Abstract
Background
Mammography is accused of having low sensitivity and specificity in dense breast parenchyma. Also, women with dense breasts show an increased risk of developing breast cancer. Breast ultrasound has been used for several years for a better characterization of breast lesions. Contrast-enhanced mammography and tomosynthesis are relative novel imaging techniques that have been implicated in breast cancer detection and diagnosis. We aimed to compare breast tomosynthesis, contrast-enhanced mammography, and breast ultrasound as complementary techniques to mammography in dense breast parenchyma.
Results
The study included 37 patients with 63 inconclusive mammography breast lesions. They all performed contrast-enhanced mammography, single-view tomosynthesis, and breast ultrasound. Mammography had a sensitivity of 83%, a specificity of 48%, a positive predictive value of 68%, a negative predictive value of 68%, and a diagnostic accuracy of 68%. Contrast-enhanced mammography had a sensitivity of 89%, a specificity of 89%, a positive predictive value of 91%, a negative predictive value of 86%, and a diagnostic accuracy of 89%. Tomosynthesis had a sensitivity of 86%, a specificity of 81%, a positive predictive value of 86%, a negative predictive value of 81%, and a diagnostic accuracy of 84%. Breast ultrasound had a sensitivity of 97%, a specificity of 85%, a positive predictive value of 90%, a negative predictive value of 96%, and a diagnostic accuracy of 92%.
Conclusion
Breast ultrasound, tomosynthesis, and contrast-enhanced mammography showed better performance compared to mammography in dense breasts. However, ultrasound being safe with no radiation hazards should be the second step modality of choice after mammography in the assessment of mammography dense breasts. Adding tomosynthesis to mammography in screening increases its sensitivity. Contrast-enhanced mammography should be reserved for cases with inconclusive sonomammographic results.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Breast cancer is a major health problem and a leading cause of death among women in Egypt. Early detection of breast cancer improves the outcomes and survival rate [1].
Breast density is used to reflect breast tissue composition and different x-ray attenuation characteristics of fat and glandular tissues. Women with heterogeneously or extremely dense breast tissue are considered to have high mammographic density [2].
Women with dense breasts encounter two major problems as increased breast density decreases the sensitivity and specificity of mammography and the dense breast itself is a risk factor for developing breast cancer [3].
Mammography is accused of having low sensitivity and specificity in dense breast parenchyma owing to a decrease in the contrast between a tumor and surrounding breast tissue, and superimposed breast tissues may obscure lesions [2].
Contrast-enhanced mammography (CEM) is one of the relatively novel imaging modalities. It provides low-energy images comparable to mammographic images and post-contrast recombined images to evaluate tumor neovascularity. CEM allows better evaluation of calcifications by their visualization on low-energy images combined with enhancement criteria on the contrast-enhanced recombined images [4,5,6].
Digital breast tomosynthesis (DBT) provides quasi-3D imaging of the breast, so it reduces the superimposition of breast tissue and improves cancer detection. Previous studies showed that DBT improved the sensitivity, specificity, and accuracy of full-field digital mammography by reducing the recall rates and increasing the cancer detection rates [7, 8].
Breast ultrasound has the advantages of being relatively available, inexpensive, and well-tolerated by women without exposure to ionizing radiation. The addition of breast ultrasound as a supplemental test to mammography in women with dense breast tissue showed an incremental cancer detection [9].
We aimed to compare breast tomosynthesis, contrast-enhanced mammography (CEM), and breast ultrasound as complementary techniques to mammography in dense breast parenchyma
Methods
The study was a prospective study carried over 7 months with an additional 18 months (3 follow-up studies every 6 months) when close follow-up was requested to confirm the diagnosis in some of the cases. The study included 37 patients with 63 inconclusive mammography breast lesions. Ten patients had bilateral lesions, and 14 patients had multiple lesions in the same breast. Their ages ranged from 27 to 69 years (mean age 46.8 ± 9.88 SD). They all performed mammography, single MLO view tomosynthesis, contrast-enhanced spectral mammography, and breast ultrasound. The final diagnosis was either by histopathological analysis of biopsy samples (for 39 lesions), fine-needle aspiration cytology, or close follow-up (for 24 lesions assigned BI-RADS 2); the study was approved by the ethical committee, and informed written consent was taken from all subjects. Image interpretation was done by a single consultant radiologist with more than 10 years of experience in advanced breast imaging mammography techniques (Figs. 1, 2, 3, and 4).
