Abstract
Background
To investigate the value of semi-quantitative and quantitative parameters (PI-RADS score, T2WI score, ADC, Ktrans, and Kep) based on multiparametric MRI (mpMRI) or biparametric MRI (bpMRI) combined with prostate specific antigen density (PSAD) in detecting clinically significant prostate cancer (csPCa).
Methods
A total of 561 patients (276 with csPCa; 285 with non-csPCa) with biopsy-confirmed prostate diseases who underwent preoperative mpMRI were included. Prostate volume was measured for calculation of PSAD. Prostate index lesions were scored on a five-point scale on T2WI images (T2WI score) and mpMRI images (PI-RADS score) according to the PI-RADS v2.1 scoring standard. DWI and DCE-MRI images were processed to measure the quantitative parameters of the index lesion, including ADC, Kep, and Ktrans values. The predictors of csPCa were screened by logistics regression analysis. Predictive models of bpMRI and mpMRI were established. ROC curves were used to evaluate the efficacy of parameters and the model in diagnosing csPCa.
Results
The independent diagnostic accuracy of PSA density, PI-RADS score, T2WI score, ADCrec, Ktrans, and Kep for csPCa were 80.2%, 89.5%, 88.3%, 84.6%, 58.5% and 61.6%, respectively. The diagnostic accuracy of bpMRI T2WI score and ADC value combined with PSAD was higher than that of PI-RADS score. The combination of mpMRI PI‑RADS score, ADC value with PSAD had the highest diagnostic accuracy.
Conclusions
PI-RADS score according to the PI-RADS v2.1 scoring standard was the most accurate independent diagnostic index. The predictive value of bpMRI model for csPCa was slightly lower than that of mpMRI model, but higher than that of PI-RADS score.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Prostate cancer (PCa) is the second most common male cancer with the highest incidence in Western countries [1]. Multiparametric magnetic resonance imaging (mpMRI) is an efficient non-invasive tool for the diagnosis, staging, and monitoring of PCa [2]. The prostate imaging reporting and data system (PI-RADS) is a 5-point scale used to predict the possibility of clinically significant prostate cancer (csPCa) based on the findings of mpMRI, which includes T2-weighted imaging (T2WI), diffusion weighted imaging (DWI), and dynamic contrast-enhanced (DCE) [3]. However, its diagnosis is based on the subjective and semi-quantitative results of mpMRI. The latest PI-RADS v2.1which update of PI-RADS v2.0 in 2019, still does not incorporate clinical data and quantitative parameters, and shows no significant value in DEC imaging. Moreover, the current system does not cover suggestions for PI-RADS category 3 lesions and MRI follow-up [3, 4]. Studies have found no significant difference in the diagnostic efficiency for csPCa between biparametric magnetic resonance imaging (bpMRI) and mpMRI [5, 6]. Use of the combination of PI-RADS scores, patient’s age, prostate specific antigen (PSA) level, and prostate specific antigen density (PSAD) has been shown to increase the detection rate of csPCa, thus providing a more evaluable reference for clinical decision-making [7, 8]. In the present study, the subjects were assigned into csPCa group and non-csPCa group based on the pathological findings. Regarding the limitations of PI-RADSv2.1, we assessed the csPCa-predicting potential of the biparametric and multiparametric models, involving the semi-quantitative and quantitative parameters of mpMRI and bpMRI (e.g., PI-RADS scores and T2 weighted image [T2WI] score according to the latest PI-RADS v2.1 scoring standard, apparent diffusion coefficient [ADC], volume transfer constant between blood plasma and the extracellular extravascular space [Ktrans], rate constant between the extracellular extravascular space and the blood plasma [Kep]) and clinical parameters (PSAD).
