Abstract
With the goal of finding a new way to reduce population densities of Bemisia tabaci biotype Q in greenhouses, seven repellent volatile chemicals and their combinations were screened. The mixture of DLCO (D-limonene, citral and olive oil (63:7:30)) had a better cost performance(SC50 = 22.59 mg/ml)to repel whiteflies from settling than the other mixtures or single chemicals. In the greenhouse, in both the choice test and the no-choice tests, the number of adult whiteflies that settled on 1% DLCO-treated tomato plants was significantly lower than those settling on the control plants for the different exposure periods (P < 0.01). In the choice test, the egg amount on the treated tomato plants was significantly lower (P < 0.01) than that on the control plants, but there was no significant difference (P > 0.05) between the number of eggs on treated and control plants in the no-choice test. Compared with the controls, 1% DLCO did not cause significantly statistic mortality rates (P > 0.05) out of different living stages of B. tabaci. The tests for evaluating the repellent efficacy, showed that a slow-releasing bottle containing the mixture had a period of efficacy of 29 days, and the application of this mixture plus a yellow board used as a push-pull strategy in the greenhouse was also effective.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The whitefly Bemisia tabaci (Gennadius) biotype Q (Hemiptera: Aleyrodidae) is a serious pest of tomato plants worldwide. Both adults and larvae cause direct damage by continual sap sucking and by excreting honeydew, which accumulates on the tomato plant, causing chlorosis of infested leaves and the growth of sooty mold. B. tabaci also causes substantial indirect damage as an efficient vector of plant pathogenic viruses, primarily Gemini viruses1 and the tomato yellow leaf curl virus-Israel (TYLCV-Is)2. Members of Geminiviridae interfere with plant photosynthesis, which seriously reduces productivity and product market value (fruit and fiber)3.
B. tabaci is a species complex that includes many genetically differentiated populations, of which the B and Q biotypes are widely distributed. The Q biotype was first recognized in the south of Spain and Portugal4, and many studies have demonstrated that B. tabaci biotype Q has a higher tolerance to extreme temperatures and a higher resistance to insecticides than the biotype B5,6,7,8,9. Biotype Q has a worldwide distribution that includes Morocco10, Egypt11, Israel12, Croatia13, Montenegro14, Italy15 and China16. Because of its extensive host range and ability to transmit a relatively large number of plant viruses, the Q biotype is of particular concern17. As demonstrated by recent research, whitefly biotype Q has caused severe damages in many regions of China18,19.
Although chemical control is widely used for the management of B. tabaci, whitefly has developed resistance to a number of insecticides20. Furthermore, the excessive use of insecticides has resulted in the elimination of natural enemies of whitefly16, leading to an ecological crisis in tomato production systems. This situation has led to the development of integrated pest management (IPM) strategies, in which biological control plays a central role21.
Various petroleum oils and plant-derived oils have repellent activities against insect pests22, demonstrating the potential for their use in IPM. The essential oil of Citrus limon (L.) was reported to repel the dengue vector Aedes albopictus (Skuse)23. The essential oils of Eucalyptus citriodora Hook and Tagetes lucida Cavanilles. grown in Colombia demonstrated repellency against Sitophilus zeamais (Motschulsky) at doses between 0.063 and 0.503 ml/cm2 24. Ginger oil has insect growth regulator and antifeedant activity against Spilosoma obliqua (Walker) larvae25. The essential oil of oriental arborvitae Platycladus orientalis (L.) Franco appears to have potential as a control agent against cowpea weevil (Callosobruchus maculatus Fab.) and rice weevil (Sitophilus oryzae L.) adults26. D-limonene from celery (Coriandrum sativum L.) and geranyl nitrile from Malabar spinach (Basella alba L.) reduced whitefly colonization significantly16. Limonene, extract from celery (C. sativum L.), was able to repel whitefly, and used in a “push–pull” strategy to control B. tabaci27. Citronellal oil and olive oil could reduce settling of whitefly adults28. Citral could be used to control Grapholita molesta (Lepidoptera: Tortricidae Busck) by mating disruption29. α-Pinene had the repellency against stripped flea beetle (Hyllotrata striolata F)30. Considering the reason of economic and repellent efficiency, some repellent volatile chemicals on above were chosen for this research.
