by flow cytometry. Among the Jurkat cells studied, 62.5% expressed both, whereas 35.3% and 1.5% expressed only the X4 and the R5 co-receptors, respectively. It is difficult to conclude as to the impact of the percentage of expression of the co-receptors on our comparative data with the HIV-1/O parental virus. However, our comparison between the HIV-1/OM recombinant virus and its HIV-1/M parental virus is validated since both of them share the same envelope and therefore the same tropism.
In order to eliminate any bias linked to the inter-manipulation variability of the cell culture, each kinetic of each virus was carried out in triplicate and the reproducibility of the values validated our comparisons. Overall, a few exceptionally outlying values were revealed, but the minimum and maximum values found in each triplicate remained close.
The choice of replication monitoring tools was decisive for our objective of comparing replicative potential. For P24 antigen production, the INNOTEST® HIV Antigen mAb kit (Fujirebio) was used because the sensitivity factor was the same for HIV-1/M and HIV-1/O20 and in order to avoid a possible misquantification linked to HIV-1/O antigenic diversity21. In addition, on each supernatant, RT activity was determined with the Cavidi method, which quantifies viral enzymatic activity without being impacted by viral diversity22. Moreover, the quantification of RT activity appears to be more representative of the replicative potential of the strain than the P24 antigen produced during replication, which corresponds to an accumulation of the protein over time.
The use of these two markers highlighted different behaviors depending on the virus and on the marker. Thus, the comparison of the kinetics of RT activity showed that the HIV-1/M parental virus replicated more quickly and more intensely than the HIV-1/OM recombinant and HIV-1/O parental viruses, but with an intermediate behavior of the recombinant virus. Since this recombinant strain did not correspond to a strain identified in vivo, the time to successfully initiate replication cycles was unknown. Thus, the culture supernatants were not removed and the cells were not washed after infection on day 0, so as not to risk inhibiting viral replication. We therefore cannot exclude that the observed differences could be conditioned by the levels of P24 antigen and RT activity in the culture supernatant on day 0, with an impact on the time it takes for each virus to reach its peak intensity but no impact on the levels that are reached. The use of three distinct parameters (the time of reaching the peak, the intensity of this peak and the viral growth rate) and of the same methodology for each virus (HIV-1/M, HIV-1/O and HIV-1/OM) allowed to limit the impact of the accumulation of P24 antigen and RT activity in the culture supernatants.
In fact, the results did not overlap completely either with those obtained with the HIV-1/M parental virus, or with the HIV-1/O parental virus. The P24 antigen production by the HIV-1/M parental virus was also greater than that of the other two viruses, with always an intermediate behavior of the HIV-1/OM recombinant virus.
Our results showed that the HIV-1/M parental virus had a significantly higher replicative potential than the HIV-1/O parental virus, as described in vitro by Arien et al.18. On the other hand, our results differed from the hypotheses put forward by Peeters et al.13. Indeed, after co-culture of peripheral blood mononuclear cells (PBMCs) from a patient co-infected with an HIV-1/MO recombinant form and the HIV-1/M parental form from which it was derived, only the recombinant virus was detected. The team suggested a replicative superiority of the HIV-1/MO recombinant virus than its HIV-1/M parental virus13, but the recombination profile described by Peeters et al. was [Mgag/pol-Oenv], which could explain our discordant results. Thus, our results showed, whatever the marker, the clear replicative superiority of the HIV-1/M parental virus compared to the recombinant virus, even if the monitoring curves for RT activity and for P24 antigen production were not completely superimposable. However, the HIV-1/OM recombinant virus seems to have certain advantages over its HIV-1/O parental virus, but it was not possible, in our study, to demonstrate a systematic better replicative potential of the HIV-1/OM recombinant virus than its parental viruses.
