Abstract
Purpose
This study sought to identify the prevalence and factors associated with atrial fibrillation (AF) in older patients with obstructive sleep apnea (OSA) in China.
Methods
This was an explorative cross-sectional study. Between January 2015 and October 2017, we continuously recruited 1285 older patients with OSA who underwent overnight polysomnography from sleep centers of multiple hospitals. They were assessed using 12-lead ECG or 24-h dynamic ECG, and their baseline demographics, clinical characteristics, sleep parameters, and medical history were determined. Multivariate binary logistic regression analysis was used to investigate the factors related to AF in these older patients with OSA.
Results
The clinician classified 122 (9.5%) patients as having AF. The prevalence of AF significantly increased with age (P < 0.05) but did not significantly differ between the mild, moderate, and severe OSA groups. Additionally, the prevalence of paroxysmal AF was 7.2% among the overall study population, and it increased with OSA severity or advanced age (P < 0.05). Persistent AF was noted in 2.3% participants, and the prevalence also increased with age. The logistic regression analysis showed that age (OR = 1.054, 95%CI: 1.027–1.018, P < 0.001), history of drinking (OR = 1.752, 95%CI: 1.070–2.867, P < 0.05), chronic heart disease (OR = 1.778, 95%CI: 1.156–2.736, P < 0.01), diabetes mellitus (OR = 1.792, 95%CI: 1.183–2.713, P < 0.01), and reduced diastolic function (OR = 2.373, 95%CI = 1.298–4.337, P < 0.01) were relevant to AF among participants with OSA.
Conclusion
The prevalence of AF is significantly common in older patients with OSA. Age, history of drinking, chronic heart disease, diabetes mellitus, and reduced diastolic function are independently related to AF in these patients.
Similar content being viewed by others
Introduction
Obstructive sleep apnea (OSA), the most common type of sleep apnea, is an independent risk factor for cardiovascular diseases and all-cause mortality [1,2,3]. Moreover, patients with OSA were frequently diagnosed with hypertension [4], diabetes mellitus (DM) [5], and stroke [6]. Several clinical studies have revealed that OSA is highly prevalent (60–90%) in patients with atrial fibrillation (AF) [7,8,9]. OSA promotes the occurrence and recurrence of AF, which is the most common clinically observed form of arrhythmia, due to the repetitive cycles of intermittent hypoxia causing an imbalance of cardiac autonomic modulation [9]. Moreover, older patients with OSA are more prone to developing AF [10, 11].
The prevalence of OSA among patients with AF, including those who underwent catheter ablation has been reported. However, the prevalence and factors associated with AF in OSA patients are not clearly studied. Therefore, we performed this large-scale, explorative cross-sectional study to assess the prevalence and risk factors of AF among older patients with OSA.
Methods
Study Population
This is an explorative observational multi-center study, a total of 1290 patients were continuously recruited from the PLA General Hospital(n = 313), Peking University International Hospital(n = 242), Peking University People's Hospital(n = 242), Beijing Chaoyang Hospital(n = 337), 960th Hospital of PLA(n = 48), and the affiliated Hospital of Gansu University of Chinese Medicine(n = 112) between January 2015 and October 2017. Our inclusion criteria were as follows:1) age ≥ 60 years; 2) diagnosis of OSA by a clinician based on overnight polysomnography; 3) assessment using 12-lead ECG or 24-h dynamic ECG; 4) The blood was collected for blood biochemistry and blood routine examination. The exclusion criteria were as follows: hyperthyroidism, severe electrolyte disorder, history of malignant tumors; and mental disorders. The study flowchart is presented in Fig. 1. Immediately after, the subjects with prior history of valvular atrial fibrillation (n = 5) were also excluded according to our study purpose. Ultimately, 1285 older patients with OSA were included in the analysis, and any ethnic heterogeneity didn’t exist among participants due to that they were both Han Chinese. This study was carried out in accordance with the Declaration of Helsinki. The ethics committee of PLA General Hospital approved the study (S2019-352–01). Written informed consent was available from all participants.