Material and methods
Patients
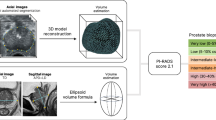
Since 2019, the imaging and clinical data were retrospectively collected from 634 patients who underwent prostate mpMRI at our hospital due to the increase in PSA level, and were confirmed by prostate biopsy or RP(198, 31%) between June 2015 and December 2020. The mpMRI was performed before or four weeks after biopsy to minimize the effect of artefacts induced by blood pooling within the gland. A total of 561 patients (age range 28–92 years; median 67 years) were included after excluding those who had history of treatment, incomplete data, such as PSA without specific value(> 100 ng/ml), or overlapping features with other tumors. Of them, 285 (50.8%) were assigned to the non-csPCa group and 276 (49.2%) to the csPCa group. The study flowchart is presented in Fig. 1.
This study was a retrospective cohort study approved by the institutional review board and complied with HIPAA. The requirement for informed consent was waived off by the human investigation committee at our institution.
MRI Protocol
MRI was performed with a 3.0-T scanner (Siemens MAGNETOM Verio or Prisma, German) using a 16-channel phased-array body coil. The protocol included axial and sagittal T2WI, axial DWI and DCE in accordance with PI-RADS v2.1. b-values used for DWI included 0, 1000, and 2000 mm/s2. ADC map was automatically calculated from b-values of 0 and 1000 mm/s2. The MRI scanning parameters are presented in Tables 1 and 2.
Image analysis
Prostate volume was measured according to the PI-RADS v2.1 [3] standard for the calculation of PSAD. Prostate index lesions of each patient were scored on a five-point scale on T2WI images (T2WI score) and mpMRI images (PI-RADS score) by a senior radiologist blinded to the pathological results and having 12 years of MRI experience (having read more than eighty thousand patients’ MRI images, these included about 1,500 prostate MRI), according to the PI-RADS v2.1 scoring standard.
All images were sent to a workstation. Specific software was used to process DW and DCE images (4D-Tissu). A senior radiologist analyzed all the mpMRI images that qualified the inclusion criteria to identify index prostate cancer foci. The mean ADC, Kep, and Ktrans values were evaluated on a selected region of interest encompassing as much of the inner part of the lesion as possible without contacting the edges (Figs. 2 and 3).
Parameter diagram of DCE measurements in prostate cancer. A Early dynamic contrast enhanced image shows avid enhancement within the anterior lesion (arrows). B The signal intensity-time curve of ROI3 shows a plateau of rapid rise and slow fall, while ROI1.2.4 shows an inflow curve of slow rise. E The Ktrans and Kep values of the index lesion are 1.110/min and 1.279/min. C, D, F shows the Ktrans and Kep values in the normal area of bilateral transition zone and right peripheral zone
Reference standard
Each patient underwent both systematic biopsy (with an average of 12 random samples from the entire prostate gland) and target biopsy (with at least three samples obtained from each lesion identified by MRI). Target sampling was performed with an MRI/TRUS fusion, alternately using the cognitive technique or dedicated software, coupled with various commercially-available ultrasound tools. For patients undergoing radical prostatectomy (RP) after puncture, pathological results obtained after RP were used as the gold standard for diagnosis. Post-RP specimens were sectioned at 4–6 mm intervals, and stained with hematoxylin and eosin (H&E). A pathologist recorded the presence or absence of PCa, tumor location, and determined the tumor Gleason score (GS) of biopsy specimens. GS was calculated according to the 2014 International Society of Urological Pathology Modified Gleason Grading System [9]. The definition of csPCa was a tumor with GS ≥ 7, or GS = 3 + 3 plus tumor size ≥ 0.5 mL [10]. Tumor size was calculated from mpMR images, most commonly T2WI. When multiple foci of PCa were found, the focus with the highest GS was considered as the index lesion.
