Abstract
Background
Foreign body granulomas are postoperative inflammatory reactions to surgical materials within the body. Traditionally, sutures have been the leading cause of foreign body granulomas in the abdomen, commonly referred to as “suture granuloma”. However, the use of polymer clips in modern surgeries has introduced new challenges, and reports of intra-abdominal foreign body granulomas caused by surgical clips are limited. Herein, we present a case of an intra-abdominal foreign body granuloma formed due to polymer clips.
Case presentation
A 45-year-old woman who underwent laparoscopic resection for sigmoid colon adenocarcinoma subsequently developed a suspected lymph node recurrence during follow-up. Imaging showed an enlarging mass adjacent to the inferior mesenteric artery with increased fluorodeoxyglucose uptake. Laparoscopic surgical resection revealed the formation of a foreign body granuloma in response to the polymer clips.
Conclusions
This case suggests that nonabsorbable polymer clips can induce granulomatous reactions postoperatively, and the appearance of lymph node recurrence may be foreign body granulomas.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Foreign body granulomas are inflammatory reactions that may occur postoperatively when surgical materials are left inside the body. Conventionally, silk sutures are the primary cause of intra-abdominal foreign body granulomas [1,2,3,4,5,6,7]. They can be incidentally discovered on computed tomography (CT) images in patients who have undergone cancer surgery, occasionally leading to a misdiagnosis of cancer recurrence [1,2,3,4,5,6]. Such misinterpretations can result in unnecessary invasive treatments, such as systemic chemotherapy [8] or high-risk extended operations, including pancreatoduodenectomy [9].
Currently, polymer clips are commonly used for vessel ligation, particularly in laparoscopic and robotic surgeries. Despite their increased use, reports of intra-abdominal foreign body granulomas caused by surgical clips are limited. Combined with a review of the previous literature, we present a case in which suspected lymph node recurrence after sigmoid colon adenocarcinoma resection turned out to be a foreign body granuloma formed due to polymer clips.
Case presentation
A 45-year-old woman with no significant medical history or allergies was admitted to our hospital with severe microcytic anemia. Subsequent colonoscopy revealed a type 2 tumor in the sigmoid–descending junction, which was pathologically diagnosed as a papillary to well-differentiated tubular adenocarcinoma. Combined with contrast-enhanced CT findings, a preoperative diagnosis of sigmoid colon cancer without metastasis (cT2N0M0, cStage I according to the Union for International Cancer Control TNM classification) was established, and laparoscopic left hemicolectomy with D2 lymph node dissection was performed. Surgical findings indicated no signs of metastatic disease, liver nodules, or peritoneal implants. First, the left colon was mobilized using a medial approach, scoring the mesentery between Gerota’s and Toldt’s fasciae. The inferior mesenteric artery (IMA) and superior rectal artery (SRA) were skeletonized by dissecting the lymphatic adipose tissue along them. The left colic artery (LCA) and the first sigmoid artery (S1) were then ligated with polymer Hem-O-Lok® clips (Teleflex), while the SRA was preserved (Fig. 1). The inferior mesenteric vein (IMV) was ligated in the same manner at the level of the LCA. The colon was fully mobilized for transection at the splenic flexure and sigmoid colon. After the resection, extracorporeal functional end-to-end anastomosis was performed. Later, the pathological sample was reported to be a carcinoma with an adenoma component (pT1N0M0, pStage I). Of the total 20 #241 and #242 lymph nodes examined, none exhibited malignancy. The patient was discharged on postoperative day 7, and her tumor marker test at 3 months postoperatively was insignificant, with a carcinoembryonic antigen (CEA) level of 4.2 ng/mL. Subsequent monitoring every 3 months, including CT and lower gastrointestinal endoscopy, revealed no evidence of recurrence, except for a slightly elevated CEA of 6.7 ng/mL, likely due to smoking resumption.
