Abstract
Physical activity is regarded as favorable to health but effects across the spectrum of human disease are poorly quantified. In contrast to self-reported measures, wearable accelerometers can provide more precise and reproducible activity quantification. Using wrist-worn accelerometry data from the UK Biobank prospective cohort study, we test associations between moderate-to-vigorous physical activity (MVPA) – both total MVPA minutes and whether MVPA is above a guideline-based threshold of ≥150 min/week—and incidence of 697 diseases using Cox proportional hazards models adjusted for age, sex, body mass index, smoking, Townsend Deprivation Index, educational attainment, diet quality, alcohol use, blood pressure, anti-hypertensive use. We correct for multiplicity at a false discovery rate of 1%. We perform analogous testing using self-reported MVPA. Among 96,244 adults wearing accelerometers for one week (age 62 ± 8 years), MVPA is associated with 373 (54%) tested diseases over a median 6.3 years of follow-up. Greater MVPA is overwhelmingly associated with lower disease risk (98% of associations) with hazard ratios (HRs) ranging 0.70–0.98 per 150 min increase in weekly MVPA, and associations spanning all 16 disease categories tested. Overall, associations with lower disease risk are enriched for cardiac (16%), digestive (14%), endocrine/metabolic (10%), and respiratory conditions (8%) (chi-square p < 0.01). Similar patterns are observed using the guideline-based threshold of ≥150 MVPA min/week. Some of the strongest associations with guideline-adherent activity include lower risks of incident heart failure (HR 0.65, 95% CI 0.55–0.77), type 2 diabetes (HR 0.64, 95% CI 0.58–0.71), cholelithiasis (HR 0.61, 95% CI 0.54–0.70), and chronic bronchitis (HR 0.42, 95% CI 0.33–0.54). When assessed within 456,374 individuals providing self-reported MVPA, effect sizes for guideline-adherent activity are substantially smaller (e.g., heart failure HR 0.84, 95% CI 0.80–0.88). Greater wearable device-based physical activity is robustly associated with lower disease incidence. Future studies are warranted to identify potential mechanisms linking physical activity and disease, and assess whether optimization of measured activity can reduce disease risk.
Similar content being viewed by others
Introduction
Physical activity may have important health benefits1, though the effects of physical activity across the range of human disease are poorly quantified. Examining relations between activity and disease risk may provide a comprehensive understanding of the benefits of physical activity, a modifiable lifestyle behavior. Past studies have generally assessed physical activity using self-report data2,3,4,5, which are subject to recall bias and correlate only modestly with measured energy expenditure6. Studies that have measured activity using wearable sensors have commonly quantified proprietary step counts7 or continuous acceleration8,9, which may be difficult to contextualize in terms of consensus activity recommendations, which typically recommend specific quantities of moderate-to-vigorous physical activity (MVPA)1,10,11. Past studies have also typically assessed only a limited set of outcomes (e.g., mortality)9,12, or focused only on prevalent disease13.
To address these challenges, we examine a unique, large prospective cohort study, the UK Biobank, which comprises over 90,000 individuals wearing wrist-worn triaxial accelerometers for one week. Our aim is to systematically identify associations between physical activity and a comprehensive array of incident human diseases, both to inform future research investigating potential causal mechanisms and to guide preventive efforts leveraging physical activity to reduce disease incidence. The use of wearable accelerometer-based physical activity measurements allows for precise and reproducible8 ascertainment of physical activity, quantified as minutes of MVPA and also classified into binary categories divided at a guideline-recommended threshold of ≥150 min of MVPA per week1,10,11. Utilizing linkage to national health records, we comprehensively assess associations between measured activity and longitudinal incidence of roughly 700 conditions spanning the full spectrum of human disease. We observe that device-measured activity is associated with lower risk of more than 350 incident conditions spanning the full spectrum of human disease, and that measured activity is a stronger indicator of disease risk as compared to self-reported activity obtained within the same population. Our findings will inform future work to identify mechanisms linking physical activity and disease, and suggest that efforts to optimize measured physical activity may result in lower disease incidence.
Results
Measured activity sample
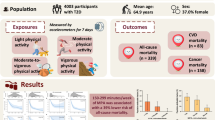
After removing individuals whose accelerometer measurements failed quality control metrics, we performed disease association testing in 96,244 individuals (Fig. 1). The mean age was 62 ± 8 years and 56% were female. Individuals had a median MVPA of 135 min/week (quartile 1:60, quartile 3:250) and 46% of individuals achieved guideline-recommended levels. MVPA distributions are shown in Supplementary Fig. 1. Detailed sample characteristics are shown in Table 1.
Depicted is a graphical overview of the study. Within 96,244 UK Biobank participants who wore a wrist-worn accelerometer for one week, we tested for associations between objectively measured moderate-to-vigorous physical activity and 697 incident diseases. We performed analogous association testing in 456,374 UK Biobank participants who provided self-reported activity data. There was overlap between the two analysis samples, as depicted by the Venn diagram in the center.