Baseline Evaluation
Demographic data and clinical characteristics of all patients were collected by the researchers, including sex, age, nationality, height, weight, body mass index [BMI was defined as weight in kilograms divided by height in meters squared and was expressed in units of kg/m2. According to the WHO definition: obese, BMI ≧ 30; overweight, 25 ≤ BMI < 30; normal weight, 18.5 ≤ BMI < 25; underweight, BMI < 18.5], blood pressure, history of smoking (was ascertained on the basis of self-reported history of cigarette smoking, according to whether smoking consecutive or accumulated more than six months or not) [12], and history of drinking (was defined as drinking at least once a week over a year, and currently having drank or quit drinking for less than three years). As recommended by current guidelines, the E/A ratio is the ratio of the early (E) to late (A) ventricular filling velocities, and reduced diastolic function was defined as: E/A ≤ 0.7 or deceleration time (DT) > 260 ms; or E/A between 0.7–1.5 and peak early diastolic velocity (e′) < 7 cm/s; or E/A > 1.5 and e′ < 7 cm/s or DT < 140 ms [13]. as well as data on sleep parameters [including the apnea–hypopnea index (AHI), the oxygen desaturation index (ODI), total sleep time (TST) the time under saturation of 90%, the mean pulse oxygen saturation(MSpO2), the lowest pulse oxygen saturation(LSpO2)], blood index (blood of participants were collected under the conditions of at least 6 h of fasting and 4 h of drinking) included total bilirubin, direct bilirubin, total cholesterol, triglyceride, high density lipoprotein cholesterol(HDL-C), low density lipoprotein cholesterol(LDL-C), urea, serum uric acid (SUA), blood glucose, RBC, WBC, PLTs, Hb, and medical history. These data were used to screen patients who met the inclusion and exclusion criteria in this study.
PSG Examination
PSG is the gold standard of OSA diagnosis. All participants underwent full overnight PSG (from 21: 00 to 7: 00 the next day) in sleep laboratories of sleep centers of different hospitals (after clinical stabilization during hospitalization). The sleep parameters of all patients were recorded using a laboratory‐based PSG instrument (Compumedics, Melbourne, Australia), including electroencephalography (EEG), electrooculography (EOG), electrocardiography (ECG), nasal-oral airflow, chest and abdominal wall motion, arterial oxygen saturation, and body position [14]. Analysis of sleep tests was carried out according to the American Academy of Sleep Medicine 2017 guidelines [14]. The PSG records of patients in each hospital were manually analyzed and manually calibrated twice by technically certified and experienced technicians, neither of whom had knowledge of demographics and clinical characteristics. Then, a professional sleep physician would analyze and interpret the report. Apnea was defined as the continuous cessation of airflow for more than 10 s, whereas hypopnea was defined as a 30% or greater drop in flow for 10 s or longer associated with ≥ 4% oxygen desaturation (obstructive sleep apnea if respiratory efforts were present) [15]. AHI was defined as the number of apnea and hypopnea events per hour during sleep. OSA severity was defined according to AHI based on the criteria [15] as follows: mild OSA, 5 ≤ AHI < 15; moderate OSA, 15 ≤ AHI < 30; and severe OSA, AHI ≥ 30.
ECG Examination
For all patients enrolled in the study who were assessed using12-lead ECG or 24-h dynamic ECG, the clinical diagnosis of AF was based on the ACC/AHA/ESC 2016 guidelines [16]. The key electrocardiographic findings of AF are a loss of P waves and replacement by fibrillatory waves; erratic activation of the ventricles resulting in an irregular, rapid heart rate (usually 90 to 170 bpm); and a narrow QRS complex, unless other conduction abnormalities coexist [17].
Statistical Analysis
All data were analyzed using SPSS version 20.0 (SPSS Inc, Chicago, IL, USA). Metrological data were first tested for normality and homogeneity of variance. Normally distributed metrological data are expressed as the mean ± standard deviation (SD), and one-way analysis of variance or the t test was used for comparisons between groups. Metrological data that did not meet the criterion for normal distribution are expressed as the median (interquartile range), and non-parametric tests were used for comparisons between groups. Count data are expressed as percentage (%), and chi-square tests were used for comparisons between groups. Multivariate analyses were performed using binary logistic regression, the results of which are expressed as odds ratio (OR) with 95% confidence interval (CI). The difference was considered statistically significant when P < 0.05.