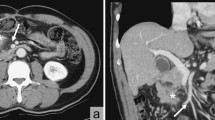
A follow-up CT, 2 years postoperatively, revealed a 10 mm mass adjacent to the IMA (Fig. 2a). Although the CEA was 6.1 ng/mL with no elevation, a further positron emission tomography (PET)/CT was recommended considering the possibility of lymph node recurrence. It showed increased fluorodeoxyglucose (FDG) uptake, with a maximum standardized uptake value (SUVmax) of 10.6 (Fig. 2b). No anastomotic recurrence was detected on colonoscopy; however, a repeat CT scan 3 months later showed that the mass had grown to 15 mm (Fig. 3a, b), suspicious of recurrence. Considering the initial cancer’s pStage I status and slight decrease in CEA to 5.1 ng/mL during this period, a differential diagnosis of benign lesions such as a desmoid tumor was considered. Core needle biopsy was deemed impossible because of the location of the mass, leading to laparoscopic surgical resection for a definitive diagnosis. The decision for surgical resection was made with anticipation of potential infiltration into the duodenum, necessitating extensive resection, possibly including duodenectomy. Intraoperatively, a solid mass was identified within the mesocolon on the lateral side of the duodenum (Fig. 4a). The mass adhered to the surrounding tissue, making it difficult to determine its border. An extensive dissection was performed on the anterior surface of the abdominal aorta and around the IMA to expose the LCA root. The two polymer clips used in the initial surgery for LCA and IMV ligation were attached to the mass (Fig. 4b). Subsequently, new clips were applied to the root of the LCA and further centrally to the IMV, and the mass was excised together with the old clips (Fig. 4c–e). The surgery lasted for 161 min with minimal blood loss. The final pathology report confirmed that a foreign body granuloma had formed in reaction to the polymer clips and was composed of granulation tissue with numerous macrophages, lymphocytes, plasma cells, and multinucleated giant cells (Fig. 5a–c). No evidence of malignancy was found, and a desmoid tumor was excluded with negative results of β-catenin. Neither mycobacteria nor fungi were identified with Ziehl–Neelsen or periodic acid–Schiff staining. The patient had a favorable postoperative course, with no evidence of recurrence at the latest follow-up after 2 months. The patient was informed of the necessity of using clips or sutures for vessel ligation, despite the potential risk of inducing granulomatous reactions.
Follow-up contrast-enhanced computed tomography (CT) and positron emission tomography (PET)/CT 2 years after the initial surgery. a Contrast-enhanced CT shows a 10 mm mass (arrow) adjacent to the inferior mesenteric artery. b PET/CT scan reveals a mass (arrow) at the same location with elevated fluorodeoxyglucose uptake, indicated by a maximum standardized uptake value of 10.6
Follow-up preoperative contrast-enhanced computed tomography (CT). Preoperative contrast-enhanced CT shows an enlarged mass (arrow) with embedded clips (arrowheads). a Axial view of the 15 mm mass, 5 mm larger than that seen in the initial follow-up CT, performed 3 months before. b Coronal view of the mass adjacent to the inferior mesenteric artery
Intraoperative findings. a Intraoperative identification of the mass (arrow) in the mesocolon lateral to the duodenum. b Visualization of the two polymer clips (arrowheads) attached to the mass (encircled with a dotted line), with the inferior mesenteric artery and root of the left colic artery (arrow) revealed. c Resection of the mass (arrow). d No residual lesion in the area, with the left colic artery (arrowhead) clipped at its origin from the inferior mesenteric artery, and preservation of the gonadal vessel (thin arrow) and ureter (thick arrow). e Illustration depicting the vessel anatomy, clips, and the mass. New clips (drawn in yellow) are applied to the left colic artery and inferior mesenteric vein to excise the mass along with the old clips (drawn in black)
Histopathological examinations of the resected specimen. a Hematoxylin and eosin staining revealing scattered multinucleated giant cells (arrowhead) (*200). b CD68 staining indicating numerous macrophages (*100). c CD138 staining showing abundant plasma cells (*100). The specimen exhibits granulation tissue characteristics
Discussion
We identified two noteworthy clinical issues: nonabsorbable polymer clips can induce a granulomatous reaction, and clinical differentiation from lymph node recurrence can be challenging, especially in patients with a history of cancer.