Associations between measured activity and incident disease
At a median follow-up of 6.2 years (quartile 1:5.7, quartile 3:6.7), MVPA was associated with risk of 373 out of 697 (54%) incident diseases tested at an FDR of 1% (Fig. 2a). Of the significant associations, 367 (98%) indicated a lower risk of disease with greater MVPA (hazard ratio [HR] range 0.70–0.98 per 150-min increase in weekly MVPA). Some of the strongest associations included lower risks of atherosclerosis (HR 0.57, 95% CI 0.44–0.74), type 2 diabetes (HR 0.74, 95% CI 0.70–0.79), chronic bronchitis (HR 0.44, 95% CI 0.37–0.53), and depression (HR 0.84, 95% CI 0.79–0.88) (Supplementary Fig. 2). Among all significant associations with lower disease risk, most conditions represented cardiac (16%), digestive (14%), endocrine/metabolic (10%), and respiratory diseases (8%), although associations were observed in all categories tested (chi-square p < 0.01, Supplementary Figs. 3 and 4). The distribution of effect sizes varied by disease category, with the lowest median hazard ratios (i.e., largest effects) observed for endocrine/metabolic, respiratory, and infectious diseases (Fig. 2b).
a Plots the negative log10 p value for the association between accelerometer-measured moderate-to-vigorous physical activity (MVPA) and incident disease (grouped by category on the x-axis) in Cox proportional hazards models adjusted for age, sex, body mass index, Townsend Deprivation Index, smoking status, alcohol use, anti-hypertensive medication use, systolic blood pressure, and diastolic blood pressure, with darker shaded points meeting significance at a false discovery rate (FDR) of 1% (horizontal red line). Upward-facing triangles represent higher risk (hazard ratios >1), while downward-facing triangles represent lower risk (hazard ratio <1). P values smaller than 1 × 10−20 are displayed as 1 × 10−20 for graphical purposes. b Shows the distribution of hazard ratios per 150-min increase in MVPA per week observed across each disease category (x-axis), with the center line depicting the within-category median hazard ratio, the bounds of the box representing quartile 1 to quartile 3, and the whiskers extending 1.5 interquartile ranges beyond the box. Categories are arranged by increasing median hazard ratio, from lowest (left) to highest (right). c Compares the median within-category hazard ratio observed with guideline-adherent activity (i.e., ≥150 min of MVPA per week1,10,11) defined using accelerometer-measured (circles) versus self-reported (triangles) MVPA. Included in the comparison are 323 diseases for which there was a nominally significant association (p < 0.05) with both exposure definitions and an effect size suggesting a lower risk of disease.
There were six associations between greater MVPA and higher risk of disease (HR range 1.08–1.24), and all represented injuries/poisonings, musculoskeletal, and dermatologic conditions. Examples included greater risk of disorders of muscle, ligament, and fascia (HR 1.09, 95% CI 1.03–1.15) and fracture of the radius or ulna (HR 1.09, 95% CI 1.02–1.15).
Overall, we obtained similar results when categorizing MVPA at the ≥150 min per week threshold recommended in consensus guidelines1,10,11 (306 associations with lower risk of disease, HR range 0.11–0.91, Supplementary Fig. 5). Some of the strongest associations with guideline-based physical activity included lower risks of heart failure (HR 0.65, 95% CI 0.55–0.77), type 2 diabetes (HR 0.64, 95% CI 0.58–0.71), cholelithiasis (HR 0.61, 95% CI 0.54–0.70), and chronic bronchitis (HR 0.42, 95% CI 0.33–0.54). Plots of the 5-year cumulative risk of these four conditions demonstrated consistent and substantial separation of longitudinal disease incidence on the basis of accelerometer-derived guideline-adherent activity (Fig. 3). Multivariable adjusted cumulative risk curves for men and women were similar and are shown in Supplementary Figs. 6 and 7.
Depicted is the 5-year cumulative risk of heart failure, type 2 diabetes, cholelithasis, and chronic bronchitis, stratified guideline-adherent activity according to accelerometer-measured moderate-to-vigorous physical activity (MVPA, top panels) and self-reported MVPA (bottom panels). In each plot, individuals are grouped into binary categories according to the guideline-based threshold of ≥150 min of MVPA/week. Red, pink, blue, and purple strata represent individuals meeting guideline-based levels, and the teal stratum represents individuals not meeting guideline-based levels1,10,11. In each plot, the number remaining at risk over time is depicted below. Representative diseases were selected from the four categories having the greatest enrichment for associations with activity, where each disease was significantly associated with both accelerometer-measured and self-reported activity at a false discovery rate of 1%.