Results
Baseline Characteristics
A total of 1285 OSA patients were included for data analysis (Fig. 1), including 305 (23.7%) with mild OSA, 385 (30%) with moderate OSA, and 595 (46.3%) with severe OSA. Age, male, weight, BMI, systolic blood pressure (SBP), AHI, ODI, the time under saturation of 90%, MSpO2, LSpO2, TG, HDL-C, creatinine, SUA, WBC, hypertension, gastroesophageal reflux disease (GERD), coronary heart disease (CHD), carotid atherosclerosis, diabetes mellitus (DM), and paroxysmal AF demonstrated significant differences among groups(P < 0.05, Table 1). Compared with non-AF OSA patients, participants diagnosed with AF were found to be older (69.6 VS 66, years), had a higher SBP(140 mmHg VS 131 mmHg), and accounted for a higher proportion of smokers (34.4% VS 21,5%) and drinkers (23.8% VS 11.0%, P both < 0.05). The scores for sleep parameters including AHI (32.2 times/h VS 27.2 times/h) and ODI (26.95 times/h VS 22.20 times/h) were higher in AF than non-AF patients (P < 0.05), but LSpO2 was lower in the AF group (77% VS 80%, P < 0.05). triglyceride (1.32 mmol/L VS 1.39 mmol/L) was lower and creatinine (76 mmol/L VS 72 mmol/L) was higher in AF than non-AF patients(P < 0.05). Additionally, those with AF had a higher proportion of CHD (46.7% VS 21.2%), cerebrovascular disease (27.0% VS 16.7%), carotid atherosclerosis (41.8% VS 24.0%), DM (41% VS 23%), peripheral vascular disease (PVD, 9.0% VS 4.5%), chronic obstructive pulmonary disease (COPD,16.4% VS 6.2%), chronic kidney disease (CKD, 9.0% VS 3.5%), and reduced diastolic function (21.3% VS 5.1%, P < 0.05); other indexes did not significantly differ between the two groups (Table 2).
Prevalence of AF in older patients with OSA
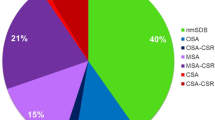
There were 122 (9.5%) patients with clinically classified AF. Of these, the prevalence of AF in males was higher than that in females: 10.2% in males vs. 8.4% in females. Moreover, the prevalence of AF significantly increased with age: 7.0% in patients aged 60–70 years vs. 14.9% in patients aged 71–96 years. The prevalence of AF tended to increase with the severity of OSA, but there was no significant difference between the three groups (Fig. 2). The prevalence of paroxysmal AF was 7.2% among the total study population, and it significantly increased with OSA severity or advanced age (P < 0.05). Similarly, the prevalence of AF in males was higher than that in females (7.7% vs. 6.5%; Fig. 3). Furthermore, 2.3% participants were found to have persistent AF, and although the prevalence also increased with age, it did not significantly differ between the mild, moderate, and severe OSA groups (Fig. 4).
Factors associated with AF in older patients with OSA
In multivariate binary logistic regression analysis, age (OR = 1.055, 95%CI: 1.028–1.082), history of drinking (OR = 1.795, 95%CI: 1.102–2.923), CHD (OR = 1.834, 95%CI: 1.196–2.812), DM (OR = 1.756, 95%CI: 1.163–2.653), and reduced diastolic function (OR = 3.033, 95%CI: 1.757–5.234) were independently related to AF in participants after adjustments for smoking, SBP, AHI, ODI, LSpO2, TG, creatinine, cerebrovascular disease, carotid atherosclerosis, PVD, COPD, and CKD (Table 3).
