Abstract
Although considerable progress has been made in the molecular biology of Colorectal cancer (CRC), novel approaches are still required to uncover the detailed molecular mechanism of CRC. We aim to explore the potential negatively regulated miRNA-mRNA pairs and investigate their regulatory roles so as to elaborate the potential roles of the critical proteins in the signaling pathways enriched by the differential target genes of negatively regulated miRNA in CRC. Firstly, the differential miRNA-mRNA pairs were selected, followed by pairs of miRNA and their target genes. The obtained relationships were subjected to do functional enrichment analysis and those enriched in CRC pathways were chose to further construct a protein interaction network. Finally, we analyzed the regulatory roles of these relationships and constructed a regulatory network of negatively regulated miRNA and mRNA relationships. A total of 372 pairs of miRNA-mRNA were found and 108 target genes of miRNA were obtained. Three miRNAs including hsa-mir-23b, hsa-mir-365-1 and hsa-mir-365-2 showed significant influence on prognosis of CRC patients. To conclude, the miRNA/mRNA deregulations pairs identified in this study have high potentials to be further applied in diagnosis and treatment of CRC.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is the third leading cause of cancer-related death worldwide1 and recently accumulated evidence indicates that CRC is a genetically heterogeneous and complicated disease caused by abnormalities in gene structure and/or expression2. It is now generally accepted that CRC mainly develops through two different genetic pathways, of which one is the chromosomal instability pathway characterized by the involvement of APC, p53 and k-ras genes, by 18q allelic loss and by aneuploidy DNA content, while the other is a pathway involving microsatellite instability (MSI)3. Although considerable progress has been made in molecular biology, novel approaches are still required to uncover the detailed molecular mechanism of CRC.
Recently, microarray tools have been enriched by the development of platforms for the analysis of miRNA expression4. miRNAs consists of an abundant class of endogenous, small non-coding RNAs at a length of 18–25 nucleotides, which can repress protein translation by binding to the target mRNAs. It is reported there are over 700 miRNA sequences in the human genome5 by the latest version of miRBase (release 13.0, March 2009). In addition, miRNAs have been mainly studied in the field of oncological research and more and more evidence shows that altered miRNA regulation may involve in the pathogenesis of cancers through regulating the translation of tumor suppressors and oncogenes6,7,8. In addition, miRNAs can target up to several hundred mRNAs, which makes them very powerful regulators and an aberrant miRNA expression can disturb a multitude of cell signaling pathways and profoundly influence cancer onset and progression. Changes in the expression of miRNAs have been observed in a variety of human tumors. Although the expression differences can not represent causal events of carcinogenesis necessarily, yet these changes may regulate some genes that are very important during the process of tumor pathogenesis and may contribute to the classification and prognosis of tumors. These alterations of miRNA expression7,8 have now been detected in various solid tumors and hematological malignancies, such as CRC. As is known, the main function of mammalian miRNAs is post-transcriptionally regulating their target mRNAs, indicating the combination of mRNAs expression and miRNAs may represent the transcriptional program which describes the normal and tumor tissue characteristics more accurately. At present, there are two approaches applied to study the connection between miRNAs and CRC, including functional and profiling studies. Anyhow, it is showed that the expression profiles of miRNAs have the same potentiality to identify the biomarkers as profiling of their mRNA or protein counterparts. This enables prognosis prediction, therapy response and distinguishing some kinds of disease like CRC.
In this study, we analyzed the expression data of miRNAs and mRNAs of CRC in (The Cancer Genome Atlas) TCGA database so as to excavate the potential deregulated miRNA/mRNA and expound the possible roles of the critic proteins in the signaling pathways enriched by the differential target genes of the deregulated miRNA, hoping to better understand the pathogenesis and make contributes to the diagnosis or therapy of CRC.
Results
Differential genes and miRNA
The information of expression value of 20531 genes and 680 miRNAs in a total of 253 samples was obtained after pre-processing the TCGA expression data with the detailed flowcharts shown in Fig. 1. Then the differential genes and miRNAs were screened by SAM and limma algorithms was/were shown in Fig. 2. A total of 4937 differentially expressed genes were selected by both the algorithms, among which 2974 of genes were up-regulated while 1963 of genes were down-regulated in cancer samples, accounting for 60.23% and 39.77% of the total differential genes respectively. There were 118 differentially expressed miRNAs, among which 57 were up-regulated while 61down-regulated in cancer samples, accounting for 48.31% and 51.69% of the total differential miRNAs (Table 1). In order to better characterize the differential miRNAs to distinguish between cancer and normal samples, the differential miRNAs were further used to do clustering analysis. The differential miRNAs could distinguish the normal and cancer samples effectively, as shown in Fig. 3.
