Abstract
Background
Hepatocellular carcinoma (HCC) is the first cause of cancer in Egypt. Recently, HCC developed post direct-acting antivirals (DAAs) differ in some characteristics from those developed without DAAs exposure regarding the biological features and behavior of HCC. We aimed to assess the epidemiological, clinical, laboratory, and radiological findings besides the biological behavior of HCC patients post DAAs in comparison to HCC not exposed to DAAs. An analytic cross-sectional research was performed at the National Liver Institute which is a tertiary multidisciplinary HCC center. Subjects included hepatitis C virus patients and were allocated into two groups: group I included 2036 HCC cases post-DAA treatment and group II included 6338 HCC cases who did not receive DAAs. Subjects were examined to evaluate clinical, laboratory, and radiological findings. Tumor staging was done using the BCLC staging system.
Results
Group II showed a more advanced Child–Pugh score, FIB-4 index, and MELD score than Group I (P = 0.001). The multiplicity of hepatic focal lesions was elevated in group I than in group II (P = 0.033). AFP level was significantly elevated in group I than in group II (p = 0.012). Portal vein invasion was significantly elevated in group I than in group II patients (P = 0.001). Extrahepatic spread of HCC was significantly elevated in group I than in group II (P = 0.001). Infiltrative lesions were significantly elevated in group I than in group II (P = 0.002).
Conclusion
Our study detected that the behavior in HCC post DAAs treatment is more aggressive in respect of the number of lesions, PV invasion; local and distant metastasis, and serum AFP level than in patients unexposed to DAAs. Strict surveillance in cirrhotic patients treated with DAA should be followed according to the international guidelines for early diagnosis and treatment of HCC.
Similar content being viewed by others
Background
Hepatocellular carcinoma (HCC) represents the fifth most common cancer and is the second leading cause of cancer-related death. HCC is most frequently diagnosed in established liver cirrhosis cases caused by chronic hepatitis C virus infection (CHC), chronic hepatitis B virus infection (CHB), non-alcoholic steatohepatitis (NASH), or non-alcoholic fatty liver disease (NAFLD), alcohol, and exposure to aflatoxin in the diet [1].
HCC is the most prevalent cancer in Egypt's general population, but it is the second most prevalent cancer among females, behind breast cancer. Egypt had high rates of HCC which may be due to high HCV prevalence post unsafe intravenous antibilharzial injections (tartar emetic). Overall HCV seroprevalence among the 48,345,948 persons tested was 4.61% (95% CI 4.61–4.62) with almost all infections due to genotype 4 (GT4) [2, 3].
Globally, the use of direct-acting antivirals (DAAs) for the treatment of HCV has resulted in elevated rates of sustained viral response (SVR) and reached > 95% in most strains and eligibility to treat cases who were unable to tolerate earlier interferon-based therapies [4]. Fifty million individuals were screened for virus C infection and more than 4 million HCV-infected patients were treated by DAAs in the national program of HCV eradication in Egypt between 2014 and 2020 [5]. There is controversy regarding the associated risk of HCC incidence and recurrence and the behavior of HCC after DAA therapy [6, 7].
We aimed to evaluate the epidemiological, clinical, laboratory, and radiological findings and biological behavior of HCC patients post DAAs in comparison to HCC with no prior exposure to DAAs.
Methods
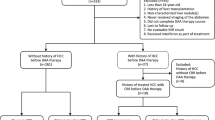
This analytic cross-sectional study of 8374 HCC patients was presented to our multidisciplinary HCC committee (A tertiary center, the HCC unit receives and treats more than 3000 new HCC patients annually and has an interventional radiology unit, transplantation unit; One of the largest HCC units in Middle EAST and Africa) in National Liver Institute from January 2015 till the end of October 2019. The study has been approved by the Institutional Review Board of the National Liver Institute and informed consent was signed by all enrolled patients who were divided into two groups; group I included HCC post-DAA treatment of chronic hepatitis C patients (2036 patients) and group II included HCC patients with chronic hepatitis C infection who did not receive DAA therapy (patients’ preference) (6338 patients). All patients in group I and group II were diagnosed according to the European Association for the Study of Liver Disease (EASL) and American Association for the Study of Liver Diseases (AASLD) updated HCC guidelines and were discovered either accidentally or during routine follow-up investigations [8, 9]. Radiological investigations were done using routine abdominal ultrasonography (US) and duplex Doppler US (to detect vascular invasion) for all patients. Triphasic computed tomography (CT) including (arterial, port venous, and delayed phases) was done for 7704 of 8374 (92%) patients on Biograph MDCT 128, Siemens, Germany. Dynamic magnetic resonance imaging (MRI) was done for 888 of 8374 (10.6%) patients (arterial, portovenous, and delayed phases). MRI was done on a GE 1.5 Tesla scanner (Optima GE 450 w). Ultrasonographic guided Tru-cut liver biopsy was done for 167 of 8374 (2%) patients using 18 G Tru-cut liver biopsy needle for histopathological examination when noninvasive radiological investigations were not conclusive. The patients in our study may need more than one radiological investigation to conclude the diagnosis (Table 1). Radiological investigations were done at the diagnostic medical imaging and interventional radiology department, National Liver Institute, and were read by expert radiologists in the HCC committee.