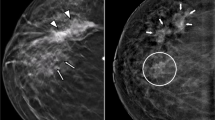
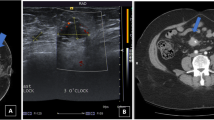
A 32-year-old patient with a history of left breast ductectomy. She presented by mastalgia and a palpable mass in the left breast. Mammography and DBT revealed clusters of pleomorphic microcalcifications in the left upper outer quadrant (a, b). CEM showed faint heterogeneous segmental non-mass enhancement in the left upper outer quadrant (c). Breast ultrasound showed microcalcifications with no associated mass lesions (d). The final diagnosis was invasive ductal carcinoma with DCIS
A 48-year-old patient came for screening. Mammography and DBT revealed spiculated mass in the right upper outer quadrant (a–c). CEM showed an intense heterogeneous enhancing mass in the right upper outer quadrant with not circumscribed spiculated margins (d, e). Breast ultrasound showed an irregular ill-defined speculated hypoechoic mass with posterior shadowing (f). The final diagnosis was invasive duct carcinoma
A 32-year-old patient presented by a palpable lump in her left breast. Mammography and DBT revealed no abnormality (a, b). CEM showed no significant contrast uptake (c). Breast ultrasound showed an irregular ill-defined hypoechoic mass within the left breast at the axillary tail region being deeply seated over the pectoralis muscle together with adjacent another hypoechoic irregular ill-defined mass (d, e). The final diagnosis was invasive duct carcinoma
A 60-year-old patient presented by a lump in the left breast. Mammography revealed focal asymmetry in the left upper inner quadrant with microcalcifications (a, b). DBT showed left breast spiculated mass with microcalcifications as well as right breast retroareolar nodule with microcalcifications (c, d). CEM showed left breast heterogeneously enhancing upper inner quadrant mass lesion with spiculated margins and surrounding multiple satellite lesions as well as right breast tiny right retroareolar homogenously enhancing mass with not circumscribed irregular margins (e, f). Breast ultrasound showed left breast irregular ill-defined mass in the left upper inner quadrant as well as right retroareolar small irregular ill-defined mass (g, h). The final diagnosis was bilateral invasive duct carcinoma
Subjects
Inclusion criteria
-
1.
Symptomatic patients with mammography dense breasts classified as C or D according to ACR BI-RADS lexicon breast density classification [10].
-
2.
Patients with mammography dense breast on screening with inconclusive mammographic findings [10].
Exclusion criteria
-
1.
Contraindication to mammography, e.g., pregnant women.
-
2.
Contraindication to intravenous contrast, e.g., patients with renal impairment, allergic patients, or those known to have a history of anaphylactic reaction to contrast media.
-
3.
Patients with breast density classified as ACR A or B.
The technique of contrast-enhanced mammography
CEM examination was performed using Senographe Essential, GE healthcare Full Field Digital Mammography machine with sonobright.
A one-shot intravenous injection (of 1.5 mL/kg) of non-ionic contrast media was performed. Two minutes after contrast administration, a pair of low-energy (23–32 KVp) and high-energy (45–49 KV) images were acquired in the mediolateral oblique (MLO) and craniocaudal (CC) views. Recombined iodine-enhanced images were obtained by the subtraction of the low from the high-energy images.
The technique of 3D tomosynthesis
For the 3D digital tomosynthesis, a single view (MLO) was obtained using Senographe Essential, GE healthcare Full Field Digital Mammography machine with sonobright. Twelve to fifteen low-dose 2D projection exposures were taken. The 3D volume of the compressed breast was reconstructed from the 2D projections in the form of a series of images (slices) through the entire breast. Images were assessed on a dedicated workstation.
The technique of breast ultrasound
Breast ultrasound was done using a LOGIQ™P5 ultrasound scanner (GE Health care) linear array electronic probe (3–9 MHz). Radial scanning of the entire breast and axillary tail of both sides was performed while the patient’s arm is relaxed and flexed behind the head. Medial lesions were scanned in the supine position, and lateral lesions, including the axilla, were scanned with the patient in the contralateral oblique position. Longitudinal and transverse images of breast lesions were obtained.
