Abstract
Background
Hepatocellular carcinoma (HCC) is classified as the sixth commonest cancer over the world and the fourth common in Egypt, representing the third leading cause of cancer-related mortality. Trans-arterial chemoembolization (TACE) is considered as one of the most effective treatment option for intermediate stage HCC. Lesions showing complete response (CR) after TACE can be classified according to their lipiodol retention pattern into complete (C-Lip, covering the entire tumor volume) or incomplete (I-Lip). The purpose of this study was to assess the prediction value of post-TACE lipiodol retention pattern on the local disease progression after complete response and thus to decide which patient should be carefully observed and considered to be incompletely treated.
Results
The study included 45 HCC lesions treated with TACE and showed complete response in the first follow-up CT performed 4–6 weeks after the procedure; 23 cases showed incomplete lipiodol retention pattern and 22 with complete lipiodol retention pattern which was carefully assessed on non-contrast CT images done 4 to 6 weeks after treatment. Follow-up CT was then performed every 3 months for at least 12 months or until local progression occurred. On the follow-up CT studies, 20 out of the 45 lesions showed persistent complete response (i.e., no local progression), while 25 of them showed local progression. Among the 25 cases with disease progression upon correlation with their lipiodol retention pattern, it was found that local progression occurred in 31.8% of tumors showed CR with complete lipiodol retention, as opposed to 78.2% of tumors showed CR with incomplete lipiodol retention with significant statistical difference and p value 0.010.
Conclusions
Lipiodol retention pattern of HCC after TACE can predict the potential tumor local outcome. Lesions with incomplete lipiodol retention are at a higher risk of local tumor progression and therefore should probably observed and can be retreated; on the other hand, tumors with complete lipiodol retention have a much lower risk of local disease tumor progression.
Similar content being viewed by others
Background
Hepatocellular carcinoma (HCC) is classified as the sixth common cancer over the world and the fourth common cancer in Egypt, representing the third leading cause of cancer-related mortality. HCC patients classified and treatment allocated according to tumor stage, liver function, and general performance status. Only 30–40% of HCC patients are candidates for curative treatment options. The other majority of HCC patients proceed for local, regional therapy or even systemic palliative treatment [1,2,3].
Trans-arterial chemoembolization (TACE) is considered as the first treatment line for intermediate stage HCC, which is involving large lesions and multinodular HCC and average normal liver function test, with no associated cancer-related symptomology, tumoral vascular invasion or extrahepatic spread. Stage B of the Barcelona Clinic Liver Cancer (BCLC) shows heterogeneous group of patients, and a sub-classification was established to properly determine the selection of patients most likely to get benefit from TACE treatment. According to this classification, the best candidates for TACE are those patients with uni- or pauci-nodular lesions without vascular extensions or tumor metastatic spread, with no symptoms and have Child–Pugh stage of ≤ B7. In those patients, the median survival following TACE is about 40–50 months in modern series. TACE is also used in patients with early-stage HCC as a bridge to liver transplantation [2,3,4].
Local regional therapy (LRT) provides a bridge for liver transplantation and has two main roles in patients with liver cirrhosis and HCC waiting for liver transplantation: first, is to achieve local disease control and second is to downstage the patients outside liver transplantation criteria to make them eligible for surgery [5].
Two different TACE techniques were used since 2004, conventional TACE (cTACE) and TACE by drug-eluting beads (DEB-TACE). cTACE combines the transcatheter chemotherapy injection using lipiodol-based emulsion associated with embolizing agent to reach the maximal cytotoxic and ischemic effects upon the tumor cell [2].
Tumor response is evaluated according to mRECIST criteria and classified into complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD). Lesions showing a CR means that there is no enhancing component on arterial phase following to the cTACE session and classified according to the lipiodol retention pattern in lesion with complete lipiodol filling (C-Lip, at which the entire tumor volume is covered by lipiodol), and incomplete lipiodol filling (I-Lip). The reappearance of contrast enhancement within the embolized lesion or within 2 cm from its margins at the arterial phase with delayed washout is considered to be local disease progression [6].
For the assessment of the treatment response, Response Evaluation Criteria in Solid Tumors (RECIST) was introduced by measurement of the tumor size. However, this neglects the fact that locoregional therapies induce tumor infarction without early changes in size. Recently, modified Response Evaluation Criteria in Solid Tumors (mRECIST) criteria were introduced and based on measuring only the viable enhancing component of the lesion in a single long-axis dimension [7].
