Abstract
This study aims to observe the hemostatic and anti-inflammatory effects of intravenous administration of tranexamic acid (TXA) in dual segment posterior lumbar interbody fusion (PLIF). The data of 53 patients with lumbar disease treated with double-segment PLIF were included in this study. The observation group was received a single-dose intravenous of TXA (1 g/100 mL) 15 min before skin incision after general anesthesia. The control group was not received TXA. The observation indicators included postoperative activated partial prothrombin time (APTT), thrombin time (PT), thrombin time (TT), fibrinogen (FIB), platelets (PLT), and postoperative deep vein thrombosis in the lower limbs, surgical time, intraoperative bleeding volume, postoperative drainage volume, transfusion rate, postoperative hospital stay, red blood cell (RBC), hemoglobin (HB), hematocrit (HCT), C-reactive protein (CRP), and erythrocyte sedimentation rate (ESR) on the 1st, 4th, 7th, and last tested day after surgery. All patients successfully completed the operation, and there was no deep vein thrombosis after operation. There was no statistically significant difference in postoperative APTT, PT, TT, FIB, PLT, surgical time, and postoperative hospital stay between the two groups (p > 0.05). The intraoperative bleeding volume, postoperative drainage volume, and transfusion rate in the observation group were lower than those in the control group, and the differences were statistically significant (p < 0.05). There was no statistically significant difference in RBC, HB, HCT, CRP, and ESR between the two groups on the 1st, 4th, 7th, and last tested day after surgery (p > 0.05). Intravenous administration of TXA in dual segment PLIF does not affect coagulation function and can reduce bleeding volume, postoperative drainage volume, and transfusion rate. Moreover, it does not affect the postoperative inflammatory response.
Similar content being viewed by others
Introduction
Posterior lumbar interbody fusion (PLIF) is currently one of the classic surgical methods for the treatment of lumbar diseases, such as lumbar disc herniation (LDH), lumbar spinal stenosis (LSS), and lumbar spondylolisthesis (LS)1,2. However, PLIF has a problem of large perioperative blood loss3,4. A previous literature had shown that the total loss of blood in dual segment PLIF could reach over 1000 mL5. Tranexamic acid (TXA), trans-4-aminomethylcyclohexanecarboxylic acid, was discovered in 1962 as a synthetic derivative of lysine6. As a plasmin inhibitor, it can inhibit the plasminogen activator by competitively inhibiting the activation of plasminogen to plasmin and play an anti-fibrinolytic effect to achieve hemostasis6. Many scholars have reported that intravenous TXA can reduce perioperative blood loss in PLIF7,8,9. The 2019 Chinese expert consensus recommended intravenous methods include single, continuous and multiple method10. Among them, intravenous of 1 g of TXA 15 min before skin incision is a typical single-dose method with a good anti-fibrinolytic effect. Besides, this application is simple to operate without interfering with intraoperative fluid, and can avoid adverse reactions between various drugs that may occur during anesthesia11.
Meanwhile, PLIF has a high risk of postoperative infection. In recent years, more and more studies have shown that in addition to the anti-fibrinolytic effect of TXA, its anti-inflammatory effect also has important clinical significance12,13. C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) are commonly used inflammatory indicators for diagnosing infection. As inflammation monitoring indexes, the specificity of CRP is 90.27%, and the sensitivity of ESR is 88.50%14. One study showed that the sensitivity of CRP and ESR in diagnosis of deep wound infection was significantly higher than those of infection-related indicators such as body temperature, local physical signs of wound, white blood cell count and neutrophil count15. However, the changes of CRP and ESR in the perioperative period of PLIF are rarely observed. In our previous study, we observed the changes of CRP and ESR during the perioperative period of single-segment PLIF with preoperative intravenous TXA16. It was unknown whether the application of single-dose TXA will affect the CRP and ESR in double-segment PLIF.
Therefore, the purpose of this study was twofold. One was to observe the hemostatic effect of intravenous method of TXA in dual segment PLIF. The second was its anti-inflammatory effect.