Discussion
Obesity is a well-known major risk factor for OSA [18], although the OSA risk conferred by obesity varies by ethnicity, with the Chinese population being particularly susceptible to weight increases [19]. In 2012, a health survey in Singapore found that, despite having lower body mass index (BMI), the Chinese population had a significantly higher prevalence of OSA (AHI ≥ 15 events/h) than the Indian and Malay population [20]. Furthermore, Chinese participants exhibited greater craniofacial bony restriction than Westerners with a higher BMI [21]. In our study, the median BMI for patients with mild, moderate, and severe OSA was 25.15, 25.76, and 27.64, respectively (3 groups both overweight, P < 0.05). A higher BMI was observed in OSA patients with than without AF (median BMI: 26.70 vs. 26.49, P > 0.05), but the difference was not statistically significant. In a large observational study of 6841 participants and a follow-up duration of 11.9 years, Cadby et al. [22] found that 455 participants who developed AF were both accepted polysomnography, and the predictors of AF identified on univariate analysis included BMI and, after multivariable adjustment, AHI (HR = 1.55, 95% CI: 1.21–2.00) was identified as an independent predictor of incident AF. There was no correlation between BMI and AF development. Gami et al. [23] found that, in participants younger than 65 years, independent predictors of incident AF included BMI (per 1 kg/m2, HR = 1.07, 95% CI 1.05 -1.10), whereas in participants aged 65 and older, heart failure, but not obesity, was a predictor of incident AF. In our study, the high prevalence of hypertension in OSA patients with and without AF could have resulted in the lack of statistically significant intergroup differences in the prevalence of hypertension. Nonetheless, in our study, the overall systolic BP was higher in OSA patients with AF than in non-AF OSA patients, and this result is in line with the result of the study by Kohno et al. [7].
Among the older participants with OSA in this study, the prevalence of AF was 9.5% and tended to further increase with OSA severity. The abovementioned results differ from those of Hendrikx [24], who reported an AF prevalence of 7.6% in patients suspected to have OSA. Furthermore, in our study, the prevalence of paroxysmal and persistent AF was 7.2% and 2.3%, respectively, and the prevalence of paroxysmal AF increased with the OSA severity. Tanaka et al. [8] reported a 72% prevalence of moderate–severe SA in patients with AF, and that the post-radiofrequency ablation AF recurrence significantly decreased with treatment of OSA [7, 8, 25]. OSA is defined as a partial or complete obstruction of the upper respiratory airway that repetitively causes dyspnea, along with intermittent hypoxemia, hypercapnia, and intrathoracic pressure changes, during nocturnal sleep [8, 26]. Nocturnal hypoxemia in OSA patients prolongs the atrial refractory period and slows atrial conduction, thereby increasing the heterogeneity of the cardiac conduction system. Moreover, repeated nocturnal apnea can promote sympathetic activation and increase the vagal tone to induce or worsen arrhythmia [25, 26]. Stroke, transient ischemic attack, and systemic embolism are the most serious consequences of AF and can significantly increase the mortality and disability rate [27].
Advanced age is a risk factor for both OSA and AF and is associated with dysfunctional and impaired immune regulation [10, 11]. Yao [10] et al. found that the prevalence of AF increases with age, especially in older populations (≥ 60 years). Consistent with the results of the above studies, we found a 5.4% increase in the risk of AF among patients for each additional year of age. Muscle relaxation and muscle strength decline could also be contributing factors in the older people population. A decrease in the upper airway muscle tone can lead to upper airway obstruction, increasing the prevalence of OSA in older people [28, 29]. The routine examination of OSA patients generally excludes 12-lead ECG or 24-h dynamic ECG; therefore, the possibility of AF in OSA patients is overlooked. We should focus on the screening of AF in older patients with OSA to prevent the development of OSA-related cardiovascular and cerebrovascular diseases.
History of drinking significantly contributes to AF occurrence. Drinking is reported to be a definite risk factor for new-onset AF and an independent predictor for recurrence of AF. A meta-analysis elaborated that moderate-severity levels of alcohol consumption are associated with an increase in AF risk, with each 1 SD per day consumed, by approximately 8% [30]. Takigawa [31] et al. demonstrated that the recurrence rate of AF after initial catheter ablation is higher in patients who consumed alcohol than in those who did not (41.9% VS 34.1%). Metabolites of alcohol have direct cardiotoxic effects and indirectly influence cardiac function by leading to disordered breathing during sleep, obesity, or nervous system diseases. Drinking affects the atrial electrophysiological properties, prolonging the intra-atrial and inter-atrial conduction, shortening the right-atrial effective refractory period, and increasing the dispersion of refractoriness, all leading to cardiac electrical remodeling and AF occurrence [32]. Given that the relationship between AF and OSA is well-documented, older OSA patients with binge drinking behaviors are highly prone to AF.