This hybrid replicative behavior of our recombinant virus could be explained by the [Ogag/pol-Menv] recombination profile chosen. Indeed, its exponential growth phase began earlier than that of the HIV-1/O parental virus, suggesting a greater infectivity of the recombinant virus. The viral envelope is known to play a key role in the recognition of receptors and co-receptors, in the early phases of infection as well as in viral fitness23,24. The envelope of the recombinant strain generated from the HIV-1/M parental virus could therefore be favorable to the recombinant virus. The predominance of group M envelopes among the HIV-1/MO recombinant forms described in vivo (7 profiles [Ogag/pol-Menv] versus 4 profiles [Mgag/pol-Oenv]14 supports this hypothesis. The production of the HIV-1/OM recombinant virus, on the other hand, appeared worse over time, possibly because the regions of the genome coding for the structural proteins and the replication enzymes came from the HIV-1/O parental virus. Indeed, the peak of RT activity of the recombinant strain was close to that of the HIV-1/O parental virus (lower by 0.2 Log10 pg/mL), as was the peak of the concentration of P24 Ag (stronger by 0.2 Log10 pg/mL). The opposite pattern of recombination, [Mgag/pol-Oenv], remains to be produced and tested, in order to confirm these hypotheses, unless this reverse virus is not viable, which is also one hypothesis.
Thus, these results should be linked to the diversity of inter-group M and O, which suggests that not all the recombinant forms are necessarily possible and that they do not necessarily all have the same profile. Due to the low availability of different PIMCs, in terms of genetic diversity (HIV-1/M subtypes and HIV-1/O subgroups), and to technical difficulties to generate others patterns (data not shown), this work shows a single representative of HIV-1/M, subtype B, associated with a single representative of HIV-1/O, subgroup T. To date, there is no HIV-1/MO recombinant form described in vivo that shows this association. Our previous study of all URF_MOs revealed that the genetic diversity of the mosaic fragments matched the molecular epidemiology in Cameroon, with a clear predominance of HIV-1/M CRF02_AG (48%) and of HIV-1/O sub-group H (84%). Indeed, among these 19 URF_MOs, 8 (42%) combine HIV-1/M subtype CRF02_AG with HIV-1/O subgroup H and only 3 (16%) combine HIV-1/M subtype CRF02_AG with HIV-1/O subgroup T. This atypical combination of parental viruses might have had an impact on our results and could not be representative of emerging factors in local situation. However, the main objective was to compare the HIV-1/OM recombinant strain to its HIV-1/M and HIV-1/O parental forms, in order to determine if this emerging form could have enhanced virological properties; which is already informative in our model. Moreover, the RBF206 form did not seem to be the most representative of HIV-1/O, since its Vpu protein has the ability to counteract Tetherin, as described in HIV-1/M forms25. Its replicative potential, due to this particularity of Vpu, could therefore be greater than that of the other variants of the O group and explain the small difference in replication observed compared to the HIV-1/OM recombinant virus.
The HIV-1/OM recombinant virus produced and studied in this work may not be the most representative of the recombinant forms described in vivo but constitutes the first built and functional HIV-1/OM chimera. This work, based on our previous studies and our knowledge about HIV-1/MO recombination, offers many perspectives for studies, especially to evaluate the potential emergence of such recombinant HIV, taking into account genetic diversity in particular. Nevertheless, this HIV-1/OM recombinant virus has highlighted many hypotheses about the impact of recombination on replicative potential and has demonstrated once again the significant plasticity of HIV genomes and their infinite possibility of evolution.
