Abstract
Gestational diabetes is characterized by hyperglycaemia diagnosed during pregnancy. Gestational and pregestational diabetes can have deleterious effects during pregnancy and perinatally. The baby's weight is frequently above average and might reach macrosomia (≥ 4 kg), which can reduce pregnancy time causing preterm births, and increase foetal-pelvic disproportion which often requires delivery by caesarean section. Foetal-pelvic disproportion due to the baby’s weight can also cause foetal distress resulting in lower Apgar scores. To analyse the association between pregestational and gestational diabetes with maternal and foetal risk. We conducted a retrospective cohort study in women pregnant between 2012 and 2018 in the region of Lleida. Regression coefficients and 95% confidence intervals (CI) were used. The multivariate analysis showed statistically significant associations between pregestational diabetes and: prematurity (OR 2.4); caesarean section (OR 1.4); moderate (OR 1.3), high (OR 3.3) and very high (OR 1.7) risk pregnancies; and birth weight ≥ 4000 g (macrosomia) (OR 1.7). In getational diabetes the multivariate analysis show significant association with: caesarean section (OR 1.5); moderate (OR 1.7), high (OR 1.7) and very high (OR 1.8) risk pregnancies and lower 1-minuto Apgar score (OR 1.5). Pregestational and gestational diabetes increase: pregnancy risk, caesarean sections, prematurity, low Apgar scores, and macrosomia.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Gestational diabetes (GD) is defined by hyperglycaemia diagnosed during pregnancy1. Maternal factors associated with gestational diabetes are increasing, mainly: the rise of overweight and obesity in women, risks factors to develop type 2 diabetes and gestational diabetes; and the older average age of the mothers in the pregnancy of their first child.
GD is the most common pregnancy-associated disorder, with potential obstetric and perinatal consequences. Therefore, antenatal care in women with GD require hospital specialists instead of primary care health checks.
A study published in 2021 by Wdowiak et al.2 showed that overweight of the mother before pregnancy correlates with high birth weight of the baby. This was confirmed in the review by Catalano et al.3. A study on hyperglycaemia and adverse pregnancy outcomes by Metzger et al.,4 which comprised data from over 23,000 women, found that the prevalence of macrosomia was 6.7%, 10.2% and 20.2% in 17,244 non-obese women without GD, 2,791 non-obese women with GD, and 935 obese women with GD, respectively. Women with obesity without GD had a 13.6% higher risk of macrosomia (defined as a birth weight of 4000 g and over) than non-obese women. Adverse pregnancy outcomes are more common in women with pregestational diabetes compared to GD, according to a systematic review by Malaza et al. Complications include cesarean section, preterm birth, congenital anomalies, pre-eclampsia, neonatal hypoglycemia, macrosomia, neonatal intensive care unit admission, stillbirth, Apgar score, large for gestational age, induction of labor, respiratory distress syndrome, and miscarriages5.
Insulin is an anabolic hormone that regulates foetal growth6,7. Maternal hyperglycaemia induces hyperglycaemia and hyperinsulinemia in the foetus. This stimulates anabolism and consequently the development of muscle, adipose, and connective tissue. The combination of hyperglycaemia and hyperinsulinemia cause an increase in storage of foetal fat and protein which results in macrosomia6.
Macrosomia is defined by a foetal weight by gestational age above the 90th percentile, or equal/over 4000 g8. This weight can shorten pregnancy time causing prematurity. It can also cause foetal-pelvic disproportion, which requires more caesarean sections and can result in lower Apgar scores.
The common etiological mechanism in pregestational and gestational diabetes is insulin resistance. Both cause hyperglycaemia in pregnancy and both have been associated with adverse effects in pregnancy. With this study conducted in the health region of Lleida, we aim to analyse the prevalence of pregestational and gestational diabetes, of high-risk pregnancies and of complications in the baby.
Methodology
Design and data collection
Retrospective, cohort observational study in pregnant women between 2012 and 2018 in the health region of Lleida.
Data from women who had delivered at the hospital Arnau de Vilanova between 1-1-2012 and 31-12-2018 were obtained through the CMBD database (Minimum Data Set) of the electronic medical records database e-CAP, and from electronic prescriptions of the Catalan Health Service.
This study is part of the ILERPREGNANT project. The main objective of ILERPREGNANT is to analyse the prevalence of different conditions, therapeutic prescription and pharmacological adherence during pregnancy9.