Method
Object of research
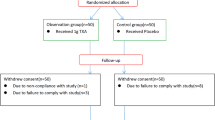
The data of patients with lumbar disease treated with PLIF from 2020.10 to 2023.4 were retrospectively collected. The inclusion criteria included a. The preoperative diagnosis was LDH, LSS and LS, and conservative treatment was ineffective. b. The American Society of Anesthesiologists rated it as Level 2. c. The scope of operation was two-segment. d. The medical records meeting the research requirements were complete. Exclusion criteria included a. There was infectious disease before the operation. b. There were blood system diseases before operation. c. There was a history of lumbar surgery. d. There was a lumbar fracture. e. There was deep venous thrombosis (DVT) of lower limbs before operation. f. There was a history of smoking or diabetes mellitus. g. Cerebrospinal fluid leakage or dural injury occurred during operation. A total of 53 cases were included in this study, including 20 males and 33 females, aged 34–80 years, with an average of (59.736 ± 11.339) years. The patients agreed to the surgery and the study was approved by the Ethics Committee of the General Hospital of Pingmei Shenma Medical Group (No. 2021004). According to whether the single-dose intravenous TXA was administered or not, they were divided into an observation group of 27 cases and a control group of 26 cases.
The calculation of sample size (N) comes from the above formula. N is the sample size, σ is the estimated standard deviation of two populations, and d is the difference between the means of two groups, α = 0.05, β = 0.2. This study conducted a double-sided test, and the table showed that \({z}_{\alpha }=1.96\), \({z}_{\beta }=0.84\). According to the results of prothrombin time (PT) in previous literature, the control group was 13.34 ± 0.99 and the observation group was 12.34 ± 0.79 after treatment. Based on the above, it is estimated that \({\sigma }^{2}=1.60\), calculated as 25 people per group, considering a 5% dropout rate, it is 26 people, which means the minimum sample size for each group is 26 people. A total of 53 people were included in this study, meeting the research needs.
The demographic data included age, gender, body mass index (BMI), disease type, activated partial thrombin time (APTT), PT, thrombin time(TT), fibrinogen (FIB), platelets (PLT), red blood cell (RBC), hemoglobin (HB), hematocrit (HCT), CRP, and ESR. There was no significant difference in the demographic data between the two groups (p > 0.05) (Table 1).
Research method
All patients were under general anesthesia. The observation group was given a single administration method, that was, intravenous single-dose of TXA (1 g/100 mL) was used 15 min before skin incision after general anesthesia. The control group was not given TXA. The surgical positions of the two groups were all in the prone position, and the operations were the same, both of which were standard double-segment PLIF. Two negative pressure drainage tubes were placed postoperatively and were removed when the volume was less than 50 mL/24 h. They received similar routine treatment measures after operation, such as antibiotics, glucocorticoids, dehydration, anticoagulation, nutritional support, wound dressing change, and monitoring of DVT of lower limbs.
Data collection
The observation indicators included postoperative APTT, PT, TT, FIB, PLT, and DVT in the lower limbs, surgical time, intraoperative bleeding volume, postoperative drainage volume, transfusion rate, postoperative hospital day, RBC, HB, HCT, CRP, and ESR on the 1st, 4th, 7th, and last tested day after surgery.
Statistical analysis
Data analysis was performed via SPSS statistical software, version 22.0. If the data follows a normal distribution, it is expressed as mean ± standard deviation, and t-test is used for comparison between the two groups. Otherwise, use M [P25; P75] to represent and compare between the two groups using Mann Whitney U test. Count data is compared between groups using chi square test. P < 0.05 indicates a statistical difference. The trend of changes in CRP and ESR during the perioperative period, represented by the median, was plotted using an Excel table.
Ethics approval and consent to participate
This study had been performed in accordance with the Declaration of Helsinki, and was approved by the Ethics Committee of the General Hospital of Pingmei Shenma Medical Group, and the reference number was 2021004. All authors confirmed that informed consent was obtained from all subjects.
Result
All patients successfully completed the operation. There was no DVT occurred after the surgery. There was no significant difference in postoperative APTT, PT, TT, FIB and PLT between the two groups (p > 0.05) (Table 2).
There was no statistically significant difference in surgical time and postoperative hospital stay of the two groups (p > 0.05). The intraoperative bleeding volume, postoperative drainage volume, and transfusion rate in the observation group were lower than those in the control group, and the difference was statistically significant (p < 0.05). There was no significant difference in RBC, HB, and HCT between the two groups on 1st, 4th, 7th, and last tested day after surgery (p > 0.05) (Table 3).
