Abstract
Background
The clinical impact of neutralizing antibodies against interferon-beta (NAb) is controversial. Their presence can lead to a decrease in interferon-beta (IFNβ) efficacy. Fatigue reported in patients with multiple sclerosis (MS) may be associated with an unfavorable clinical course. We conducted a prospective multicentre study to assess the association between response to IFNβ, NAb and fatigue.
Methods
Patients with relapsing-remitting MS on IFNβ treatment were included. During the second year of treatment, the patients were analyzed for NAb status and non-response criteria to IFNβ (number of relapses ≥1 during the follow-up period, increase in the Expanded Disability Status Scale ≥0.5). The score on the Modified Fatigue Impact Scale (MFIS pathological if score ≥35) was noted for each patient.
Results
Of the 176 patients included: 22.3% were NAb positive, 54.5% presented non-response criteria to IFNβ, and 57.4% had a pathological MFIS score. Fatigue was increased in NAb + patients (p = 0.0014) and they were more likely to present non-response criteria to IFNβ (p = 0.041) than NAb- patients. Multivariate logistic regression analysis showed that the presence of NAb was related to fatigue (p = 0.0032) and denoted disease activity in these patients (p = 0.026).
Conclusions
This study demonstrates the impact of NAb on the non-clinical response to IFNβ. Fatigue assessment is an indicator of IFNβ responsiveness and a predictive biomarker of deterioration on patient’s neurological status.
Similar content being viewed by others
Background
Immunomodulatory treatment with interferon-beta (IFNβ) is a first-line treatment for patients with relapsing-remitting multiple sclerosis (MS). As with any therapy derived from human recombinant proteins, this treatment has immunogenic properties [1]. Neutralizing antibodies (NAb) against IFNβ develop in 2% to 46% of treated patients [2]-[14]. This large variability in observed NAb prevalence can be explained by the more immunogenic character of IFNβ-1b compared with IFNβ-1a [13], the increased prevalence of NAb with multi-weekly injections [9], the more immunogenic nature of subcutaneous administration as opposed to intramuscular administration [8],[13],[14], the dose treatment [7],[11] and the different follow-up durations. Moreover, a positive NAb status may be reversible over time [12],[13],[15]. The origin of the reversibility of NAb status is unknown but the hypothesis of a re-establishment of immune tolerance after a breakdown period with IFNβ-treatment is possible [16]. While it is recognized that NAb has a negative impact on magnetic resonance imaging (MRI), the effect of NAb on clinical outcome remains a subject of debate to this day. Indeed, some studies have found conflicting results regarding the impact of NAb on the clinical response to IFNβ [2],[4],[5],[8]-[12],[14],[17],[18]. The variability of the results about the impact of NAb may depend on the statistical approaches used in these studies, which should consider that many of the NAb positive patients revert to a NAb negative status over time [19]. Another factor, that can impact on NAb status is the neutralizing assay used in these different studies [19]. The interpretation of the NAb status is consequently difficult for the clinician to analyze. This has given rise to recommendations aimed at facilitating the decision as to whether to test for NAb or not [19].
Fatigue is a symptom reported by 53-92% of patients with MS and is one of its most disabling symptoms [20]-[23]. Direct involvement of immunological factors has been suggested as a pathophysiological mechanism responsible for fatigue during MS [24],[25]. Moreover, the intensity of fatigue (physical and psychosocial fatigue), was statistically correlated with the EDSS (Expanded Disability Status Scale) and physical fatigue was a prognostic marker of a worsening of the disability status after a follow-up period of three years [26],[27].
We hypothesized that fatigue could be predictive of non-response to treatment with IFNβ. Accordingly, we studied the association between response to IFNβ, fatigue and the presence of NAb.
Methods
Inclusion criteria
To be included patients had to be ≥18 years, IFNβ naive, with an EDSS ≤5.0, and fulfilling the clinical criteria for treatment with IFNβ, i.e., patients with a clinically isolated syndrome (CIS) with an active inflammatory process severe enough to need intravenous corticosteroids, if alternative diagnoses had been excluded, and if these patients were considered at high risk of developing clinically definite MS, or patients with relapsing-remitting MS with at least two relapses within the last two years [28].