Materials and methods
Parental infectious molecular clones and chimeric infectious molecular clones
Selection of parental forms and positioning of breakpoint
Given the reverse genetics approach that we aimed to implement, two parental infectious molecular clones (PIMCs) were used as parental viruses: pRBF206 and p89.6. RBF206 is an HIV-1/O subgroup T form isolated and characterized in our laboratory, with an R5 tropism. The corresponding PIMC pRBF206 was obtained as previously described26. For HIV-1/M, 89.6 belongs to the subtype B and shows an X4 tropism. The PIMC p89.6 was obtained from the National Institute for Biological Standards and Control (NIBSC) Centralized Facility for AIDS Reagent (CFAR). To generate the chimeric infectious molecular clones (CIMC), we first designed the recombination patterns and chose a breakpoint, on the basis of our previous study14. The recombination patterns were chosen to reproduce the two major chimeric genome profiles described in vivo: [Ogag/pol-Menv] and [Mgag/pol-Oenv], and to generate the recombinant viruses HIV-1/OM and HIV-1/MO, respectively. Given the recombination hotspot identified in vivo14, the breakpoint was positioned in a conserved region identified in the vpr gene of HIV-1/M and HIV-1/O genomes (Fig. 1). These two chimeric sequences were sent to the Genecust society to synthesize the corresponding CIMCs, but only pHIV-1/OM was successfully obtained.
Cell lines
Human HEK-293T adherent cells, obtained from the CFAR (NIBSC), were seeded at 37 °C with 5% CO2 in DMEM + GlutaMAX™ (Gibco, Germany) containing 10% heat inactivated fetal calf serum (dFCS) (Eurobio Scientific, France) and gentamicin (50 µg/mL). Cells were maintained twice a week with trypsinization and one-third re-seeding. Jurkat.CD4-CCR5, obtained from the CFAR (NIBSC), is a CD4+ T non-adherent cell line with endogenous CXCR4 and stably transfected to express human CCR5. Cells were maintained in RPMI-1640 medium (Lonza, Swiss) supplemented with 10% dFCS, gentamicin (50 µg/mL) and geneticin G418 (500 µg/mL), at 37 °C with 5% CO2, and were one-fifth re-seeded twice a week.
Production of parental and recombinant viruses from PIMC and CIMC
The parental and recombinant viruses were produced during a single experiment (Fig. 4). For this, the two PIMCs (pRBF206 and p89.6) and the one CIMC (pOM-TB-2016) were rescued using a two-step reverse genetic system as follows. The day before transfection (D-3), 750 000 HEK293T cells were counted and seeded in a 6-well plate. The next day (D-2), HEK293T cells at 60–80% confluency were transfected with 1 µg of pHIV-1/M, pHIV-1/O, or pHIV-1/OM, using JetPrime™ (Polyplus Transfection, France), according to manufacturer’s instructions. At D0, a co-culture step was performed with 1⋅106 Jurkat.CD4-CCR5 and at D2, supernatants were transferred to T25 flasks. Between D4 and D43, a fraction of each cell-free supernatant was removed, tested by quantifying p24 antigen (VIDAS® HIV P24 II, BioMérieux, France), frozen and replaced by culture medium (RPMI + dFCS + gentamicin + geneticin), in order to remain at the same volume throughout the culture.
General principle of the production of HIV-1/OM recombinant viruses from a CIMC. The capacity of CIMCs to produce the corresponding recombinant strains was evaluated using a protocol based on the transfection of HEK293T cells (for the production of viruses) followed by a co-culture step with the Jurkat cell line (for the viral amplification).
The produced stock of HIV-1/M, HIV-1/O and HIV-1/OM supernatants was constituted by pooling if the concentration of p24 antigen was greater than 3 Log10pg/mL. The three viral pools were characterized by quantifying the p24 antigen (VIDAS® HIV P24 II, BioMérieux), the viral load (Xpert® HIV-1 Viral Load, Cepheid, USA) and the infectious virus titer by the 50% cell culture infectious dose (TCID50) Montefiori method, on TZM-bl cells. Finally, to eliminate any contamination of one virus by another, four nested RT-PCRs amplifying the Vif-Vpu region were performed as a quality control on each viral pool, as previously described10. The presence of a breakpoint in the vpr gene was investigated using homologous ([M-M] and [O-O]) and heterologous ([M-O] and [O-M]) combinations of HIV-1/M and HIV-1/O group-specific primers, followed by Sanger sequencing.