Image analysis and interpretation of mammography
Each lesion was evaluated regarding the site and type (mass, architectural distortion, focal asymmetry ± calcifications). We assigned each lesion a BI-RADS category according to the 2013 BI-RADS Atlas [10]. The interpreting breast image radiologist was aware of the clinical data but was blind to the final pathology results.
Image analysis and interpretation of contrast-enhanced mammography
Lesions were characterized according to the 2013 MRI BI-RADS lexicon morphology descriptors as no specific lexicon is still present for CESM [10]. Identified lesions were first classified into enhancing and non-enhancing. Enhancing lesions were then classified into mass or non-mass. When an enhancing mass lesions were identified, they were characterized according to their margins (circumscribed, not circumscribed irregular or not circumscribed, spiculated), the intensity of enhancement (mild, moderate, and severe), and the pattern of internal enhancement (homogenous, heterogeneous, or ring enhancement).
• When non-mass enhancing lesions were identified, they were further characterized according to their distribution (focal, ductal, segmental, regional, or parenchymal), the pattern of internal enhancement (homogenous or heterogeneous), and the intensity of enhancement (mild, moderate, and severe). We assigned a BI-RADS category for each lesion in reference to the BI-RADS atlas 2013 [10] as there is no standardized BI-RADS lexicon to CEM, guided by the results of clinical data and CEM findings but blinded to the final pathological diagnosis.
Image analysis and interpretation of tomosynthesis
Each lesion was evaluated regarding the site and type (mass, architectural distortion, focal asymmetry ± calcifications). We assigned a BI-RADS category for each lesion in reference to the BI-RADS atlas 2013 [10], guided by the results of clinical data and tomosynthesis findings but blinded to the final pathological diagnosis.
Image analysis and interpretation of breast ultrasound
Each lesion was evaluated regarding its shape, boundary, margin, echopattern, and posterior acoustic features, calcifications, and axillary lymph nodes. We assigned a BI-RADS category for each lesion according to ultrasound BI-RADS atlas 2013 [10], guided by the results of clinical data and breast ultrasound findings but blinded to the final pathological diagnosis.
Statistical analysis
-
Data were statistically described in terms of range, mean standard deviation (SD), frequencies (number of cases), and percentages when appropriate.
-
Accuracy was represented using the terms sensitivity, specificity, positive predictive value, negative predictive value, and overall accuracy, the likelihood ratio of a positive test, and the likelihood ratio of a negative test.
Results
In the current study, 34/37 (92%) cases were assigned an ACR score of C and 3/37 (8%) cases were assigned an ACR score of D according to the ACR breast density classification.
Upon correlation with the final diagnosis, 27/63 (43%) were benign lesions and 36/63 (57%) were malignant lesions. Within the “malignant lesions” group, 35/36 (97%) lesions were invasive duct carcinomas and 1/36 (3%) lesion was DCIS and associated IDC. The distribution of different pathological entities within the “benign lesions” group is demonstrated in Table 1.
The mammography findings in the studied lesions are demonstrated in Table 2. Mammography BI-RADS category was assigned for each lesion where 19/63 (30%) lesions were considered benign, classified as BI-RADS 1, 2, and 3, while 44/63 (70%) lesions were considered malignant classified as BI-RADS 4 or 5. After revising the results of pathology and/or close follow-up, 30 lesions were true positives, 14 lesions were false positives, 6 lesions were false negatives, and 13 lesions were true negatives. Diagnostic indices of mammography were a sensitivity of 83%, a specificity of 48%, a positive predictive value of 68%, a negative predictive value of 68%, and a diagnostic accuracy of 68%. This was shown in Table 3.
Regarding CEM findings, 46/63 (70%) lesions showed contrast uptake and 17/63 (30%) lesions did not show contrast uptake. Enhancing lesions were classified into 32/46 (68%) enhancing mass lesions and 14/46 (32%) enhancing non-mass lesions. Upon correlation with the results of pathology and/or close follow-up, 3/32 (10%) enhancing mass lesions and 11/14 (78.6%) enhancing non-mass lesions were benign while 29/32 (90%) enhancing mass lesions and 3/14 (21.4%) enhancing non-mass lesions were malignant. Thirteen out of 17 (68%) non-enhancing lesions were benign, and only 4/17 (32%) non-enhancing lesions were malignant. Accordingly, a BI-RADS category was assigned for each lesion. Twenty-eight out of 63 (44%) lesions were benign (BI-RADS 1, 2, and 3), and 35/63 (56%) lesions were malignant (BI-RADS 4 and 5). After revising the pathology results, 31 lesions were true positives, 3 were false positives, 4 lesions were false negatives, and 24 were true negatives. So, contrast-enhanced spectral mammography had a sensitivity of 89%, a specificity of 89%, a positive predictive value of 91%, a negative predictive value of 86%, and a diagnostic accuracy of 89%. This was shown in Table 3.