Many studies suggested that mRECIST overestimates the post-treatment tumor response compared to the pathological assessment of necrosis. In cTACE this could happened due to hyper-attenuation on CT from the lipiodol can mask underlying hyper-enhancement. Therefore, many authors put recommendations for using MRI to assess tumor response. However, many interventional teams still use CT for monitoring their patients believing that lipiodol retention is informative with a good correlation to pathological necrosis [8,9,10].
The purpose of the study was to assess the prediction value of lipiodol retention pattern on post-cTACE CT scan for local disease progression of HCC after complete response and thus to decide whether HCC lesions with incomplete lipiodol retention pattern have a higher risk of local progression and should be considered to be incompletely treated. Our aim was also to improve accuracy of reporting HCCs response after cTACE and consequently aiding in the treatment plan.
Methods
The study was a prospective cross-sectional analytic study performed over 30 patients with liver cirrhosis secondary to chronic viral hepatitis and having 45 HCC lesions diagnosed according to LIRADS criteria as LR-4 and LR-5 underwent cTACE aiming to assess the prediction value of lipiodol retention pattern on post-TACE CT scan for local disease progression after complete response. The study was conducted at the radiology department of our institution in the period between March 2022 and March 2023 on 30 patients with 45 HCC lesions, underwent a first cTACE session and came for follow-up to assess treatment response and to plan the next step in management. The study was IRB approved. Written informed consent was obtained from all patients shared in the study.
Inclusion criteria
Patients candidates for TACE according to Barcelona clinic liver cancer (BCLC) guidelines and underwent cTACE for at least 1 HCC. Tumor showed complete response (CR) based on mRECIST criteria in the first post-therapy follow-up CT which was performed 4–6 weeks after trans-arterial chemoembolization.
Exclusion criteria
Patients with impaired renal functions. Patients with contraindication to TACE or underwent other treatment procedures. Lesions showed response other than complete response (CR) according to mRECIST criteria.
TACE procedure
The cTACE procedures were performed via senior expert intervention radiologist with 16 years of experience at intervention unit via right femoral approach under digital subtraction images or cone beam 3D CT angiography. The treatment done via injection of chemotherapy and emulsified oil contrast (Lipiodol) and associated embolization of the arterial feeder was performed.
Triphasic CT examination protocol
Post-contrast triphasic CT study of the abdomen was performed using multi-detector CT (128 detector LightSpeed VCT; GE Healthcare). Multiple phases were obtained as follows: non-contrast phase, followed by iodinated water soluble IV contrast injection (about 350 mg/mL). The contrast dose injected was 2 mL/kg injected at a rate of 4 mL/s. Post-contrast early arterial obtained about 20 s after injection, late arterial obtained about 40 s after injection, and venous phase obtained about 80 s after injection. The acquisition thickness was about 2.5 mm. A 5 min delayed phase was then obtained.
Follow-up post-interventional CT was then performed 4–6 weeks after treatment and proper categorization of the pattern of lipiodol retention was assessed at the non-contrast phase images. Follow-up CT scan with multi-phases was performed every 3 months. Each tumor was followed until local progression occurred. If the ablation bed revealed no recurrence or local disease progression on last available follow-up CT scan over period of at least 12 months after TACE, then this is considered to be a persisting CR.
CT images interpretation
Triphasic CT images pre- and post-cTACE images were reviewed in consensus by three expert radiologists with 11, 16 and 17 years of experience in abdominal imaging. Images analyzed via picture archiving and communication system (PACS). The following items were assessed and reported for each treated lesion: (a) the longest diameter; (b) the presence of delayed enhancing tumoral capsule; (c) tumoral portal vein thrombosis; (d) location of the lesions within the lover segments according to Couinaud’s classification; (e) the pattern of lipiodol retention on 1st follow-up CT performed 4–6 weeks after TACE session. To ensure covering the whole tumor volume, the follow-up study compared to the pre-cTACE CT.
Lipiodol retention pattern was considered as a complete retention if the volume of entire lesion was filled by lipiodol as compared to the surrounding liver on non-contrast phase. The lipiodol accumulation was considered complete even if it appears homogenous or patchy as long as the whole tumor showed lipiodol accumulation. While lipiodol retention pattern was categorized as incomplete retention if the lesion volume showed partial accumulation not covering the whole tumor volume on non-contrast phase at post-TACE study.