Statistical analysis
According to the pathological results after biopsy, patients were divided into two groups, i.e., csPCa and non-csPCa. Between-group differences with respect to each parameter (PSA density, PI-RADS score, T2WI score, ADC, Ktrans, and Kep) were assessed using the Independent-samples U test. The predictors of csPCa were screened by logistics regression analysis. In case of multicollinearity, logistic regression analysis was performed using the likelihood ratio forward method to screen variables in the model. mpMRI and bpMRI predictive models were established and receiver operating characteristic (ROC) curves were plotted to evaluate the efficiency of each parameter and the model in diagnosing csPCa. The diagnostic performance was compared using the DeLong test. Two-tailed P values < 0.05 were considered indicative of statistical significance.
Results
All the 561 cases were confirmed by biopsies and eligible for this study. The non-csPCa group comprised of 285 patients, including 168 with benign prostatic hyperplasia (BPH), 49 with prostatitis, 20 with prostate intraepithelial neoplasia (PIN), and 48 with clinically insignificant Pca (ciPCa). The CsPCa group comprised of 276 patients, including 69 with International Society of Urological Pathology (ISUP) grade 2, 77 with ISUP grade 3, 60 with ISUP grade 4, and 70 with ISUP grade 5 prostate cancer (Fig. 4).
Representative case of prostate cancer An 80-year-old man with PSA 73.2 ng/mL, PSAD 0.77 ng/mL2, and Gleason score 3 + 4 prostate cancer confirmed after RP. A, B Axial T2WI and FS-T2WI sequences show a T2 hypointense nodule (arrows) involving the left peripheral zone with extraprostatic extension; T2WI score = 5. C Diffusion-weighted image (b = 2000) shows a markedly hyperintense signal (arrows) corresponding to (A) and (B). D ADC map image shows focal hypointense signal corresponding to (C), ADC value of the lesion is 0.829 × 10-3mm2/s; DWI PI-RADS = 5. E Early dynamic contrast enhanced image shows avid enhancement within the anterior lesion (arrows), DCE MRI PI-RADS = positive. PI-RADS score = 5. F, G The Ktrans and Kep values of the anterior lesion are 0.345/min and 1.249/min, respectively. H Gross morphology of the RP specimen. (I) Microscopic pathological view of the index lesion
Normality test of semi-quantitative and quantitative parameters
The one-sample Kolmogorov–Smirnov test showed non-normal distribution of PI-RADS score, T2WI score, ADC, Ktrans, Kep, PSAD, and patient’s age (P < 0.001 for all).
Mann–Whitney U test of semi-quantitative and quantitative parameters
The csPCa group had significantly higher PSAD, PI-RADS score, T2WI score, Ktrans and Kep, but significantly lower ADC compared to the non-csPCa group (P < 0.05 for all). No significant difference in age was detected between csPCa and non-csPCa group (P = 0.099, Table 3).
Univariable logistic regression analysis of semi-quantitative and quantitative parameters and their respective diagnostic efficiency
Univariable logistic regression analysis showed significant differences between csPCa and non-csPCa groups with respect to PI-RADS score, T2WI score, ADC, and PSAD (P < 0.05 for all), but not with respect to Ktrans, Kep and ADC reciprocal (ADCrec). Furthermore, receiver operating characteristic (ROC) curve analysis revealed that PI-RADS score showed the highest diagnostic efficiency for csPCa, followed by T2WI score, ADCrec, PSAD, Kep, and Ktrans, in that order (P = 0.000 for all, Fig. 5, Table 4).
Kendall’s tau-b correlation coefficient showed a significant correlation between PI-RADS score and T2WI score (t = 0.769, P < 0.001). To avoid multicollinearity among variables, PI-RADS score and T2WI score were separately introduced into the biparametric and multiparametric models.
Multivariable logistic regression analysis of Semi-quantitative and quantitative parameters
The biparametric model involving ADC measured by bpMRI plus T2WI scores and PSAD, and the multiparametric model involving ADC measured by mpMRI plus PI-RADS score and PSAD were created by binary logistic regression.