Yellow boards covered with adhesive are widely used as an insect control tool that can attract and kill whiteflies and maintain their populations at tolerable levels for approximately 30 days31. However, satisfactory results with yellow boards were only obtained with low densities of whitefly31. Nonetheless, combining yellow boards with repellent oils may bolster the pest-reducing capability of yellow boards by employing a ‘push’ and ‘pull’ strategy for better control of B. tabaci.
The objectives of this experiment were to test the efficacy of seven volatile chemicals and their mixtures as repellents for B. tabaci biotype Q and to identify the most effective chemical or mixture. Additionally, the chemicals were tested for contact toxicity toward B. tabaci biotype Q, and the optimal methods of applying monomers or mixtures plus yellow boards as a push-pull strategy in the greenhouse were evaluated.
Results
Laboratory bioassays
All seven volatile chemicals caused avoidance in the whitefly, whereas the 2% Tween 20 did not. All seven volatile chemicals caused avoidance within 10 min, and the AIs peaked between 30 and 40 min (Fig. 1).
Of all the volatile chemicals, limonene and citral were similar to D-limonene base upon comparisons of fiducial limits of SR50 (Table 1). D-limonene had a smaller SR50 value than lemonile, citronellal, α-Pinene, and olive oil. The combination of D-limonene and citral with olive oil in the ratio of 63:7:30 and 81:9:10 had a smaller SR50 values than D-limonene, but for economic reasons (as a kind of dietary fat32, olive oil had the lowest price among all the chemicals used in this experiment), the ratio of 63:7:30 was chosen for subsequent experiments.
Toxicity toward different developmental stages
Compared with the controls, 1% DLCO did not cause significantly statistic mortality rates (P > 0.05) when eggs (F = 0.77; df = 2, 6; P = 0.51), larval stages (F = 1.31; df = 2, 6; P = 0.34), pupae (F = 0.44; df = 2, 6; P = 0.66) and adults (F = 2.30; df = 2, 6; P = 0.18) were treated.
Choice and no-choice tests
In the choice test, the number of adult whiteflies that settled on 1% DLCO-treated tomato plants was significantly lower than those settling on the control plants for the different exposure periods (T-test, P < 0.01) (Fig. 2). The mean number of adult whiteflies on the treated plants ranged from 16.8 to 21.7 per plant, which was 55.5–61.5% lower than the number on control plants (ranged from 65.7 to 75.9) (Fig. 2). After 36 h, the number of eggs laid on the treated tomato plants (71.6 ± 9.8) was 57% lower than that on the control plants (264.2 ± 20.0) (t = −24.44; df = 4; P < 0.01).
In the no-choice tests, the number of adult whiteflies on tomato plants treated with 1% DLCO ranged from 27.6 to 34.0, which was 18.4 to 23.8% lower than the number on control plants for the different exposure periods (T-test, P < 0.01) (Fig. 5). After 36 h, no significant difference between the number of eggs on treated (275.4 ± 39.4) and control plants (265.1 ± 34.4) was found (t = −0.45; df = 4; P = 0.68).
Repellent effect
Two methods (tomato plants sprayed with 1% DLCO or slow-releasing bottles containing 1% DLCO hung over tomato plants) were compared, and a 3-way ANOVA was used to do the statistics from the data of treatment, distance, time and interactions. Results showed that treatment, distance, time and interactions affected the number of adult whiteflies significantly (P < 0.01) (Table 2).
In the spraying treatment, there were significantly differences between number of adults on plants at different distances from day one to day five (P < 0.01). The number of adult whiteflies on the center tomato plant (0 m) was significantly reduced on the first day comparing with the other plants at different distances (F = 89.6; df = 6, 14; P < 0.01) (Fig. 3A). More whiteflies settled on tomato plants at 0.4 m than on the center plants (Fig. 3A), but there were no significant differences between the number of whiteflies on tomato plants located at distances further than 0.4 m on the first day (Fig. 3A). On the second day, there were still significant differences between the number of adult whiteflies on the center tomato plant (0 m) and other tomato plants at different distances (F = 56.66; df = 6, 14; P < 0.01) (Fig. 3B). The number of adults on plants at 0.4 m was not significantly different from that on plants at 0.8 m on the second day (Fig. 3B). Differences were not detected between distances on the sixth day (F = 2.47; df = 6, 14; P = 0.08) (Fig. 3D). Thus, the spraying treatment had an effective repellent range of approximately 0.4 m from the center for five days.