Study design
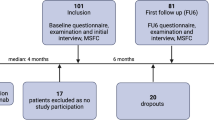
This was a prospective, multicentre study, in the neurological department of three hospitals in France: Strasbourg, Rennes and Nancy. Patients fulfilling the inclusion criteria underwent two specific consultations. The initial consultation, called the “inclusion consultation”, was performed when IFNβ was initiated. During this inclusion consultation, patient consent was obtained and the pre-IFNβ EDSS was assessed. The choice of the IFNβ was at the discretion of the clinician: intramuscular IFNβ-1a (Avonex®, Biogen Idec, Cambridge, MA, USA), subcutaneous IFNβ-1a (22 or 44 μg REBIF®, Merck Serono, Geneva, Switzerland), subcutaneous IFNβ-1b (BETAFERON®, Bayer Schering, Berlin, Germany; EXTAVIA®, Novartis, Dorval, Canada). A second consultation, called the “follow-up consultation”, was used to assess the clinical response to IFNβ, the fatigue experienced, the severity of the flu-like syndrome and the patient’s mood. This “follow-up consultation” happened during the second year of treatment. This time for this consultation was chosen to systematically test patients for NAb, as recommended by the experts panel recommendations [19]. During this “follow-up” consultation, patients and physicians were not aware of the result of NAb status. A written informed consent for participation in the study was obtained from all patients. Because the protocol of this study did not modify the classical clinical practice, it was not necessary, in accordance with the French law at the beginning of the study, to require the approval of a specific medical ethic committee. Nevertheless, this study was approved by the Neurology Department of Nancy University Hospital and was conducted according to the criteria of the Helsinki Declaration. Moreover, the use of the database was approved by the CNIL (Commission Nationale de l’Informatique et des Libertés – National Commission on Information Technology and Liberties) and the CCTIRS (Comité Consultatif sur le Traitement de l’Information en matière de Recherche dans le domaine de la Santé – Consultative Committee on Clinical Research Data Management).
Evaluation of response to IFNβ treatment
The response to IFNβ was assessed at a clinical level only. Non-response to IFNβ between the inclusion and follow-up consultations was defined as the occurrence of at least one MS relapse and/or an increase in EDSS ≥0.5, confirmed after six months. A MS relapse was defined as the occurrence, the recurrence or the worsening for more than 24 hours of neurological symptoms and usually ending in a partial or complete remission [29],[30]. A patient was considered to be an IFNβ responder if there was no relapse or worsening of EDSS during follow-up period.
Evaluation of fatigue
Fatigue was assessed during the follow-up consultation with the French version of the MFIS [23],[31]. The MFIS is a self-administered questionnaire, validated in French, which explores fatigue experienced during the previous four weeks through a set of 21 items. Each item is marked from zero (“never”) to four (“almost always”). The final score thus ranges from zero to 84. In our study, a patient was considered tired if his MFIS score was ≥35.
Evaluation of the severity of the flu-like syndrome
Under IFNβ, fatigue may be secondary to the flu-like syndrome. Consequently, the flu-like syndrome was also evaluated in this study, using items 13 to 16 from the MSTCQ (Multiple Sclerosis Treatment Concern Questionnaire) [32]. The MSTCQ is a self-administered questionnaire assessing the overall tolerability of IFNβ. Items 13–16 assess more specifically the flu-like syndrome. Each of these four items is marked from zero to five. The final score thus ranges from zero to 20.
Evaluation of mood
As a depressive syndrome can be correlated with fatigue during MS, we also assessed mood [26]. For this we used the MADRS (Montgomery and Asberg Depression Rating Scale) which is a hetero-administered questionnaire consisting of 10 items, marked from zero to six and validates in French [33],[34]. The final score thus ranges from zero to 60.
Testing for neutralizing antibodies against IFNβ
NAb testing was systematically carried out by the hormonology department of the University Hospital of Rennes using a method called “Luciferase Reporter Gene Assay” [35]. NAb titers were calculated using the Kawade-Grossberg formula in TRU (Ten fold Reduction neutralizing Unit)/mL [36]. A titer just under 20 TRU/mL was considered negative; between 20 and 100 TRU/mL and 20 to 400 TRU/mL weakly to moderately positive for IFNβ-1a and IFNβ-1b respectively; and over 100 TRU/mL and 400 TRU/mL highly positive for IFNβ-1a and IFNβ-1b respectively. There was no other NAb testing to confirm NAb positive status.
Statistical analyses
All statistical analyses were performed using the SAS 9.2® software. All figures were created using GraphPad Prism 5®. Bivariate analyses were expressed as mean difference +/- SEM (Standard Error Mean). The quantitative variables were compared by Student t-test or Man-Whitney test and qualitative variables by chi-square test or Fisher’s exact test. The correlation between variables was determined by calculating the Spearman coefficient. The multivariate analysis, to eliminate confounding factors, used logistic regression models. This model was preferred to a survival model because all data were available and collected on the anniversary date of 2 years precisely. A p < 0.05 was considered statistically significant.