For most conditions, risk of disease was lowest at higher MVPA levels (Fig. 4 and Supplementary Fig. 8). Nevertheless, for a few conditions enriched within certain disease categories (e.g., musculoskeletal, injuries/poisonings), risk was lowest at intermediate MVPA levels. The pattern of associations observed for alternative MVPA thresholds (i.e., ≥75 and ≥300 MVPA min/week) was generally similar to that seen at the ≥150 min per week threshold, although the total number of significant associations at ≥300 MVPA min/week was smaller (Supplementary Fig. 9).
Depicted is the relative hazard of incident disease according to quintile of accelerometer-measured moderate-to-vigorous physical activity (MVPA) and grouped by disease category. Each disease is represented by four points, with each point representing the hazard ratio associated with a given quintile of MVPA (quintile 2 = red, quintile 3 = orange, quintile 4 = light green, quintile 5 = dark green), as compared to the lowest quintile (quintile 1) as the referent. MVPA volumes corresponding to each quintile are shown in the legend. The hashed horizontal line depicts a hazard ratio of one (i.e., equal hazard to quintile 1). A single value below 0.1 was rounded to 0.1 for graphical purposes (bottom right).
Self-reported activity
Within 456,374 UK Biobank participants providing questionnaire-based activity data (Table 1 and Fig. 1), self-reported MVPA was also generally associated with lower risk of disease, although there were fewer associations overall (Supplementary Figs. 3, 4, 10, and 11). As compared to accelerometer-derived MVPA, self-reported MVPA had several more associations indicating a higher risk of disease, enriched for musculoskeletal conditions (e.g., localized osteoarthrosis, HR 1.02, 95% CI 1.01–1.04) and injuries/poisonings (e.g., joint/ligament sprain, HR 1.02, 95% CI 1.02–1.03) (Supplementary Figs. 3,4, 10, and 11). When assessing guideline-adherent activity, many of the associations observed with accelerometer-derived activity were again seen, but effect sizes were smaller. For example, self-reported guideline-adherent activity was also associated with lower risks of heart failure (HR 0.84, 95% CI 0.80–0.88), type 2 diabetes (HR 0.85, 95% CI 0.83–0.87), cholelithiasis (HR 0.86, 95% CI 0.84–0.89), and chronic bronchitis (HR 0.70, 95% CI 0.67–0.75) (Figs. 2c and 3). Association results for diseases related to both accelerometer-measured and self-reported guideline-adherent activity are summarized in Table 2.
Secondary analyses
We repeated disease association testing using overall mean acceleration, which demonstrated a similar pattern of associations as accelerometer-derived MVPA (Supplementary Fig. 12). Greater vigorous activity was also generally associated with lower disease risk, although the total number of associations was smaller (Supplementary Figs. 13–15). The observed pattern of associations between measured MVPA and incident disease was consistent across subgroups of age (i.e., age <55, age 55–64, and age ≥65, Supplementary Fig. 16). Associations with measured MVPA were similar when only hospital data (i.e., excluding general practitioner data) were used to define outcomes (n = 343 significant associations, Supplementary Fig. 17). There were a greater number of significant associations in models without BMI, blood pressure, or anti-hypertensive use as covariates (n = 500, Supplementary Fig. 18). In a landmark analysis in which person-time began two years after accelerometer wear, associations with measured MVPA were similar, although the total number was somewhat smaller (n = 259 significant associations, Supplementary Fig. 19). Disease-level association results from the primary and secondary analyses are available in the Supplementary Data.
Discussion
In summary, within over 90,000 individuals wearing a wrist-based activity sensor over the course of one week, we quantified associations between measured physical activity (quantified as MVPA) and future risk of nearly 700 diseases. We observed strong associations between physical activity and hundreds of conditions enriched for cardiac, digestive, endocrine/metabolic, respiratory and other diseases. Achievement of guideline-adherent activity levels was overwhelmingly associated with lower disease risk. At the same time, greater MVPA was associated with progressively lower disease risk both below and above guideline-based thresholds. Although self-reported MVPA was also associated with lower disease risk, both the total number of associations and magnitude of effect sizes were greater using accelerometer-measured MVPA. Overall, our results prioritize a number of diseases for future study to better define mechanisms by which physical activity may affect disease risk, and suggest that wearable sensors may be important tools for evaluating efforts to modify disease risk using physical activity.
Our findings support and extend previous observations linking physical activity with lower risk of disease. Multiple studies have demonstrated associations between greater physical activity and lower risk of disease, primarily cardiometabolic conditions2,14. Importantly, however, most previous studies have relied on self-reported data, which are subject to recall bias and measurement imprecision. In contrast, wearable accelerometers provide a mechanism for precise and reproducible activity measurement. Recently, accelerometer-measured physical activity has been associated with lower risks of cardiovascular12 and neurological disease15, as well as overall mortality9. Our findings extend prior results by quantifying associations with accelerometer-measured activity across a broad spectrum of disease, exploring physical activity dose-response relationships, and benchmarking the relative utility of device-measured versus self-reported activity as a marker of disease risk.