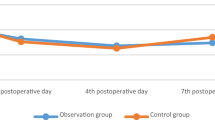
There was no significant difference in CRP and ESR between the two groups on 1st, 4th, 7th, and last tested day after surgery (p > 0.05). The comparison was shown in Table 4, Figs. 1 and 2.
Discussion
Although there have been many studies on the safety and hemostatic effects of TXA in PLIF. Most scholars reported that intravenous TXA in PLIF could effectively reduce perioperative blood loss without increasing the risk of DVT17,18,19. However, some researchers still believed that TXA could inhibit the dissolution of fibrin, thereby increasing the risk of DVT20. Therefore, if studying the intravenous use of TXA in PLIF, its safety had to be considered. In this study, all the operations of groups were successfully completed, and there were no significant abnormalities in postoperative APTT, PT, TT, FIB, and PLT. And there was no occurrence of DVT after surgery. Therefore, this indicated that intravenous TXA was safe and feasible in dual segment PLIF.
In our early study, we found that preoperative intravenous administration of TXA in single segment PLIF could reduce intraoperative bleeding and postoperative drainage16. This study found that intravenous application of TXA in dual segment PLFI could also reduce intraoperative bleeding, postoperative drainage, and transfusion rate. This was similar to the research results of Gao et al.21. Generally speaking, reducing intraoperative bleeding also reduces the need for intraoperative hemostasis. This will make the field of view of the surgical area clearer, more conducive to surgical operations, and ultimately reduce surgical time. However, different scholars have different results in terms of surgical time. Qiao et al. found that the application of TXA in PLIF could shorten surgical time22. This study found that the application of TXA did not reduce the operative time, which was similar to the results of Wang et al.23. It was worth noting that different results were obtained in the research of Sun et al.24. They applied 1g of TXA intravenously 15 min before the PLIF surgery, which extended the surgical time compared to the control group who did not use TXA. The reasons for the inconsistency in the above surgical time were related to the following points. One was related to the sample size of the study. When the sample size was small, it may inevitably cause bias in the research results. The second was related to the cooperation between surgical techniques and surgical teams. In theory, a decrease in intraoperative bleeding and postoperative drainage volume will maintain high levels of postoperative RBC, HB, and HCT. However, our study yielded different results. There was no significant difference in RBC, HB, and HCT measured at different time after surgery between the two groups. We believed that there might be two reasons for this result. Firstly, although TXA had a hemostatic effect, its half-life was short, and its sustained hemostatic effect was only a few hours. Secondly, although the control group had a large amount of intraoperative bleeding, the transfusion rate was higher. These combined results resulted in the control group maintaining higher levels of RBC, HB, and HCT. Meanwhile, the above might also be reasons for the similar postoperative hospital stay between the two groups.
With the successful application of TXA as a hemostatic in orthopedic surgery in recent years, more and more studies have shown that TXA has potential anti-inflammatory effects25,26,27,28. Therefore, we observed the anti-inflammatory effect of single dose intravenous injection of TXA on dual segment PLIF. CRP and ESR are commonly used indicators in clinical practice to monitor inflammatory reactions. CRP is an acute phase protein synthesized by hepatocytes with high specificity and sensitivity. When the body is stimulated by inflammation, it can mobilize complement and immune cells to clear pathogens and injured cells29. The half-life of CRP is 15 h, and the CRP value of 99% of healthy people is less than 10 mg/L30. One study found that CRP could be 10 to 60 times higher than the normal value when it was elevated, and then decreased rapidly, generally 14 days after surgery, to a normal value of 6.76 mg/L31. ESR is also one of commonly used clinical indicators to monitor postoperative infection after PLIF32. When the body suffers from infection, tissue damage, necrosis, or some disease activity, progression, and deterioration, the plasma resistance of RBC after they overlap each other in a "coin" shape decreases, thereby accelerating ESR33. ESR will increase within one week after lumbar spine surgery, and return to the baseline level in two to three weeks, but more than 25 mm/h34. A study has shown that combined monitoring of CRP and ESR in spinal surgery conduced to improve the diagnosis rate of postoperative infection35.