Post-treatment response of the tumor and local progression were evaluated, and each lesion was individually categorized according to mRECIST into complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD) (Table 1). The study selectively focused on the lesions show complete response. Therefore, patients without a CR were excluded from the study. Each single lesion was followed until local disease progression occurred (by periodic post-treatment CT study every 3 months), local progression defined as appearance of an arterially enhancing nodular areas with subsequent corresponding washout on portal or delayed phases within or along the margins of the previously embolized lesion. If the treated lesion showed no local disease progression on the post-treatment CT study follow-up (for at least 12 months after the cTACE), the lesion was considered to be a persisting complete response.
Statistical method and analysis
A Chi square (χ2) test were performed for comparison between different study groups. The association between patterns of lipiodol retention and local disease progression was assessed by means of odds ratio (OR). The obtained results were considered statistically significant when P value was less than 0.05. The data was analyzed using Statistical Package for the Social Sciences (SPSS) version 28.
Results
The study included a total of 30 patients with 45 HCC lesions. The patients’ ages ranged from 40 to 72 years (mean 63.80 ± 10.02 SD). Twenty seven patients were males (90%) and three patients were females (10%).
Eighteen patients had solitary HCC lesion (60%), nine patients had two lesions (30%) and three patients had three lesions (10%). The size of the treated lesions before the TACE procedure ranged between 3.2 and 6.1 cm with mean size 4.2 cm ± 0.08.
Response to TACE and lipiodol retention pattern
Forty five of the 45 HCCs (100%) were classified as CR on first CT performed 4–6 weeks after TACE according to mRECIST criteria.
Lipiodol retention within the lesion was considered to be complete in 22 lesions (48.9%), and incomplete in 23 lesions (51.1%). On follow-up CT studies, 20 lesions showed persistent CR which means no local progression (44.4%) and 25 lesions showed local progression (55.6%).
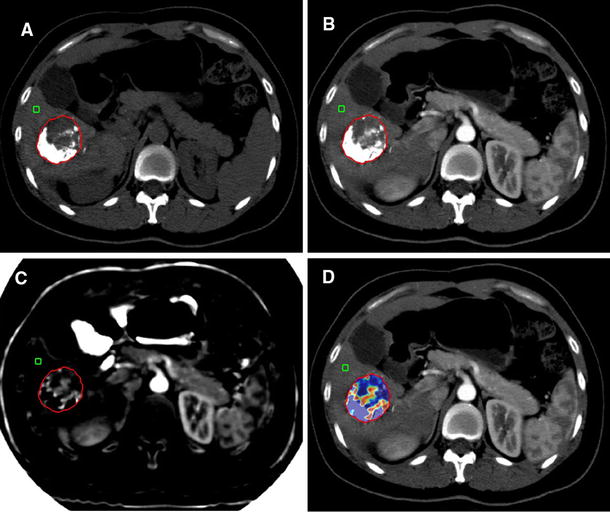
Among the 22 lesions with complete lipiodol retention pattern, 15 out of the 22 (68.2%) showed no local progression (persistent CR) (Fig. 1), while seven out of 22 (31.8%) showed local progression (Fig. 2). Among the 23 lesions with incomplete lipiodol retention pattern, five out of the 23 (21.8%) showed no local progression (persistent CR) (Fig. 3), while 18 out of 23 (78.2%) showed local progression (Fig. 4). A significant statistical difference was found with p value 0.010 (Table 2).
A 60-year-old male patient diagnosed with HCC and underwent a first session of cTACE. a Non-contrast and b arterial phase image of post-TACE CT showed complete lipiodol retention within the lesion on a precontrast image with no enhancement on arterial phase image denoting complete response. c Arterial phase and d delayed phase image of the follow-up CT study showing regression in the size of the lesion with no arterial enhancement or washout consistent with persistent complete response
A 64-year-old male patient diagnosed with HCC and underwent a first session of cTACE. a Non-contrast and b arterial phase image of post-TACE CT showed complete lipiodol retention covering the entire volume of the lesion on a precontrast image with no enhancement on arterial phase image denoting complete response. c Arterial phase and d delayed phase image of the follow-up CT study showing regression in the amount of lipiodol within the lesion and newly developed areas of arterial enhancement and delayed wash out consistent with local disease progression
A 53-year-old male patient diagnosed with HCC and underwent a first session of cTACE. a Non-contrast and b arterial phase image of post-TACE CT showed incomplete lipiodol retention within the lesion on a precontrast image with no enhancement on arterial phase image denoting complete response. c Arterial phase and d delayed phase image of the follow-up CT study showing no arterial enhancement or washout consistent with persistent complete response
A 60-year-old male patient diagnosed with HCC and underwent a first session of cTACE. a Non-contrast and b arterial phase image of post-TACE CT showed complete lipiodol retention covering the lesion on a precontrast image with no enhancement on arterial phase image denoting complete response. c Arterial phase and d delayed phase image of the follow-up CT study showing regression in the amount of lipiodol and newly developed small nodular areas of arterial enhancement and delayed wash out consistent with local disease progression
Factors related with local progression
Age
Among the studied patients, mean age of patients whom lesions showed local progression on follow-up was 63.82 ± 11.94 years old, while mean age of patients whom lesions showed persistent complete response (i.e., no local progression) is 63.77 ± 7.22 years old. No statistically significant difference was noted (P value 0.2).
