Abstract
Background
Clinical amyopathic dermatomyositis is characterized by cutaneous symptoms but lacks muscle symptoms. Anti-melanoma differentiation-associated gene 5 antibodies are frequently found in Japanese patients with clinical amyopathic dermatomyositis. Patients with rapidly progressive interstitial lung disease with positive anti-melanoma differentiation-associated gene 5 antibodies have poor prognoses, and majority of them are treated with combination immunosuppressive therapy; however, the best treatment is yet to be determined.
Case presentation
A 52-year-old Asian male patient presented with a chief complaint of dyspnea on exertion. He had a typical skin rash and rapidly progressive interstitial pneumonia. Additionally, anti-melanoma differentiation-associated gene 5 antibodies were detected; therefore, he was diagnosed with dermatomyositis-associated interstitial pneumonia. Respiratory failure worsened despite administering steroid pulse therapy, tacrolimus, and cyclophosphamide. Consequently, plasma exchange was performed on day 13 of admission. After a slight improvement, the patient’s respiratory failure worsened. Thus, cyclophosphamide was replaced by tofacitinib on day 28. Although respiratory failure improved and the progression of interstitial pneumonia seemed under control, βD-glucan level increased and Aspergillus antigen was detected on day 49. Micafungin and voriconazole were administered, but the patient succumbed to worsening respiratory failure on day 61. The pathological autopsy revealed multiple nodular lesions with cavity formation in both lungs and the presence of Aspergillus with severe neutrophilic infiltration and necrosis, which supported the diagnosis of invasive pulmonary aspergillosis.
Conclusion
The patient with anti-melanoma differentiation-associated gene 5 antibody-related rapidly progressive interstitial lung disease, whose disease was difficult to control after the administration of triple immunosuppressive therapy (steroids, tacrolimus, and cyclophosphamide), showed good response with tofacitinib. Unfortunately, the patient died of invasive pulmonary aspergillosis owing to severe immunosuppression; thus, the signs of complications should be promptly detected.
Similar content being viewed by others
Background
Clinical amyopathic dermatomyositis (CADM) is characterized by cutaneous symptoms but lacked muscle symptoms. Anti-melanoma differentiation-associated gene 5 (MDA5) antibodies are frequently found in Japanese patients with CADM. Patients with rapidly progressive interstitial lung disease (RP-ILD) with positive anti-MDA5 antibodies have poor prognoses, and the majority of them are treated with combination immunosuppressive therapy, but the best treatment has yet to be determined [1, 2]. Tofacitinib has been recently reported to be effective in anti-MDA5 antibody-positive DM but with an immunosuppression side effect [3, 4]. Herein, we report a case of death owing to invasive pulmonary aspergillosis, although tofacitinib controlled the RP-ILD.
Case presentation
A 52-year-old Asian male patient presented to his local doctor with dyspnea on exertion lasting 1 month and was diagnosed with interstitial pneumonia. He was a non-smoker with a history of hypertension, dyslipidemia, and allergic rhinitis. He had Gottron’s sign on the dorsal surfaces of hands and elbows, reverse Gottron’s sign on both palms, and Shawl sign on the upper back. The patient had mild grasping pain in both thighs, but manual muscle strength testing was normal. Subpleural consolidation shadows in both lungs tended to enlarge during 2 weeks and DM-related rapidly progressive interstitial pneumonia was suspected because of the skin findings. Steroid pulse therapy, tacrolimus at 5 mg, and cyclophosphamide at 800 mg were administered. The patient was transferred to our hospital on day 8 of admission because he was positive for anti-MDA5 antibody and was considered to have refractory disease.
Laboratory findings (Table 1) included creatine kinase within the normal range, markedly elevated ferritin at 2465 ng/mL, and positive anti-MDA5 antibody (enzyme-linked immunosorbent assay, antibody titer 5600). Additionally, lactate dehydrogenase was elevated at 416 U/L and C-reactive protein (CRP) was elevated at 2.88 mg/dL.