Our results suggest that achievement of guideline-recommended physical activity levels may be an important marker of substantially lower risk of a wide range of future diseases. Whether assessed as a continuous variable or dichotomized at a guideline-recommended threshold, greater MVPA was associated with lower risk of over 350 diseases. Although associations were enriched for cardiac, digestive, endocrine/metabolic, and respiratory conditions, the breadth of associations observed spanned every category tested. For example, individuals meeting guideline-recommended levels of accelerometer-derived MVPA had a 32% lower risk of an incident sleep disorder, which is consistent with randomized trial evidence supporting a beneficial role of exercise on sleep16. Similarly, guideline-recommended MVPA was associated with a 40% lower risk of cholelithiasis, which may be related to beneficial effects on gut motility17. Despite evaluating nearly 700 conditions, we observed only six significant associations in which activity was related to higher disease risk, with each representing a musculoskeletal disorder, injury, or dermatologic condition (e.g., corns and calluses), and potentially attributable to use-related degeneration.
Importantly, greater levels of measured activity—even if not to the point of achieving guideline-recommended levels—may be beneficial. When assessing dose-response relationships within the roughly 350 diseases significantly associated with measured MVPA, achievement of progressively higher quintiles of MVPA appeared monotonically associated with lower disease risk, with the lowest risk observed in the highest quintile. Similarly, we observed that achievement of activity at thresholds below (≥75 min of MVPA/week) and above (≥300 min of MVPA/week) the guideline-based threshold of ≥150 min1,10,11 were associated with lower disease risk. Importantly, current guidelines are largely based on self-reported activity data1,10,11, and the information content of accelerometer-derived activity may differ substantively from self-reported measures, potentially in a manner dependent upon which specific activities are being quantified18. As a result, future work is needed to define appropriate thresholds for accelerometer-derived activity. Nevertheless, our findings suggest that, in general, greater device-measured activity levels appear broadly associated with lower risks of most human diseases.
Our results suggest that wearable accelerometer-based activity measurement may play an important role in future public health efforts focused on physical activity. Prior reports have suggested that self-reported activity may be biased5, and therefore a weak surrogate for measured activity6. Indeed, recent evidence suggests that device-based activity measures may be a more powerful predictor of cardiovascular outcomes19 and mortality5 than self-reported activity within the same population. We similarly observed substantially stronger effect sizes and more substantial risk stratification when assessing guideline-adherent activity using accelerometer-derived as opposed to self-reported MVPA. Interestingly, although the distribution of significant associations was qualitatively similar using accelerometer-derived versus self-reported data, self-reported data suggested a considerably greater number of associations indicating greater disease risk, primarily comprising musculoskeletal conditions and injuries/poisonings. Future work is warranted to better understand potential differences in information content between measured and self-reported physical activity, and identify optimal methods for leveraging activity measurement to prevent disease.
Our study should be considered in the context of design. First, physical activity was measured for only one week. It is possible that a longer duration of monitoring would have led to more accurate classification of activity. Second, our comparisons between measured and self-reported activity may be susceptible to biases introduced by varying follow-up time and temporal differences in exposure ascertainment. Third, since our aim was to identify diseases for which physical activity may plausibly affect risk to generate hypotheses and inform future studies, we controlled for substantive confounders and took steps to explore reverse causality (e.g., 2-year blanking analysis). Nevertheless, we acknowledge that our findings reflect associations, that residual confounding and reverse causality may exist, and that robust causal inference analyses are warranted. Fourth, reliance on diagnosis codes to define outcomes introduces potential bias whereby individuals with lower physical activity levels may have more frequent healthcare contact leading to a higher likelihood of incident disease diagnosis. Fifth, although we performed data processing in accordance with previous practices using wrist-based accelerometers8,20,21,22, exposure classification in accelerometer-based studies may differ substantially based on processing choices made23. Sixth, in addition to overall MVPA, we assessed the guideline-recommended MVPA threshold of ≥150 min/week1,10,11 given that it is commonly used in clinical practice and has been associated with outcomes previously in accelerometer studies across multiple populations21,22,24,25. Nevertheless, we acknowledge that current activity guidelines are largely based on self-reported data, evidence to support a particular threshold of accelerometer-measured activity is limited, and optimal levels of device-measured activity may differ. Indeed, our findings generally suggest that greater MVPA levels are increasingly beneficial, whether below or above the 150 MVPA min/week threshold. Seventh, some of our subgroup analyses (e.g., age <55 years) may be underpowered due to lower event rates. Eighth, we report findings from a single community-based sample comprising comparatively healthy individuals from the UK26 whose activity habits may have been influenced by the awareness of being monitored, and therefore our results may not generalize to other populations.