Number of lesions
Among the 20 lesions that showed no local progression (persistent CR) on follow-up CT up to 1 year after treatment, 12 patients had single lesion and 4 patients had 2 lesions. While among the 25 lesions that showed local progression on follow-up CT, 6 patients had single lesion, 5 patients had 2 lesions and 3 patient had 3 lesions. No significant statistical difference was found (P value 0.05).
Relation between numbers of the lesions and lipiodol retention pattern
Among the 22 HCC lesions showed complete lipiodol retention pattern on post-TACE CT, 11 patients had single lesion, four patients had two lesions and one patient had three lesions. Among the 23 HCC lesions showed incomplete lipiodol retention pattern on post-TACE CT, seven patients had single lesion, five patients had two lesions and two patients had three lesions. No significant statistical difference was found (P value 0.1).
Relation between size of the lesions and lipiodol retention pattern
The mean size of the lesions among the 22 HCC lesions with complete lipiodol retention pattern was 4.1 cm ± 0.2 SD. While the mean size of the 23 HCC lesions with incomplete lipiodol retention pattern was 5.2 cm ± 0.4 SD. Based on Pearson’s correlation coefficient, the results show that there is a negative significant correlation between size of the lesion and lipiodol retention pattern with \(r=-0.553\) and \({p}_{{\text{value}}}<0.001\), which indicates that the larger the lesion the higher the incidence of I-lip. Based on Mann–Whitney U test which is a nonparametric statistical test used to compare two samples/groups. The results show that there is a statistical significant difference between the size of the lesion of I-Lip group and the size of the lesion of C-Lip group with statistic value \(U=85.50\) and \({p}_{{\text{value}}}<0.001\).
Relation between size of the lesions and local progression on follow-up
Based on Pearson’s correlation coefficient, the results show that there is a high negative significant correlation between size of the lesion and local progression on follow-up with \(r=-0.758\) and \({p}_{{\text{value}}}<0.001\), which indicates that the larger the lesion the higher the incidence of local progression. Based on Mann–Whitney U test which is a nonparametric statistical test is used to compare two samples/groups. The results show that there is a statistical significant difference between the size of the lesion of local progression group and the size of the lesion of no local progression group with statistic value \(U=30\) and \({p}_{{\text{value}}}<0.001\)
Discussion
Hepatocellular carcinoma (HCC) is a main health problem in Egypt, representing the third leading cause of cancer-related mortality. Only 30–40% of HCC patients are candidates for curative options of treatment. The other majority of HCC patients proceed for local, regional therapy or even systemic palliative treatment [1,2,3].
In this study, we demonstrated that among the 45 treated HCC lesions showing complete response (CR) according to mRECIST criteria on post-TACE CT studies, 22 of them showed complete lipiodol retention pattern within the lesion, while 23 lesions showed incomplete lipiodol retention pattern. On follow-up CT studies, 20 HCC lesions showed persistent complete response (i.e., no local progression), while 25 of them showed local progression. Upon correlation to their lipiodol retention pattern on post-TACE CT studies, we found that local progression only occurred in 31.8% of tumors with a CR and complete lipiodol retention, as opposed to the majority of tumors (78.2%) with a CR and incomplete lipiodol retention.
Many different authors studied the therapeutic effect and prognosis of lipiodol retention pattern after cTACE intervention. The study done by Burgio et al. [8] reported that there is a high risk of local progression of HCCs viable lesions along the incomplete lipiodol retention bed after first intervention by cTACE session even if there is a complete response (CR) when assessed by mRECIST criteria, so these lesions should be considered to be incompletely treated.
Also, Stark et al. [11] reported a strong correlation between lipiodol deposition and enhancing tumor volume (ETV) on the pre-treatment MRI and post-treatment MRI follow-up. On post-treatment study follow-up, the regions of the tumor filled by lipiodol showed higher rates of ETV reduction than the regions not filled by lipiodol (p = 0.0475).