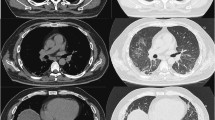
Imaging findings included (Fig. 1) infiltrative shadows predominantly on the peripheral sides of both lungs in chest radiography and consolidation shadows and ground glass opacities around bronchial vascular bundles and just subpleural in all lung lobes, with indentation changes in computed tomography (CT). No obvious honeycombing was observed.
a, b Computed tomography revealed consolidation shadows and ground glass opacities around bronchial vascular bundles and just below the pleura in all lung lobes, with indentation changes, on day 14 before admission. There was no obvious honeycombing. c, d Ground glass opacities are enhanced and the consolidation shadows are worsening on day 1 of admission. e, f Consolidation shadows and ground glass opacities were improving on day 23 of admission
Clinical course after transfer to our hospital: the patient had typical skin, arthralgia, and inflammatory findings, but no muscle weakness. A skin biopsy confirmed pathological findings consistent with DM and a muscle asymptomatic DM (CADM) diagnosis with positive anti-MDA5 antibodies. The patient’s resting oxygen saturation was 96% on room air and oxygen therapy at 3 L/minute was used during exertion upon transfer to our hospital on day 8 of admission.
Plasma exchange was started on day 13 when the patient required 5 L/min of oxygen therapy during exertion. Respiratory failure briefly improved and anti-MDA5 antibody and ferritin levels slightly decreased after three plasma exchange cycles (Fig. 2). Respiratory failure worsened again, requiring 10 L/min of oxygen therapy during exertion, thus tofacitinib was started instead of cyclophosphamide on day 28. The patient’s oxygenation during exertion improved to 3 L/min, the same level as at the start of treatment, and anti-MDA5 antibodies and ferritin remained unchanged after starting tofacitinib, thus the disease was thought to be under control.
However, CRP was elevated on day 49. In addition, βD-glucan was 220.3 pg/mL and Aspergillus antigen was positive on day 51. Chest computed tomography (CT) scan revealed multiple nodules with a halo sign and air-crescent sign, and the patient was diagnosed with invasive pulmonary aspergillosis (Fig. 3). No Aspergillus was detected in the blood culture, but Aspergillus fumigatus was found in the sputum. Tacrolimus and tofacitinib were discontinued and micafungin was started; however, respiratory failure rapidly progressed. Voriconazole was also added; however, the patient died 10 days after the clinical diagnosis of invasive pulmonary aspergillosis.
Pathological autopsy findings are shown in Fig. 4. The autopsy was performed after obtaining informed consent from the family. The lung weighed 770 g and 540 g on the right and left, respectively, and multiple nodular lesions with a cavity were found in some areas. A high degree of neutrophilic infiltration and necrotic Aspergillus growth were observed histologically, which was consistent with invasive pulmonary aspergillosis. Nodular lesions were observed in the kidneys and thyroid gland, and Aspergillus organisms with surrounding necrotic tissue were observed from the same areas. DM was not observed in the iliopsoas muscle, suggesting the treatment effect. The lung was suggestive of the usual interstitial pneumonia pattern in the background, suggesting that Aspergillus infection caused diffuse alveolar damage, resulting in respiratory failure and death.
Discussion and conclusion
RP-ILD with positive anti-MDA5 antibodies has a poor prognosis, and the best therapy is yet to be established although most initial combination therapies include steroids, calcineurin inhibitors, cyclophosphamide, and rituximab [2, 5,6,7]. The 6-month mortality rates of approximately 50% have been reported despite aggressive immunosuppressive therapy [3, 4]. Additionally, plasma exchange and polymyxin B immobilized fiber column direct hemoperfusion (PMX-DHP) have been tried in patients’ refractory to therapy, but with no consensus [8, 9]. The best therapy is still to be established.
Kurasawa et al. listed a ferritin level of ≥ 1000 ng/mL before the treatment, lung field shadows in all lung lobes, and worsening shadows despite the treatment as poor prognostic factors in DM-associated interstitial pneumonia [4]. Our patient met all these factors, especially the very high ferritin level (2465 ng/mL), which increased to 6411 ng/mL on day 8. Steroid pulse therapy, tacrolimus, and cyclophosphamide were administered to the patient as a multidrug combination immunosuppressive therapy, but the treatment was not effective. Plasma exchange was only effective by temporarily removing antibodies or cytokines, and improvement in the underlying clinical condition is difficult to conclude. Of 15 patients with RP-ILD associated with anti-MDA5 antibody, 5 died of respiratory failure within 90 days, while the remaining 10 survived for > 5 years [10].