With the slow-releasing bottles, the numbers of whitefly adults that settled on plants at different distances had been keeping significant differences from the first day to day 29 (P < 0.01). On the first day the adult whiteflies settled more on tomato plants at 1.2 m than on the center plants (F = 129.03; df = 6, 14; P < 0.01), but there were no significant differences between the number of whiteflies on tomato plants located at distances further than 1.2 m (Fig. 3E). Number of whiteflies on tomato plants was significantly different on day two (F = 880.75; df = 6, 14; P < 0.01) (Fig. 3F). At day 29, there were no significant differences between 1.2 m and more distant plants. After 30 days, all the plants had a similar number of adult whiteflies (F = 1.69; df = 6, 14; P = 0.20) (Fig. 3H). Thus, the slow-releasing bottle had an effective repellent range of 0.8–1.2 m from the center, and the period of repellence was approximately 29 days.
Push-pull strategy application
The population densities of adult whiteflies on tomato plants were significantly lower in the two treatments than in the control and were also significantly different between the two treatments (F = 38.48; df = 2, 6; P < 0.01). The average number of adults on each plant in treatment one (slow-releasing bottles containing 1% DLCO plus yellow boards hung over tomato plants) was 17.5 ± 3.8, whereas it was 29.1 ± 2.7 for treatment two (yellow boards only) and 47.0 ± 4.5 for the control plot.
Discussion
Previous research has indicated that ginger oil can protect tomato seedlings from whitefly settling and oviposition, and this repellency of ginger oil was attributed to its odor33. The research of Sertkaya (2010) showed that essential oils obtained from medicinal plants such as thyme (Thymbra spicata subsp. spicata L.), fennel (Foeniculum vulgare Mill.) and rosemary (Rosmarinus officinalis L.) were repellent to cotton whitefly adults34. Although not all of the tested volatile chemicals in the present study have been previously investigated with regard to repellent and deterrent activities against B. tabaci, the potential of most of them to be repellents of other insects is known. For example, the oils of lemongrass (Cymbopogon citratus L) and citronella (Cymbopogon winteriana L) produced strong irritant and repellent responses in Aedes aegypti (Diptera: Culicidae)35, and Cymbopogon spreng (L.) essential oil repelled Myzus persicae (Hemiptera: Aphididae) on field tobacco36. Limonene diluted 500 times had a 62% greater deterrent effect on adults than controls, repelling whitefly egg-laying by more than 80% in the greenhouse27. D-limonene and geranyl nitrile were reported to reduce whiteflies on cucumber16. In contrast, olive oil, was less effective in reducing adult whitefly settling than other compounds28, but the combination of D-limonene, citral and olive oil was effective with a ratio of 63:7:30 in the current study.
The direct airborne repellent assay, which was improved, was used in this experiment to identify and evaluate a relatively suitable volatile chemical mixture, DLCO. It has the advantage of direct repulsion to an airborne chemical vapor and less time required for pest control comparing with trap assays (a former way to test the repellent effect)37,38. However, the assay monitors only the airborne vapor of materials rather than a contact chemical response because the screen net separates the adult whiteflies. Although the toxicity bioassays of the mixture toward eggs, larvae, pupae and adults showed that the compounds were safe for the whitefly, additional toxicity and repellency tests are necessary.
Phytotoxicity is likely to be a problem in the application of volatile chemicals to tomato plants. Indeed, cotton seed oil sprayed directly over the top of plants caused severe damage39, and the concentration of oil spray droplets on the youngest developing leaves resulted in injury. In this greenhouse research, a few necrotic spots appeared on some of the treated leaves at concentrations above 1%, while at concentrations up to 10%, severe wilting and death of the entire treated plant occurred. For this reason, the 1% concentration was used in the toxicity bioassays and the greenhouse choice and no-choice tests.