Another study by Varzaneh et al. [12] showed strong statistically significant correlation between the predicted tumor necrosis and calculated image based volumetric necrosis on post-embolization follow-up MRI study (r = 0.889, p < 0.001) and between predicted tumor necrosis and pathological calculated tumor necrosis (r = 0.871, p < 0.001) and this put conclusion that the calculated volume of enhancement on pre-TACE MRI and the post-TACE oil deposition in CT may accurately predict necrosis in treated HCC lesions.
On the other hand, Langenbach et al. [13] stated that the amount of Lipiodol in post-interventional CT did not correlate with post-therapy tumor response, and therefore do not allow sufficient prediction of the tumor response after treatment.
Certain authors recommending the use of MR imaging instead of CT for proper assessment of the post-TACE tumor response. Hunt et al. [14] demonstrated that MRI has more diagnostic accuracy than CT study (55% vs. 43% respectively) in assessment of tumor viability following to TACE of hepatic focal lesion in transplanted patients. As expected, MR imaging is more accurate than CT in detection of the viable tumoral remnants because MRI interpretation is not affected by lipiodol retention. Moreover, the accuracy of MRI study can be enhanced and improved by the use of image subtraction technique.
Nevertheless, the level of evidence supporting these statements remains low, because the limited studies and small sample sizes. Moreover, certain authors have reported a strong correlation between tumor devascularization on MR imaging and the amount of lipiodol retention on CT [15, 16].
However, MR machine is not available or easily accessible as CT, more cost than CT with many limitation and contraindications to MRI (i.e., pace makers or claustrophobia). Thus, CT scan still done routinely in post-TACE patient as follow-up [17, 18].
The study has some limitations. First, the number of tumors included in the study was fairly small. Our inclusion criteria can provide an explanation for this. We only included patients who had undergone a first session of cTACE because we did not want to introduce bias by including previously treated tumors. Due to the fact that the majority of HCC diagnoses were made using imaging criteria, we were also unable to connect local progression and lipiodol retention with tumor grade. In addition, we did not investigate overall survival. Our study’s objective was not to establish a link between lipiodol retention patterns and long-term outcomes, but rather to improve the understanding of the local response of tumors treated with cTACE. Also MRI was not performed for our patients, we recommend further larger studies with comparison between the results of CT and MRI regarding local progression after TACE.
Conclusions
The lipiodol retention pattern of HCCs after TACE can predict the potential local outcome. Tumors with incomplete lipiodol retention are at a high risk of local progression and therefore should probably be retreated; on the other hand, tumors with complete lipiodol retention have a much lower risk of local disease progression.
Availability of data and materials
All the datasets used and analyzed in this study are available with the corresponding author on reasonable request.
Abbreviations
- BCLC:
-
Barcelona clinic liver cancer
- CR:
-
Complete response
- cTACE:
-
Conventional transcatheter arterial chemoembolization
- DEB:
-
Drug-eluting beads
- HCC:
-
Hepatocellular carcinoma
- IRB:
-
Institutional review board
- mRECIST:
-
Modified Response Evaluation Criteria in Solid Tumors
- MRI:
-
Magnetic resonance imaging
- PACS:
-
Picture archiving and communication system
- PD:
-
Progressive disease
- PR:
-
Partial response
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- SD:
-
Stable disease
- TACE:
-
Transcatheter arterial chemoembolization
References
Rashed WM, Kandeil MAM, Mahmoud MO et al (2020) Hepatocellular Carcinoma (HCC) in Egypt: a comprehensive overview. J Egypt Natl Canc Inst 32(1):1–11
Raoul JL, Forner A, Bolondi L, Cheung TT et al (2019) Updated use of TACE for hepatocellular carcinoma treatment: How and when to use it based on clinical evidence. Cancer Treat Rev 72:28–36
Gnutzmann D, Kortes N, Sumkauskaite M et al (2018) Transvascular therapy of hepatocellular carcinoma (HCC), status and developments. Minim Invasive Ther Allied Technol 27(2):69–80
Galle PR, Forner A, Llovet JM et al (2018) EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 69(1):182–236
Dioguardi Burgio M, Garzelli L, Cannella R et al (2023) Hepatocellular carcinoma: optimal radiological evaluation before liver transplantation. Life 13(12):2267