Characterization of in vitro replicative potential
The replication kinetics of the parental and recombinant viruses were performed in triplicate on the Jurkat.CD4-CCR5 line. For this, 5⋅106 cells were infected with each virus (HIV-1/M, HIV-1/O or HIV-1/OM) at a multiplicity of infection (MOI) of 0.01, and in 5 mL of medium (RPMI + dFCS + Gentamicin + Geneticin). Between the first day (D1) and the last day (D30) of culture, 400 μL of supernatant were taken then frozen 6 days out of 7 and replaced by 400 μL of new culture medium, in order to maintain the same volume throughout the culture. The kinetics were measured on the basis of the RT activity (Lenti RT activity, Cavidi, Sweden) and the concentration of P24 antigen (INNOTEST® HIV Antigen mAb, Fujirebio, Belgium) of each supernatant, for each triplicate of each virus, retrospectively. The mean values of the RT activity and of the concentration of P24 antigen of each triplicate were calculated (in Log10pg/mL) for each point of the kinetics and the peak was defined for each kinetic.
Replication profile comparison
In order to highlight a possible difference between the HIV-1/OM recombinant virus and its HIV-1/M and HIV-1/O parental viruses, three parameters were considered: the time of reaching the peak (in days), the intensity of this peak (in Log10pg/mL) and the growth rate (in Log10/day). The growth rate was defined as the slope of the line connecting the point following the lowest point, reflecting the start of replication, and the highest point, corresponding to the replication peak.
Data availability
All relevant data are within the manuscript but if needed, any supplementary data are available from the DYNAMCURE research team, at the University of Rouen Normandie (alice.moisan@chu-rouen.fr).
References
Plantier, J.-C. et al. HIV-1 group M superinfection in an HIV-1 group O-infected patient. AIDS 18, 2444–2446 (2004).
Heyndrickx, L., Alary, M., Janssens, W., Davo, N. & van der Groen, G. HIV-1 group O and group M dual infection in Bénin. Lancet 347, 902–903 (1996).
Takehisa, J. et al. Various types of HIV mixed infections in Cameroon. Virology 245, 1–10 (1998).
Vergne, L. et al. Biological and genetic characteristics of HIV infections in Cameroon reveals dual group M and O infections and a correlation between SI-inducing phenotype of the predominant CRF02_AG variant and disease stage. Virology 310, 254–266 (2003).
Yamaguchi, J. et al. HIV infections in northwestern Cameroon: identification of HIV type 1 group O and dual HIV type 1 group M and group O infections. AIDS Res. Hum. Retroviruses 20, 944–957 (2004).
De Oliveira, F. et al. Detection of numerous HIV-1/MO recombinants in France. AIDS 32, 1289–1299 (2018).
De Oliveira, F. et al. Multiple HIV-1/M + HIV-1/O dual infections and new HIV-1/MO inter-group recombinant forms detected in Cameroon. Retrovirology 14, 1 (2017).
Brand, D. et al. First identification of HIV-1 groups M and O dual infections in Europe. AIDS 18, 2425–2428 (2004).
Ngoupo, P. A. Fréquence et profil génétique des doubles infections VIH-1/M+O et formes recombinantes VIH-1/MO au Cameroun (Normandie Université, 2016).
Ngoupo, P. A. et al. First evidence of transmission of an HIV-1 M/O intergroup recombinant virus. AIDS 30, 1–8 (2016).
Vessière, A. et al. First evidence of a HIV-1 M/O recombinant form circulating outside Cameroon. AIDS 24, 1079–1082 (2010).
Takehisa, J. et al. Human immunodeficiency virus type 1 intergroup (M/O) recombination in Cameroon. J. Virol. 73, 6810–6820 (1999).
Peeters, M. et al. Characterization of a highly replicative intergroup M/O human immunodeficiency virus type 1 recombinant isolated from a Cameroonian patient. J. Virol. 73, 7368–7375 (1999).