First, polymer clips can cause foreign body granulomas several months after surgery, as any foreign body within the body can trigger a granulomatous reaction [10]. An intravital experiment on mice skin proposed the following sequence of events: macrophages bind to the surface of the foreign body and differentiate into giant cells; once these giant cells fuse together, they produce vascular endothelial growth factor-A. This in turn recruits fibroblasts to encapsulate the foreign body in the collagen matrix [11]. Our review of case reports on PubMed using keywords “foreign body granuloma” and “colon cancer or colorectal cancer” revealed that silk sutures were primarily responsible for most cases of foreign body granuloma following colorectal cancer surgery [1,2,3,4,5, 7, 12,13,14]. Table 1 shows the recent English case reports of foreign body granuloma after colorectal cancer surgery with the present case indicated as No. 11 for comparison. Other reports of foreign body granuloma after general surgery mentioned those elicited by polypropylene sutures, surgical sponges, and metal clips [12, 15, 16]. One letter has described a case similar to ours, in which a polymer Hem-O-Lok® clip caused a foreign body granuloma [17]. This demonstrates the significance of categorizing such cases as “clip granulomas” for future recognition.
In the present case, in contrast to the clips applied on the LCA and IMV, the clip on the S1 did not cause noticeable granulation. The factors distinguishing these outcomes remain unknown; our hypothesis is that the clip on the LCA, being slightly apart from the bifurcation, potentially had more contact with the surrounding tissues and thus induced a more robust inflammatory response.
Second, a mass resembling a lymph node recurrence may be a granuloma formed in reaction to vessel clips. Both malignant tumors and inflammatory granulomas exhibit increased FDG accumulation. A meta-analysis of focal colorectal incidental FDG uptake revealed substantial overlap in the average standardized uptake values among malignant, premalignant, and benign lesions [18]. Our literature review on intra-abdominal foreign body granulomas suggested a median SUVmax value of 8.55 (5.04–14.6) (Table 1). Although the cutoff value for an intra-abdominal mass is unknown, a previous review recommended using SUVmax > 11.4 to distinguish adenocarcinoma from other etiologies of focal colonic uptake, with 80% sensitivity and 82% specificity [19]. This implied a potentially higher SUVmax for malignant lesions than for benign lesions. Additionally, certain features of PET/CT scans aid in differentiation. One such feature is ring-shaped FDG uptake in foreign body granulomas, which reflects the thick wall of the granuloma and the encapsulated foreign body [20]. Another indication is that cancer cells show increased uptake in the later phase, whereas benign tumors do not [21]. In our case, the clips were not completely encapsulated by the granuloma but rather attached to it, resulting in an atypical homogenous appearance. Furthermore, a dual-time PET/CT scan was not performed, making it impossible to distinguish benign granulomas from lymph node recurrence.
Another differential diagnosis of a lymph node recurrence mimic is desmoid tumor, which is a benign fibroblastic neoplasm that grows slowly. Up to 30% of the cases are associated with previous injuries or surgery [22], which can make it challenging to distinguish them from cancer recurrence, particularly in patients with a history of surgical resection. We have identified several English case reports of intra-abdominal desmoid tumors after colorectal cancer surgery, which were detected from 6 to 36 (median, 18) months post-surgery [23,24,25,26,27,28,29]. However, PET/CT was performed only in three cases, all showing the SUVmax values below 5 [23, 27, 28]. Upon the detection of an enlarging mass on postoperative cancer follow-up CT, it is imperative to raise concerns regarding recurrence. However, alternative possibilities must be discussed, especially in low-risk situations, such as pStage I colon cancer (4.4% recurrence rate after curative resection) [30]. In our case, the suspicion of a desmoid tumor was unique (Table 1) and was possibly attributable to the early postoperative period and low recurrence risk.