Participants
Women who had delivered between 1-1-2012 and 31-12-2018. Pregnancy data are included from the date of the last period until the date of birth. As such, data from 2011 were taken into account for pregnant women with a birth date in 2012 but with a last period date in 2011. Pregnant women who do not belong to the health region of Lleida were excluded. To verify the representativity of the sample, the percentage of births studied (births registered at the University Hospital Arnau de Vilanova in Lleida) was calculated with respect to the total number of births in the health region of Lleida according to the data obtained from the Statistical Institute of Catalonia (Idescat) database (Table 1).
The main variable recorded was presence of gestational diabetes or previous diabetes, a dichotomous qualitative variable defined by an abnormal O'Sullivan test at weeks 24 and 28 of pregnancy (according to GEDE, 2014), verified by an Oral Glucose Tolerance test (OGT). Extraction of medical records in e-CAP with the code for gestational diabetes (ICD-10 code O24.9).
Other variables taken into account were: risk of the pregnancy; duration of the pregnancy (miscarriage, preterm, term, prolonged); caesarean section; birth weight (< 2500 g = underweight, between 2500 g and 3999 g = normal weight, and ≥ 4000 g = macrosomia), 1-minute and 5-minute Apgar score; and preeclampsia9.
Ethics
This study was approved by the Clinical Research Ethics Committee (CREC) of the Institut de Recerca IDIAP Jordi Gol (code 19/194-P). It follows the tenets of the Declaration of Helsinki. The information was extracted from centralized medical files in the e-CAP database by the Health Research and Assessment Management Department. Informed consent from participants was not needed. The variables in the e-CAP database were processed anonymously and with all the guarantees of confidentiality established by the National Law and Regulation 2016/679 of the European Parliament and Council on the protection of individuals with regard to the use of personal information. The need of Informed consent was waived by “Idiap Jordi Gol i Gurina comitè”.
Human and animal rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent
The databases from which the data were obtained are based on opt-out presumed consent and data are anonymized. If a patient decides to opt out, their data is excluded from the database. The need of Informed consent was waived by the Clinical Research Ethics Committee (CREC) of the Institut de Recerca IDIAP Jordi Gol.
Results
Study population
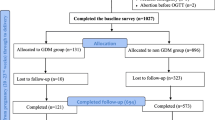
The sample consisted of 21,375 pregnant women who had given birth at the Hospital Arnau de Vilanova in Lleida between 2012 and 2018 (both included). Women who did not have a personal identification code (CIP) (n = 1625) were excluded from the group, as well as women with insufficient data in the clinical record. The final sample consisted of 17,177 patients (Fig. 1).
Characteristics of the study population
The prevalence of pregnant women with diabetes was 8.2%, 1.6% pregestational (diabetes mellitus type 1 N = 4, diabetes mellitus type 2 N = 271) and 6.6% gestational (N = 1123). A total of 87.2% pregnancies ended in term deliveries, 5.9% were preterm infants, 2.5%, prolonged pregnancies, and 4.4% ended in miscarriage. Preeclampsia occurred in 0.9% of pregnancies. Caesarean section was performed in 17.3% deliveries. Most babies (87.2%) had a normal weight at birth (2500–4000 g), 6% had low weight (< 2500 g), and 6.8% had macrosomia (> 4000 g). Low 1-min and 5-min Apgar scores (< 7) were found in 2.5% and 0.8% infants, respectively (Table 2).
Factors associated with diabetes during pregnancy
The following factors were associated with diabetes during pregnancy:
The proportion of caesarean sections was higher in women with gestational diabetes 25.6%) or pregestational diabetes (24%) than in women with no diabetes (16.6%). Preterm pregnancies were 7.7% in women with gestational diabetes, 12.8% in pregestational diabetes compared to 5.7% in women with no diabetes. A larger proportion of babies with a birth weight over 4000 g (macrosomia) had mothers with diabetes (8.4% gestational diabetes and 11.8% pregetational diabetes), compared to mothers with no diabetes (6.5%). The prevalence of 1-min Apgar < 7 was of 3.9% in case of mothers with gestational diabetes, 4.1% in case of pregestational diabetes and 2.4% in case of mothers without diabetes (Table 3).