The greenhouse choice and no-choice tests indicated that spraying tomato plants with the screened mixture effectively reduced the settling of adult whiteflies. Although both sides of the leaves were covered thoroughly by spraying, the deposit was too thin, and evaporation could not be controlled. Because of gravity, the volatile chemical mixture on the adaxial surface of the tomato leaves was not retained as much as that on the abaxial surface. Thus, a way of retaining the volatile chemical mixture for a longer time on the plant might be necessary, particularly because evaporation tends to be higher in the open field. By comparison, the application with the slow-releasing bottle showed better coverage, and the bottle application was synergistic when combined to yellow boards.
In conclusion, this study describes a new repellent with potential for control of whiteflies. Following the approach described here, more effective avoidance agents can be identified and applied for the control of whiteflies.
Methods
Plant materials
‘Hybrid 101’ tomato (Solanum lycopersicum L.) seeds provided by the Chinese Academy of Agricultural Sciences were sown in a greenhouse in plastic pots (10 cm in diameter). The plants were grown inside net cages (120 mesh gauze) to exclude insects. The greenhouse had natural light and an average temperature of 27 ± 4 °C.
Insects
B. tabaci (biotype Q), which was used in all bioassays, was collected in June 2010 from tomato fields in the southern suburbs of Beijing, China (116.2°E, 39.5°N). The insects were reared in isolation on tomato plants under standard laboratory conditions (26 ± 2 °C, 60–70% RH and a photoperiod of 14:10 h (L:D)), and the whiteflies were not exposed to any insecticides. The identification of whiteflies (biotype Q of B. tabaci) was performed according to RAPD-PCR40.
Laboratory bioassays
The commercially volatile chemicals used in this study are described in Table 3. The bioassays were conducted from 8:00 a.m. to 6:00 p.m. in a temperature-controlled room at 27 °C and 40% RH. A binary-choice bioassay tube, which was considered as a specific performance of direct airborne repellent assay, was used to measure repulsion to volatile chemicals (Fig. 4). The bioassay arena was constructed of clear glass (45 cm in length by 3 cm in diameter) with three stoppers (left, right, and upper). The lateral stoppers were covered with a 2.5-cm-diameter filter paper liner circle with coarse fabric netting outside to prevent the whiteflies from direct contact with the chemicals (Fig. 4). Twenty microliters of volatile chemical (vol:vol, containing 2% Tween 20 in deionized water) was added to one end (left or right glass plug) of the filter paper, and a control (deionized water) was added to the other end of the test tube. One hundred B. tabaci adults of unknown sex and age were introduced into the tube from the middle hole (upper glass plug).
First, seven chemicals (1%, vol:vol) and 2% Tween 20 in deionized water were tested to identify a suitable evaluation time. After the incubation period (10 min, 20 min, 30 min, 40 min, 50 min, 60 min, 240 min and 480 min), the whiteflies were counted in the two test zones, and the avoidance index (AI)37 was calculated:

where zone A was the treatment area, and zone B was the control area. Thus, a time-dependent AI was found for each volatile chemical. Trials were repeated three times for each volatile chemical test.
To identify suitable concentrations of the volatile chemicals, a linear regression was used with two variables AI (y) and concentration (x). In this test, the seven volatile chemicals and their mixtures were tested at six concentrations (0.01%, 0.1%, 1.0%, 10%, 50%, and 70%, vol:vol, containing 2% Tween 20 in deionized water), and there were three replications for each concentration; deionized water alone served as the control. For each volatile chemical, the concentration was determined at which 50% of the whiteflies avoided settling (SC50). The SR50 values come from each SC50value of the chemicals divided by D-Limonene’s. The binary-choice bioassay tube was washed with soap and water, rinsed with 90% ethanol and dried between experiments.