Results
Included patient characteristics
In all 176 patients fulfilled the inclusion criteria and participated in this study: 131 (74.4%) were females, 45 (25.6%) were males giving a sex ratio of 2.91. The average age at MS diagnosis was 34 +/- 0.8 years and at study inclusion 36 +/- 0.8 years. The mean duration of MS course before inclusion was 5.3 +/- 0.4 years. The mean EDSS at introduction of IFNβ was 1.7 +/- 0.1 and the mean number of relapses before treatment 2.4 +/- 0.1. At inclusion consultation, 51 patients were CIS with only one MS relapse. The median time from the inclusion consultation to the follow-up consultation was 411 days. Twenty-eight patients (16.1%) received intramuscular IFNβ-1a, 47 (27%) subcutaneous IFNβ-1b and 99 (56.9%) subcutaneous IFNβ-1a. Thirty-nine patients (22.3%) had developed a positive NAb status (NAb+) in the second year of treatment. Ninety-six patients (54.5%) were classified as non-responders to IFNβ between the two consultations (relapse ≥1 or ΔEDSS ≥0.5) and 101 patients (57.4%) were tired at the follow-up consultation.
NAb + patients characteristics
The 39 NAb + patients were compared with the 136 patients who remained NAb- during follow-up (Table 1). Both groups of patients were similar in terms of age at MS diagnosis (NAb-: 33.8 +/- 0.9 years vs. NAb+: 35.2 +/- 1.5 years, p = 0.48, Student t-test), age at treatment initiation (35.5 +/- 0.9 years vs. 37.3 +/- 1.6 years, p = 0.32, Student t-test), duration of disease progression (5.3 +/- 0.5 years vs. 4.9 +/- 0.9 years, p = 0.66, Student t-test), EDSS at inclusion consultation (1.7 +/- 0.1 vs. 1.8 +/- 0.2, p = 0.66, Student t-test) and number of relapses before inclusion consultation (2.4 +/- 0.1 vs. 2.4 +/- 0.2, p = 0.98, Student t-test). There was also no statistically significant difference between the two groups regarding the IFNβ type (intramuscular IFNβ-1a: 25 (18.5%) vs. 3 (7.9%) patients; subcutaneous IFNβ-1a: 77 (57%) vs. 22 (57.9%) patients; subcutaneous IFNβ-1b SC: 33 (24.4%) vs. 13 (34.2%) patients, p = 0.21, Fisher’s exact test). In contrast, there was a statistically significant difference in terms of sex with an F/M sex ratio of 2.32 in the NAb- group vs. 8.75 in the NAb + group (p = 0.012, Fisher’s exact test).
Relationship between NAb titer and clinical response to IFNβ
We compared the NAb- group with the NAb + group and studied the impact of NAb on the response to IFNβ. On bivariate analysis, we found a statistically significant difference during the follow-up period in terms of number of relapses (NAb-: 0.6 +/- 0.1 vs. NAb + 1.0 +/- 0.2, p = 0.016, Student t-test) and EDSS variation (ΔEDSS = 0.1 +/- 0.1 vs. 0.4 +/- 0.1, p = 0.035, Student t-test) (Figure 1a and b). Thus, the ratio Responder/Non-Responder to IFNβ is statistically different between the two groups (Responder/Non-Responder: 67/69 (49.3%/50.7%) vs. 12/27 (30.8%/69.2%), p = 0.041, Chi-square test) (Figure 1c).
Comparison of NAb- and NAb + groups in terms of response to IFNβ treatment during the follow-up period. a. Mean number of relapse during follow-up period between NAb- and NAb + groups. b. EDSS variation during follow-up period between NAb- and NAb + groups. c. Number of responder and non responder in NAb- and NAb + groups. *p <0.05.
Multivariate analysis demonstrated that non-response to IFNβ is the only factor explaining a NAb + status (OR = 2.4, 95% CI: 1.1-5.3; p = 0.026).