Tomosynthesis findings among studied lesions are demonstrated in Table 4. BI-RADS category was given for each lesion according to morphology descriptor; 27/63 (43%) lesions were benign (BI-RADS 1, 2, and 3) and 36/63 (57%) lesions were malignant (BI-RADS 4 and 5). After revising the results of pathology and/or close follow-up, 31 lesions were true positives, 5 lesions were false positives, 5 lesions were false negatives, and 22 lesions were true negatives. Tomosynthesis had a sensitivity of 86%, a specificity of 81%, a positive predictive value of 86%, a negative predictive value of 81%, and a diagnostic accuracy of 84%. This was shown in Table 3.
Regarding ultrasound findings, 50/63 (79%) lesions presented by mass lesions while 13/63 (31%) presented by non-mass lesions. Mass lesions were further assessed regarding shape, margins, echogenicity, and posterior acoustic enhancement and/or shadowing. BI-RADS category was given for each lesion according to ultrasound morphology descriptors; 24/63 (38%) lesions were considered benign (BI-RADS 1, 2, and 3), and 39/63 (62%) lesions were considered malignant. After revising the results of pathology and/or close follow-up, 35 were true positives, 4 were false positives, 1 lesion was false negative, and 23 were true negatives. So, breast ultrasound had a sensitivity of 97%, a specificity of 85%, a positive predictive value of 90%, a negative predictive value of 96%, and a diagnostic accuracy of 92%. This was shown in Table 3.
Discussion
According to the 2013 American College of Radiology BI-RADS lexicon classification of breast density, ACR C indicates that the breasts are heterogeneously dense, which may obscure small mass while ACR D indicates that the breast is extremely dense [10]. Although mammogram is the gold standard technique in the detection and diagnosis of breast cancer, the increased mammographic breast density poses a real challenge in diagnosis owing to the remarkably decreased mammography sensitivity resulting in a considerable number of false-negative mammograms [11,12,13]. On the other hand, the increased breast density may also increase the false-positive mammograms resulting in an unnecessary increase in the recall rates. Adding to this, the increased breast density impacts the risk of breast cancer development being a strong independent risk factor as reported in several studies [14, 15].
In the past few years, the number of breast imaging modalities addressing the problem of the mammography dense breast has increased; thus, the implementation of sound practice has become a real challenge. Out of these new modalities, contrast-enhanced mammography and 3D digital tomosynthesis have gained high appreciation.
In the current study, we compared the performance of contrast mammography, 3D tomosynthesis, and breast ultrasound in the mammographic dense breast. The study included 37 patients with 63 breast lesions assigned as BI-RADS C (34/37, 92%) or BI-RADS D (3/37, 8%).
As regards mammography findings, the commonest presentation was mass lesions (25/63, 40%) followed by breast asymmetry(8/63, 28%). Other lesions included indeterminate calcifications (5/63, 8%) and mass/asymmetry with calcifications (2/63, 3%) while no lesions were identified in 13/63 (21%). Mammography had a sensitivity of 83%, a specificity of 48%, a positive predictive value of 68%, a negative predictive value of 68%, and a diagnostic accuracy of 68%. As reported in several previous studies, evidence suggests that the sensitivity and specificity of mammography are decreased in breasts with a higher density. Chiu et al. [16] reported a sensitivity of 62.8% in the dense breast as compared to 82% in the fatty breast. A drop in specificity was also reported with an increased breast density (89.6% in the dense breast as compared to 96.5% in the fatty breast). The calculated diagnostic indices in our study did not completely match those reported in a cohort study performed by Von Euler-Chelpin et al. [17] on 54,997 participants. In this study, they compared the sensitivity and specificity of mammography as compared to breast density. Although the calculated sensitivity dropped from 80% in the homogeneous fatty breast down to 41% in the dense breast, the specificity of mammography was not significantly changed.