A study has showed that in orthopedic surgery, the anti-inflammatory effect of TXA was shown by the lower CRP level of patients who received TXA compared with those who not or small dose36. However, the specific anti-inflammatory mechanism of TXA is still unclear. Different researchers put forward different opinions according to their research results. Later et al.37 studied the mRNA expression level of 114 inflammatory genes before and after operation, and found that TXA could reduce the expression of inflammatory genes. Kong et al.38 believed that the anti-inflammatory mechanism of TXA may be that it could protect PLT by reducing the degradation of PLT membrane glycoprotein receptors by plasmin. By protecting PLT, the activation, adhesion and aggregation of PLT can be inhibited, and at the same time, it could effectively inhibit the aggregation of leukocytes and the activation of endothelial cells, so as to reduce the inflammatory reaction. Although fibrinolysis and inflammation are independent processes in many aspects, it is likely that there is a close relationship between fibrinolysis and inflammation system39. Among them, fibrinolytic enzyme plays an important role in the process of connecting the fibrinolytic system and the inflammatory system, and it can stimulate the release of cytokines and other inflammatory mediators of different cell types to promote inflammation40. In addition, fibrinolytic enzyme can activate the body's complement system by cracking the complement protein while playing the role of thrombolysis. After the activation of the complement system, it can cause and strengthen the inflammatory response through chemotaxis, anaphylaxis, kininoid action and other ways41. However, we found that there were three problems, when study the anti-inflammatory mechanism of TXA. First, there were few reports on the anti-inflammatory effects of TXA, and most of them are analyzed around joint surgery. Second, when reviewing previous studies, it was found that the dosage, frequency and timing of TXA were not completely consistent, and these factors might affect the anti-inflammatory effect of TXA. Third, there were many indicators used to display inflammatory reaction, such as interleukin, tumor necrosis factor, CRP and ESR, while different inflammatory factors may reflect different inflammatory reactions. Therefore, the anti-inflammatory mechanism of TXA still needs further study.
In this study, preoperative intravenous administration of 1 g of TXA did not significantly affect the postoperative CRP and ESR. This is similar to our previous results in single-segment PLIF with the same dose and administration method16. The research results of Yuan et al.20 showed that the postoperative inflammation indicators of patients who received TXA were lower than those of the control group who did not receive TXA, and the improvement of inflammation indicators after multiple used of TXA was better than that of single dose used. Meanwhile, a study has also shown that as the dosage of TXA increases, its anti fibrinolytic effect was enhanced, which might have potential anti-inflammatory effects42. However, this study yielded different results, with no significant difference in postoperative CRP and ESR between the two groups. There may be two reasons for the different results mentioned above. On the one hand, we only used 1 g of TXA once before surgery, which belonged to low dose single application, and might not reach the sign of causing detectable anti-inflammatory reaction. Because the anti-inflammatory effect of TXA in the perioperative period had a significant positive correlation with the times and dose of TXA39. On the other hand, the use of antibiotics during the perioperative period might mask the anti-inflammatory effect of TXA, which was difficult to be detected.
Conclusion
In summary, through this study, intravenous infusion of TXA is safe and feasible in dual segment PLIF. It does not affect coagulation function and can reduce intraoperative bleeding, postoperative drainage, and transfusion rate. Moreover, it does not affect the postoperative inflammatory response, especially CRP and ESR. However, this study also has some limitation, as it is a retrospective and relatively small sample size study. In the future, further research will be conducted.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- TXA:
-
Tranexamic acid
- PLIF:
-
Posterior lumbar interbody fusion
- APTT:
-
Activated partial thromboplastin time
- PT:
-
Prothrombin time
- TT:
-
Thrombin time
- FIB:
-
Fibrinogen
- PLT:
-
Platelets
- RBC:
-
Red blood cell
- HB:
-
Hemoglobin
- HCT:
-
Hematocrit
- CRP:
-
C-reactive protein
- ESR:
-
Erythrocyte sedimentation rate
- BMI:
-
Body mass index
- LSS:
-
Lumbar spinal stenosis
- LDH:
-
Lumbar disc herniation
- LS:
-
Lumbar spondylolisthesis
- DVT:
-
Deep venous thrombosis
References
Mobbs, R. J., Phan, K., Malham, G., Seex, K. & Rao, P. J. Lumbar interbody fusion: Techniques, indications and comparison of interbody fusion options including PLIF, TLIF, MI-TLIF, OLIF/ATP, LLIF and ALIF. J. Spine Surg. (Hong Kong) 1, 2–18. https://doi.org/10.3978/j.issn.2414-469X.2015.10.05 (2015).
