Abstract
The relationship between urinary collecting system invasion (UCSI) and oncological outcomes in renal cell carcinoma (RCC) patients has attracted extensive attention recent years. However, the reports were inconsistent and remain controversial. Thus, we performed a systematic literature search of PubMed, Embase, Web of Science and The Cochrane Library databases to identify relevant studies up to June 2015 and conducted a standard meta-analysis of survival outcomes. 17 studies containing 9012 RCC patients satisfied the inclusion criteria. Pooled HRs for overall survival (OS) and recurrence-free survival (RFS) were 1.45 (95% CI, 1.26–1.66, P < 0.001) and 2.27 (95% CI, 1.54–3.34, P < 0.001), respectively. Further subgroup analysis suggested that UCSI was significant associated with poor cancer-specific survival (CSS) in stage T1–T2 RCC (HR = 2.05, 95% CI: 1.43–2.96, P < 0.001) but not in stage T3–T4 tumors (HR = 1.08, 95% CI: 0.63–1.85, P = 0.771). Current evidence revealed that UCSI has a significant negative impact on OS and RFS in RCC patients and could be used to predict CSS especially in localized RCC. Thus, RCC patients with UCSI should be paid more attention by clinician and pathologist and require close follow up for their poor prognosis.
Similar content being viewed by others
Introduction
Renal cell carcinoma (RCC) is the third most common genitourinary tumor, which represents about 3% of all human cancers1. Epidemiological data demonstrate that the incidence of RCC has steadily increased in recent years2. The increase may be due to the widespread use of non-invasive imaging techniques, which allow the early detection of small renal masses3,4. With approximately 25–30% of patients found to have metastases at presentation and 20–30% of patients occurs relapse after surgical resection5, RCC patients should be closely watched and stratified to categories with different risk of recurrence, progression and survival. The stratification can significantly improve postoperative patients’ counseling, selection of individualized treatment, planning of appropriate follow-up schedules and the design of clinical randomized controlled trials. Currently the Tumor-Nodes-Metastasis (TNM) staging system remains the most widely accepted system for RCC classification, which describe the anatomic extent of RCC and relate it to the prognosis6. However, patients with comparable tumor characteristics can experience significantly different even opposed clinical outcomes. Thus, it is necessary for the staging system to be continuously assessed and updated as new research data are available.
Invasion of urinary collecting system (UCSI), which used to be included in the first edition of American Joint Committee on Cancer (AJCC) TNM staging system in 1978, is no longer considered as a criterion in the subsequent versions7. However, some recent studies have focused on the prognostic value of UCSI in RCC and suggested inconsistent and controversial results. Anderson et al.8 found that the presence of UCSI was independently associated with higher overall and disease-specific mortality in patients undergoing nephrectomy for locally invasive RCC. Brookman-Amissah et al.9 also demonstrated that collecting system invasion was independently associated with a significant decline in cancer-specific survival and was associated with simultaneous metastatic spread at the time of surgery and multilocular dissemination. On the other hand, Waalkes et al.10 and Schrader et al.11 suggested that UCSI was disqualified as individual prognostic factor for RCC and did not advocate the inclusion of UCSI into upcoming TNM staging systems.
Understanding the relationship between UCSI and RCC outcomes is very important for the prognostic models establishing. To derive a more precise evaluation of the prognostic significance of UCSI in RCC patients, we systematically review published relevant studies and carried out a meta-analysis by standard techniques.
Results
Study characteristics
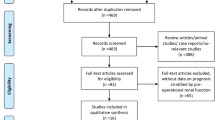
A total of 486 potential relevant studies were retrieved from our initial literature search in the aforementioned databases. Using literature manager software (Endnote), 112 duplicated papers were excluded. After carefully screening titles and abstracts of identified records, 329 studies were excluded for reasons such as apparent irrelevant studies, case reports, conference abstracts, editorials and review articles. Of the remaining 45 studies selected for full text evaluation, 28 studies that belonged to duplicated publication, or failed to offer sufficient data (HRs with corresponding 95% CI) were excluded. Finally, 17 studies met our eligibility criteria and were included in the meta-analysis (Fig. 1). The characteristics and information of these included studies were shown in Table 1. The 17 studies contained 9012 RCC patients including 1008 UCSI, which were all diagnosed by histopathological methods. These RCC patients came from different countries (China, United State, Brazil, Korea, Italy, Egypt, Germany and France) with the duration of follow-up of more than 12 months. Of the eligible 17 studies, 12 studies8,9,10,11,12,13,14,15,16,17,18,19 containing 7006 patients were carried out to investigate the impact of UCSI on the CSS of RCC patients, 4 studies8,18,20,21 containing 2086 patients to investigate the OS and 4 studies17,22,23,24 containing 1157 patients reported the RFS, respectively. Assessment of quality scores by NOS demonstrated that the scores of included studies ranged from 7 to 9, which were considered adequate for the following meta-analysis.