NAb impact on the different scales
Bivariate analysis showed a trend for NAb + patients to have a higher MADRS score (4.8 +/- 0.7) than NAb- patients (3.6 +/- 0.4; p = 0.14; Student t-test) (Figure 2a). NAb + patients tend to be less affected than NAb- patients by the flu-like syndrome as assessed by items 13 to 16 of the MSTCQ (5.9 +/- 0.8 vs. 7.2 +/- 0.4; p = 0.086; Student t-test) (Figure 2b). In terms of fatigue on the MFIS score, there was a significant difference between the NAb- group (34.2 +/- 1.7) and the NAb + group (47.3 +/- 2.9; p = 0.0002; Student t-test) (Figure 2c). Also, the ratio No fatigue patients/Fatigue patients was significantly different between the NAb- group (67/69) and the NAb + group (8/31; p = 0.0014, Chi-square test) (Figure 2d).
Comparison between NAb- and NAb + groups on the different scales at the time of the follow-up consultation. a. Mean MADRS score at follow-up consultation between NAb- and NAb + groups. b. Mean MSTCQ score at follow-up consultation between NAb- and NAb + groups. c. Mean MFIS score at follow-up consultation between NAb- and NAb + groups. d. Number of fatigue and no fatigue in NAb- and NAb + groups. **p <0.01, ***p <0.001.
Relationship between response to IFNβ and fatigue
Bivariate analysis did not reveal any significant differences but only a clear tendency for fatigue patients to be non-responders to IFNβ (Responder/Non-Responder: 41/60 (40.6%/59.4%)) compared with patients without fatigue (39/36 (52.0%/48.0%); p = 0.13; Chi-square test) (Figure 3).
NAb titer and fatigue intensity
Among the 39 NAb + patients, the mean NAb titer was 878 +/- 346 TRU/mL. Our study did not reveal any correlation between the NAb titer and MFIS score (Spearman coefficient r = 0.14; p = 0.38) (Figure 4a).
Fourteen patients (35.9%) were classified as weakly or moderately positive and 25 patients (64.1%) had a strong positive titer. The mean MFIS score for patients with a low to moderate positive titer was statistically similar to the score for patients with a strong positive titer (respectively 47.5 +/- 4.7 and 47.2 +/- 3.8; p = 0.98; Man-Whitney test) (Figure 4b).
A positive NAb titer is related to female sex and fatigue and is inversely related to the intensity of flu-like syndrome
Multivariate analysis demonstrated a statistically significant association between NAb + and fatigue (OR = 4.6; 95% CI: 1.7 to 12.8; p = 0.0032). There was also a statistically significant relation between the female sex and NAb + (OR = 3.4; 95% CI: 1.05 to 11.0; p = 0.04). Moreover, the intensity of the flu-like syndrome was inversely correlated to the probability of developing a NAb + titer (OR = 0.88; 95% CI: 0.80-0.97; p = 0.01).
CIS subgroup of patients
Among the 51 CIS patients at inclusion consultation, 23 (45.1%) converted into a clinically definite MS. These 23 patients were compared to the 28 CIS patients without conversion to a clinically definite MS. There was no statistically significant difference between these two groups in terms of Nab status, EDSS score, fatigue, depression and flu-like syndrome intensity (Table 2).
Discussion
Our study demonstrates the impact of NAb on the non-clinical response to IFNβ in relapsing-remitting MS patients. The clinical impact is characterized both by the existence of relapses during follow-up and by the worsening of EDSS at the end of the follow-up period. Our hypothesis that fatigue could be predictive of a deterioration of the patient’s neurological status can be tentatively confirmed. Nevertheless, this hypothesis is supported by the demonstration of a link between the presence of NAb and fatigue, NAb being involved in non-response to IFNβ. In addition, our study suggests that women are at greater risk of developing NAb. We show that the presence of the flu-like syndrome during follow-up is predictive for the absence of NAb.
The non-response to IFNβ in these patients reflects both the number of relapses during follow-up and changes in EDSS. To our knowledge, only one study to date has demonstrated the negative role of these two clinical parameters but this study only included 78 patients [10]. Other studies have found divergent results and are summarized in Table 3 [2],[4],[5],[8]-[12],[14],[17],[18].
These differences in results can be explained by methodological differences from one study to another, especially in the duration of the follow-up period. The median follow-up of patients in our study was 411 days which is a short follow-up duration. Thus, it could be envisaged that the clinical impact of NAb on the non-response to IFNβ is expressed mainly at the beginning of treatment and that the negative clinical impact of NAb would tend to decrease over time. This hypothesis is supported by the possibility of a NAb reversibility [12],[13],[15]. However, we can conclude that the presence of NAb is a marker for therapeutic ineffectiveness to IFNβ at the beginning of treatment.