Dioguardi Burgio M, Sartoris R, Libotean C et al (2019) Lipiodol retention pattern after TACE for HCC is a predictor for local progression in lesions with complete response. Cancer Imaging 19:1–9
Osman MF, Farag ASA, Samy HA, El-Baz TM, Elkholy SF (2021) Role of multislice computed tomography 3D volumetric analysis in the assessment of the therapeutic response of hepatocellular carcinoma after transarterial chemoembolization. Egyp J Radiol Nuclear Med 52(1):1–10
Miszczuk MA, Chapiro J, Geschwind JFH et al (2020) Lipiodol as an imaging biomarker of tumor response after conventional transarterial chemoembolization: prospective clinical validation in patients with primary and secondary liver cancer. Transl Oncol 13(3):100742
Dioguardi Burgio M, Ronot M, Bruno O et al (2016) Correlation of tumor response on computed tomography with pathological necrosis in hepatocellular carcinoma treated by chemoembolization before liver transplantation. Liver Transpl 22:1491–1500. https://doi.org/10.1002/lt.24615.[PubMed][CrossRef][GoogleScholar]
Kloeckner R, Otto G, Biesterfeld S, Oberholzer K, Dueber C, Pitton MB (2010) MDCT versus MRI assessment of tumor response after transarterial chemoembolization for the treatment of hepatocellular carcinoma. Cardiovasc Intervent Radiol 33:532–540. https://doi.org/10.1007/s00270-009-9728-y.[PubMed][CrossRef][GoogleScholar]
Stark S, Wang C, Savic LJ et al (2020) Automated feature quantification of Lipiodol as imaging biomarker to predict therapeutic efficacy of conventional transarterial chemoembolization of liver cancer. Sci Rep 10(1):18026
Najmi Varzaneh F, Pandey A, Aliyari Ghasabeh M et al (2018) Prediction of post-TACE necrosis of hepatocellular carcinoma usingvolumetric enhancement on MRI and volumetric oil deposition on CT, with pathological correlation. Eur Radiol 28:3032–3040
Langenbach MC, Vogl TJ, Said G et al (2021) Lipiodol as a predictive indicator for therapy response to transarterial chemoembolization of hepatocellular carcinoma. Cancer Biother Radiopharm. https://doi.org/10.1089/cbr.2020.4137
Hunt SJ, Yu W, Weintraub J et al (2009) Radiologic monitoring of hepatocellular carcinoma tumor viability after transhepatic arterial chemoembolization: estimating the accuracy of contrast-enhanced cross-sectional imaging with histopathologic correlation. J Vasc Interv Radiol 20(1):30–38
Kwan SW, Fidelman N, Ma E et al (2012) Imaging predictors of the response to transarterial chemoembolization in patients with hepatocellular carcinoma: a radiological-pathological correlation. Liver Transpl 18(6):727–736
Shim JH, Han S, Shin YM et al (2013) Optimal measurement modality and method for evaluation of responses to transarterial chemoembolization of hepatocellular carcinoma based on enhancement criteria. J Vasc Interv Radiol 24(3):316–325
Bargellini I, Vignali C, Cioni R et al (2010) Hepatocellular carcinoma: CT for tumor response after transarterial chemoembolization in patients exceeding Milan criteria—selection parameter for liver transplantation. Radiology 255(1):289–300
Schima W, Ba-Ssalamah A, Kurtaran A et al (2007) Post-treatment imaging of liver tumours. Cancer Imaging 7(Special issue A):S28
Acknowledgements
Not applicable.
Funding
Not applicable (no funding).
Author information
Authors and Affiliations
Contributions
MNG performed the CT assessment, statistical analysis and manuscript editing. BEM put the idea of the study, participation in the image analysis and manuscript editing. AMH involved in data collection, CT assessment and performed the statistical analysis. SFE is the editor of the manuscript and involved in participation in data collection and CT assessment. MMS involved in participation in data collection, manuscript editing and the CT analysis. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Written informed consent was signed by all patients before the CT examination. The study is approved by the medical committee of the faculty of medicine Cairo University. Reference no. MS-225-2022.
Consent for publication
All patients included in this research gave written informed consent to publish the data contained within this study.
Competing interests
The authors declare that they have no financial or non-financial competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Grace, M.N., Mahmoud, B.E., Abdel-Latif, A.M.H. et al. The prediction value of lipiodol retention pattern on post-TACE CT scan for local progression of HCC after complete response. Egypt J Radiol Nucl Med 55, 69 (2024). https://doi.org/10.1186/s43055-024-01242-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43055-024-01242-x