The multivariate analysis showed statistically significant associations between pregestational diabetes and: prematurity (OR 2.4); caesarean section (OR 1.4); moderate (OR 1.3), high (OR 3.3) and very high (OR 1.7) risk pregnancies; and birth weight ≥ 4000 g (macrosomia) (OR 1.7) (Fig. 2). In gestational diabetes the multivariate analysis show significant association with: caesarean section (OR 1.5); moderate (OR 1.7), high (OR 1.7) and very high (OR 1.8) risk pregnancies and lower 1-minuto Apgar score (OR 1.5) (Fig. 3).
Discussion
In this retrospective study, prevalence of gestational and pregestational diabetes were 6.6% and 1.6%, respectively. Pregestational diabetes in pregnancy is associated with higher rates of prematurity, caesarean section and macrosomia, and gestational diabetes is associated with caesarean section, lower 1-min Apgar scores and risk of pregnancy (moderate, high and very high).
Preterm births account for 75% of neonatal mortality and almost 50% of long-term neurological morbidity10,11,12,13. In this study, the prevalence of preterm births was 8.6% in patients with diabetes compared to 5.7% in patients without diabetes. Similarly, a case–control study found a higher incidence of foetal distress, macrosomia, small for gestational age and preterm infants in mothers with GD compared to the control group14.
Furthermore, a meta-analysis that evaluated the effects of glucose intolerance (GI) that does not reach the criteria for gestational diabetes, observed an increase in caesarean sections, babies large for gestational age (LGA), preeclampsia, preterm births and low Apgar scores in women with GI15.
Regarding the increased risk of caesarean section, the Hyperglycaemia and Adverse Pregnancy Outcome (HAPO) study reported an increased risk in women with elevated glucose concentrations4. Analysing the characteristics and outcomes of pregnant women with gestational diabetes according to insulin sensitivity, Benhalima et al.16 observed that patients with gestational and insulin-resistant diabetes presented higher rates of preterm births (8.5% vs. 4.7%, p = 0.030), need for induction of labour (42.7% vs. 28.1%, p < 0.001), total caesarean sections (28.7% vs. 19.4%, p = 0.00) and emergency caesarean sections (16.0% vs. 9.7%, p = 0.010) compared with women without diabetes. In the current study, the proportion of caesarean sections was also higher in women with than without diabetes (25.4% vs. 16.6%, p < 0.001).
Our data agree with the literature, although the percentage of caesarean sections varies in different studies. Moore et al.17 analyse the cultural perception of the caesarean section and suggest that it is possible to reduce its rates. Powe et al.18 analyse gestational diabetes in relation to insulin sensitivity, reporting a higher number of caesarean sections (33.3% vs 15.2%) in patients with impaired insulin sensitivity, even after adjusting for BMI.
Regarding Apgar scores < 7, they have been associated with gestational and pregestational diabetes5,19,20 in various studies, and with higher rates of respiratory distress and admission to neonatal intensive care units. Preda et al.18 reports a 1-min Apgar < 7 in 7.8% of mothers with GD compared to 0% in mothers without GD.
Other studies have also shown macrosomia as a significant adverse effect in gestational and pregestational diabetes21,22,23,24. The longitudinal Lawlor study associated diabetes with macrosomia25. Zeng et al.26 corroborated the association between gestational and pregestational diabetes with macrosomia. Since they found a significant association between glycaemia one year before pregnancy and macrosomia, they proposed to perform a rapid glucose test during check-ups in women before they become pregnant. Other factors also associated with macrosomia are obesity, age of the pregnant mother, body mass index, hypertension, and smoking8,27.
Limitations
We believe that the sample obtained from a large population database has avoided a possible selection bias in this observational study. Since this is a retrospective study, some variables might not be well recorded in the medical history, i.e., socio-demographic data, and data regarding control of diabetes during pregnancy which can be related to complications, such as HbA1c or glycaemic control. It is currently unclear if current GD treatment guidelines can completely prevent long-term adverse effects. However, some studies have shown that treatment of GD based on different thresholds can reduce the incidence of macrosomia and other unfavourable perinatal outcomes28,29,30,31.
In our study, we have not differentiated the different subtypes of gestational diabetes. This is relevant, since we observe that women with high insulin resistance show worse metabolic parameters during complications of pregnancy, while the phenotype and outcomes of women with insulin-sensitive gestational diabetes are more similar to women without diabetes16. We believe that these clinical phenotypes may also be associated with the complications of gestational diabetes.