The first biparametric model involving ADC measured by bpMRI plus T2WI score and PSAD is shown below:
\(Logit \left(P\right)= -1.925+0.494\times PSAD+1.006 \times T2WI\,scores-2.434 \times ADC\ ;\) in which, the independent variables, including T2WI score (OR = 2.734; 95% CI, 2.199–3.398), ADC (OR = 0.088; 95% CI, 0.029–0.266), and PSAD (OR = 1.639, 95% CI, 1.223–2.197) were all statistically significant (P < 0.05 for all).
The model could predict 83.8% of csPCa cases, with a positive predictive value of 83.4% and a negative predictive value of 84.2% (χ2 = 363.055, P < 0.001).
The second multiparametric model involving ADC measured by mpMRI plus PI-RADS score and PSAD is shown below:
\(Logit \left(P\right)= -2.212+0.441\times PSAD+1.120 \times PI-RADS\, scores-2.350 \times ADC\ ;\) in which, the independent variables, including PI-RADS scores (OR = 3.064; 95% CI, 2.428–3.866), ADC (OR = 0.095; 95% CI, 0.032–0.288), and PSAD (OR = 1.554, 95% CI, 1.170–2.064) were all statistically significant (P < 0.05 for all).
The model predicted 85.2% of csPCa cases with a positive predictive value of 86.4% and a negative predictive value of 84.1% (χ2 = 376.368, P < 0.001).
Diagnostic efficiency of the biparametric and multiparametric models in diagnosing csPCa compared with that of PI-RADS
The Areas under curve (AUC) of the multiparametric model was significantly higher than those of the biparametric model and PI-RADS (Delong test P < 0.05, Fig. 6). The multiparametric model showed the highest Youden index, followed by the biparametric model (Table 5).
ROC curve of each model and PI-RADS score in the diagnosis of csPCa PIS, the 5-point results of PI-RADS v2.1 assessed by senior physicians; PRE1, the biparametric model involving ADC measured by bpMRI plus T2WI scores and PSAD; PRE2, the multiparametric model involving ADC measured by mpMRI plus PI-RADS scores and PSAD; ROC, receiver operating characteristic; PI-RADS, prostate imaging reporting and data system
Thirty cases were selected to verify the accuracy of the prediction model. The sigmoid function was used for the conversion. The average P values (the probability of csPCa occurrence) were 69.87% and 71.36% in the csPCa group, and 16.22% and 15.31% in the non-csPCa group. The top 10 representative ROI verification results are presented in Table 6.
Discussion
In the present study, the PI-RADS v2.1 based on the mpMRI findings showed relatively high accuracy (89.5%), sensitivity (78.9%), and specificity (86.6%) for diagnosis of csPCa, which is consistent with those findings in previous studies [11, 12]. According to a meta-analysis [12], the diagnostic sensitivity and specificity of PI-RADS v2.1 for csPCa were 87% and 74%, respectively. However, a study by Westphalen et al. [13] suggested relatively low positive predictive value of PI-RADS v2.1 for prostate MRI (49%; 95% CI, 40–58%) due to the strong subjectivity in the process of scoring. The independent diagnostic efficiency of quantitative parameters, like ADC (84.6%) and PSAD (80.2%), are relatively low, but these parameters are more objective and measurable, due to the well-recognized standards and measurement repeatability. Pepe et al. [14] found that ADC evaluation could support clinicians in decision making of patients with PI-RADS score 3 at risk for csPCa, for increase the ROC from 0.71 to 0.81. Marco [15] found that PSAD can help detected mpMRI false negative csPCa. And several studies have shown that PI-RADS combined with ADC or PSAD significantly enhances the diagnostic accuracy and positive predictive value of csPCa, thus avoiding unnecessary biopsy [16,17,18]. DCE MRI is an established mpMRI sequence for assessing prostate cancer, which highlights hemodynamic changes in cancer lesions and measures quantitative parameters that reflect microvascular perfusion (e.g., Kep, Ktrans) [19]. As a single predictor, the odds ratios (OR) of Kep (0.987, P > 0.05) and Ktrans (0.794, P > 0.05) in the present study were closer to 1, suggesting that they presented no contribution to the predictive model of csPCa, which is consistent with previous findings [20, 21]. We created predictive models based on MR imaging data, quantitative parameters, and clinical indicators, which not only significantly enhance the diagnostic accuracy for csPCa, but also objectively identify the cancer lesion. The results were similar with those from Liying Han [22]. Its area under curve value of the combined model (0.911) was also higher than those of ADC, PSAD, and PI-RADS v2.0 (0.887, 0.861, and 0.859, respectively).