This suggests that patients’ unresponsive to initial therapy should be given different or intensified treatment. Tofacitinib is a JAK1/3 inhibitor that is effective in treating rheumatoid arthritis or ulcerative colitis. The JAK/STAT signaling pathway is activated by interferons causing interferon-stimulated gene transcription, including MDA5. Tofacitinib reduces MDA5 expression and activation by inhibiting this pathway [11]. A successful response case to tofacitinib in anti-MDA5 antibody-associated RP-ILD was first reported in Japanese patients with refractory disease [4]. Chen et al. reported a significantly higher survival rate in patients treated with tofacitinib plus glucocorticoids compared to conventional therapy with glucocorticoids, tacrolimus, and cyclophosphamide [3]. Several similar cases have been reported in recent years, and we decided to treat this patient with tofacitinib because he was considered refractory to the therapy. Since tofacitinib is approved only for refractory rheumatoid arthritis and moderate-to-severe ulcerative colitis in Japan, we obtained approval for its off-label use in the hospital and explained the risk and benefit to the patient and obtained his consent.
Unlike the prior worsening of respiratory failure, oxygenation improved upon the administration of tofacitinib. Anti-MDA5 antibody and ferritin levels remained unchanged after tofacitinib initiation, which could have suppressed the elevation in their levels. Thus, the disease was thought to be under control. Anti-MDA5 antibodies, ferritin, and interleukin-18 have been reported as useful indicators for treatment response in RP-ILD of DM [12]. The patient remained stable for approximately 3 weeks after treatment, but respiratory failure progressed owing to the development of invasive pulmonary aspergillosis, which ultimately did not save his life. The risk of thrombosis and infection was increased during the treatment of collagen disease with tofacitinib [13]. Additionally, patients with positive anti-MDA-5 antibodies being treated with the immunosuppressive combination therapy of steroid, tacrolimus, and cyclophosphamide had a higher rate of cytomegalovirus reactivation, and patients with anti-MDA-5 antibodies and interstitial pneumonia are at increased risk for pneumocystis pneumonia [14].
Other JAK inhibitors (baricitinib) were reported as risk factors for the development of pulmonary aspergillosis when used for coronavirus disease 2019 pneumonia although an association between DM-associated interstitial pneumonia or tofacitinib and pulmonary aspergillosis was not reported [15]. Determining the tofacitinib administration as the direct cause in this patient is impossible, but the development of invasive pulmonary aspergillosis was strongly suspected due to immunosuppression, including tofacitinib. The presence of Aspergillus organisms in the kidneys and thyroid gland at pathological autopsy supports the presence of a high degree of immunosuppression. Regularly monitoring βD-glucan and cytomegalovirus is important when such a strong immunosuppressive therapy is administered. No references were found that recommend the use of fungal prophylaxis in cases similar to this one, and early identification and treatment of these infections as early as possible are directly related to prognosis. Additionally, further case series and clinical trials are expected to clarify the efficacy and safety of tofacitinib in the treatment of CADM.
The patient with anti-MDA5 antibody-related RP-ILD whose disease was difficult to control after the administration of triple immunosuppressive therapy (steroids, tacrolimus, and cyclophosphamide) was under control with tofacitinib. The patient died of invasive pulmonary aspergillosis owing to severe immunosuppression, thus signs of complications should be promptly detected.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CADM:
-
Clinical amyopathic dermatomyositis
- CRP:
-
C-reactive protein
- CT:
-
Computed tomography
- MDA5:
-
Anti-melanoma differentiation-associated gene 5
- PMX-DHP:
-
Polymyxin B immobilized fiber column direct hemoperfusion
- RP-ILD:
-
Rapidly progressive interstitial lung disease
References
Yen TH, Tseng CW, Wang KL, Fu PK. Combination therapy with rituximab, tofacitinib and pirfenidone in a patient with rapid progressive interstitial lung disease (RP-ILD) due to MDA5 antibody-associated dermatomyositis: a case report. Medicina. 2021;57:1358.
Tsuji H, Nakashima R, Hosono Y, Imura Y, Yagita M, Yoshifuji H, et al. Multicenter prospective study of the efficacy and safety of combined immunosuppressive therapy with high-dose glucocorticoid, tacrolimus, and cyclophosphamide in interstitial lung diseases accompanied by anti-melanoma differentiation-associated gene 5-positive dermatomyositis. Arthritis Rheumatol. 2020;72:488–98.