The risk of developing AF is 1.792-fold higher in DM patients than in those without DM, indicating that DM is another factor related to AF development and also closely related to OSA. Staszewsky [33] et al. showed that the risk of AF increases by 32% in patients with DM. Similarly, Papazoglou [34] et al. reported the DM-associated increase in AF risk of 40%. Huang [35]et al. revealed that the prevalence of DM is 16.8% in nonvalvular AF patients. Zhang [36] et al. reported the prevalence of OSA as 60% in hospitalized patients with type 2 DM. DM can affect the prognosis of AF patients, and OSA is closely related to insulin resistance or metabolic disorders. OSA increases sympathetic activity, disrupting the hypothalamus–pituitary–adrenal axis, thereby increasing the risk of metabolic disorders in individuals with OSA. Secondly, intermittent hypoxia in OSA causes oxidative stress, which can directly and indirectly promote inflammation, leading to insulin resistance and impaired beta cell function, setting the stage for DM [37]. Given the close association among OSA, DM, and AF, the prevalence of AF in patients with DM patients was higher than that in patients without DM in this study.
Cardiovascular diseases including CHD and reduced diastolic function were associated with AF among participants. The repeated hypoxemia and hypercapnia in OSA lead to oxidative stress and inflammation. Reactive oxygen species activate nuclear factor-kappa B, which leads to increased production of C-reactive protein, tumor necrosis factor-α, and interleukin-6, along with adhesion molecules such as E selectin, CD15, and CD32 that can cause endothelial damage, CHD, heart failure. And hypoxemia of OSA can decrease the oxygen of cardiomyocytes and cause systolic and diastolic dysfunction [2]. CHD and reduced diastolic function commonly result in abnormal autonomic modulation to affect the stability of cardiac electrophysiology and cardiac pump function to promote the development of AF [38]. It can be seen that cardiovascular diseases played an important role in the AF associated with older OSA patients.
This study was a multicenter, large-sample, explorative observational study, and we have attempted to include as many relevant factors as possible in our analysis. However, there were a few limitations that we assessed the risk factors of in the AF group and the non-AF group of OSA patients without including healthy controls. Secondly, this was a cross-sectional study to investigate the baseline among all participants. consequently, it was difficult to establish a causal relationship between OSA and AF. Finally, the study population was restricted to Chinese, which may have led to unexpected selection bias.
Conclusion
The prevalence of AF was in older patients with OSA was higher in our study than in similar previous studies. Age, drinking history, CHD, DM, and reduced diastolic function are dependent risk factors for AF. It is important to determine the risk of AF in older patients with OSA because it would reduce the occurrence of major cardiovascular events in these patients.
Availability of data and material
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- OSA:
-
Obstructive sleep apnea
- AF:
-
Atrial fibrillation
- BMI:
-
Body mass index
- SBP:
-
Systolic blood pressure
- DBP:
-
Diastolic blood pressure
- AHI:
-
The apnea–hypopnea index
- ODI:
-
The oxygen desaturation index
- TST:
-
Total sleep time
- MSpO2:
-
The mean pulse oxygen saturation
- LSpO2:
-
The lowest pulse oxygen saturation
- TG:
-
Triglyceride
- RBC:
-
Red blood cell
- WBC:
-
White blood cell
- PLTs:
-
Platelets
- Hb:
-
Hemoglobin
- GERD:
-
Gastroesophageal reflux disease
- CHD:
-
Coronary heart disease
- DM:
-
Diabetes mellitus
- PVD:
-
Peripheral vascular disease
- COPD:
-
Chronic obstructive pulmonary disease
- CKD:
-
Chronic kidney disease
References
Valenza MC, Baranchuk A, Valenza-Demet G, et al. Prevalence of risk factors for atrial fibrillation and stroke among 1210 patients with sleep disordered breathing. Int J Cardiol. 2014;174(1):73–6.
Khattak HK, Hayat F, Pamboukian SV, et al. Obstructive Sleep Apnea in Heart Failure: Review of Prevalence, Treatment with Continuous Positive Airway Pressure, and Prognosis. Tex Heart Inst J. 2018;45(3):151–61.