The various IFNβ available for the clinician have different immunogenicity properties [7]-[9],[11],[13],[14]. In terms of non-response to IFNβ induced by NAb, our study would point towards the use of the least immunogenic IFNβ, i.e., intramuscular IFNβ-1a. To our knowledge, no study has compared the therapeutic response to the different forms of IFNβ on the annualized relapse rate and EDSS progression, except the work of Etemadifar et al. which was on a small population of 90 patients [37]. In the same vein, our study found that the risk of developing NAb was higher for women than for men. This finding could suggest a better clinical response to IFNβ in male patients. Few studies have focused on the clinical response to IFNβ according to gender. One study, by Rudick et al., found no difference in intramuscular IFNβ-1a efficacy between men and women but this result may be biased due to the low prevalence of NAb in the study as intramuscular IFNβ-1a is the least immunogenic IFNβ [38].
Fatigue could be the sign of a negative clinical course of MS and may even be the only manifestation of an authentic relapse [39]. In our work, we found a clear trend for non-responder patients to IFNβ to be more tired than responder patients. It has already been suggested in the past that fatigue is a prognostic marker for an unfavorable neurological outcome [27]. We demonstrate a highly significant relation between fatigue and Nab + status. As Nab + status is related to non-response to IFNβ, we believe that fatigue is a clinical marker for non-response to IFNβ. Thus, it would seem appropriate to propose that tired patients under IFNβ be tested for NAb and, if a NAb + status is detected, to consider an alternative therapy. Thereby, in 2008, Capobianco et al. demonstrated the advantage of a treatment change to glatiramer acetate in NAb + patients with a gain of 20.1 months without relapse in this setting [40]. Our work demonstrates that the intensity of fatigue is not correlated to the value of the NAb titer. Thus, in the hypothesis that fatigue is a reflection of non-response to IFNβ treatment, a high NAb titer would not denote a poorer clinical response than a low or a moderate titer.
Nevertheless, this study demonstrates that fatigue can also occur in patients with NAb negative status. It leads to consider that fatigue during MS is not only related with NAb status. Indeed, there are several dimensions to MS-related fatigue [41],[42]. Primary fatigue is directly related to the pathophysiological mechanisms associated with MS, including direct involvement of immunological factors [24],[25]. Secondary fatigue is explained by the direct effects of the disease (sleep disorder, reactive depression, analgesic treatment of nociceptive pain, etc.).
Interestingly, the occurrence of the flu-like syndrome under IFNβ seems to be prognostic for NAb- status. Indeed, the intensity of the flu-like syndrome is inversely correlated to the risk of being NAb+. The flu-like syndrome is one of the major causes for patients to stop a long-term IFNβ treatment. Hartung et al. found a similar result in their study: a clear trend in NAb- patients to stop their treatment with IFNβ compared with NAb + patients due to the adverse effects of the treatment which were more prevalent in the former [12]. Therefore, the existence of a flu-like syndrome during the follow-up of patients receiving IFNβ is a good prognostic factor for the absence of NAb and, paradoxically, would suggest that treatment should be maintained. However, this result should be interpreted with caution because the flu-like syndrome in our study was assessed by a subset of the MSTCQ-score. This score, though validated to assess the overall adverse effects of a treatment with IFNβ, has not yet been validated for all the subsets.
Our study has some limitations. Firstly, this work has an open non randomized study design that may influence the results and lead to a selection bias. Furthermore, the work focuses on a relatively small population of 176 patients with a median follow-up just under 14 months. These two parameters give our study a low statistical power to clearly demonstrate a significant relation between fatigue and non-response to IFNβ. The link between fatigue and adverse neurological outcome in the work of Debouverie et al. was demonstrated after a 3-year follow-up [27]. We believe that a longer follow-up on a larger population would allow us to confirm this correlation. In addition, non-response to IFNβ can also be evaluated radiologically and not only clinically. It is possible that some of our patients who were considered IFNβ responders could have a radiological activity of the disease. This might be a bias in our study in the classification of patients. Moreover, it would be interesting to know for NAb + patients the time period spent with NAb- status, that can also impact the interpretation of the results [19].
Conclusion
Our study establishes the impact of NAb on the non-response to IFNβ in patients with relapsing-remitting MS. Moreover, there is a tripartite relation between response to IFNβ, fatigue and NAb status. NAb positive status is correlated with an increased prevalence of fatigue during follow-up. Although the link between fatigue and non-response to IFNβ cannot be demonstrated directly because of a lack of statistical power, we found a clear trend to suggest a link between these two parameters. Thus, fatigue in patients treated with IFNβ constitutes a good clinical marker for non-response to treatment. Detection of fatigue by a validated scale should lead the clinician to test for NAb status and possibly to propose an alternative therapeutic treatment to IFNβ.