To establish a definitive diagnosis, excisional biopsy is recommended for deep lesions to minimize the risk of cancer dissemination during incisional biopsy. Indeed, almost all previous intra-abdominal cases were conclusively diagnosed via surgical resection (Table 1) [1,2,3,4,5,6,7,8,9, 12,13,14]. Only a few exceptions involving core needle biopsies were found in cases where a benign tumor was strongly suspected based on the typical imaging findings of a foreign body granuloma [16]. In cases where sacrificing surrounding organs is required for complete mass resection, considering intraoperative frozen section diagnosis from partial resection can help minimize the extent of surgery. In conclusion, if percutaneous biopsy is impractical due to the location of the mass and the possibility of cancer recurrence cannot be entirely ruled out, opting for excisional biopsy is the best possible choice. This proactive approach, as employed in our case, aims to remove the mass before it infiltrates surrounding organs, thereby minimizing the extent of necessary resection. This strategy not only facilitates a more accurate diagnosis, but also contributes to a more conservative treatment plan for the patient.
Conclusions
This case report highlights the importance of considering foreign body granulomas in patients who have undergone surgery with polymer clips. It also emphasizes that lymph node recurrence mimics might be foreign body granulomas. Although surgical resection and pathological examination are essential for a definitive diagnosis, this awareness can guide clinicians to achieve accurate assessments and deliver appropriate explanation and treatment to their patients. Further research is warranted to explore the predisposing factors that make patients more susceptible to foreign body granulomas and determine the specific surgical materials that provoke robust granulomatous reactions so as to plan strategic preparations for alternative devices.
Availability of data and materials
Data presented in this case report are available from the corresponding author upon reasonable request.
Abbreviations
- CT:
-
Computed tomography
- IMA:
-
Inferior mesenteric artery
- SRA:
-
Superior rectal artery
- LCA:
-
Left colic artery
- S1:
-
First sigmoid artery
- IMV:
-
Inferior mesenteric vein
- CEA:
-
Carcinoembryonic antigen
- PET:
-
Positron emission tomography
- FDG:
-
Fluorodeoxyglucose
- SUVmax:
-
Maximum standardized uptake value
References
Kim SW, Shin HC, Kim IY, Baek MJ, Cho HD. Foreign body granulomas simulating recurrent tumors in patients following colorectal surgery for carcinoma: a report of two cases. Korean J Radiol. 2009;10:313–8. https://doi.org/10.3348/kjr.2009.10.3.313.
Matsuura S, Sasaki K, Kawasaki H, Abe H, Nagai H, Yoshimi F. Silk suture granuloma with false-positive findings on PET/CT accompanied by peritoneal metastasis after colon cancer surgery. Int J Surg Case Rep. 2016;28:22–5. https://doi.org/10.1016/j.ijscr.2016.09.002.
Martínez-Martínez A, García-Espinosa J. Non-absorbable suture granuloma mimicking a retroperitoneal metastatic implant in a patient with sigmoid neoplasm. Rev Esp Enferm Dig. 2016;108:579–80.
Huang SF, Chiang CL, Lee MH. Suture granuloma mimicking local recurrence of colon cancer after open right hemicolectomy: a case report. Surg Case Rep. 2021;7:164. https://doi.org/10.1186/s40792-021-01251-2.
Takano Y, Haruki K, Tsukihara S, Abe T, Koyama M, Ito D, et al. Suture granuloma with hydronephrosis caused by ileostomy closure after rectal cancer surgery: a case report. Surg Case Rep. 2021;7:210. https://doi.org/10.1186/s40792-021-01303-7.
Uehara H, Hamada M, Hatta M, Sekimoto M, Noda Y, Minami K, et al. Two cases of 18F-FDG-PET/CT positive Schloffer tumor following curative surgery of colon cancer. Clin Case Rep. 2022;10: e6741. https://doi.org/10.1002/ccr3.6741.