Heat map of microarray results of the differential miRNA.
The right longitudinal axis showed the names of miRNA; the left longitudinal axis showed the clustering information of miRNA; the upper abscissa axis showed the clustering information of samples. Red represented the up-regulated miRNA while green represented the down-regulated miRNA. The clustering of samples were mainly divided into two major clusters, one was the normal tissue samples and the other was cancer tissue samples; the clustering of miRNA was the same with that of samples, one was the up-regulated miRNA in cancer tissues while the other was the down-regulated miRNA.
Functional enrichment analysis of the differential targeted genes
108 target genes of miRNA were obtained and there were a total of 2093 validated target genes through searching all the differential miRNA in miRWalk2.0 database. Pearson correlation tests of the 108 miRNA and their 2093 target genes were calculated respectively and the pairs of miRNA- target genes with negative correlation value p < 0.05 were screened. There were 377 pairs which contained 202 genes and 76 miRNA. In order to study the regulation effects of the significant negative relationships, 202 genes were selected to map into the DAVID database and subjected to functional enrichment analysis. After GO enrichment and KEGG pathway analysis for the genes, we found the biological processes involved mainly included the positive regulation of the main macromolecule metabolic process, positive regulation of biosynthetic process, positive regulation of macromolecule biosynthetic process, positive regulation of gene expression and so on, among which 52 genes were enriched in 20 KEGG related pathway and 36 genes were enriched in 13 tumor-associated pathways, including pathways in cancer, prostate cancer, chronic myeloid leukemia and CRC. In addition, 30 genes were enriched in 5 signaling pathways, including MAPK signaling pathway, ErbB signaling pathway and the like; wherein the expression of critical protein ErbB1 in ErbB signaling pathway was significantly down-regulated (Fig. 4), while 23 genes were enriched in focal adhesion and cell cycle pathway and the expression of key protein CDK2 in cell cycle pathway was up-regulated (Fig. 4).
Screening of disease related miRNAs
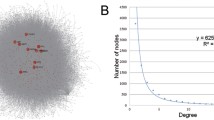
With the aim of further elucidating the roles of miRNA-mRNA relationships, the miRNA-mRNA pairs where all the genes enriched in colorectal pathway were selected; then a total of 30 corresponding miRNAs were screened, finally 137 genes associated with these miRNAs were selected, which constituted 231 pairs of miRNA-mRNA with miRNA. The selected 30 miRNAs were all subjected to do survival analysis using Kaplan-Meier method, among which hsa-mir-23b, hsa-mir-365-1 and hsa-mir-365-2 showed significant influence on prognosis, as shown in Fig. 5. All these 3 miRNAs were downregulated in cancer samples, while the one with relatively high expression in the downregulated samples would have a bad prognosis. Then 13 genes in the CRC-related miRNA-mRNA pairs were subjected to do enrichment analysis and the results showed that the main biological processes involved in included promoting the regulation of metabolic processes of large molecules, promoting the regulation of cell bio-synthesis, promoting the regulation of bio-synthesis, promoting the regulation of gene expression and so on. 42 genes were included in the KEGG pathway, among which 32 genes participated in cancer pathways, while 26 genes were associated with 5 signaling pathway and 21 genes were involved in focal adhesion, cell cycle, adhesive connection, which contained all the pathways that the original 202 genes were enriched in, indicating the selected 137 genes could represent the 202 genes with regulation procures shown in Fig. 6. Then the 137 genes were mapped to the String database and used to construct a PPI interaction network by STRING. A total of 387 pairs of protein interactions with reliability scores greater than 0.4 were selected, among which 96 nodes, accounting for 70.1% of all disease-related genes. As demonstrated in Fig. 7, the protein interaction network of these genes presented a highly aggregated state. High aggregation is an essential characteristic of biological networks. As could be seen from Fig. 7, the majority of genes in a strong interaction with each other were significantly down-regulated. And they formed two clusters, one was associated with cancer, the majority of them were down-regulated; while the other was associated with cell cycle and most of them were up-regulated. Furthermore we calculated the topology parameters of the network, as shown in Fig. 8A–E. From A we could see the distribution of network node degrees followed a pattern of power law network; from the shortest path in B, the average aggregation coefficient in C and proximity to the center in D, we could also see that they meet the characteristics of small-world networks.