Toxicity toward developmental stages
Egg, larva and pupa bioassays. Tomato plants (see above) with three leaves were placed in a net cage (35 cm × 35 cm × 35 cm, 120 mesh gauze) and inoculated with 300 adult whiteflies for egg, larva and pupa bioassays. Tomato leaves infested with 0–2-day-old eggs, larvae or pupae were sprayed with 4 ml per leaf of a 1% volatile chemical mixture (D-limonene: citral: olive oil at 63:7:30) with 2% Tween 20 (referred to henceforth as 1% DLCO), deionized water (control 1), or 2% Tween 20 diluent (control 2). The cumulative mortality of eggs and larvae had been recording for 18 days after the application; the cumulative pupal mortality had been recording for 10 days after the pupae emerged. One tomato plant was treated and checked for each treatment for each bioassay, and all bioassays were repeated three times with totally 27 different net cages and 27 different plants35.
Adult bioassays. A single tomato plant (free of whiteflies) with three leaves was sprayed with 4 ml of 1% DLCO, deionized water (control 1), or 2% Tween 20 diluent (control 2). After air drying, each of the treated tomato plant was placed respectively in one net cage (35 cm × 35 cm × 35 cm, 120 mesh gauze), and 300 adult whiteflies were introduced into each net cage. Adult mortality was recorded after 7 days. The experiment was repeated three times with a total of 9 different net cages and 9 different plants35.
Choice and no-choice tests
Treated and control tomato plants with three leaves were used as hosts for whiteflies and air dried at room temperature for 1 h before use33.
Choice tests. Thirty-two tomato plants were placed in one net cage (7 m × 2 m × 1.5 m, 120 mesh gauze) in the greenhouse and divided evenly into two groups: a treatment group sprayed with 1% DLCO and a control group sprayed with 2% Tween 20. The distance between each tomato plant was 40 cm in each group, and the distance between the two groups was 4 m41. 1800 adult whiteflies were released into the center of the cage. The whiteflies that settled on both sides of the leaf of each tomato plant were counted at 0.5, 1, 2, 3, 4, 24, and 36 h after release. The eggs on both sides of the leaf were counted under a dissecting microscope at 20 x magnification 36 h after the whiteflies were released. The experiment was repeated three times with different plants.
No-choice tests. Tomato plants were placed in net cages (35 cm × 35 cm × 35 cm, 120 mesh gauze) in the greenhouse; each cage contained one tomato plant. Two cages were used for the treatments, and two cages were used for controls. The treated plants were sprayed with 1% DLCO, and the control plants were sprayed with 2% Tween in distilled water. The cages were assigned at random in the greenhouse, and 60 adult whiteflies were released into each cage. On both sides of the leaf of each tomato plant the adults were counted at 0.5, 1, 2, 3, 4, 24, and 36 h after release by direct observation, the eggs were counted under a dissecting microscope at 20 x magnification 36 h after the whiteflies were released. The experiment was repeated three times on three alternate days with different plants.
Avoidance indexes (AIs) were calculated according to the number of adults or eggs in the treatments and controls in the choice and no-choice tests.
Repellent effect
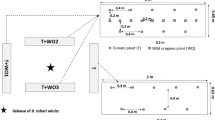
Nineteen tomato plants were placed in one circular arena (5 m diameter × 1.5 m height, 120 mesh gauze) which was used for each treatment or control (Fig. 5). The distance between adjacent tomato plants was 0.4 m. For treatment one, a tomato plant was sprayed with 4 ml of 1% DLCO at the center of the arena; for treatment two, one slow-releasing bottle with 16 ml DLCO hung on the center plant (The bottle is made of polymer material, repellents can across the molecular gap of the polymer material slowly, which can extend the period of repellents. The bottle volume for the experiment is 16 ml).
Approximately 1800 adult whiteflies were released into the circular arena for each treatment or control, and the whiteflies were counted daily after release on each plant. The ranges and lengths of time of the repellent effect of each volatile chemicals were determined42. The experiments were repeated three times with different plants.