In recent years, a large number of scholars have proposed that prostate MRI without DCE (bpMRI) may replace PI-RADSv2.1 based on mpMRI as a non-invasive monitoring means for csPCa [23,24,25]. Comparing the results of a meta-analyses [24], the diagnostic sensitivity (87%, 95%CI: 78%-93%) and specificity (72%, 95%CI: 56%-84%) of mpMRI for csPCa were not significantly different from bpMRI (sensitivity: 84%, 95%CI: 80% to 88%, specificity 75%, 95%CI: 68% to 81%). However, in our study, the diagnostic efficiency and positive predictive value of multiparametric model were significantly higher than those of the biparametric model. That’s consistent with PI-RADS v2.1, when bpMRI is performed and DCE data are not obtained, transition zone (TZ) assessment remains unchanged, and the proportion of men with PI-RADS assessment category 3 may increase [3]. Similarly, Greer et al. [26] found that DCE-MRI was conducive to enhance the diagnostic efficiency for csPCa, and the abnormal findings on DCE-MRI significantly increased the detection rate of PI-RADS v2.0 in categories 2–5 (A total of 163 patients with 654 lesions were evaluated). Therefore, mpMRI can be recommended to avoid misdiagnosis of csPCa, particularly suitable for prostate cancer risk groups.
Although the diagnostic efficiency of multiparametric model for csPCa was superior to that of the biparametric model, the complex procedure may challenge junior physicians or physicians in low-level hospitals with fewer cases. In the present study, the negative predictive value of the biparametric model was comparable to that of the multiparametric model (84.2% vs. 84.1%). Moreover, considering the risks associated with intravenous injection of contrast agents, and low economic and time cost of dynamic contrast-enhanced MRI, we think biparametric model might be more appropriate for early screening of csPCa.
This was a retrospective cohort study with an expanded sample size. Biopsy was not performed prior to mpMRI. Most of the involved subjects were pathologically diagnosed as prostate diseases by MRI/ultrasound fusion-guided biopsy or pathological examination after radical prostatectomy. This helped improve the reliability of our findings. In addition, this is first study to compare the characteristics and clinical value of the biparametric and multiparametric models involving MRI imaging data, quantitative parameters, and clinical indicators with those of PI-RADS v2.1. We identified the critical role of DCE-MRI in diagnosing csPCa, which can make up for the limitations of PI-RADS v2.1. Collectively, the biparametric and multiparametric models were found to be useful tools for selecting the optimal MRI and for planning the therapeutic strategy. However, its accuracy still requires to be verified by a larger sample from multiple centers. The latest research [27, 28] showed that MRI scoring with the Prostate Imaging for Recurrence Reporting assessment based on mpMRI could provide structured, reproducible, and accurate evaluation of local recurrence after definitive therapy for prostate cancer. Meanwhile, Pepe’s study shows PSMA PET/CT demonstrated good accuracy in the diagnosis of csPCa, which was not inferior to mpMRI (77.5% vs. 73.7%) [29]. These will be the focus of further research.
Some limitations of our study should be acknowledged. First of all, this study focused on the diagnosis of csPCa without taking into consideration the stage of cancer lesions. We chose the index lesion of csPCa because it contains lethal progenitor cells that determine the progression and metastasis of prostate cancer [30, 31]. Second, there were no independent studies on the diagnostic efficacy of transitional zone and peripheral zone lesions in this study. Third, this was a single-center retrospective cohort study. Our results should be further validated in a larger, multi-center prospective study.