Authors' contributions
CL performed the statistical analysis. SP, GE and JDS participated in the design of the study. CM carried out the biological analysis of neutralizing antibodies against interferon-beta. MD conceived of the study, and participated in its design and coordination and helped to draft the manuscript. PM participated in the design of the study, funding, and writing the manuscript. All authors read and approved the final manuscript.
Abbreviations
- CIS:
-
Clinically isolated syndrome
- EDSS:
-
Expanded disability status scale
- IFNβ:
-
Interferon-beta
- MADRS:
-
Montgomery and asberg depression rating scale
- MFIS:
-
Modified fatigue impact scale
- MS:
-
Multiple sclerosis
- MSTCQ:
-
Multiple sclerosis treatment concern questionnaire
- MRI:
-
Magnetic resonance imaging
- NAb:
-
Neutralizing antibodies against interferon-beta
- OD:
-
Odd ratio
- SC:
-
Subcutaneous
- SEM:
-
Standard error mean
- TRU:
-
Ten fold Reduction neutralizing Unit
- vs:
-
Versus
References
Schellekens H, Casadevall N: Immunogenicity of recombinant human proteins: causes and consequences. J Neurol. 2004, 251 (Suppl 2): II4-II9.
Neutralizing antibodies falling on treatment of multiple sclerosis with interferon beta-1b: experience falling on the first three years. Neurology. 1996, 47 (4): 889-894.
Jacobs LD, Cookfair DL, Rudick RA, Herndon RM, Richert GR, Salazar AM, Fischer JS, Goodkin DE, Granger CV, Simon JH, Alam JJ, Bartoszak DM, Bourdette DN, Braiman J, Brownscheidle CM, Coats ME, Cohan SL, Dougherty DS, Kinkel RP, Mass MK, Munschauer FE, Priore RL, Pullicino PM, Scherokman BJ, Whitham RH: Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis: the Multiple Sclerosis Collaborative Research Group (MSCRG). Ann Neurol. 1996, 39 (3): 285-294.
Rudick RA, Simonian NA, Alam JA, Campion M, Scaramucci JO, Jones W, Coats ME, Goodkin DE, Weinstock-Guttman B, Herndon RM, Mass MK, Richert JR, Salazar AM, Munschauer FE, Cookfair DL, Simon JH, Jacobs LD: Incidence and significance of neutralizing antibodies to interferon beta-1a in multiple sclerosis: Multiple Sclerosis Collaborative Research Group (MSCRG). Neurology. 1998, 50 (5): 1266-1272.
Randomised double-blind placebo-controlled study of interferon beta-1a in relapsing/remitting multiple sclerosis. Lancet. 1998, 352 (9139): 1498-1504.
Jacobs LD, Beck RW, Simon JH, Kinkel RP, Brownscheidle CM, Murray TJ, Simonian NA, Slasor PJ, Sandrock AW: Intramuscular interferon beta-1a therapy initiated falling on a first demyelinating event in multiple sclerosis. CHAMPS Study Group. N Engl J Med. 2000, 343 (13): 898-904.
Clanet M, Radue EW, Kappos L, Hartung HP, Hohlfeld R, Sandberg-Wollheim M, Kooijmans-Couthino MF, Tsao EC, Sandrock AW: A randomized, double-blind, dose-comparison study of weekly interferon beta-1a in relapsing MS. Neurology. 2002, 59 (10): 1507-1517.
Panitch H, Goodin DS, Francis G, Chang P, Coyle PK, O’Connor P, Monaghan E, Li D, Weinschenker B: Randomized, comparative study of interferon beta-1a treatment regimens in MS: the EVIDENCE Trial. Neurology. 2002, 59 (10): 1496-1506.
Sorensen PS, Ross C, Clemmesen KM, Bendtzen K, Frederiksen JL, Jensen K, Kristensen O, Petersen T, Rasmussen S, Ravnborg M, Stenager E, Koch-Henriksen N: Clinical importance of neutralising antibodies against interferon beta in patients with relapsing-remitting multiple sclerosis. Lancet. 2003, 362 (9391): 1184-1191.