Push-pull strategy application
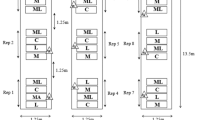
The greenhouses (72 m × 10 m × 2.5 m) were located in Tangshan, Hebei Province, China (118.11°E, 39.36°N). The row spacing of the tomato plants was 80 cm, and the plant spacing within rows was 40 cm. Two treatment plots (treatment one, slow-releasing bottles (1% DLCO) plus yellow boards hung over tomato plants; treatment two, only yellow boards hung over tomato plants) and one control plot (no slow releasing bottles or yellow boards) were used, and each of these was located in a separate room (72 m × 8 m; Fig. 6). The distance between the bottles was within double the repellent’s effective range (2 m) (Fig. 6). The door of each room was open to allow wild whiteflies to enter. The greenhouse tests were repeated three times with different plants.
The adult whiteflies that settled on the tomato plants in the treatment and control plots were counted every five days using a five-point sampling method42. For each sampling point, six leaves (three located in the upper layer of the plant and three in the bottom layer) were checked to record adult whiteflies. The total number of sampled leaves was 720 for each treatment (average of 4 weeks of sampling) in the greenhouse.
Statistical analyses
Linear regression, paired T-tests, ANOVAs followed by Tukey HSD (Honestly Significant Difference) tests were used to analyze the number of whitefly behavioral responses to volatile chemicals in the laboratory or greenhouse bioassays16. All statistical analyses were performed with SPSS (version 16.0 for Windows, SPSS Inc., Chicago, IL, USA) or PoloPlus (version 2.0 for Windows, LeOra Software).
Additional Information
How to cite this article: Du, W. et al. A Primary Screening and Applying of Plant Volatiles as Repellents to Control Whitefly Bemisia tabaci (Gennadius) on Tomato. Sci. Rep. 6, 22140; doi: 10.1038/srep22140 (2016).
References
Brown, J. K. & Czosnek, H. Whitefly transmission of plant viruses. Adv. Bot. Res. 36, 65–100 (2002).
Polston, J. E. Tomato yellow leaf curl virus-Economic impact, In Hopkins, K. (Eds), Post-global crop protection compendium. CAB International, Wallingford, United Kingdom, pp. 89–90 (2001).
Oliveira, M. R. V., Henneberry, T. J. & Anderson, P. History, current status, and collaborative research projects for Bemisia tabaci . Crop Prot. 20, 709–723 (2001).
Guirao, P., Beitia, F. & Cenis, J. C. Biotype determination of Spanish populations of Bemisia tabaci (Hemiptera: Aleyrodidae), Bull. Entomol. Res. 87, 587–593 (1997).
Elbert, A. & Nauen R. Resistance of Bemisia tabaci (Homoptera: Aleyrodidae) to insecticides in southern Spain with special reference to neonicotinoids. Pest Manag. Sci. 56, 60–64 (2000).
Roditakis, E., Roditakis, N. E. & Tsagkarakou, A. Insecticide resistance in Bemisia tabaci (Homoptera: Aleyrodidae) populations from Crete. Pest Manag. Sci. 61, 577–582 (2005).
Karunker, I. et al. Over-expression of cytochrome P450 CYP6CM1 is associated with high resistance to imidacloprid in the B and Q biotypes of Bemisia tabaci (Hemiptera: Aleyrodidae). Insect Biochem. Molec. 38, 634–644 (2008).
Mahadav, A., Kontsedalov, S., Czosnek, H. & Ghanim, M. Thermotolerance and gene expression following heat stress in the whitefly Bemisia tabaci B and Q biotypes. Insect Biochem. Molec. 30(10), 668–676 (2009).
Longhurst, C. et al. Cross-resistance relationships of the sulfoximine insecticide sulfoxaflor with neonicotinoids and other insecticides in the whiteflies Bemisia tabaci and Trialeurodes vaporariorum . Pest Manag. Sci. 69, 809–813 (2013).
Monci, F. et al. Spread of tomato yellow leaf curl virus-Sar from the Mediterranean basin: presence in the Canary Islands and Morocco. Plant Dis. 84, 490 (2000).
De Barro, P. J., Driver, F., Trueman, J. W. H. & Curran. J. Phylogenic relationship of world populations of Bemisia tabaci (Gennadius) using ribosomal ITS1. Mol. Phylogenet. Evol. 16, 29–36 (2000).