In summary, we performed phenome-wide association testing for incident disease using a unique resource of accelerometer-measured physical activity obtained within over 90,000 individuals. We observed that device-measured activity—defined as continuous MVPA as well as according to guideline-based thresholds—is associated with lower risk of more than 350 incident conditions spanning the full spectrum of human disease. Measured activity was a stronger indicator of disease risk as compared to self-reported activity obtained within the same population. Our findings prioritize future work to identify potential mechanisms linking physical activity and disease, and suggest that optimization of measured physical activity levels may provide a mechanism to reduce future disease incidence.
Methods
Study population
The UK Biobank is a prospective cohort of 502,629 participants enrolled between 2006–201027. Briefly, 9.2 million individuals aged 40–69 years living within 25 miles of 22 assessment centers in the UK were invited, and 5.4% participated in the baseline assessment. Questionnaires and physical measures were collected at recruitment, and all participants are followed for outcomes through linkage to national health-related datasets. All participants provided written informed consent. The UK Biobank was approved by the UK Biobank Research Ethics Committee (reference# 11/NW/0382). The use of UK Biobank data (application 17488) was approved by the local Mass General Brigham Health Institutional Review Board.
Accelerometer-derived physical activity
Between February 2013–December 2015, 236,519 UK Biobank participants were invited to wear a wrist-worn accelerometer for one week, of whom 106,053 agreed to participate and 103,695 submitted data8. Participants were sent an Axivity AX3 (Newcastle upon Tyne, UK) wrist-worn triaxial accelerometer. The sensor captured continuous acceleration at 100 Hz with dynamic range of ±8 g.
As described previously, acceleration signals were calibrated to local gravity8. Sample data were combined into 5-s epochs, with each epoch represented by the average vector magnitude. Non-wear-time was identified as consecutive stationary episodes ≥60 min in duration in which all three axes had standard deviation <13.0 mg28. Epochs representing non-wear-time were imputed based on the average of similar time-of-day vector magnitude and intensity distribution data points on different days. We excluded individuals with insufficient wear-time to support imputation (<72 h of wear-time or no wear data in each one-hour period of the 24-h cycle), and whose signals were insufficient for calibration8.
The primary accelerometer-derived exposure was min of moderate-to-vigorous physical activity (MVPA), defined as the sum of 5-s epochs where mean acceleration was ≥100 mg21,29. As performed previously, we extracted MVPA in bouts (5-min periods where ≥80% of epochs met the MVPA threshold), which reduces misclassification of random wrist movement as MVPA20,21, and results in activity estimates in the UK Biobank which align more closely with expectations from UK population-based surveys22. We then classified whether MVPA levels met thresholds recommended in guidelines from the World Health Organization10, American College of Cardiology/American Heart Association1, and European Society of Cardiology11. Secondary exposures included overall mean acceleration, a surrogate for global physical activity that has been validated against energy expenditure30. For a minority of individuals (n = 2316, 2%) contributing >72 h but less than a full week of wear-time, we extrapolated the observed MVPA rate to one week to account for variable wear-time and to facilitate categorization of weekly guideline-based activity.
Self-reported physical activity
Self-reported physical activity data were obtained at enrollment in over 450,000 UK Biobank participants using the short-form international physical activity questionnaire (IPAQ)31. We quantified self-reported activity as weekly minutes of MVPA to mirror our accelerometer-based analyses, which quantified total time spent performing activities of moderate or greater intensity.
Additional exposures
Age, sex, body mass index (BMI), blood pressure, and anti-hypertensive medication use were assessed at the study visit most closely preceding accelerometer wear (accelerometer analysis) or IPAQ administration (self-reported activity analysis). Tobacco and alcohol use were obtained using standardized questionnaires. Alcohol use was quantified as total grams of alcohol consumed per week. The Townsend Deprivation Index was measured as a surrogate for socioeconomic deprivation32. As performed previously33, self-reported educational history and degree/qualification status were converted to years of educational attainment, and diet quality was classified as poor, intermediate, or good in accordance with responses to dietary questionnaries34. Individuals with missing BMI (0.2%) and blood pressure (0.1%) were excluded. For a minority of individuals (11%) who reported alcohol use frequency but not volume, we assumed the median volume observed at the reported frequency. A small number of individuals who did not report smoking status (0.002%) or medication use (0.4%) were assumed to be never smokers and not exposed to blood pressure medications, respectively. The sample median education level was assumed for individuals who did not report an education history (0.8%), and an intermediate diet quality for those who did not report sufficient dietary data (0.3%).
Outcomes
We defined diseases using v1.2 of the Phecode Map35, a set of 1867 disease definitions arranged into clinically meaningful groups and identified using standardized sets of International Classification of Disease, 9th and 10th revision codes. Phecode definitions can be found at https://phewascatalog.org/. Diagnostic code sources included hospital data through linkage to national health-related datasets, as well as outpatient general practitioner visit data through linkage to electronic health records. Since general practitioner data are available only for a subset of UK Biobank participants (45%), we performed secondary analyses considering only hospital data. In our incident disease analyses of accelerometer-derived variables, person-time started at the end of accelerometer measurement and ended at an event, death, or last follow-up, whichever came first. In our incident disease analyses of self-reported activity, person-time started at enrollment and was otherwise constructed similarly. The date of last follow-up varied according to availability of linked health data and was therefore defined as March 31, 2021 for participants enrolled in England and Scotland, and February 28, 2018 for participants enrolled in Wales.