Meta-analysis
Of the 12 studies that referred to CSS, there was apparent inter-study heterogeneity (I2 = 59.2%, P = 0.005). Thus, a random effect model was performed to calculate the pooled HR and corresponding 95% CI. As shown in Fig. 2, the combined HR of these studies revealed that UCSI was associated with poorer CSS in RCC patients (HR = 1.24, 95% CI: 1.01–1.50, P = 0.036). To explore the source of significant heterogeneity, meta-regression analysis and subgroup analysis were performed by patients ethnicity, study number, tumor stage and analysis style. The results showed that tumor stage might have significant association with the heterogeneity (P = 0.041), while other factors did not (Table 2). In addition, subgroup analysis showed that the combined HR estimate for CSS in Caucasian was 1.25 (95% CI, 1.01–1.54, P < 0.001). For RCC patients in low stage and under multivariate analyses, UCSI was also significant associated with poor CSS (P < 0.001 and P = 0.035, respectively), which indicated that UCSI might be an independent cancer-special outcome prognostic factor, especially in low stage RCC patients.
No evident inter-study heterogeneity was observed in the 4 studies that focused on OS (I2 = 0%, P = 0.67). Thus, a fixed model was applied to pool the results. The combined HR for OS was 1.45 (95% CI, 1.26–1.66, P < 0.001), indicating that UCSI was associated with worse OS in patients with RCC (Fig. 3). In addition, sensitivity analysis was conducted by sequential omission of individual studies, which did not significantly influence the results and confirmed the credibility of outcomes.
As shown in Fig. 4, four studies were eligible for examining the relationship between the UCSI and RFS of RCC. A fixed effect model was selected because there was no evident heterogeneity among the four studies (I2 = 7.8%, P = 0.354). The pooled results (HR = 2.27, 95% CI: 1.54–3.34, P < 0.001) indicated that UCSI had an adverse impact on the RFS of RCC patients who received surgical treatment. The further sensitivity analysis did not alter the significance of combined HR, which validated the credibility of results.
Publication bias
Funnel plots, Begg’s test and Egger’s test were conducted to assess the publication bias in our meta-analysis of included studies. As shown in Fig. 5, there was no evident asymmetry in the funnel plots. In addition, the results from Begg’s test (P value) and Egger’s test (intercept with corresponding 95% CI, P value) for the included studies evaluating the survival outcomes were Pbegg = 0.304, intercept 1.25 with 95% CI −0.86 to 3.36, Pegger = 0.216 (CSS); Pbegg = 0.308, intercept −1.13 with 95% CI −3.31 to 1.04, Pegger = 0.154 (OS); Pbegg = 1.000, intercept −2.76 with 95% CI −21.49 to 15.98, Pegger = 0.592 (RFS), respectively. Therefore, the aforementioned evidences suggested a low probability of publication bias.
Discussion
The relationship between UCSI and oncological outcomes in RCC patients has attracted extensive attention and been widely debated, however, the reports remain controversial and there has yet to be a consensus on whether UCSI should be included in the following AJCC staging system25,26. Thus, we systemically review the published studies that evaluated the impact of UCSI on RCC survival and conducted a standard meta-analysis to clarify the prognostic value of UCSI in patients with RCC.
In the present research, based on the inclusion and quality assessment criteria, 17 studies were eligible and the HRs of cumulative survival rates were summarized quantitatively by meta-analysis techniques. Our results indicated that UCSI had a significant negative impact on OS and RFS of RCC patients who underwent surgically treatment. Interesting, by subgroup analysis, renal pelvis invasion was significantly associated with poor disease-specific prognosis in stage T1–T2 RCC (HR = 2.05, 95% CI: 1.43–2.96, P < 0.001) but not in stage T3–T4 tumors (HR = 1.08, 95% CI: 0.63–1.85, P = 0.771), which means that UCSI could predict cancer-specific mortality in organ-confined rather than in advanced tumors. The inconsistent prognostic influence suggests that pathological features such as perinephric fat involvement or vein invasion might be more important cancer-specific outcome predictors than UCSI in locally advanced tumors. On the other hand, it also suggests that collecting system invasion, together with tumor size, could be an additional useful prognostic variable for CSS in localized RCC.