Moisan, A. et al. Evolution of HIV-1 groups M and O: genetic comparative analysis of 23 HIV-1/MO inter-group recombinant forms. (2017).
Arenas, M., Lorenzo-Redondo, R. & Lopez-Galindez, C. Influence of mutation and recombination on HIV-1 in vitro fitness recovery. Mol. Phylogenet. Evol. 94, 264–270 (2016).
Rousseau, C. M. et al. Extensive intrasubtype recombination in South African human immunodeficiency virus type 1 subtype C infections. J. Virol. 81, 4492–4500 (2007).
Burke, D. S. Recombination in HIV: An important viral evolutionary strategy. Emerg. Infect. Dis. 3, 253–259 (1997).
Ariën, K. K. et al. The replicative fitness of primary human immunodeficiency virus type 1 (HIV-1) Group M, HIV-1 Group O, and HIV-2 isolates. J. Virol. 79, 8979–8990 (2005).
Geuenich, S., Kaderali, L., Allespach, I., Sertel, S. & Keppler, O. T. Biological signature characteristics of primary isolates from human immunodeficiency virus type 1 Group O in ex vivo human tonsil histocultures. J. Virol. 83, 10494–10503 (2009).
Fransen, K., Beelaert, G. & van der Groen, G. Evaluation of four HIV antigen tests. J. Virol. Methods 93, 189–193 (2001).
Ly, T. D. et al. The variable sensitivity of HIV Ag/Ab combination assays in the detection of p24Ag according to genotype could compromise the diagnosis of early HIV infection. J. Clin. Virol. 55, 121–127 (2012).
Braun, J. et al. A new quantitative HIV load assay based on plasma virion reverse transcriptase activity for the different types, groups and subtypes. AIDS 17, 331 (2003).
Marozsan, A. J. et al. Differences in the fitness of two diverse wild-type human immunodeficiency virus type 1 isolates are related to the efficiency of cell binding and entry. J. Virol. 79, 7121–7134 (2005).
Palmer, S. et al. Multiple, linked human immunodeficiency virus Type 1 drug resistance mutations in treatment-experienced patients are missed by standard genotype analysis. J. Clin. Microbiol. 43, 406–413 (2005).
Mack, K. et al. Efficient Vpu-mediated tetherin antagonism by an HIV-1 Group O strain. J. Virol. 91, e02177-e2216 (2017).
Kluge, S. F. et al. Nef proteins of epidemic HIV-1 Group O strains antagonize human tetherin. Cell Host Microbe 16, 639–650 (2014).
Acknowledgements
We wish to thank the staff of the laboratory of Virology of Rouen University Hospital (especially Fanny Lermechain) and the members of the DYNAMICURE research team for the cloning experiments, especially Gaetan Riou, from Inserm UMR1234—PANTHER Laboratory, for his help in flow cytometry experiments. We are grateful to Nikki Sabourin-Gibbs, Rouen University Hospital, for her help in editing the manuscript and to Denys Brand, from Tours University, for his rigorous proofreading.
Author information
Authors and Affiliations
Contributions
A.M., T.M. and J.C.P. conceived and designed the analysis. A.M. performed the experiments, collected the data and prepared the figures. A.M., F.T., E.A.G., T.M. and J.C.P. contributed data or analysis tools. A.M., M.V. and J.C.P. performed the analysis. E.A.G., T.M. and J.C.P. supervised the experiments. A.M. and J.C.P. wrote the main manuscript text. All the authors have read approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moisan, A., Tombette, F., Vautrin, M. et al. In vitro replicative potential of an HIV-1/MO intergroup recombinant virus compared to HIV-1/M and HIV-1/O parental viruses. Sci Rep 14, 1730 (2024). https://doi.org/10.1038/s41598-024-51873-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-51873-3
- Springer Nature Limited