(A) PPI networks of disease related genes, red represented the up-regulated genes while green represented the down-regulated genes, each of side represented PPI correlations; (B) distribution of the degrees; (C) distribution of the shortest path; (D) average aggregation coefficient; (E) the proximity to the center.
Construction of regulation networks of miRNA-mRNA
Eight disease and also eight normal samples were randomly selected to do principal component analysis in miRNA and genes of disease-related miRNA-mRNA so as to distinguish between normal and disease samples with results shown in Fig. 9. In order to further investigate the role of miRNA in miRNA-mRNA pairs, we chose 30 miRNAs to calculate their changes in all disease samples, as shown in Fig. 10A. The majority changes of miRNAs were less than 0.25, the miRNA hsa-mir-149 with change exceeding 0.25 was selected to execute survival analysis and observe the effect of its expression in different samples on the prognosis. The survival and treatment information of the corresponding samples in TCGA were downloaded respectively and they were divided into two groups based on expression level of hsa-mir-149, one with high expression within the group, while the other with low expression with the final survival curve demonstrated in Fig. 10B. From which we could see the survival rates of the samples with high expression within the group were higher than that of the ones with low expression at 500–800 days; however, at 800–2500 days the survival rates were lower than that of the ones with low expression within the group and the survival rates were higher than that of the ones with low expression within the group again after 2800 days. Ultimately a miRNA-mRNA regulatory network was successfully constructed by the regulation interaction of miRNA-mRNA (Fig. 11).
(A) principal component analysis of the miRNA in disease-related miRNA-mRNA pairs; (B) principal component analysis of the mRNA in disease-related miRNA-mRNA pairs. Green points and red points represented the randomly selected 8 normal samples and 8 disease samples respectively. The abscissa axis and longitudinal axis indicated the scores of the first principal component and the second principal component of every sample respectively.
Discussion
In the study, a total of 372 pairs of important miRNA/mRNA were found and the potential functional relations of which were suitable for verification by an experiment. A number of such miRNA/mRNA showed they may play critical roles during the regulation of some genes, especially those expressed in cancer. For example, miR-125b inhibits the formation of tumor vessels by suppressing the expression of VE-cadherin9; the miRNAmiR-34 family members are important activity mediators for the p53 gene, which plays a key role in oncogenesis. Ectopic expression of miR-34 in breast cancer cells results in decreased proliferation, invasion and induces apoptosis. Decreased expression of miR-34 was observed in breast tumors and non-small cell lung cancer10. In addition, the expression level of miR-221 that correlates with the expression of p53 can be regarded as a prognostic marker; miR-141 is a new biomarker that can be used for diagnostics of CRC with distant metastasis along with the tumor specific antigen CEA6.
In order to study the roles of the miRNA-mRNA pairs in cancer, we selected 137 genes and found that they participated in a series of biological processes and signaling pathways including MAPK signaling pathway, ErbB signaling pathway, Cell cycle and so on. The expressions of certain proteins in the MAPK signaling pathway such as MAP2K4, MAPK1, MAP3K2 and so on were all abnormal. As we known, the MAPK signaling pathway is a highly conserved intercellular signaling system present in multicellular organisms and plays an essential role in cancer progression11. And a number of miRNAs have been reported to be associated with the MAPK/ERK pathway in different experimental systems and tumors, some of which have been reported to directly target components of MAPK signaling, but most appear to target the downstream players of MAPK signaling, such as proteins regulating the cell cycle and migration12,13,14. Furthermore, the critical protein ErbB1 in ErbB signaling pathway was down-regulated.