Contrast-enhanced mammography uses a dual-energy technique performed after contrast administration to identify and characterize lesions based on angiogenesis, as well as morphologic features and density [5]. Also, low-energy images of CEM could detect microcalcifications, architectural distortion, and non-enhancing lesions [18].
Kamal et al. [19] analyzed the morphology and enhancement characteristics of breast lesions on CEM. Their retrospective study included 168 consecutive patients with 211 breast lesions. Enhancement was observed in 145/211 lesions (68.7%): 42/145 (29%) benign and 103/145 (71%) malignant lesions (p value ≤ 0.001). They concluded that the morphology and enhancement characteristics of breast lesions on CESM are reliable in differentiating between benign and malignant breast lesions
In the current study, 46/63 (70%) lesions showed contrast uptake. We applied the morphology descriptors to differentiate between benign and malignant lesions with correlation with final pathology, out of which 14/46 (30%) were benign and 32/46 (70%) were malignant, while 17/63 (30%) lesions did not show contrast uptake in CEM, out of which four lesions were malignant upon correlation with final pathology. These four lesions were out of contrast mammographic view as they were very deep on the pectoralis muscle at the axillary tail region.
CEM improves the sensitivity and specificity of mammography because it provides functional information in addition to morphology. So, adding contrast to mammograms in the current study improved the diagnostic indices with a calculated sensitivity of 89%, a specificity of 89%, a positive predictive value of 91%, a negative predictive value of 86%, and a diagnostic accuracy of 89% for CESM.
Our results were comparable to a study performed by Sung et al. [20]; their study included 904 CEM studies for women of high breast cancer risk. They concluded that sensitivity of contrast agent–enhanced mammography was 87.5% (compared with 50.0% for digital mammography) with a specificity of 93.7%.
Sorin et al. [21] performed a retrospective study discussing the role of CEM in women with dense breasts; they stated that CEM may be a valuable supplemental screening modality for women who have dense breasts as it increases the sensitivity of breast cancer detection.
Another study was done by Mori et al. [22] comparing the diagnostic accuracy of CEM to digital mammography in dense breasts. Their study demonstrated a significantly higher sensitivity, specificity, and accuracy of CEM compared to standard mammography.
Initial studies suggest that, when tomosynthesis is used in conjunction with standard 2-dimensional digital mammography as a screening test, DBT can reduce recall rates and increase cancer detection rates [23].
The main advantage of tomosynthesis is its ability to diminish the masking effect of tissue overlap and structure noise usually encountered with 2D mammography [24].
In our study, tomosynthesis had a sensitivity of 86%, a specificity of 81%, a positive predictive value of 86%, a negative predictive value of 81%, and a diagnostic accuracy of 84%.
Our results were comparable with Bian et al. [25] and Asbeutah et al. [26] where they also found that DBT showed increased sensitivity and specificity compared to mammography in dense breasts.
Phi et al. [27] performed a meta-analysis on DBT in dense breasts for screening or diagnosis. They concluded that DBT increased cancer detection rate in screening and diagnosis; in diagnosis, DBT increased sensitivity but not specificity.
In 2016, Rafferty et al. [28] performed a study including 452,320 examinations: 278,906 were digital mammography alone and 173,414 digital mammographies plus tomosynthesis. They concluded that adding DBT to digital mammography increased the cancer detection rate more in women with heterogeneously dense breasts than in those with either non-dense breasts or extremely dense breasts. The reduction in recall rate was also greatest in the heterogeneously dense subgroup.
Skaane et al. [29] conducted a prospective study on DM versus DBT and DM in screening. They found that the addition of digital breast tomosynthesis to digital mammography resulted in significant gains in sensitivity and specificity.
Breast ultrasound had been used for many years in the characterization of breast lesions. The J-start prospective randomized control study of ultrasonography has shown increase sensitivity and detection rates of early cancers [30].
In our study, breast ultrasound had a sensitivity of 97%, a specificity of 85%, a positive predictive value of 90%, a negative predictive value of 96%, and a diagnostic accuracy of 92%.
Our results matched with Nandan et al. [31], a retrospective study that focused on charts from the Cancer Institute of Guyana to assess the role of ultrasound in screening young women. They concluded that ultrasonography is effective and sensitive in the diagnosis of breast cancer. It is also effective in diagnosing benign breast diseases in younger women with dense breast tissue.