Malucchi S, Sala A, Gilli F, Bottero R, Di Sapio A, Capobianco M, Bertolotto A: Neutralizing antibodies reduce the efficacy of betaIFN during treatment of multiple sclerosis. Neurology. 2004, 62 (11): 2031-2037.
Freedman MS, Francis GS, Sanders EA, Rice GP, O’Connor P, Comi G, Duquette P, Metz L, Murray TJ, Bouchard JP, Abramsky O, Pelletier J, O’Brien F: Randomized study of once-weekly interferon beta-1Ia therapy in relapsing multiple sclerosis: three-year data from the OWIMS study. Mult Scler. 2005, 11 (1): 41-45.
Hartung HP, Freedman MS, Polman CH, Edan G, Kappos L, Miller DH, Montalban X, Barkhof F, Petkau J, White R, Sahajpal V, Knappertz V, Beckmann K, Lanius V, Sandbrink R, Pohl C: Interferon β-1b-neutralizing antibodies 5 years after clinically isolated syndrome. Neurology. 2011, 77 (9): 835-843.
Hegen H, Schleiser M, Gneiss , Di Pauli F, Ehling R, Kuenz B, Lutterotti A, Berger T, Deisenhammer F: Persistency of neutralizing antibodies depends on titer and interferon-beta preparation. Mult Scler. 2012, 18 (5): 610-615.
Sato DK, Nakashima I, Fukazawa T, Shimizu Y, Tomizawa Y, Yokoyama K, Misu T, Creeke PI, Farrell R, Giovannoni G, Itoyama Y, Fujihara K, Aoki M: Neutralizing antibodies are associated with a reduction of interferon-β efficacy during the treatment of Japanese multiple sclerosis patients. Tokohu J Exp Med. 2012, 228 (2): 85-92.
Sorensen PS, Koch-Henriksen N, Ross C, Clemmensen KM, Bendtzen K: Appearance and disappearance of neutralizing antibodies during interferon-beta therapy. Neurology. 2005, 65 (1): 33-39.
Van der Meide PH, Schellekens H: Anti-cytokine autoantibodies: epiphenomenon or critical modulators of cytokine action. Biotherapy. 1997, 10 (1): 39-48.
Francis GS, Rice GP, Alsop JC: Interferon beta-1a in MS: results following development of neutralizing antibodies in PRISMS. Neurology. 2005, 65 (1): 48-55.
Kappos L, Clanet M, Sandberg-Wollheim M, Radue EW, Hartung HP, Hohlfeld R, Xu J, Bennett D, Sandrock A, Goelz S: Neutralizing antibodies and efficacy of interferon beta-1a: a 4-year controlled study. Neurology. 2005, 65 (1): 40-47.
Polman CH, Bertolotto A, Deisenhammer F, Giovannoni G, Hartung HP, Hemmer B, Killestein J, McFarland HF, Oger J, Pachner AR, Petkau J, Reder AT, Reingold SC, Schellekens H, Sorensen PS: Recommendations for clinical use of data on neutralising antibodies to interferon-beta therapy in multiple sclerosis. Lancet Neurol. 2010, 9 (7): 740-750.
Flachenecker P, Kümpfel T, Kallmann B, Gottschalk M, Grauer O, Rieckmann P, Trenkwalder C, Toyka KV: Fatigue in multiple sclerosis: a comparison of different rating scales and correlation to clinical parameters. Mult Scler. 2002, 8 (6): 523-526.
Boërio D, Lefaucheur JP, Hogrel JY, Créange A: Pathophysiology and treatment of fatigue in multiple sclerosis. Rev Neurol. 2006, 162 (3): 311-320.
Krupp L: Fatigue is intrinsic to multiple sclerosis (MS) and is the most commonly reported symptom of the disease. Mult Scler. 2006, 12 (4): 367-368.
Fisk JD, Pontefract A, Ritvo PG, Archibald CJ, Murray TJ: The impact of fatigue on patients with multiple sclerosis. Can J Neurol Sci. 1994, 21 (1): 9-14.
Heesen C, Nawrath L, Reich C, Bauer N, Schulz KH, Gold SM: Fatigue in multiple sclerosis: an example of cytokine mediated sickness behavior?. J Neurol Neurosurg Psychiatry. 2006, 77 (1): 34-39.
Flachenecker P, Bihler I, Weber F, Gottschalk M, Toyka KV, Rieckmann P: Cytokine mRNA expression in patients with multiple sclerosis and fatigue. Mult Scler. 2004, 10 (2): 165-169.