Mitsuyoshi M, Nagata J, Akiyama Y, Yamauchi M, Sannomiya H, Kohi S, et al. Laparoscopic approach for port site mass diagnosed as a Schloffer tumor following surgery of ascending colon cancer. Asian J Endosc Surg. 2024;17: e13267. https://doi.org/10.1111/ases.13267.
Kakimoto Y, Matsuoka N, Ushimaru Y, Suzuki H, Endo Y, Kato Y. A case of a foreign body granuloma composed of a suture which was diagnosed as a false-positive tumor on PET/CT. Nihon Rinsho Geka Gakkai Zasshi (J Jpn Surg Assoc). 2013;74:459–62. https://doi.org/10.3919/jjsa.74.459.
Miyake K, Matsuda G, Oshi M, Kogure Y, Den K, Tsuchiya N, et al. Foreign body granuloma caused by surgical suture material mimicking lymph node recurrence detected by delayed phase FDG-PET/CT imaging. Jpn J Gastroenterol Surg. 2016;49:58–65. https://doi.org/10.5833/jjgs.2015.0014.
Pagán AJ, Ramakrishnan L. The formation and function of granulomas. Annu Rev Immunol. 2018;36:639–65. https://doi.org/10.1146/annurev-immunol-032712-100022.
Dondossola E, Holzapfel BM, Alexander S, Filippini S, Hutmacher DW, Friedl P. Examination of the foreign body response to biomaterials by nonlinear intravital microscopy. Nat Biomed Eng. 2016;1:0007. https://doi.org/10.1038/s41551-016-0007.
Nihon-Yanagi Y, Ishiwatari T, Otsuka Y, Okubo Y, Tochigi N, Wakayama M, et al. A case of postoperative hepatic granuloma presumptively caused by surgical staples/clipping materials. Diagn Pathol. 2015;10:90. https://doi.org/10.1186/s13000-015-0291-3.
Pantiora EV, Kontis EA, Michalaki V, Primetis E, Vezakis A, Polydorou A, et al. Granuloma mimicking local recurrence on PET/CT after liver resection of colorectal liver metastasis: a case report. Cureus. 2016;8: e717. https://doi.org/10.7759/cureus.717.
Asano E, Furuichi Y, Kumamoto K, Uemura J, Kishino T, Usuki H, et al. A case of Schloffer tumor with rapid growth and FDG-PET positivity at the port site of laparoscopic sigmoidectomy for colon cancer. Surg Case Rep. 2019;5:116. https://doi.org/10.1186/s40792-019-0677-7.
Yazyi FJ, Canullan CM, Baglietto NF, Klappenbach RF, Alonso Quintas F, Alvarez Rodriguez J, et al. Schloffer’s tumor: case report and review of the literature. Int J Surg Case Rep. 2014;5:1234–7. https://doi.org/10.1016/j.ijscr.2014.10.044.
Nakajo M, Jinnouchi S, Tateno R, Nakajo M. 18F-FDG PET/CT findings of a right subphrenic foreign-body granuloma. Ann Nucl Med. 2006;20:553–6. https://doi.org/10.1007/BF03026820.
Fan X, Zan R, Bian L, Chen Y. Hem-O-Lok clips may lead to misdiagnosis in postoperative follow-up of tumor patients: a case report. Asian J Surg. 2021;44:1123–4. https://doi.org/10.1016/j.asjsur.2021.05.041.
Treglia G, Taralli S, Salsano M, Muoio B, Sadeghi R, Giovanella L. Prevalence and malignancy risk of focal colorectal incidental uptake detected by (18)F-FDG-PET or PET/CT: a meta-analysis. Radiol Oncol. 2014;48:99–104. https://doi.org/10.2478/raon-2013-0035.
Pencharz D, Nathan M, Wagner TL. Evidence-based management of incidental focal uptake of fluorodeoxyglucose on PET-CT. Br J Radiol. 2018;91:20170774. https://doi.org/10.1259/bjr.20170774.