This study had few limitations such as the interpretation of images was done by a single radiologist and the absence of an appropriate BI-RADS lexicon for CEM examination. A standardized lexicon of morphology descriptors seen on CEM would provide the optimal analysis and reporting of enhancing lesions detected in the breast.
Conclusion
Breast ultrasound, tomosynthesis, and contrast-enhanced mammography showed better performance compared to mammography in dense breasts. However, ultrasound being safe with no radiation hazards should be the second step modality of choice after mammography in the assessment of mammography dense breasts. Adding tomosynthesis to mammography in screening increases its sensitivity. Contrast-enhanced mammography should be reserved for cases with inconclusive sonomammographic results.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BI-RADS:
-
Breast Imaging-Reporting and Data System
- CEM:
-
Contrast-enhanced mammogram
- CC:
-
Craniocaudal
- DBT:
-
Digital breast tomosynthesis
- DCIS:
-
Ductal carcinoma in situ
- IDC:
-
Invasive ductal carcinoma
- MLO:
-
Mediolateral oblique
- SPSS:
-
Statistical Package for the Social Sciences
- 3D:
-
Three dimensional
References
Mostafa AAE, Eltomey MA, Elaggan AM et al (2019) Automated breast ultrasound (ABUS) as a screening tool: initial experience. Egypt J RadiolNucl Med 50:37
Nazari SS, Mukherjee P (2018) An overview of mammographic density and its association with breast cancer. Breast cancer 25(3):259–267
Thigpen D, Kappler A, Brem R (2018) The role of ultrasound in screening dense breasts-a review of the literature and practical solutions for implementation. Diagnostics 8(1):20
Patel BK, Lobbes MBI, Lewin J (2018) Contrast enhanced spectral mammography: a review. Semin Ultrasound CT MR 39(1):70–79
Perry H, Phillips J, Dialani V et al (2019) Contrast-enhanced mammography: a systematic guide to interpretation and reporting. AJR Am J Roentgenol 212(1):222–231
Houben IP, Vanwetswinkel S, Kalia V et al (2019) Contrast-enhanced spectral mammography in the evaluation of breast suspicious calcifications: diagnostic accuracy and impact on surgical management. ActaRadiol 60(9):1110-1117.
Yi A, Chang JM, Shin S et al (2018) Detection of noncalcified breast cancer in patients with extremely dense breasts using digital breast tomosynthesis compared with full-field digital mammography. The British journal of radiology 92(1093):20180101
Phi XA, Tagliafico A, Houssami N et al (2018) Digital breast tomosynthesis for breast cancer screening and diagnosis in women with dense breasts - a systematic review and meta-analysis. BMC Cancer 18(1):380
Klevo GA, Collado-Mesa F, Net JM et al (2017) Utility of supplemental screening with breast ultrasound in asymptomatic women with dense breast tissue who are not at high risk for breast cancer. The Indian journal of radiology & imaging 27(1):52–58
Sickles EA, D’Orsi CJ, Bassett LW, et al. (2013) ACR BI-RADS® mammography. In: ACR BI-RADS® atlas, breast imaging reporting and data system. Reston, VA, American College of Radiology
Price ER, Hargreaves J, Lipson JA, Sickles EA, Brenner RJ, Lindfors KK et al (2013) The California breast density information group: a collaborative response to the issues of breast density, breast cancer risk, and breast density notification legislation. Radiology 269(3):887–892
D’Orsi CJSE, Mendelson EB, Morris EA( 2013). ACR BI-RADS® atlas, breast imaging reporting and data system. Reston, VA: American College of Radiology.