Statistical analysis
Associations between accelerometer-derived MVPA (per 150 min/week, or approximately one standard deviation of the MVPA distribution) and incident disease were assessed using Cox proportional hazards regression. Individuals with prevalent disease at the time of exposure ascertainment were excluded from incident analyses of that disease. Given our aim to broadly identify plausible associations between MVPA and disease, and our intent to assess hundreds of disease outcomes simultaneously, we selected a uniform set of potential confounding variables to adjust for in our models. Specifically, we adjusted for age, sex, BMI, Townsend Deprivation Index, smoking status, alcohol use, anti-hypertensive medication use, systolic blood pressure, and diastolic blood pressure. Additional models were constructed using (a) adherence to standard physical activity guidelines (≥150 min of MVPA/week1,10,11) on the basis of accelerometer-derived MVPA, (b) overall mean acceleration, (c) self-reported MVPA, and (d) adherence to standard physical activity guidelines (≥150 min of MVPA/week1,10,11) on the basis of self-reported data, as secondary exposures of interest. To prevent model instability, only diseases with ≥120 events were tested (i.e., at least 10 events per variable in the primary model36). Given our interest in associations between physical activity and incident disease, as well as the age distribution of the sample, we excluded pregnancy conditions and congenital anomalies (n = 6 conditions after applying the minimum event filter), resulting in a total of 697 conditions tested in the primary analysis. To assess the robustness of potential associations to residual confounding, we report E-values37, which represent the minimum strength of association on the risk ratio scale that a potential confounder would need to have with the exposure and outcome to nullify the observed association. As suggested previously37, we present E-values for both the effect estimate and the limit of the confidence interval closer to the null.
To assess the ability of guideline-adherent activity to stratify risk of incident disease, we plotted the 5-year cumulative risk of heart failure, type 2 diabetes, cholelithiasis, and chronic bronchitis (exemplar conditions from each of the four disease categories having the greatest number of associations with physical activity), stratified at the guideline-based threshold of ≥150 min/week of MVPA. We also generated adjusted risk curves by plotting the predicted risk outputs from Cox models adjusted for each covariate included in the primary models, and stratified by guideline-adherent activity separately for men versus women. In these models, covariates were set to the sex-specific mean value (continuous variables), or most commonly observed value (categorical variables). To assess dose-response relationships between MVPA and disease risk, we plotted the relative hazard of incident disease for each quintile of MVPA, as compared to the lowest quintile, for each disease having a significant association with MVPA.
We performed several secondary analyses. First, we fit analogous models assessing for associations between measured MVPA and incident disease within age subgroups (i.e., <55, 55–64, and ≥65 years), approximating tertiles of the sample distribution. Second, we assessed associations with measured MVPA using alternative cutoffs of activity (i.e., ≥75 min and ≥300 min of MVPA/week), with the latter threshold representing activity levels recommended by the World Health Organization for additional health benefit10. Third, we performed analogous association testing between minutes of vigorous physical activity (mean acceleration >430 mg19,29) and disease. Fourth, to assess whether associations between activity and disease may have been driven by reverse causation, we repeated association testing of measured MVPA and incident disease in a landmark analysis in which person-time began two years after measured activity exposure. Fifth, we repeated association testing considering only hospital data (i.e., not general practitioner data) to define incident disease. Sixth, since BMI, systolic blood pressure, and diastolic blood pressure may serve as mediators of the effect of MVPA on diseases as opposed to confounders, we repeated association testing with these variables removed.
Except where otherwise specified, all analyses were performed using R v4.038 (packages: ‘survival’, ‘data.table’, ‘fdrtool’). All p value thresholds were corrected for multiplicity by targeting a relatively stringent FDR threshold of 1%39. Tail-area based FDR thresholds were derived utilizing a generalized approach leveraging a modified Grenander distribution-based algorithm as described previously and implemented in R package ‘fdrtool’40. Directed acyclic graphs for the primary and secondary models are shown in Supplementary Fig. 20.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
UK Biobank data are freely available for research purposes by application (https://www.ukbiobank.ac.uk/enable-your-research/register). Phecode-based outcomes developed for the current study will be returned to the UK Biobank for future research use within 6 months of publication.
Code availability
Data processing scripts used to perform the analyses described herein can be found at (https://github.com/shaankhurshid/acceleration_phewas). Detailed summary data supporting major study results are provided in the Supplementary Data.
References
Arnett, D. K. et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association task Force on clinical practice Guidelines. Circulation 140, e596–646 (2019).