Pittion-Vouyovitch S, Debouverie M, Guillemin F, Vandenberghe N, Anxionnat R, Vespignani H: Fatigue in multiple sclerosis is related to disability, depression and quality of life. J Neurol Sci. 2006, 243 (1-2): 39-45.
Debouverie M, Pittion-Vouyovitch S, Brissart H, Guillemin F: Physical dimension of fatigue correlated with disability change over time in patients with multiple sclerosis. J Neurol. 2008, 255 (5): 633-636.
Polman CH, Reingold SC, Edan G, Filippi M, Hartung HP, Kappos L, Lublin FD, Metz LM, McFarland HF, O'Connor PW, Sandberg-Wollheim M, Thompson AJ, Weinshenker BG, Wolinsky JS: Diagnostic criteria for multiple sclerosis: 2005 revisions to the “McDonald Criteria”. Ann Neurol. 2005, 58 (6): 840-846.
Matthews WB: Symptoms and signs. McAlpine’s Multiple Sclerosis. Edited by: Matthews WE. 1991, Churchill Livingstone, Edinburgh, 61-63. 2
Schumacher GA, Beebe G, Kibler RF, Kurland LT, Kurtzke JF, McDowell F, Nagler B, Sibley WA, Tourtellotte WW, Willmon TL: Problems of experimental trials of therapy in multiple sclerosis. Ann NY Acad Sci. 1965, 122: 552-568.
Debouverie M, Pittion-Vouyovitch S, Louis S, Guillemin F: Validity of a French version of the fatigue impact scale in multiple sclerosis. Mult Scler. 2007, 13 (8): 1026-1032.
Cramer JA, Cuffel BJ, Divan V, Al-Sabbagh A, Glassman M: Patient satisfaction with an injection device for multiple sclerosis treatment. Acta Neurol Scand. 2006, 113 (3): 156-162.
Montgomery SA, Asberg M: A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979, 134: 382-389.
Pellet J, Bobon DP, Mormont I, Lang F, Massardier A: Étude Princeps de Validation Française de la MADRS: Sous-Échelle Dépression de la CPRS. Congrès de psychiatrie et de neurologie de langue française Reims. 1980
Lam R, Farrell R, Aziz T, Gibbs E, Giovannoni G, Grossberg S, Oger J: Validating parameters of a luciferase reporter gene assay to measure neutralizing antibodies to IFNbeta in multiple sclerosis patients. J Immunol Methods. 2008, 336 (2): 113-118.
Grossberg SE, Kawade Y, Kohase M, Yokoyama H, Finter N: The neutralization of interferons by antibody. I. Quantitative and theoretical analyses of the neutralization reaction in different bioassay systems. J Interferon Cytokine Res. 2001, 21 (9): 729-742.
Etemadifar M, Janghorbani M, Shaygannejad V: Comparison of Betaferon, Avonex, and Rebif in treatment of relapsing-remitting multiple sclerosis. Acta Neurol Scand. 2006, 113 (5): 283-287.
Rudick RA, Kappos L, Kinkel R, Clanet M, Phillips JT, Herndon RM, Sandrock AW, Munschauer FE: Gender effects on intramuscular interferon beta-1a in relapsing-remitting multiple sclerosis: analysis of 1406 patients. Mult Scler. 2011, 17 (3): 353-360.
Flachenecker P, Meissner H: Fatigue in multiple sclerosis presenting as acute relapse: subjective and objective assessment. Mult Scler. 2008, 14 (2): 274-277.
Capobianco M, Rizzo A, Malucchi S, Sperli F, Di Sapio A, Oqqero A, Zaffaroni M, Ghezzi A, Bertolotto A: Glatiramer acetate is a treatment option in neutralising antibodies to interferon-beta-positive patients. Neurol Sci. 2008, 29 (Suppl 2): S227-S229.
Kos D, Kerckhofs E, Nagels G, D'Hoogue MB, Ilsbroukx S: Origin of fatigue in multiple sclerosis: review of the literature. Neurorehabil Neural Repair. 2008, 22 (1): 91-100.
Andreasen AK, Stenager E, Dalgas U: The effect of exercise therapy on fatigue in multiple sclerosis. Mult Scler. 2011, 17 (9): 1041-1054.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Manceau, P., Latarche, C., Pittion, S. et al. Neutralizing antibodies and fatigue as predictors of low response to interferon-beta treatment in patients with multiple sclerosis. BMC Neurol 14, 215 (2014). https://doi.org/10.1186/s12883-014-0215-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12883-014-0215-y