To the best our knowledge, it is the first time that a comprehensive and standard meta-analysis has evaluated the association between UCSI and survival of kidney cancer. From our systematic review of 17 published studies including 9012 patients, the invasion of collecting system by RCC was unusual (11.2%), particularly in small masses. However, its prognostic value should not be ignored because the accurate determination of prognosis after surgery is highly important for both the planning of surveillance program and adequate adjuvant therapy. Our meta-analysis results quantity the impact of UCSI to be a negative prognostic factor. Several potential reasons for RCC patients with UCSI had a poor survival have been proposed but still not very clearly. A study by Klatte et al.23 showed that there was a tendency for an association between collecting system invasion and microvascular invasion and the biological aggressive of these tumors lead to poor survival. Besides, Waalkes et al.10 demonstrated that UCSI was significantly associated with increased frequencies of lymphatic and visceral metastasis at diagnosis, which indicated the invasion of renal pelvis might be a high risk factor for cancer recurrence.
There were also several limitations in our meta-analysis. First, although 17 eligible studies involving 9012 subjects were included in this systematic review, most of them were retrospective studies, which might render the results less reliable. Second, marked heterogeneity of studies was seen in pooled-analysis of CSS (I2 = 59.2%). By using subgroup analysis and meta-regression analysis, we found that the heterogeneity of CSS pooled-analysis may have been mostly due to different tumor stage among the included studies. When the analyses were performed separately according to low and high stage, the prognostic role of UCSI were significance and insignificance, respectively. Besides, several factors such as patients’ baseline characteristics (study size, gender, age, pathological subtype) and duration of follow up might also contribute to part of heterogeneity. Third, there were only four studies investigated OS and RFS of RCC even by a comprehensive literature search, which might inevitably increase the risk of random error, therefore more large prospective studies are needed to further confirm our findings. Finally, despite the well-recognized advantages of systematic review and meta-analysis, the results were affected by the quality of included studies and the reporting bias that papers with null or nonsignificant results were more difficult to be published than those with significant results might be unavoidable27.
Currently collecting system invasion is not considered as a criterion for tumor staging in the latest TNM staging system, thus some clinical pathologists fail to consistently describe this parameter in their reports even if present. In light of the absence of both centralized pathology and standardized system for classifying invasion, it might be argued that only extensive invasion into the collecting system was identified, while the less obvious or microscopic may have been missed, therefore leading to a selection bias of more aggressive tumors28. Further prospective evaluation of the relevance of collecting system invasion as a prognosticator is warranted and the complex prognostic implication of UCSI especially in organ-confined RCC might be one of the next challenges to be addressed by more high-quality studies in the future, which requires concise histopathological description on collecting system invasion to be rendered by pathological report.
In conclusion, our systematics review and meta-analysis of current evidence suggest that UCSI has a significant negative impact on OS and RFS in RCC patients and could be used to predict cancer-specific mortality in localized RCC. Thus, this pathological parameter might be recommended to consider for further TNM classification revisions and to improve the validly of new prognostic nomograms. Given the available data it does seem reasonable to conclude that RCC patients with collecting system invasion should be paid more attention by clinician and pathologist and require close follow up for their poor prognosis.
Methods
Literature search
This meta-analysis was conducted according to the guideline of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)29. The detailed checklist of PRISMA 2009 was available in the supplementary data.
Electronic databases (PubMed, Embase, Web of Science and The Cochrane Library) were searched for published studies that investigated the relationship between UCSI and RCC prognosis up to June 2015. The search strategy included the following terms through MeSH headings, keywords and text words: “collecting system invasion/involvement” or “renal pelvis invasion” or “pelvicaliceal invasion” combined with “renal cancer” or “renal cell carcinoma” or “kidney cancer”. Two independent investigators (Chen and Li) assessed the titles and abstracts of published studies. In addition, we manually reviewed the references cited in the relevant studies for possible inclusions. There was no language limitation existed in the search process.
Eligibility criteria
The criteria for inclusion in our meta-analysis were set out as the following: (1) studies that confirmed the UCSI by histopathological examination; (2) studies analyzing the relationship between UCSI and RCC prognosis; (3) studies with the median follow-up not less than 12 months; (4) studies that reported overall survival (OS) or cancer-special survival (CSS) or recurrence-free survival (RFS) with hazard ratio (HR) and corresponding 95% confidence interval (CI) or studies that provided sufficient information to achieve an estimated HR and 95% CI by using the methods reported by Tierney et al.30.
Studies were excluded if they (1) were case reports, conference abstract, editorials or review articles; (2) investigated RCC cases fewer than 80 patients; (3) lacked sufficient data to estimate the HR and 95% CI. When multiple published papers by the same authors were retrieved, the most informative publication was included to avoid incorporating duplicated data.