Horowitz, A. R. et al. Biotype Q of Bemisia tabaci identified in Israel, Phytoparasitica. 31, 94–98 (2003).
Zanic, K., Cenis, J. L., Kacic, S. & Katalinic, M. Current Status of Bemisia tabaci in coastal Croatia. Phytoparasitica. 33, 60–64 (2005).
Skaljac, M. et al. Diversity and localization of bacterial symbionts in three whitefly species (Hemiptera: Aleyrodidae) from the east coast of the Adriatic Sea. Bull. Entomol. Res. 103, 48–59 (2013).
Demichelis, S., Bosco, D., Manino, A., Marian, D. & Caciagli, P. Distribution of Bemisia tabaci (Hemiptera: Aleyrodidae) biotypes in Italy. Ca. Entomol. 132, 519–527 (2000).
Zhao, Q. et al. Reducing whiteflies on cucumber using intercropping with less preferred vegetables. Entomol. Exp. Appl. 150, 19–27 (2014).
Navas-Castillo, J., Camero, R., Bueno, M. & Moriones, E. Severe yellowing outbreaks in tomato in Spain associated with infections of Tomato chlorosis virus. Plant Dis. 84, 835–837 (2000).
Chu, D. et al. Comparative analysis of population genetic structure in Bemisia tabaci (Gennadius) biotypes B and Q based on ISSR marker. Agr. Sci. China. 7, 1348–1354 (2008).
Pu, G., Yu, K. & Cui, X. Study on survival characteristic of B-biotype and Q-biotype Bemisia tabaci by treatment of high and low temperatures. China Plant Prot. 29, 12–14 (2009).
Palumbo, J. C., Horowitz, A. R. & Prabhaker, N. Insecticidal control and resistance management for Bemisia tabaci . Crop Prot. 20, 739–765 (2001).
Naranjo, S. E. Conservation and evaluation of natural enemies in IPM systems for Bemisia tabaci . Crop Prot. 20, 835–852 (2001).
Larew, H. G. & Locke, J. C. Repellency and toxicity of a horticultural oil against whiteflies on chrysanthemum. Hort. Sci. 25, 1406–1407 (1990).
Giatropoulos, A. et al. Evaluation of bioefficacy of three citrus essential oils against the dengue vector Aedes albopictus (Diptera: Culicidae) in correlation to their components enantiomeric distribution. Parasitol. Res. 111, 2253–2263 (2012).
Nerio, L. S., Verbel, O. J. & Stashenko, E. E. Repellent activity of essential oils from seven aromatic plants grown in Colombia against Sitophilus zeamais Motschulsky (Coleoptera). J. Stored Prod. Res. 45, 212–214 (2009).
Agarwal, M., Walia, S., Dhingra, S. & Khambay, B. P. S. Insect growth inhibition, antifeedant and antifungal activity of compounds isolated/derived from Zingiber officinale Roscoe (ginger) rhizomes. Pest Manag. Sci. 57, 289–300 (2001).
Hashemi, S. M. & Safavi, S. A. Chemical constituents and toxicity of essential oils of oriental arborvitae, Platycladus orientalis (L.) franco, against three stored-product beetles. Ch. J. Agr. Res. 72, 188–194 (2012).
Li, Y. F. et al. Identification of plant chemicals attracting and repelling whiteflies. Arthropod-Plant Interact. 8, 183–190 (2014).
Schuster, D. J., Thompson, S., Ortega, L. D. & Polston, J. E. Laboratory evaluation of products to reduce settling of sweetpotato whitefly adults. J. Econ. Entomol. 102, 1482–1489 (2009).
Faraone, N., D’Errico, G., Caleca, V., De Cristofaro, A. & Trimble, R. M. Electrophysiological and behavioral responses of oriental fruit moth to the monoterpenoid citral alone and in combination with sex pheromone. Environ Entomol. 42(2), 314–322 (2013).
Sundufu, A. J., Gong, H. & Huang, S. Repellency of α-pinene against stripped flea beetle, Hyllotreta striolata (F). Acta Ecol. Sin. 23(2), 303–307 (2002).