Drager LF, McEvoy RD, Barbe F, et al. Sleep Apnea and Cardiovascular Disease: Lessons From Recent Trials and Need for Team Science. Circulation. 2017;136(19):1840–50.
Ahmad M, Makati D, Akbar S. Review of and Updates on Hypertension in Obstructive Sleep Apnea. Int J Hypertens. 2017;2017:1848375.
Huang T, Lin BM, Stampfer MJ, et al. A Population-Based Study of the Bidirectional Association Between Obstructive Sleep Apnea and Type 2 Diabetes in Three Prospective U.S. Cohorts. Diabetes Care. 2018;41(10):2111–9.
Lyons OD, Ryan CM. Sleep Apnea and Stroke. Can J Cardiol. 2015;31(7):918–27.
Kohno T, Kimura T, Fukunaga K, et al. Prevalence and clinical characteristics of obstructive- and central-dominant sleep apnea in candidates of catheter ablation for atrial fibrillation in Japan. Int J Cardiol. 2018;260:99–102.
Tanaka N, Tanaka K, Hirao Y, et al. Home Sleep Apnea Test to Screen Patients With Atrial Fibrillation for Sleep Apnea Prior to Catheter Ablation. Circ J. 2021;85(3):252–60.
Ben Halima M, Sammoud K, Ben Amar J, et al. Prevalence and predictors of Sleep Apnea in Atrial Fibrillation patients. Tunis Med. 2020;98(12):1031–8.
Yao Y, Liu F, Wang Y, et al. Lipid levels and risk of new-onset atrial fibrillation: A systematic review and dose-response meta-analysis. Clin Cardiol. 2020;43(9):935–43.
Morales CR, Hurley S, Wick LC, et al. In-home, self-assembled sleep studies are useful in diagnosing sleep apnea in the elderly. Sleep. 2012;35(11):1491–501.
World Health Organization. Guidelines for controlling and monitoring the tobacco epidemic (World Health Organization, 1998).
Nagueh SF, Smiseth OA, Appleton CP, et al. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2016;17(12):1321–60.
Kapur VK, Auckley DH, Chowdhuri S, et al. Clinical Practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: An American Academy of Sleep Medicine Clinical Practice Guideline. J Clin Sleep Med. 2017;13(3):479–504 (Published 2017 Mar 15).
Berry RB, Budhiraja R, Gottlieb DJ, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med. 2012;8(5):597–619.
Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace. 2016;18(11):1609–78.
Gutierrez C, Blanchard DG. Diagnosis and Treatment of Atrial Fibrillation. Am Fam Physician. 2016;94(6):442–52.
Tuomilehto H, Seppä J, Uusitupa M. Obesity and obstructive sleep apnea–clinical significance of weight loss. Sleep Med Rev. 2013;17(5):321–9.
Lim DC, Pack AI. Obstructive Sleep Apnea: Update and Future. Annu Rev Med. 2017;14(68):99–112.
Tan A, Cheung YY, Yin J, et al. Prevalence of sleep-disordered breathing in a multiethnic Asian population in Singapore: a community-based study. Respirology. 2016;5:943–50.
Lee RW, Vasudavan S, Hui DS, et al. Differences in craniofacial structures and obesity in Caucasian and Chinese patients with obstructive sleep apnea. Sleep. 2010;33:1075–80.
Cadby G, McArdle N, Briffa T, Hillman DR, Simpson L, Knuiman M, Hung J. Severity of OSA is an independent predictor of incident atrial fibrillation hospitalization in a large sleep-clinic cohort. Chest. 2015;148(4):945–52.
Gami AS, Hodge DO, Herges RM, Olson EJ, Nykodym J, Kara T, Somers VK. Obstructive sleep apnea, obesity, and the risk of incident atrial fibrillation. J Am Coll Cardiol. 2007;49(5):565–71.
Hendrikx T, Sundqvist M, Sandström H, et al. Atrial fibrillation among patients under investigation for suspected obstructive sleep apnea. PLoS One. 2017;12(2):e0171575.
Abumuamar AM, Newman D, Dorian P, et al. Cardiac effects of CPAP treatment in patients with obstructive sleep apnea and atrial fibrillation. J Interv Card Electrophysiol. 2019;54(3):289–97.