Elliott, A. D. et al. Association between physical activity and risk of incident arrhythmias in 402 406 individuals: evidence from the UK Biobank Cohort. Eur. Heart J. https://doi.org/10.1093/eurheartj/ehz897 (2020).
Yu, E. et al. Diet, lifestyle, biomarkers, genetic factors, and risk of cardiovascular disease in the nurses’ health studies. Am. J. Public Health 106, 1616–1623 (2016).
Mishima, R. S. et al. Self-reported physical activity and atrial fibrillation risk: a systematic review and meta-analysis. Heart Rhythm 18, 520–528 (2021).
Tikkanen, E., Gustafsson, S. & Ingelsson, E. Associations of fitness, physical activity, strength, and genetic risk with cardiovascular disease: longitudinal analyses in the UK biobank study. Circulation 137, 2583–2591 (2018).
Garriguet, D., Tremblay, S. & Colley, R. C. Comparison of physical activity adult questionnaire results with accelerometer data. Health Rep. 26, 11–17 (2015).
Lin, H. et al. Association of habitual physical activity with cardiovascular disease risk. Circ. Res. 127, 1253–1260 (2020).
Doherty, A. et al. Large scale population assessment of physical activity using wrist worn accelerometers: the UK biobank study. PLoS ONE 12, e0169649 (2017).
Strain, T. et al. Wearable-device-measured physical activity and future health risk. Nat. Med. 26, 1385–1391 (2020).
Guidelines Review Commitee. Global Recommendations on Physical Activity for Health. (WHO, 2010).
Piepoli, M. F. et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice: the sixth joint task force of the European Society of Cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of 10 societies and by invited experts)developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 37, 2315–2381 (2016).
Ramakrishnan, R. et al. Accelerometer measured physical activity and the incidence of cardiovascular disease: evidence from the UK Biobank cohort study. PLoS Med. 18, e1003487 (2021).
Barker, J. et al. Physical activity of UK adults with chronic disease: cross-sectional analysis of accelerometer-measured physical activity in 96 706 UK biobank participants. Int. J. Epidemiol. dyy294. https://doi.org/10.1093/ije/dyy294 (2019).
Kraigher-Krainer, E. et al. Association of physical activity and heart failure with preserved vs. reduced ejection fraction in the elderly: the Framingham Heart Study. Eur. J. Heart Fail. 15, 742–746 (2013).
Wu, P.-F. et al. Assessment of causal effects of physical activity on neurodegenerative diseases: a Mendelian randomization study. J. Sport Health Sci. 10, 454–461 (2021).
Yang, P.-Y., Ho, K.-H., Chen, H.-C. & Chien, M.-Y. Exercise training improves sleep quality in middle-aged and older adults with sleep problems: a systematic review. J. Physiother. 58, 157–163 (2012).
Peters, H. P., De Vries, W. R., Vanberge-Henegouwen, G. P. & Akkermans, L. M. Potential benefits and hazards of physical activity and exercise on the gastrointestinal tract. Gut 48, 435–439 (2001).
Troiano, R. P., McClain, J. J., Brychta, R. J. & Chen, K. Y. Evolution of accelerometer methods for physical activity research. Br. J. Sports Med. 48, 1019–1023 (2014).
Khurshid, S. et al. Accelerometer-derived physical activity and risk of atrial fibrillation. Eur. Heart J., https://doi.org/10.1093/eurheartj/ehab250 (2021).
da Silva, I. C. et al. Physical activity levels in three Brazilian birth cohorts as assessed with raw triaxial wrist accelerometry. Int. J. Epidemiol. 43, 1959–1968 (2014).
Menai, M. et al. Accelerometer assessed moderate-to-vigorous physical activity and successful ageing: results from the Whitehall II study. Sci. Rep. 7, 45772 (2017).
Khurshid, S. et al. Accelerometer-derived physical activity and risk of atrial fibrillation. Eur. Heart J. 42, 2472–2483 (2021).
Migueles, J. H. et al. Comparability of published cut‐points for the assessment of physical activity: implications for data harmonization. Scand. J. Med. Sci. Sports, https://doi.org/10.1111/sms.13356 (2018).
O’Neal, W. T. et al. Objectively measured physical activity and the risk of Atrial Fibrillation (from the REGARDS Study). Am. J. Cardiol. 128, 107–112 (2020).
Smith, L. et al. Levels and patterns of self‐reported and objectively‐measured free‐living physical activity among prostate cancer survivors: a prospective cohort study. Cancer 125, 798–806 (2019).
Fry, A. et al. Comparison of sociodemographic and health-related characteristics of UK biobank participants with those of the general population. Am. J. Epidemiol. 186, 1026–1034 (2017).