Conclusions
PI-RADS score was the most accurate independent diagnostic index. The predictive value of bpMRI model for csPCa was slightly lower than that of mpMRI model, but higher than that of PI-RADS score. Created bpMRI and mpMRI models for diagnosing csPCa, can overcome the limitations of PI-RADS v2.1 and facilitate treatment decision-making. BpMRI might be more appropriate for early screening of csPCa, and mpMRI for avoiding missed diagnosis.
Availability of data and materials
The data and materials used during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- PCa:
-
Prostate cancer
- mpMRI:
-
Multiparametric magnetic resonance imaging
- PI-RADS:
-
Prostate imaging reporting and data system
- csPCa:
-
Clinically significant prostate cancer
- DEC:
-
Dynamic contrast-enhanced
- bpMRI:
-
Biparametric magnetic resonance imaging
- PSAD:
-
Prostate specific antigen density
- TRUS:
-
Transrectal Ultrasound
- MRF-TB:
-
MRI-US fusion targeted biopsy
- RP:
-
Radical prostatectomy
- T2WI:
-
T2 weighted image
- ADC:
-
Apparent diffusion coefficient
- Ktrans :
-
Volume transfer constant between blood plasma and the extracellular extravascular space
- Kep :
-
Rate constant between the extracellular extravascular space and the blood plasma
- GS:
-
Gleason score
- ROC:
-
Receiver operating characteristic
- ciPCa:
-
Clinically insignificant Pca
- ISUP:
-
International Society of Urological Pathology
- AUC:
-
Areas under curve
- OR:
-
Odds Ratios
References
Paladini A, Cochetti G, Colau A, et al. The challenges of patient selection for prostate cancer focal therapy: a retrospective observational multicentre study. Curr Oncol. 2022;29:6826–33. https://doi.org/10.3390/curroncol29100538.
Stavrinides V, Giganti F, Trock B, et al. Five-year outcomes of magnetic resonance imaging-based active surveillance for prostate cancer: a large cohort study. Eur Urol. 2020;78:443–51. https://doi.org/10.1016/j.eururo.2020.03.035.
Turkbey B, Rosenkrantz AB, Haider MA, et al. Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur Urol. 2019;76(2019):340–51. https://doi.org/10.1016/j.eururo.2019.02.033.
Ullric T, Schimmöller L. Perspective: a critical assessment of PI-RADS 2.1. Abdom Radiol (NY). 2020;45:3961–8. https://doi.org/10.1007/s00261-020-02424-7.
Woo S, Suh CH, Kim SY, et al. Head-to-Head Comparison Between Biparametric and Multiparametric MRI for the Diagnosis of Prostate Cancer: A Systematic Review and Meta-Analysis. AJR Am J Roentgenol. 2018;211:W226–w241. https://doi.org/10.2214/ajr.18.19880.
Gan JM, Kikano EG, Smith DA, et al. Clinically Significant prostate cancer detection after a negative prebiopsy MRI examination: comparison of biparametric versus multiparametric MRI. AJR Am J Roentgenol. 2022;218:859–66. https://doi.org/10.2214/ajr.21.26569.
Iwamoto H, Izumi K, Kadomoto S, et al. A novel screening strategy for clinically significant prostate cancer in elderly men over 75 years of age. Asian J Androl. 2021;23:36–40. https://doi.org/10.4103/aja.aja_39_20.
Bhat NR, Vetter JM, Andriole GL, et al. Magnetic resonance imaging-defined prostate-specific antigen density significantly improves the risk prediction for clinically significant prostate cancer on biopsy. Urology. 2019;126:152–7. https://doi.org/10.1016/j.urology.2018.12.010.
Epstein JI, Amin MB, Reuter VE, et al. Contemporary Gleason Grading of Prostatic Carcinoma: An Update With Discussion on Practical Issues to Implement the 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma. Am J Surg Pathol. 2017;41:e1–7. https://doi.org/10.1097/pas.0000000000000820.