We have analysed the outcomes in relation to diabetes during pregnancy, however undertaking subgroup analyses, considering factors such as BMI, age, and ethnicity, could provide more detailed insights into the risks linked with gestational and pregestational diabetes.
Despite these limitations, since we have extracted a large sample from a universal health system, we believe that our data reliably reflect the complications of diabetes in our environment.
Conclusions
In conclusion, screening and diagnosis of diabetes mellitus during and before pregnancy is crucial for an appropriate management that prevents maternal and neonatal complications and reduces the chronic cardio-metabolic risk of mother and child.
Our study shows that pregestational and gestational diabetes correlate with a larger incidence of prematurity, caesarean sections, worse 1-min Apgar scores, and macrosomia.
Obesity and diabetes continue a global upward trend with negative effects on patients and society. Adhering to clinical guidelines holds significant importance for clinicians, facilitating early counseling to pregnant women concerning risk factors and requisite interventions in diabetes mellitus32.
Data availability
The data used in this study are only available for the participating researchers, in accordance with current European and national laws. Thus, the distribution of the data is not allowed. However, researchers from public institutions can request data from SIDIAP. Further information is available online (https://www.sidiap.org/index.php/en/solicituds-en).
References
American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of medical care in diabetes-2021. Diabetes Care 44(Suppl 1), S15-33 (2021).
Wdowiak, A. et al. Effect of excessive body weight and emotional disorders on the course of pregnancy and well-being of a newborn before and during COVID-19 pandemic. J. Clin. Med. 10(4), 656 (2021).
Catalano, P. M. & Shankar, K. Obesity and pregnancy: Mechanisms of short term and long term adverse consequences for mother and child. BMJ 356, j1 (2017).
HAPO Study Cooperative Research Group et al. Hyperglycaemia and adverse pregnancy outcomes. N. Engl. J. Med. 358(19), 1991–2002 (2008).
Malaza, N. et al. A systematic review to compare adverse pregnancy outcomes in women with pregestational diabetes and gestational diabetes. Int. J. Environ. Res. Public Health 19(17), 10846. https://doi.org/10.3390/ijerph191710846 (2022).
Ornoy, A., Becker, M., Weinstein-Fudim, L. & Ergaz, Z. Diabetes during pregnancy: A maternal disease complicating the course of pregnancy with long-term deleterious effects on the offspring. A clinical review. Int. J. Mol. Sci. 22(6), 2965 (2021).
Underwood, L. E. & D’Ercole, A. J. Insulin and insulin-like growth factors/somatomedins in foetal and neonatal development. Clin. Endocrinol. Metab. 13(1), 69–89 (1984).
Kc, K., Shakya, S. & Zhang, H. Gestational diabetes mellitus and macrosomia: A literature review. Ann. Nutr. Metab. 66(Suppl 2), 14–20 (2015).
Siscart, J. et al. Adherence to treatment for hypothyroidism in pregnancy and relationship with thyrotropin control: A retrospective observational cohort study. BMC Pregnancy Childbirth 22(1), 168 (2022).
Arizmendi, J., Pertuz, V. C., Colmenares, A., Hoyos, G. D. & Palomo, T. Diabetes gestacional y complicaciones neonatales. Revista Med. 20(2), 50–59 (2012).
Genaro VM, Palmira MSJ. Morbilidad y mortalidad materna y perinatal de la diabetes gestacional en una población mexicana. Eur. Sci. J. ESJ. [cited 2022 May 23];10(6). https://eujournal.org/index.php/esj/article/view/2733. (2014).
Bougherara, L., Hanssens, S., Subtil, D., Vambergue, A. & Deruelle, P. Diabetes gestacional. EMC Ginecología-Obstetricia 54(1), 1–11 (2018).
Physical activity and exercise during pregnancy and the postpartum period: ACOG committee opinion, Number 804. Obstet. Gynecol. 2020;135(4):e178–88.
Zhuang, W. et al. Adverse effects of gestational diabetes-related risk factors on pregnancy outcomes and intervention measures. Exp. Ther. Med. 20(4), 3361–3367 (2020).
Bidhendi Yarandi, R., Vaismoradi, M., Panahi, M. H., Gåre Kymre, I. & Behboudi-Gandevani, S. Mild gestational diabetes and adverse pregnancy outcome: A systemic review and meta-analysis. Front. Med. (Lausanne) 8, 699412 (2021).