Littlejohns, T. J., Sudlow, C., Allen, N. E. & Collins, R. UK Biobank: opportunities for cardiovascular research. Eur. Heart J. 40, 1158–1166 (2019).
van Hees, V. T. et al. Estimation of daily energy expenditure in pregnant and non-pregnant women using a wrist-worn tri-axial accelerometer. PLoS ONE 6, e22922 (2011).
Hildebrand, M., Van Hees, V. T., Hansen, B. H. & Ekelund, U. Age group comparability of raw accelerometer output from wrist- and hip-worn monitors. Med. Sci. Sports Exerc. 46, 1816–1824 (2014).
White, T., Westgate, K., Wareham, N. J. & Brage, S. Estimation of physical activity energy expenditure during free-living from Wrist Accelerometry in UK Adults. PLoS ONE 11, e0167472 (2016).
Craig, C. L. et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 35, 1381–1395 (2003).
Townsend, P., Phillimore, P. & Beattie, A. Health and Deprivation: Inequality and the North (Croom Helm, 1988).
Okbay, A. et al. Polygenic prediction of educational attainment within and between families from genome-wide association analyses in 3 million individuals. Nat. Genet. 54, 437–449 (2022).
Bhattacharya, R. et al. Association of diet quality with prevalence of Clonal Hematopoiesis and adverse cardiovascular events. JAMA Cardiol. 6, 1069–1077 (2021).
Wu, P. et al. Mapping ICD-10 and ICD-10-CM codes to phecodes: workflow development and initial evaluation. JMIR Med. Inform. 7, e14325 (2019).
Peduzzi, P., Concato, J., Feinstein, A. R. & Holford, T. R. Importance of events per independent variable in proportional hazards regression analysis. II. Accuracy and precision of regression estimates. J. Clin. Epidemiol. 48, 1503–1510 (1995).
VanderWeele, T. J. & Ding, P. Sensitivity analysis in observational research: introducing the E-value. Ann. Intern. Med. 167, 268–274 (2017).
R Core Team. R: A language and Environment for Statistical Computing (R Foundation for Statistical Computing Vienna, Austria, 2015).
Korthauer, K. et al. A practical guide to methods controlling false discoveries in computational biology. Genome Biol. 20, 118 (2019).
Strimmer, K. A unified approach to false discovery rate estimation. BMC Bioinform. 9, 303 (2008).
Acknowledgements
V.N. is supported by NIH T32HL007604. P.T.E. is supported by the grants from the NIH 1RO1HL092577, the European Union MAESTRIA 965286 and the American Heart Association 18SFRN34110082. E.J.B. is supported by R01HL092577, R01HL141434, R01AG066010; 1R01AG066914, 2U54HL120163, and American Heart Association 18SFRN34110082. L.-C.W. is supported by American Heart Association post-doctoral fellowship 18SFRN34110082. Prior to his employment at Novartis and during this work S.A.L. was supported by NIH grants R01HL139731, R01HL157635 and American Heart Association 18SFRN34250007.
Author information
Authors and Affiliations
Contributions
S.K.—conceptualization, data curation, formal analysis, methodology, writing (original draft). L.-C.W.—methodology, writing (review and editing). V.N.—methodology, writing (review and editing). J.P.P.—methodology, writing (review and editing). R.A.V.—writing (review and editing). M.A.A.—writing (review and editing). E.J.B.—writing (review and editing). P.T.E.—writing (review and editing). S.A.L.—conceptualization, methodology, supervision, writing (review and editing).
Corresponding author
Ethics declarations
Competing interests
S.A.L. is a full-time employee of Novartis as of July 18, 2022. S.A.L. has received sponsored research support from Bristol Myers Squibb, Pfizer, Boehringer Ingelheim, Fitbit, Medtronic, Premier, and IBM, and has consulted for Bristol Myers Squibb, Pfizer, Blackstone Life Sciences, and Invitae. P.T.E. receives sponsored research support from Bayer AG and IBM Health and he has consulted for Bayer AG, Novartis, MyoKardia, and Quest Diagnostics. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khurshid, S., Weng, LC., Nauffal, V. et al. Wearable accelerometer-derived physical activity and incident disease. npj Digit. Med. 5, 131 (2022). https://doi.org/10.1038/s41746-022-00676-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41746-022-00676-9
- Springer Nature Limited
This article is cited by
-
Accelerometer-measured physical activity, sedentary behavior, and risk of incident pelvic organ prolapse: a prospective cohort study in the UK Biobank
International Journal of Behavioral Nutrition and Physical Activity (2024)
-
Deep learning of movement behavior profiles and their association with markers of cardiometabolic health
BMC Medical Informatics and Decision Making (2024)
-
Self-supervised learning for human activity recognition using 700,000 person-days of wearable data
npj Digital Medicine (2024)
-
Sensing health: a bibliometric analysis of wearable sensors in healthcare
Health and Technology (2024)
-
Understanding activity and physiology at scale: The Apple Heart & Movement Study
npj Digital Medicine (2024)