Narkiewicz-Jodko, J. & Kania, S. The efficiency of yellow boards and selective insecticides in control of Trialeurodes vaporariorum on tomatoes. Bull. Oepp-eppo Bull. 22, 499–502 (1992).
Kontou, N., Psaltopoulou, T., Panagiotakos, D., Dimopoulos, M. A. & Linos, A. The mediterranean diet in cancer prevention: a review. J Med Food. 14(10), 1065–1078(2011).
Zhang, W., Mcauslane, H. J. & Schuster, D. J. Repellency of Ginger Oil to Bemisia argentifolii (Homoptera:Aleyrodidae) on Tomato. J. Econ. Entomol. 97, 1310–1318 (2004).
Sertkaya, E., Kaya, K. & Soylu, S. Chemical compositions and insecticidal activities of the essential oils from several medicinal plants against the cotton whitefly, Bemisia tabaci. Asian J. Chem. 22, 2982–2990 (2010).
Kontsedalov, S. et al. Toxicity of spiromesifen to the developmental stages of Bemisia tabaci biotype B. Pest Manag. Sci. 65, 5–13 (2009).
Tyaig, B. K., Shah, A. K. & kaul, B. L. Evaluation of repellent activities of Cymbopogon essential oil against mosquito vectors of malaria, filafriasis and dengue fever in India. Phytomedicine. 5, 324–329 (1998).
Kwon, Y. et al. Drosophila TRPA1 channel is required to avoid the naturally occurring insect repellent citronellal. Curr. Biol. 20, 1672–1678 (2010).
Reeder, N. L., Ganz, P. J.,. Carlson, J. R. & Saunders, C. W. Isolation of a deet-insensitive mutant of Drosophila melanogaster (Diptera: Drosophilidae). J. Econ. Entomol. 94, 1584–1588 (2001).
Butler, G. D. & Henneberry, T. J. Sweetpotato whiteflies migration, population increase, and control on lettuce with cottonseed oil sprays. Southwest. Entomol. 14, 287–293 (1989).
Qiu, B. L. et al. Identification of three major Bemisia tabaci biotypes in China based on morphological and DNA polymorphisms. Prog. Nat. Sci. 19, 713–718 (2009).
Togni, P. H. B., Laumann, R. A., Medeiros, M. A. & Sujii, E. R. Odour masking of tomato volatiles by coriander volatiles in host plant selection of Bemisia tabaci biotype B. Entomol. Exp. Appl. 136, 164–173 (2010).
Hagstrum, D. W. Using five sampling methods to measure insect distribution and abundance in bins storing wheat. J. Stored Prod. Res. 36, 253–262 (2000).
Acknowledgements
This study was supported by the Professional Science & Technology Foundation (award no. 201103002) of the Ministry of Agriculture of China. We thank professor Pei Liang for his help on laboratory bioassays.
Author information
Authors and Affiliations
Contributions
W.D. and Y.Q. wrote the main manuscript text and prepared all figures. X.H. and Y.W. revised the manuscript and added some data. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Du, W., Han, X., Wang, Y. et al. A Primary Screening and Applying of Plant Volatiles as Repellents to Control Whitefly Bemisia tabaci (Gennadius) on Tomato. Sci Rep 6, 22140 (2016). https://doi.org/10.1038/srep22140
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep22140
- Springer Nature Limited
This article is cited by
-
Attraction of whitefly Aleurodicus cocois mediated by cashew volatiles
Phytoparasitica (2022)
-
Essential oils from two aromatic plants repel the tobacco whitefly Bemisia tabaci
Journal of Pest Science (2022)
-
Volatile Organic Compounds as Insect Repellents and Plant Elicitors: an Integrated Pest Management (IPM) Strategy for Glasshouse Whitefly (Trialeurodes vaporariorum)
Journal of Chemical Ecology (2020)
-
Herbivore- and MeJA-induced volatile emissions from the redroot pigweed Amaranthus retroflexus Linnaeus: their roles in attracting Microplitis mediator (Haliday) parasitoids
Arthropod-Plant Interactions (2018)