Chen Z, Wang X, Ye S. Tofacitinib in amyopathic dermatomyositis-associated interstitial lung disease. N Engl J Med. 2019;381:291–3.
Kurasawa K, Arai S, Namiki Y, Tanaka A, Takamura Y, Owada T, et al. Tofacitinib for refractory interstitial lung diseases in anti-melanoma differentiation-associated 5 gene antibody-positive dermatomyositis. Rheumatol. 2018;57:2114–9.
Kawasumi H, Gono T, Kawaguchi Y, Yamanaka H. Recent treatment of interstitial lung disease with idiopathic inflammatory myopathies. Clin Med Insights Circ Respir Pulm Med. 2015;9(suppl 1):9–17.
Nakashima R, Hosono Y, Mimori T. Clinical significance and new detection system of autoantibodies in myositis with interstitial lung disease. Lupus. 2016;25:925–33.
Ogawa Y, Kishida D, Shimojima Y, Hayashi K, Sekijima Y. Effective administration of rituximab in anti-MDA5 antibody-positive dermatomyositis with rapidly progressive interstitial lung disease and refractory cutaneous involvement: a case report and literature review. Case Rep Rheumatol. 2017;2017:5386797.
Endo Y, Koga T, Suzuki T, Hara K, Ishida M, Fujita Y, et al. Successful treatment of plasma exchange for rapidly progressive interstitial lung disease with anti-MDA5 antibody-positive dermatomyositis: a case report. Medicine. 2018;97: e0436.
Ichiyasu H, Sakamoto Y, Yoshida C, Sakamoto K, Fujita R, Nakayama G, et al. Rapidly progressive interstitial lung disease due to anti-MDA-5 antibody-positive clinically amyopathic dermatomyositis complicated with cervical cancer: successful treatment with direct hemoperfusion using polymyxin B-immobilized fiber column therapy. Respir Med Case Rep. 2017;20:51–4.
Hozumi H, Fujisawa T, Nakashima R, Johkoh T, Sumikawa H, Murakami A, et al. Comprehensive assessment of myositis-specific autoantibodies in polymyositis/dermatomyositis-associated interstitial lung disease. Respir Med. 2016;121:91–9.
Nombel A, Fabien N, Coutant F. Dermatomyositis with anti-MDA5 antibodies: bioclinical features, pathogenesis and emerging therapies. Front Immunol. 2021;12: 773352.
Gono T, Sato S, Kawaguchi Y, Kuwana M, Hanaoka M, Katsumata Y, et al. Anti-MDA5 antibody, ferritin and IL-18 are useful for the evaluation of response to treatment in interstitial lung disease with anti-MDA5 antibody-positive dermatomyositis. Rheumatol (Oxf Engl). 2012;51:1563–70. https://doi.org/10.1093/rheumatology/kes102.
You H, Xu D, Zhao J, Li J, Wang Q, Tian X, et al. JAK inhibitors: prospects in connective tissue diseases. Clin Rev Allergy Immunol. 2020;59:334–51.
Mehta P, Machado PM, Gupta L. Understanding and managing anti-MDA 5 dermatomyositis, including potential COVID-19 mimicry. Rheumatol Int. 2021;41:1021–36.
Horiuchi H, Utada S, Shinomiya Y, Miyagawa T, Sogo A, Niida S, et al. Chronic pulmonary aspergillosis during convalescence from severe COVID-19 treated with oral itraconazole: a report of two cases. Cureus. 2022;14: e27281.
Acknowledgements
The authors would like to thank Enago (www.enago.jp) for the English language review.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
YI carried out study conceptualization, investigation, resources, data curation, writing—original draft, review and editing preparation, and visualization; TY carried out study conceptualization, investigation, resources, data curation, writing—original draft, review and editing preparation, visualization, and supervision; TH, TN, TT, TI, SK, YA, YM, AS, and HN carried out investigation, resources, and data curation; MH carried out investigation, resources, data curation, and visualization; HC carried out supervision and project administration.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The need for approval was waived by our ethics review board.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Imai, Y., Yorozuya, T., Hatakeyama, T. et al. Management of anti-melanoma differentiation-associated gene 5 antibody-induced refractory dermatomyositis complicated by interstitial pneumonia using tofacitinib and its outcomes: a case report. J Med Case Reports 18, 471 (2024). https://doi.org/10.1186/s13256-024-04793-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13256-024-04793-9