Riaz S, Bhatti H, Sampat PJ, et al. The Converging Pathologies of Obstructive Sleep Apnea and Atrial Arrhythmias. Cureus. 2020;12(7):e9388.
Healey JS, Amit G, Field TS. Atrial fibrillation and stroke: how much atrial fibrillation is enough to cause a stroke? Curr Opin Neurol. 2020;33(1):17–23.
Hwang CH. Immediate muscle strengthening by an end-effector type gait robot with reduced real-time use of leg muscles: A case series and review of literature. World J Clin Cases. 2019;7(19):2976-85.
Rukhadze I, Fenik VB, Benincasa KE, et al. Chronic intermittent hypoxia alters density of aminergic terminals and receptors in the hypoglossal motor nucleus. Am J Respir Crit Care Med. 2010;182(10):1321–9.
Gallagher C, Hendriks JML, Elliott AD, et al. Alcohol and incident atrial fibrillation - A systematic review and meta-analysis. Int J Cardiol. 2017;246:46–52.
Takigawa M, Takahashi A, Kuwahara T, et al. Impact of Alcohol Consumption on the Outcome of Catheter Ablation in Patients With Paroxysmal Atrial Fibrillation. J Am Heart Assoc. 2016;5(12):e004149.
Zhang H, Ruan H, Rahmutula D, et al. Effect of acute and chronic ethanol on atrial fibrillation vulnerability in rats. Heart Rhythm. 2020;17(4):654–60.
Staszewsky L, Cortesi L, Baviera M, et al. Diabetes mellitus as risk factor for atrial fibrillation hospitalization: Incidence and outcomes over nine years in a region of Northern Italy. Diabetes Res Clin Pract. 2015;109(3):476–84.
Papazoglou AS, Kartas A, Samaras A, et al. Prognostic significance of diabetes mellitus in patients with atrial fibrillation. Cardiovasc Diabetol. 2021;20(1):40.
Huang B, Yang Y, Zhu J, et al. Clinical characteristics and impact of diabetes mellitus on outcomes in patients with nonvalvular atrial fibrillation. Yonsei Med J. 2015;56(1):62–71.
Zhang P, Zhang R, Zhao F, et al. The prevalence and characteristics of obstructive sleep apnea in hospitalized patients with type 2 diabetes in China. J Sleep Res. 2016;25(1):39–46.
Reutrakul S, Mokhlesi B. Obstructive Sleep Apnea and Diabetes: A State of the Art Review. Chest. 2017;152(5):1070–86.
Khan AA, Lip GYH, Shantsila A. Heart rate variability in atrial fibrillation: The balance between sympathetic and parasympathetic nervous system. Eur J Clin Invest. 2019;49(11):e13174.
Acknowledgements
Thanks for every member of this team, and thanks for the support of the Chinese PLA General Hospital fund.
Funding
This study was supported by Open Subject of National Clinical Research Center for Geriatric Diseases (NCRCG-PLAGH-2018008); Military Health Care Project (19BJZ34); Youth Program for Military Medicine of Chinese PLA General Hospital (QNC19054). The funders had not directly role in the design.
Author information
Authors and Affiliations
Contributions
Huanhuan Wang, JianHua Li, Yinghui Gao, Kaibing Chen, Yan Gao, JingJing Guo, LiBo Zhao, Xiaofeng Su, Min Shi, Xiao Zou, Weihao Xu, Yabin Wang, Juan Liu, Hu Xu, Junling Lin and Xiaoxuan Kong collected the data. Huanhuan Wang, JianHua Li, and Yinghui Gao analyzed the data and wrote the manuscript draft. Lin Liu, Jiming Han and Xiaoshun Qian designed this study. All the authors contributed to the paper review. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was carried out in accordance with the Declaration of Helsinki. Written informed consent was available from all participants. The ethics committee of PLA General Hospital approved the study (S2019-352–01).
Consent for publication
Not applicable
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, H., Li, J., Gao, Y. et al. Prevalence and factors associated with atrial fibrillation in older patients with obstructive sleep apnea. BMC Geriatr 22, 204 (2022). https://doi.org/10.1186/s12877-022-02791-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12877-022-02791-4