Weinreb JC, Barentsz JO, Choyke PL, Prostate Imaging PI-RADS, Reporting -, System D, et al. Version 2. Eur Urol. 2015;69(2016):16–40. https://doi.org/10.1016/j.eururo.2015.08.052.
Gaudiano C, Rustici A, Corcioni B, et al. PI-RADS version 2.1 for the evaluation of transition zone lesions: a practical guide for radiologists. Br J Radiol. 2022;95(1131):20210916. https://doi.org/10.1259/bjr.20210916.
Park KJ, Choi SH, Kim MH, et al. Performance of Prostate Imaging Reporting and Data System Version 2.1 for Diagnosis of Prostate Cancer: A Systematic Review and Meta-Analysis. J Magn Reson Imaging. 2021;54:103–12. https://doi.org/10.1002/jmri.27546.
Westphalen AC, McCulloch CE, Anaokar JM, et al. Variability of the Positive Predictive Value of PI-RADS for Prostate MRI across 26 Centers: Experience of the Society of Abdominal Radiology Prostate Cancer Disease-focused Panel. Radiology. 2020;296:76–84. https://doi.org/10.1148/radiol.2020190646.
Pepe P, D’Urso D, Garufi A, et al. Multiparametric MRI Apparent Diffusion Coefficient (ADC) Accuracy in Diagnosing Clinically Significant Prostate Cancer. In Vivo. 2017;31:415–8. https://doi.org/10.21873/invivo.11075.
Roscigno M, Stabile A, Lughezzani G, et al. Multiparametric magnetic resonance imaging and clinical variables: Which is the best combination to predict reclassification in active surveillance patients? Prostate Int. 2020;8:167–72. https://doi.org/10.1016/j.prnil.2020.05.003.
Stevens E, Truong M, Bullen JA, et al. Clinical utility of PSAD combined with PI-RADS category for the detection of clinically significant prostate cancer. Urol Oncol. 2020;38(846):e849–846.e816. https://doi.org/10.1016/j.urolonc.2020.05.024.
Zhang KS, Schelb P, Kohl S, et al. Improvement of PI-RADS-dependent prostate cancer classification by quantitative image assessment using radiomics or mean ADC. Magn Reson Imaging. 2021;82:9–17. https://doi.org/10.1016/j.mri.2021.06.013.
Abreu-Gomez J, Walker D, Alotaibi T, et al. Effect of observation size and apparent diffusion coefficient (ADC) value in PI-RADS v2.1 assessment category 4 and 5 observations compared to adverse pathological outcomes. Eur Radiol. 2020;30:4251–61. https://doi.org/10.1007/s00330-020-06725-9.
Tan CH, Hobbs BP, Wei W, et al. Dynamic contrast-enhanced MRI for the detection of prostate cancer: meta-analysis. AJR Am J Roentgenol. 2015;204:W439–448. https://doi.org/10.2214/ajr.14.13373.
Mussi TC, Martins T, Garcia RG, et al. Are Dynamic Contrast-Enhanced Images Necessary for Prostate Cancer Detection on Multiparametric Magnetic Resonance Imaging? Clin Genitourin Cancer. 2017;15:e447–54. https://doi.org/10.1016/j.clgc.2016.10.001.
Scialpi M, Scialpi P, Martorana E. Prostate Cancer Index Lesion Detection and Volume Estimation: Is Dynamic Contrast-Enhanced MRI Really Reliable? AJR Am J Roentgenol. 2019;213:W289. https://doi.org/10.2214/ajr.19.21764.
Han L, He G, Mei Y, et al. Combining Magnetic Resonance Diffusion-Weighted Imaging with Prostate-Specific Antigen to Differentiate Between Malignant and Benign Prostate Lesions. Med Sci Monit. 2022;28:e935307. https://doi.org/10.12659/MSM.935307.