Benhalima, K. et al. Characteristics and pregnancy outcomes across gestational diabetes mellitus subtyweight based on insulin resistance. Diabetologia 62(11), 2118–2128 (2019).
Moore, M. L. Increasing caesarean birth rates: A clash of cultures?. J. Perinat. Educ. 14(4), 5–8 (2005).
Powe, C. E. et al. Heterogeneous contribution of insulin sensitivity and secretion defects to gestational diabetes mellitus. Diabetes Care 39(6), 1052–1055 (2016).
Preda, A. et al. Analysis of maternal and neonatal complications in a group of patients with gestational diabetes mellitus. Medicina (Kaunas) 57(11), 1170 (2021).
Hiersch, L. et al. Gestational diabetes mellitus is associated with adverse outcomes in twin pregnancies. Am. J. Obstet. Gynecol. 220(1), 102.e1-102.e8 (2019).
Coton, S. J., Nazareth, I. & Petersen, I. A cohort study of trends in the prevalence of pregestational diabetes in pregnancy recorded in UK general practice between 1995 and 2012. BMJ Open 6(1), e009494 (2016).
Mitanchez, D. Foetal and neonatal complications in gestational diabetes: Perinatal mortality, congenital malformations, macrosomia, shoulder dystocia, birth injuries, neonatal complications. Diabetes Metab. 36(6 Pt 2), 617–627 (2010).
Yang, G. R., Dye, T. D. & Li, D. Effects of pre-gestational diabetes mellitus and gestational diabetes mellitus on macrosomia and birth defects in Upstate New York. Diabetes Res. Clin. Pract. 155, 107811 (2019).
Gualdani, E., Di Cianni, G., Seghieri, M., Francesconi, P. & Seghieri, G. Pregnancy outcomes and maternal characteristics in women with pregestational and gestational diabetes: A retrospective study on 206,917 singleton live births. Acta Diabetol. 58(9), 1169–1176. https://doi.org/10.1007/s00592-021-01710-0 (2021).
Lawlor, D. A. et al. Association of existing diabetes, gestational diabetes and glycosuria in pregnancy with macrosomia and offspring body mass index, waist and fat mass in later childhood: Findings from a prospective pregnancy cohort. Diabetologia 53(1), 89–97 (2010).
Zeng, M. et al. Association between maternal pregestational glucose level and adverse pregnancy outcomes: A population-based retrospective cohort study. BMJ Open 11(9), e048530 (2021).
Yogev, Y. & Langer, O. Pregnancy outcome in obese and morbidly obese gestational diabetic women. Eur. J. Obstet. Gynecol. Reprod. Biol. 137(1), 21–26 (2008).
Landon, M. B. et al. A multicenter, randomized trial of treatment for mild gestational diabetes. N. Engl. J. Med. 361(14), 1339–1348 (2009).
Duran, A. et al. Introduction of IADPSG criteria for the screening and diagnosis of gestational diabetes mellitus results in improved pregnancy outcomes at a lower cost in a large cohort of pregnant women: The St. Carlos Gestational Diabetes Study. Diabetes Care 37(9), 2442–2450 (2014).
Crowther, C. A. et al. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N. Engl. J. Med. 352(24), 2477–2486 (2005).
Randall, D. A., Morris, J. M., Kelly, P. & Glastras, S. J. Are newly introduced criteria for the diagnosis of gestational diabetes mellitus associated with improved pregnancy outcomes and/or increased interventions in New South Wales, Australia? A population-based data linkage study. BMJ Open Diabetes Res. Care 9(1), e002277 (2021).
Queensland Clinical Guidelines. Gestational diabetes mellitus (GDM). Guideline No. MN21.33-V6-R26. Queensland Health. http://health.qld.gov.au/qcg. (2022).
Acknowledgements
The authors would like to acknowledge Dr. Miquel Butí for his valuable contribution and support to design and create the data base.
Author information
Authors and Affiliations
Contributions
J.S and M.S. conducted the data analysis, J.A and D.P. developed the study protocol and contributed to discussion. M.O. and M.S. wrote and edited the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oros Ruiz, M., Perejón López, D., Serna Arnaiz, C. et al. Maternal and foetal complications of pregestational and gestational diabetes: a descriptive, retrospective cohort study. Sci Rep 14, 9017 (2024). https://doi.org/10.1038/s41598-024-59465-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-59465-x
- Springer Nature Limited