Miyake KK, Nakamoto Y, Mikami Y, Ishizu K, Saga T, Higashi T, et al. F-18 FDG PET of foreign body granuloma: pathologic correlation with imaging features in 3 cases. Clin Nucl Med. 2010;35:853–7. https://doi.org/10.1097/RLU.0b013e3181f483da.
Nakamoto Y, Higashi T, Sakahara H, Tamaki N, Kogire M, Doi R, et al. Delayed (18)F-fluoro-2-deoxy-d-glucose positron emission tomography scan for differentiation between malignant and benign lesions in the pancreas. Cancer. 2000;89:2547–54. https://doi.org/10.1002/1097-0142(20001215)89:12%3c2547::aid-cncr5%3e3.0.co;2-v.
Schlemmer M. Desmoid tumors and deep fibromatoses. Hematol Oncol Clin N Am. 2005;19:565–71. https://doi.org/10.1016/j.hoc.2005.03.008.
Nam KH, Kim B. Intra-abdominal desmoid fibromatosis mimicking tumour recurrence after the operation: a case series. Malays J Pathol. 2023;45:111–22.
Meyer A, Szajnbok P, Koszka AJM, Pezzutti D, Segatelli V, Monteiro J Jr. A rare sporadic pancreatic desmoid fibromatosis with splenic vein invasion diagnosed by CT scan-guided core needle biopsy: a case report with possible differential diagnosis from metastatic colorectal or renal cancer. J Surg Case Rep. 2021;2021: rjab257. https://doi.org/10.1093/jscr/rjab257.
Shayesteh S, Salimian KJ, Fouladi DF, Blanco A, Chu LC, Fishman EK. Pancreatic cystic desmoid tumor following metastatic colon cancer surgery: a case report. Radiol Case Rep. 2020;15:2063–6. https://doi.org/10.1016/j.radcr.2020.08.013.
Maemoto R, Miyakura Y, Tamaki S, Takahashi J, Takayama N, Fukuda R, et al. Intra-abdominal desmoid tumor after laparoscopic low anterior resection for rectal cancer: a case report. Asian J Endosc Surg. 2020;13:426–30. https://doi.org/10.1111/ases.12742.
Takada M, Okuyama T, Yoshioka R, Noie T, Takeshita E, Sameshima S, Oya M. A case with mesenteric desmoid tumor after laparoscopic resection of stage I sigmoid colon cancer. Surg Case Rep. 2019;5:38. https://doi.org/10.1186/s40792-019-0587-8.
Nakanishi K, Shida D, Tsukamoto S, Ochiai H, Mazaki J, Taniguchi H, et al. Multiple rapidly growing desmoid tumors that were difficult to distinguish from recurrence of rectal cancer. World J Surg Oncol. 2017;15:180. https://doi.org/10.1186/s12957-017-1248-7.
Mizuno R, Akiyoshi T, Kuroyanagi H, Fujimoto Y, Ueno M, Oya M, et al. Intra-abdominal desmoid tumor mimicking locoregional recurrence after colectomy in a patient with sporadic colon cancer: report of a case. Surg Today. 2011;41:730–2. https://doi.org/10.1007/s00595-010-4340-y.
Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, et al. Japanese society for cancer of the colon and rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 2020;25:1–42. https://doi.org/10.1007/s10147-019-01485-z.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
This study received no specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
SK conceived the case presentation, performed the literature review, and wrote the manuscript. YS actively engaged in surgery and assisted with manuscript revision. MH managed the treatment and contributed to the development of the case-related content. All the authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This case report adheres to the ethical principles, and written informed consent was obtained from the patient.
Consent for publications
Written informed consent was obtained from the patient for the publication of this case report and accompanying images.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kimura, S., Honda, M. & Sanmoto, Y. Polymer clip granuloma mimicking lymph node recurrence: a case report. surg case rep 10, 76 (2024). https://doi.org/10.1186/s40792-024-01881-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40792-024-01881-2