Becerra MF, Alameddine M, Zucker I, et al. Performance of Multiparametric MRI of the Prostate in Biopsy Naïve Men: A Meta-analysis of Prospective Studies. Urology. 2020;146:189–95. https://doi.org/10.1016/j.urology.2020.06.102.
Bass EJ, Pantovic A, Connor M, et al. A systematic review and meta-analysis of the diagnostic accuracy of biparametric prostate MRI for prostate cancer in men at risk. Prostate Cancer Prostatic Dis. 2021;24:596–611. https://doi.org/10.1038/s41391-020-00298-w.
Schoots IG, Barentsz JO, Bittencourt LK, et al. PI-RADS Committee Position on MRI Without Contrast Medium in Biopsy-Naive Men With Suspected Prostate Cancer: Narrative Review. AJR Am J Roentgenol. 2021;216:3–19. https://doi.org/10.2214/AJR.20.24268.
Greer MD, Shih JH, Lay N, et al. Validation of the Dominant Sequence Paradigm and Role of Dynamic Contrast-enhanced Imaging in PI-RADS Version 2. Radiology. 2017;285:859–69. https://doi.org/10.1148/radiol.2017161316.
Pecoraro M, Turkbey B, Purysko AS, et al. Diagnostic Accuracy and Observer Agreement of the MRI Prostate Imaging for Recurrence Reporting Assessment Score. Radiology. 2022;304:342–50. https://doi.org/10.1148/radiol.212252.
Ciccarese F, Corcioni B, Bianchi L, et al. Clinical Application of the New Prostate Imaging for Recurrence Reporting (PI-RR) Score Proposed to Evaluate the Local Recurrence of Prostate Cancer after Radical Prostatectomy. Cancers (Basel). 2022;14:4725. https://doi.org/10.3390/cancers14194725.
Pepe P, Pepe L, Cosentino S, et al. Detection Rate of 68Ga-PSMA PET/CT vs. mpMRI Targeted Biopsy for Clinically Significant Prostate Cancer. Anticancer Res. 2022;42:3011–5. https://doi.org/10.21873/anticanres.15785.
Liu W, Laitinen S, Khan S, et al. Copy number analysis indicates monoclonal origin of lethal metastatic prostate cancer. Nat Med. 2009;15:559–65. https://doi.org/10.1038/nm.1944.
Ahmed HU. The index lesion and the origin of prostate cancer. N Engl J Med. 2009;361:1704–6. https://doi.org/10.1056/NEJMcibr0905562.
Acknowledgements
We’d like to thank for the technical supports provided by NHC Key Laboratory of Thalassemia Medicine (Guangxi Medical University).
Funding
This work was supported by the Guangxi Zhuang Autonomous Region Health Commission Self-financed Scientific Research Project (Z20201092); and Guangxi Medical University Undergraduate Innovation and Entrepreneurship Training Program (202110598157).
Author information
Authors and Affiliations
Contributions
Xiao Feng: Project development, Data Collection, Data analysis, Manuscript writing. Xin Chen: Project development, Data Collection, Data analysis. Peng Peng: Protocol development. He Zhou: Data Collection, Data analysis. Yi Hong: Data Collection, Data analysis. Chunxia Zhu: Data Collection, Data analysis. Libing Lu: Data Collection, Data analysis. Siyu Xie: Data Collection, Data analysis. Sijun Zhang: Data Collection, Data analysis. Liling Long: design and revision of the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study has been performed in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University (NO.2022-KY-E-(283).
The requirement for informed consent was waived off by the human investigation committee of the First Affiliated Hospital of Guangxi Medical University.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Feng, X., Chen, X., Peng, P. et al. Values of multiparametric and biparametric MRI in diagnosing clinically significant prostate cancer: a multivariate analysis. BMC Urol 24, 40 (2024). https://doi.org/10.1186/s12894-024-01411-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12894-024-01411-0