To further study the function of the 30 miRNAs, they were all subjected to do survival analysis and we found hsa-mir-23b, hsa-mir-365-1 and hsa-mir-365-2 showed significant influence on prognosis. miR-365 has been reported to be involved in the carcinogenesis of CRC15 and hsa-mir-365-2 was reported to act as one of negative regulators of BCL2 through direct binding to their respective binding sites in the 3′-UTR of the human BCL2 gene16. While BCL2 was identified as an oncogene that does not promote cell proliferation but the evasion of cell death17. And previous study had proved that overexpression of hsa-mir-365-2 not only caused an increase in apoptosis but also augmented the apoptotic effect of etoposide in breast cancer MCF7 cells, indicating that hsa-mir-365-2 had therapeutic potential.
Moreover, from the expression changes of these miRNAs among various cancer samples, we found that there was little expression change in most of the miRNAs while miRNA hsa-mir-149 changed a lot. Several lines of evidence have suggested that miR149 plays multiple roles in the cell proliferation, as well as pathogenesis of the progression of various types of malignant tumors and infectious diseases18,19,20. However, the results were still in dispute21,22. The expression of MiR-149 was down-regulated in several tumors, such as NSCLC and it acted as a tumor suppressor to inhibit the oncogenes expression. For example, the expression of miR-149 was down-regulated in glioblastoma and it could restrain the proliferation and invasion of glioma cells through hindering AKT1 signaling20. Moreover, loss of miR-149 resulted in oncogenes expression increase and was related with tumor stage in astrocytomas and renal cell carcinoma23. Elevated miR-149 also played an important role in the progress of nasopharyngeal carcinoma24. Nevertheless, little was known about the role of miR-149 in CRC. Our study demonstrated the influence of miR-149 on CRC, we divided miR-149 into two groups based on its expression level to observe the effect on the prognosis of CRC. The results indicated that the miR-149 at different level had certain influence on the survival rates of patients with CRC.
As nearly half of the mRNAs codes protein are subjected to the miRNA-mediated regulation, the role of miRNAs as important regulators of significant genes for cancer has been confirmed25. Therefore, it is of importance to understand the functions of miRNA and their regulatory networks, which may provide new insights into cancer development and find new potential biomarkers and therapeutic targets. To conclude, this study may provide useful information for understanding the miRNA changes in CRC.
Materials and Methods
Data
The colorectal adenocarcinoma miRNASeq and level3 RNAseq data were downloaded from TCGA database. The data platforms for miRNASeq and RNASeqV2 were BCGSC__IlluminaHiSeq_miRNASeq and UNC__IlluminaHiSeq_RNASeqV2 respectively, which contained a total of 261 samples including 253 colon adenocarcinoma and eight normal tissue samples and they were divided into two groups. The data collection and this study were conducted in compliance with all applicable laws, regulations and policies for the protection of human subjects and any necessary approvals, authorizations, human subject assurances, informed consent documents26. The normalized level3 data were selected to remove the miRNA and mRNA undetected or their signals were 0. Fig. 4 of signaling pathway of ErbB and fig. 6 of signaling pathway of cell cycle were obtained by Kyoto Encyclopedia of Genes and Genomes27 with copyright permission.
Screening of differential genes and miRNA
Samr28 packet in R software was utilized to screen the differential genes and miRNAs between normal and cancer tissues. The threshold for screening differential genes was set at delta = 1, fold change >2 and FDR <5%. In order to ensure normal and cancer tissues could be well characterized by the selected differential genes and miRNAs, we also applied limma29 to screen the differential genes and miRNAs and the threshold was set at adj.P.Val = 0.05, fold change >2, finally the genes and miRNAs demonstrated to be differential by both of the algorithms were selected.
Target genes of the differential miRNA
MiRWalk2.030 database was used to inquiry all the target genes of the differential miRNAs and those had been validated by experiments (that was already reported before) were chosen. In addition, we took advantage of Pearson rank correlation to calculate the significant correlation between miRNAs and their target genes and filtered out the genes with difference and significant negative correlation.
Determination of the disease-related miRNA/mRNA
Firstly, the negatively regulated differential target genes were subjected to biological function enrichment analysis and the online analytic tool DAVID (Database for Annotation, Visualization and Integrated Discovery)31 was used to enrich GO function and KEGG pathways of the genes significantly up-regulated and down-regulated. GO terms and KEGG pathways with significant enrichment value FDR less than 0.05 were selected to be analyzed. Then the genes and their associated miRNAs enriched in CRC pathway were selected to constitute pairs of miRNA-mRNA seeds and searched for the corresponding negatively regulated target genes of these miRNA in all the negatively regulated genes so as to form disease-related miRNA-mRNA pairs.