Martin, B. I. et al. Trends in lumbar fusion procedure rates and associated hospital costs for degenerative spinal diseases in the United States, 2004 to 2015. Spine 44, 369–376. https://doi.org/10.1097/brs.0000000000002822 (2019).
Bouloussa, H. et al. Is it safe to perform lumbar spine surgery on patients over eighty five?. Int. Orthop. 41, 2091–2096. https://doi.org/10.1007/s00264-017-3555-6 (2017).
Machado, G. C. et al. Trends, complications, and costs for hospital admission and surgery for lumbar spinal stenosis. Spine (Phila Pa 1976). 42, 1737–1743. https://doi.org/10.1097/brs.0000000000002207 (2017).
Zhang, Y. et al. Comparison of blood loss of two different laminectomy decompression in treatment of patients with lumbar degenerative disc disease. J. PLA Med. College 37, 328–331. https://doi.org/10.3969/j.issn.2095-5227.2016.04.008 (2016).
Zhang, Y., Pan, J. & Liu, W. Application of tranexamic acid in spinal surgery. J. Gannan. Med. Univ. 39, 964–966. https://doi.org/10.3969/j.issn.1001-5779.2019.09.025 (2019).
Yan, L. et al. Impact of the tranexamic acid on bleeding amount of surgical patient with degenerative spinal disease: A randomized blinded study. Front. Surg. 8, 655692. https://doi.org/10.3389/fsurg.2021.655692 (2021).
Li, J., Wang, L., Bai, T., Liu, Y. & Huang, Y. Combined use of intravenous and topical tranexamic acid efficiently reduces blood loss in patients aged over 60 operated with a 2-level lumbar fusion. J. Orthop. Surg. Res. 15, 339 (2020).
Hao, S. et al. The effect of intravenous unit-dose tranexamic acid on visible and hidden blood loss in posterior lumbar interbody fusion: a randomized clinical trial. Sci. Rep. 13, 4714. https://doi.org/10.1038/s41598-022-27307-3 (2023).
Zhou, Z. et al. Expert consensus on the application of tranexamic acid and anticoagulant for the enhanced recovery after orthopedic surgery in China. Chin. J. Bone Joint Surg. 12, 81–88. https://doi.org/10.3969/j.issn.2095-9958.2019.02.01 (2019).
Yuan, J. et al. Effect of adequate amount of tranexamic acid before operation on blood loss and safety in posterior lumbar fusion with multiple segments. Chin. J. Blood Transfus. 34, 43–47. https://doi.org/10.13303/j.cjbt.issn.1004-549x.2021.01.014 (2021).
Wybraniec, M. T. et al. Atrial fibrillation and rapid atrial pacing are linked to enhanced inflammation and altered fibrinolysis in patients undergoing pulmonary vein isolation. Kardiologia Polska 77, 386–388. https://doi.org/10.5603/KP.a2018.0246 (2019).
Zeng, W. N. et al. Low-dose epinephrine plus tranexamic acid reduces early postoperative blood loss and inflammatory response: A randomized controlled trial. J. Bone Joint Surg. Am. 100, 295–304. https://doi.org/10.2106/jbjs.16.01585 (2018).
Wang, L. et al. Clinical significance of PCT, CRP, ESR, WBC count as predictors in postoperative early infections complications with fever after posterior lumbar internal fixation. Chin. J. Orthop. Trauma 28, 66–70. https://doi.org/10.3969/j.issn.1003-0034.2015.01.017 (2015).
Ma, X. et al. Early diagnosis of deep infection after spine surgeries. Chin. J. Spine Spinal Cord 25, 971–976. https://doi.org/10.3969/j.issn.1004-406X.2015.11.03 (2015).
Hao, S. et al. RBC, HB, HCT, CRP, and ESR at different postoperative periods after the application of intravenous unit dose transient acid in PLIF: A case control study. 9, 1032376, https://doi.org/10.3389/fsurg.2022.1032376 (2023).
Hao, S. et al. Safety and efficacy of an intravenous unit dose of tranexamic acid in posterior lumbar interbody fusion. J. Biol. Regulat. Homeostatic Agents 37, 3903–3909. https://doi.org/10.23812/j.biol.regul.homeost.agents.20233707.385 (2023).