Boyd NF, Martin LJ, Bronskill M et al (2010) Breast tissue composition and susceptibility to breast cancer. J Natl Cancer Inst 102(16):1224–1237
Huo CW, Chew GL, Britt KL et al (2014) Mammographic density-a review on the current understanding of its association with breast cancer. Breast Cancer Res Treat 144(3):479–502
Santen RJ, Boyd NF, Chlebowski RT et al (2007) Cummings S, Cuzick J, Dowsett M, et al. Critical assessment of new risk factors for breast cancer: considerations for development of an improved risk prediction model. EndocrRelat Cancer. 14(2):169–187
Chiu SY, Duffy S, Yen AM et al (2010) Effect of baseline breast density on breast cancer incidence, stage, mortality, and screening parameters: 25-year follow-up of a Swedish mammographic screening. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology 19(5):1219–1228
Von Euler-Chelpin M., Lillholm M., Vejborg I. et al.,(2019). Sensitivity of screening mammography by density and texture: a cohort study from a population-based screening program in Denmark. Breast Cancer Res21, 111
Yasin R, El Ghany EA (2019) BIRADS 4 breast lesions; comparison of contrast-enhanced spectral mammography and contrast enhanced MRI. Egypt J Nucl Med 50:34
Kamal RM, Helal M, Wessam Ret al.,(2015) Contrast-enhanced spectral mammography: impact of the qualitative morphology descriptors on the diagnosis of breast lesions. EurRadiol84 (6), P1049-1055
Sung JS, Lebron L, Keating D et al., (2019) Performance of dual-energy contrast-enhanced digital mammography for screening women at increased risk of breast cancer Radiology 293:1, 81-88
Sorin V, Yagil Y, Yosepovich A et al (2018) Contrast-enhanced spectral mammography in women with intermediate breast cancer risk and dense breasts. Am J Roentgenol 211(5):W267–W274
Mori M, Akashi-Tanaka S, Suzuki S et al (2017) Diagnostic accuracy of contrast-enhanced spectral mammography in comparison to conventional full-field digital mammography in a population of women with dense breasts. Breast Cancer 24(1):104–110,2017
Takahashi TA, Lee CI, Johnson KM (2017) Breast cancer screening: does tomosynthesis augment mammography? Cleveland Clinic Journal of Medicine 84(7):522–527
Seely JM, Alhassan T. (2018) Screening for breast cancer in 2018 – what should we be doing today? CurrOncol25(Suppl 1): S115–24.
Bian T, Lin Q, Cui C et al (2016) Digital breast tomosynthesis: a new diagnostic method for mass-like lesions in dense breasts. Breast J. 22(5):535–540
Asbeutah A, Karmani N, Asbeutah A et al (2018) Comparison of digital breast tomosynthesis (DBT) and digital mammography (DM) for detection of breast cancer in women in Kuwait. Med PrincPract. 28(1):10–15
Phi XA, Tagliafico A, Houssami N et al (2018) Digital breast tomosynthesis for breast cancer screening and diagnosis in women with dense breasts - a systematic review and meta-analysis. BMC cancer 18(1):380
RaffertyEA, DurandMA, ConantEF, et al (2016) Breast cancer screening using tomosynthesis and digital mammography in dense and nondense breasts. JAMA 2016; 315:1784–1786.
Skaane p, Bandos AL, Niklason LT et al.,(2019) Digital mammography versus digital mammography plus tomosynthesis in breast cancer screening: the Oslo tomosynthesis screening trial Radiology 291:1, 23-30
Ohuchi N, Suzuki A, Sobue T et al (2016) Sensitivity and specificity of mammography and adjunctive ultrasonography to screen for breast cancer in the Japan Strategic Anti-cancer Randomized Trial (J-START): a randomized controlled trial. Lancet 387:341–348. https://doi.org/10.1016/S0140-6736(15)00774-6
Nandan F, Alladin B (2018) The role of ultrasound as a diagnostic tool for breast cancer in the screening of younger women (age 25-38) in Guyana. Journal of Medical Diagnostic Methods. 07. https://doi.org/10.4172/2168-9784.1000273
Acknowledgements
Not applicable
Funding
No funding sources.
Author information
Authors and Affiliations
Contributions
HA and RK wrote the manuscript. MH collected the patient data and is responsible for correspondence to the journal, and AY participated in the design of the study. LB participated in its design and performed the statistical analysis. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the ethical committee of the Faculty of Medicine, Cairo University; the ethical approval number is not available. An informed written consent was taken from all subjects.
Consent for publication
All patients included in this research gave written consent to publish the data contained within this study.
Competing interests
No financial or non-financial competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Azzam, H., Kamal, R.M., Hanafy, M.M. et al. Comparative study between contrast-enhanced mammography, tomosynthesis, and breast ultrasound as complementary techniques to mammography in dense breast parenchyma. Egypt J Radiol Nucl Med 51, 148 (2020). https://doi.org/10.1186/s43055-020-00268-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43055-020-00268-1