Construction of PPI network
Firstly, In order to distinguish between cancer and normal tissues, the miRNA and mRNA in disease-related miRNA/mRNA pairs were chosen to perform principal component analysis (PCA). PCA is a mathematical algorithm (Raychaudhuri, Stuart et al. 2000), which can not only reduce the dimensionality of the data, but also retain most of the variables in the data set. Through identification of the principal components by PCA, one direction can be found and the value of the data distributed along this direction were the maximum, which reduced the data dimension. Furthermore, just a number of variables rather than thousands of variables can be enough to classify the samples by principal component analysis. Then the disease-related miRNAs were selected and their expression changes among various cancer samples were calculated, the miRNA showing the greatest change was picked up to do survival analysis so as to screen the differential disease-related miRNA among cancers. Finally, STRING32 online database was applied to obtain the protein-protein interaction relationships corresponding to the genes of disease-related miRNA/mRNA and the relationships with score coefficient greater than 0.4 were screened to build the PPI network.
Network analysis
The regulation relationships among various genes were analyzed through calculating the topological properties of the network such as distribution of network node degree, distribution of the shortest path, the average clustering coefficient and proximity to the center and so on, moreover the related genes were subjected to depth analysis. The pairs of disease-related miRNA/mRNA were used to construct molecular regulation networks.
Additional Information
How to cite this article: Zhou, X. et al. Identifying miRNA/mRNA negative regulation pairs in colorectal cancer. Sci. Rep. 5, 12995; doi: 10.1038/srep12995 (2015).
References
Jemal, A. et al. Global cancer statistics. CA-Cancer J. Clin 61, 69–90 (2011).
Dean, M. Cancer as a complex developmental disorder—nineteenth Cornelius P. Rhoads Memorial Award Lecture. Cancer Res 58, 5633–5636 (1998).
Benatti, P. et al. Microsatellite instability and colorectal cancer prognosis. Clinical Cancer Research 11, 8332–8340 (2005).
Liu, C.-G. et al. An oligonucleotide microchip for genome-wide microRNA profiling in human and mouse tissues. Proc Natl Acad Sci USA 101, 9740–9744 (2004).
Slaby, O., Svoboda, M., Michalek, J. & Vyzula, R. MicroRNAs in colorectal cancer: translation of molecular biology into clinical application. Mol Cancer 8, 102 (2009).
Garzon, R., Calin, G. A. & Croce, C. M. MicroRNAs in cancer. Annu Rev Med 60, 167–179 (2009).
Winter, J., Jung, S., Keller, S., Gregory, R. I. & Diederichs, S. Many roads to maturity: microRNA biogenesis pathways and their regulation. Nature cell biology 11, 228–234 (2009).
Esquela-Kerscher, A. & Slack, F. J. Oncomirs—microRNAs with a role in cancer. Nat Rev Cancer 6, 259–269 (2006).
Muramatsu, F., Kidoya, H., Naito, H., Sakimoto, S. & Takakura, N. microRNA-125b inhibits tube formation of blood vessels through translational suppression of VE-cadherin. Oncogene 32, 414–421, 10.1038/onc.2012.68 (2013).
Neilsen, P. M. et al. Mutant p53 drives invasion in breast tumors through up-regulation of miR-155. Oncogene 32, 2992–3000 (2013).
Liu, S. M., Lu, J., Lee, H. C., Chung, F. H. & Ma, N. miR-524-5p suppresses the growth of oncogenic BRAF melanoma by targeting BRAF and ERK2. Oncotarget 5, 9444–9459 (2014).
Ikeda, Y., Tanji, E., Makino, N., Kawata, S. & Furukawa, T. MicroRNAs associated with mitogen-activated protein kinase in human pancreatic cancer. Mol Cancer Res 10, 259–269 (2012).
Philippidou, D. et al. Signatures of microRNAs and selected microRNA target genes in human melanoma. Cancer Res 70, 4163–4173 (2010).
Paroo, Z., Ye, X., Chen, S. & Liu, Q. Phosphorylation of the human microRNA-generating complex mediates MAPK/Erk signaling. Cell 139, 112–122 (2009).
Guo, H. et al. MicroRNAs-372/373 promote the expression of hepatitis B virus through the targeting of nuclear factor I/B. Hepatology 54, 808–819, 10.1002/hep.24441 (2011).