Because the data included in our study were retrieved from published literature, ethical approval from ethics committees was not needed.
Data extraction and quality assessment
Data from each eligible study were extracted by two investigators (Chen and Gu) independently with a standardized items form. The following information, if available, were recorded: first author’ name, publication year, study region, recruitment period, sample size, median of patient age, follow-up time and survival data including OS or CSS or RFS with their HRs and corresponding 95% CI.
Study quality was scored by two reviewers (Chen and Gu) using the Newcastle Ottawa Scale (NOS), which was recommended for the assessment of non-randomized studies31. The quality of studies included the following three main categories: selection, comparability and ascertainment of outcome. The total scores were added by these three aspects and a study with more scores means a better methodological quality. We defined studies with scores more than 6 were qualified to be included in the meta-analysis. Discrepancies between investigators for the above questions were resolved through discussion.
Statistical analysis
Pooled HR with its corresponding 95% CI was calculated to evaluate the impact of UCSI on the survival of RCC patients and HR greater than one indicated a worse prognosis in patients with UCSI. The statistical heterogeneity of combined HR was conducted using Cochrane Q test and I2 metrics. When there was no evident heterogeneity existed among studies (I2 > 50% suggested obvious heterogeneity)32, we used the fixed effect model, namely Mantel-haenszel method to pool the results, otherwise, the random effect model (DerSimonian and Laird method) was applied. Potential sources of heterogeneity, if significant, were explored by using subgroup analysis and meta-regression analysis. Besides, sensitivity analysis was performed by sequential omission of individual studies to evaluate the stability of outcomes. The possibility publication bias was assessed by visual inspection of the funnel plots, Begg rank correlation test33 and Egger linear regression test34. All analyses were performed using the program STATA version 12.0 (State Corporation, College Station, TX, USA). All statistical tests were two sided and difference was considered significant when a P value < 0.05.
Additional Information
How to cite this article: Chen, L. et al. Prognostic role of urinary collecting system invasion in renal cell carcinoma: a systematic review and meta-analysis. Sci. Rep. 6, 21325; doi: 10.1038/srep21325 (2016).
References
DeSantis, C. E. et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin 64, 252–271 (2014).
Chow, W. H., Dong, L. M. & Devesa, S. S. Epidemiology and risk factors for kidney cancer. Nat Rev Urol 7, 245–257 (2010).
Murai, M. & Oya, M. Renal cell carcinoma: etiology, incidence and epidemiology. Curr Opin Urol 14, 229–233 (2004).
Patard, J. J. et al. The changing evolution of renal tumours: a single center experience over a two-decade period. Eur Urol 45, 490–493 (2004).
Crispen, P. L. et al. Lymph node dissection at the time of radical nephrectomy for high-risk clear cell renal cell carcinoma: indications and recommendations for surgical templates. Eur Urol 59, 18–23 (2011).
Ficarra, V., Galfano, A., Mancini, M., Martignoni, G. & Artibani, W. TNM staging system for renal-cell carcinoma: current status and future perspectives. Lancet Oncol 8, 554–558 (2007).
Carr, D. T. The manual for the staging of cancer. Ann Intern Med 87, 491–492 (1977).
Anderson, C. B. et al. Urinary collecting system invasion is a predictor for overall and disease-specific survival in locally invasive renal cell carcinoma. Urology 78, 99–104 (2011).
Brookman-Amissah, S. et al. Urinary collecting system invasion reflects adverse long-term outcome and is associated with simultaneous metastatic spread at the time of surgery and with multilocular dissemination during postsurgical follow-up in renal cell cancer. World J Urol 28, 103–109 (2010).
Waalkes, S. et al. Urinary collecting system invasion is no independent prognostic factor in renal cell carcinoma. World J Urol 28, 283–288 (2010).
Schrader, A. J. et al. Urinary collecting system invasion in renal cell carcinoma: incidence and long-term prognosis. Int J Urol 16, 718–722 (2009).
Brookman-May, S. et al. Collecting system invasion and Fuhrman grade but not tumor size facilitate prognostic stratification of patients with pT2 renal cell carcinoma. J Urol 186, 2175–2181 (2011).
Klatte, T. et al. Cytogenetic and molecular tumor profiling for type 1 and type 2 papillary renal cell carcinoma. Clin Cancer Res 15, 1162–1169 (2009).
Margulis, V. et al. Location of extrarenal tumor extension does not impact survival of patients with pT3a renal cell carcinoma. J Urol 178, 1878–1882 (2007).