Wang, F. et al. The effectiveness and safety of tranexamic acid in posterior lumbar interbody fusion: A placebo-controlled randomized study. Chin. J. Spine Spinal Cord 29, 422–430. https://doi.org/10.3969/j.issn.1004-406X.2019.05.06 (2019).
Xiao, K., Zhuo, X., Peng, X., Wu, Z. & Li, B. The efficacy and safety of tranexamic acid in lumbar surgery: A meta-analysis of randomized-controlled trials. Jt. Dis. Relat. Surg. 33, 57–85. https://doi.org/10.52312/jdrs.2022.432 (2022).
Yuan, Z., Wu, Y., Bao, E., Zhang, L. & Chen, X. Effects of repeated intravenous administration of tranexamic acid on inflammation and the coagulation-fibrinolytic system of patients with posterior lumbar spinal instrumentation fusion. Med. J. West China 35, 1158–1162. https://doi.org/10.3969/j.issn.1672-3511.2023.08.012 (2023).
Ga, Y., Wang, Y., Liu, Y., Yang, X. & Wu, K. Analysis on efficacy of using tranexamic acid before and after double-segment PLIF in reducing perioperative blood loss. Chongqing Med. 51, 2079–2083. https://doi.org/10.3969/j.issn.1671-8348.2022.12.022 (2022).
Li, Q., Qiao, X., Li, J. A., Liu, S. & Xiao, Y. Meta-analysis of the safety and effectiveness of tranexamic acid in the posterior lumbar interbody fusion technique. Chin. J. Spine Spinal Cord 31, 238–246. https://doi.org/10.3969/j.issn.1004-406X.2021.03.07 (2021).
Wang, Q. et al. Tranexamic acid reduces postoperative blood loss of degenerative lumbar instability with stenosis in posterior approach lumbar surgery: A randomized controlled trial. Eur. Spine J. 22, 2035–2038. https://doi.org/10.1007/s00586-013-2836-z (2013).
Sun, Z. et al. Clinical trial of tranexamic acid in posterior decompression and fusion internal fixation for the treatment of lumbar degenerative diseases. Chin. J. Clin. Pharmacol. 39, 336–340. https://doi.org/10.13699/j.cnki.1001-6821.2023.03.008 (2023).
Yu, Z., Yao, L. & Yang, Q. Tranexamic acid plus diluted-epinephrine versus tranexamic acid alone for blood loss in total joint arthroplasty: A meta-analysis. Medicine (Baltimore). 96, e7095. https://doi.org/10.1097/md.0000000000007095 (2017).
Mi, B. et al. Is combined use of intravenous and intraarticular tranexamic acid superior to intravenous or intraarticular tranexamic acid alone in total knee arthroplasty? A meta-analysis of randomized controlled trials. J. Orthop. Surg. Res. 12, 61. https://doi.org/10.1186/s13018-017-0559-2 (2017).
Wang, F. et al. The efficacy and safety of multiple-dose intravenous tranexamic acid in reducing perioperative blood loss in patients with thoracolumbar burst fracture. Clin. Neurol. Neurosurg. 193, 105766. https://doi.org/10.1016/j.clineuro.2020.105766 (2020).
Zhang, S. et al. Tranexamic acid attenuates inflammatory effect and modulates immune response in primary total knee arthroplasty: A randomized, placebo-controlled, pilot trial. Inflammopharmacology 28, 839–849. https://doi.org/10.1007/s10787-020-00695-6 (2020).
Ren, S. Dynamic changes of CRP and ESR before and after spinal internal fixation and its clinical significance. Mod. Diagn. Treat 29, 1969–1971. https://doi.org/10.3969/j.issn.1001-8174.2018.12.063 (2018).
Pelosi, P. et al. Prognostic role of clinical and laboratory criteria to identify early ventilator-associated pneumonia in brain injury. Chest 134, 101–108. https://doi.org/10.1378/chest.07-2546 (2008).
Su, X., Shang, X. & Zhang, H. The regularity of the levels of CRP and ESR in patients before and after operation of spinal internal fixation and their clinical significance. J. Cervicodynia. Lumbodynia. 36(278–282), 287. https://doi.org/10.3969/j.issn.1005-7234.2015.04.006 (2015).
Brigden, M. The erythrocyte sedimentation rate. Still a helpful test when used judiciously. Postgrad. Med. 103, 257–262, 272–254. https://doi.org/10.3810/pgm.1998.05.493 (1998).