Singh, R. & Saini, N. Downregulation of BCL2 by miRNAs augments drug-induced apoptosis–a combined computational and experimental approach. J Cell Sci 125, 1568–1578, 10.1242/jcs.095976 (2012).
Youle, R. J. & Strasser, A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 9, 47–59, (2008).
Wang, F. et al. SP1 mediates the link between methylation of the tumour suppressor miR‐149 and outcome in colorectal cancer. J Pathol 229, 12–24 (2013).
Lai, L. et al. MicroRNA-92a negatively regulates Toll-like receptor (TLR)-triggered inflammatory response in macrophages by targeting MKK4 kinase. J Biol Chem 288, 7956–7967 (2013).
Pan, S. et al. MicroRNA-149 inhibits proliferation and invasion of glioma cells via blockade of AKT1 signaling. Int J Immunopatho Pharmacol 25, 871–881 (2011).
Zhang, J., Liu, Y.-f. & Gan, Y. Lack of association between miR-149 C > T polymorphism and cancer susceptibility: a meta-analysis based on 4,677 cases and 4,830 controls. Mol Biol Rep 39, 8749–8753 (2012).
Wang, Y. et al. MicroRNA-149 inhibits proliferation and cell cycle progression through the targeting of ZBTB2 in human gastric cancer. PLoS One 7, e41693 (2012).
Li, D. et al. Grade-specific expression profiles of miRNAs/mRNAs and docking study in human grade I–III astrocytomas. Omics 15, 673–682 (2011).
Luo, Z. et al. An in silico analysis of dynamic changes in microRNA expression profiles in stepwise development of nasopharyngeal carcinoma. BMC Med Genomics 5, 3 (2012).
Chekulaeva, M. & Filipowicz, W. Mechanisms of miRNA-mediated post-transcriptional regulation in animal cells. Curr Opin Chem Biol 21, 452–460 (2009).
Cancer Genome Atlas Research, N. Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455, 1061–1068, 10.1038/nature07385 (2008).
Kanehisa, M. et al. Data, information, knowledge and principle: back to metabolism in KEGG. Nucleic Acids Res 42, D199–205 (2014).
Tusher, V. G., Tibshirani, R. & Chu, G. Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98, 5116–5121 (2001).
Diboun, I., Wernisch, L., Orengo, C. A. & Koltzenburg, M. Microarray analysis after RNA amplification can detect pronounced differences in gene expression using limma. BMC genomics 7, 252 (2006).
Dweep, H., Gretz, N. & Sticht, C. in RNA Mapping. Springer 1182, 289–305 (2014).
Dennis Jr, G. et al. DAVID: database for annotation, visualization and integrated discovery. Genome Biol 4, P3 (2003).
Jensen, L. J. et al. STRING 8—a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res 37, D412–D416 (2009).
Acknowledgements
We thank Kanehisa Laboratories for the permission to use the figures of “signaling pathway of ErbB” and “signaling pathway of cell cycle” in this article. The study is supported by the Medicine and Health Science and Technology plan projects in Zhejiang Province (2015KYB130).
Author information
Authors and Affiliations
Contributions
X.L.Z. and X.M.X. conceived and initialed the study; X.L.Z., X.M.X., J.H.W. and W.B.C. extracted the data set; X.L.Z., X.M.X., J.H.W., J.J.L. and W.B.C. performed the analysis. X.L.Z. and X.M.X. wrote the paper.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhou, X., Xu, X., Wang, J. et al. Identifying miRNA/mRNA negative regulation pairs in colorectal cancer. Sci Rep 5, 12995 (2015). https://doi.org/10.1038/srep12995
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep12995
- Springer Nature Limited
This article is cited by
-
Discovering the ‘Dark matters’ in expression data of miRNA based on the miRNA-mRNA and miRNA-lncRNA networks
BMC Bioinformatics (2018)
-
Identification of miRNA-mRNA Modules in Colorectal Cancer Using Rough Hypercuboid Based Supervised Clustering
Scientific Reports (2017)
-
SWIM: a computational tool to unveiling crucial nodes in complex biological networks
Scientific Reports (2017)
-
rs15869 at miRNA binding site in BRCA2 is associated with breast cancer susceptibility
Medical Oncology (2016)