Klatte, T. et al. Prognostic factors for renal cell carcinoma with tumor thrombus extension. J Urol 178, 1189–1195 (2007).
Shi, X. et al. Prognostic prediction and diagnostic role of intercellular adhesion molecule-1 (ICAM1) expression in clear cell renal cell carcinoma. J Mol Histol 45, 427–434 (2014).
Cho, H. J. et al. Prognostic value of capsular invasion for localized clear-cell renal cell carcinoma. Eur Urol 56, 1006–1012 (2009).
Terrone, C. et al. Prognostic value of the involvement of the urinary collecting system in renal cell carcinoma. Eur Urol 46, 472–476 (2004).
Verhoest, G. et al. Urinary collecting system invasion is an independent prognostic factor of organ confined renal cell carcinoma. J Urol 182, 854–859 (2009).
yanhon, Z., shejun, M., shaofang, W., zhiyong, L. & ying, W. Multivariate analysis of prognostic factors in renal cell carcinoma. Chinese Journal of Experimental Surgery 32, 638–640 (2015).
Palapattu, G. S. et al. Collecting system invasion in renal cell carcinoma: impact on prognosis and future staging strategies. J Urol 170, 768–772 (2003).
Ornellas, A. A., Andrade, D. M., Ornellas, P., Wisnescky, A. & de Santos Schwindt, A. B. Prognostic factors in renal cell carcinoma: Analysis of 227 patients treated at the Brazilian national cancer institute. International Braz J Urol 38, 185–194 (2012).
Klatte, T. et al. Prognostic relevance of capsular involvement and collecting system invasion in stage I and II renal cell carcinoma. BJU Int 99, 821–824 (2007).
Sameh, W. M., Hashad, M. M., Eid, A. A., Abou Yousif, T. A. & Atta, M. A. Recurrence pattern in patients with locally advanced renal cell carcinoma: The implications of clinicopathological variables. Arab Journal of Urology 10, 131–137 (2012).
Uzzo, R. G., Cherullo, E., Myles, J. & Novick, A. C. Renal cell carcinoma invading the urinary collecting system: implications for staging. J Urol 167, 2392–2396 (2002).
Moch, H. et al. Reassessing the current UICC/AJCC TNM staging for renal cell carcinoma. Eur Urol 56, 636–643 (2009).
Sutton, A. J., Song, F., Gilbody, S. M. & Abrams, K. R. Modelling publication bias in meta-analysis: a review. Stat Methods Med Res 9, 421–445 (2000).
Lake, A. M. & Chang, S. S. Kidney cancer: The prognostic value of urinary collecting system invasion. Nat Rev Urol 6, 639–640 (2009).
Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G. & Group, P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8, 336–341 (2010).
Tierney, J. F., Stewart, L. A., Ghersi, D., Burdett, S. & Sydes, M. R. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 8, 16 (2007).
Wells, G., Shea, B., O’connell, D., Peterson, J. & Welch, V. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. (2012) Available at: http://www.ohri.ca/programs/clinical_epidemiology/nosgen.pdf. (Accessed: 5 July 2015).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ 327, 557–560 (2003).
Begg, C. B. & Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 50, 1088–1101 (1994).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Acknowledgements
This work was supported by the National High Technology Research and Development Program of China (2014AA020607).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: L.C., H.L. and X.Z.; Performed the experiments: L.C. and L.G.; Analyzed the data: X.M., X.L., Y.G. and F.Z.; Contributed analysis tools: Y.F., Y.Z. and Y.X.; Wrote the paper: L.C. and X.Z.; Revised the manuscript: H.L. and L.G.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Chen, L., Li, H., Gu, L. et al. Prognostic role of urinary collecting system invasion in renal cell carcinoma: a systematic review and meta-analysis. Sci Rep 6, 21325 (2016). https://doi.org/10.1038/srep21325
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep21325
- Springer Nature Limited
This article is cited by
-
Urinary collecting system invasion on multiphasic CT in renal cell carcinomas: prevalence, characteristics, and clinical significance
Abdominal Radiology (2021)
-
Renal cell carcinoma: predicting RUNX3 methylation level and its consequences on survival with CT features
European Radiology (2019)
-
WHO/ISUP classification, grading and pathological staging of renal cell carcinoma: standards and controversies
World Journal of Urology (2018)
-
Recurrence in Localized Renal Cell Carcinoma: a Systematic Review of Contemporary Data
Current Urology Reports (2017)
-
External validation of a nomogram including the computed tomography imaging score to predict indolent renal masses
International Urology and Nephrology (2017)