Ma, F., Tian, Z., Song, X. & Wang, C. The clinical significance of combined detection of C-reactive protein, erythrocyte sedimentation rate and procalcitonin in diagnosing early postoperative infection after lumbar operation. J. Xinjiang Med. Univ. 604–607, 611. https://doi.org/10.3969/j.issn.1009-5551.2015.05.021 (2015).
Wang, H., Wang, Y., Lu, L. & Li, F. Clinical value of monitoring of CRP, PCT, ESR, and WBC in diagnosis of early infection after posterior lumbar internal fixation. Chin. J. Nosocomiol. 26, 5175–5178. https://doi.org/10.11816/cn.ni.2016-161841 (2016).
Niu, B., Wang, S. & Sun, Y. Expression of C-reactive protein and erythrocyte sedimentation rate in elderly patients patients before and after internal fixation of spine. Chin. J. Laborat. Diagn. 20, 291–293 (2016).
Okholm, S. H., Krog, J. & Hvas, A. M. Tranexamic acid and its potential anti-inflammatory effect: A systematic review. Semin. Thrombosis Hemostasis 48, 568–595. https://doi.org/10.1055/s-0042-1742741 (2022).
Later, A. F. et al. Antifibrinolytics attenuate inflammatory gene expression after cardiac surgery. J. Thorac. Cardiovasc. Surg. 145(1611–1616), 1616.e1611–1614. https://doi.org/10.1016/j.jtcvs.2012.11.042 (2013).
Kong, G., Ma, W. & Zhang, S. The effect on CRP after TKA which topically use tranexamic acid combine with glucocorticoid. Chin. J. Geriatr. Orthop. Rehabil. (Electronic Edition) 5, 317–321. https://doi.org/10.3877/cma.j.issn.2096-0263.2019.06.003 (2019).
Xu, H., Xie, J., Lei, Y., Chen, G. & Pei, F. Research progress on the anti-inflammatory effects of dexamethasone and tranexamic acid in total joint arthroplasty. Chin. J. Bone Joint 9, 439–444. https://doi.org/10.3969/j.issn.2095-252X.2020.06.009 (2020).
Burysek, L., Syrovets, T. & Simmet, T. The serine protease plasmin triggers expression of MCP-1 and CD40 in human primary monocytes via activation of p38 MAPK and janus kinase (JAK)/STAT signaling pathways. J. Biol. Chem. 277, 33509–33517. https://doi.org/10.1074/jbc.M201941200 (2002).
Barrett, C. D. et al. Tranexamic acid mediates proinflammatory and anti-inflammatory signaling via complement C5a regulation in a plasminogen activator-dependent manner. J. Trauma Acute Care Surg. 86, 101–107. https://doi.org/10.1097/ta.0000000000002092 (2019).
Zhang, S. et al. The effects of multiple intravenous tranexamic acid administrations after total knee arthroplasty on fibrinolytic activity and inflammatory response. Chin. J. Orthop. 37, 1483–1489. https://doi.org/10.3760/cma.j.issn.0253-2352.2017.23.006 (2017).
Acknowledgements
The authors thank Department of Anesthesia and Perioperative Medicine, General Hospital of Pingmei Shenma Medical Group, they contributed to the manuscript.
Funding
Project supported by Hainan Province Clinical Medical Center (No. 0202067). Scientific research project of General Hospital of Pingmei Shenma Medical Group (No. 4104022021180717).
Author information
Authors and Affiliations
Contributions
S.H. wrote the first draft of the article. S.L. and B.L. critically reviewed and edited drafts. S.L. and H.L. diagnosed the patients. S.H., S.L. and H.L. treated the patient. S.D. and X.C. have made substantial contributions to the conception and designed of the manuscript. Z.Z. followed up the patients. All authors read and approved the final manuscript. All the participants agreed to publish their identifiable data in an online, open-access journal. The authors consent to publish was obtained from the participants.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hao, S., Luo, S., Zhao, Z. et al. The hemostatic and anti-inflammatory effects of intravenous single-dose of tranexamic acid in double-segment posterior lumbar interbody fusion: a case control study. Sci Rep 14, 12800 (2024). https://doi.org/10.1038/s41598-024-62823-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-62823-4
- Springer Nature Limited