Patients were diagnosed as having liver cirrhosis when they meet more than one of the following: definite clinical signs and laboratory findings of liver cirrhosis; abdominal ultrasonographic signs suggestive of cirrhosis; or noninvasive scores suggestive of cirrhosis (FIB-4 > 3.25 and APRI > 1.0) [10]. The study was conducted in accordance with the Good Clinical Practice guidelines and the Declaration of Helsinki. Both groups include de novo HCC after ablative therapy (resection, thermal ablation, or ethanol injection). We excluded HCC cases with combined HCV and hepatitis B virus (HBV) infection, cases who developed HCC during antiviral therapy and recurrent HCC cases (all cases were naive), and cases with non-SVR for HCV infection.
Treatment regimens of oral DAAs used were sofosbuvir plus ribavirin, sofosbuvir plus simeprevir, sofosbuvir and daclatasvir, sofosbuvir plus daclatasvir and ribavirin, sofosbuvir and ledipasvir, sofosbuvir plus ledipasvir and ribavirin.
Demographic, performance status, clinical, radiological, and laboratory data of studied patients were analyzed, and histopathological investigations of liver biopsy samples were performed if needed.
Laboratory tests were done after history taking and complete clinical examination. Blood specimens were gathered for complete blood count liver and renal function tests as well as alpha-fetoprotein (AFP). Prothrombin time, concentration, and international normalized ratio (INR) were assessed. HBsAg and anti-HBc IgM and IgG were tested to exclude co-infection. Our study is a comparative one to evaluate the characterization and biological behavior of HCC lesions in the two studied groups. We compared lesions size and number, AFP level, Child–Pugh Score, performance condition by the Eastern Cooperative Oncology Group (ECOG) scale, vascular invasion, lymph node metastasis, and distant metastasis in both groups.
Statistical analysis
The collected data were organized, tabulated, and statistically analyzed using SPSS software (Statistical Package for the Social Sciences, version 21, SPSS Inc. Chicago, IL, USA). For quantitative data, the range, mean, and standard deviation were calculated. For qualitative data, which describes a categorical set of data by frequency, percentage, or proportion of each category, a comparison between two groups and more was done using the Chi-square test (X 2). Significance was adopted at p < 0.05 for interpretation of results of tests of significance.
Results
This study included 8374 patients divided into two groups, group I included 2036 HCC patients post DAAs treatment of chronic hepatitis C, and Group II included 6338 HCC patients with HCV infection not treated with DAAs.
Patients were divided into two groups; group I included 2036 (24.3%) HCC post-DAA treatment of chronic hepatitis C patients,1753(86.1%) males and 283(13.9%) females with mean age ± SD of 58.98 ± 7.67 and group II included 6338 (75.7%) HCC patients with chronic hepatitis C infection who did not receive DAA therapy, 5356 (84.5%) males and 982(15.5%) females with mean age ± SD of 59.62 ± 9.17).
Multiple hepatic focal lesions were more elevated in group I than in group II (p = 0.03). HCC lesion size was insignificantly different. Portal vein invasion was significantly elevated in group I than in group II (P = 0.001). Extrahepatic spread of HCC was significantly elevated in group I than in group II (P = 0.001) (Table 2).
Infiltrative lesions were significantly elevated in group I than in group II (P = 0.002) (Table 2, Fig. 1). Nodular (well-circumscribed) lesions were significantly present more in group II than in group I (P = 0.001) Most lesions in both groups are situated in the right lobe with no significant difference (Table 2, Figs. 1, 2, 3 and 4).
Triphasic CT scan of the abdomen in post-DAA HCC patient shows left lobe (segment III) infiltrative heterogeneously enhanced HCC lesion showing marginal enhancement in the arterial phase (A) with washout in the portovenous and delayed phases with left portal vein invasion and minimal IHBRs dilatation with multiple small scattered right lobe HCC lesions and metastatic Para-aortic and peripancreatic lymphadenopathy (D, E, F)
Triphasic CT scan of the abdomen in HCC patient not received DAA shows right lobe (segment VI) vascular HCC lesion shows enhancement in the arterial phase (A) with washout in the portovenous and delayed phases (B and C) with portal vein invasion of the right posterior branch and malignant IVC thrombosis
Triphasic CT scan of the abdomen in post-DAA HCC patient shows right lobe (segment VI) large exophytic HCC lesion shows heterogenous enhancement in the arterial phase with a large area of central breakdown (A) and washout in the portovenous and delayed phases (B and C) with right renal multiple cortical simple cysts
Regarding Child–Pugh, a group I showed a significantly higher score of Child A than group II (p = 0.002), while Child–Pugh B and C were significantly elevated in group II than group I (P = 0.048 and 0.019, respectively). The model of End Stage Liver Disease (MELD) score in group II was significantly elevated than in group I. FIB-4 has a significantly higher rate of fibrosis index in group II than in group I (p = 0.001). Serum bilirubin was significantly elevated in group II than in group I (P = 0.035), while serum albumin was significantly elevated in group I than group II (P = 0.047). INR was significantly elevated in group II than in group I (p = 0.05). Platelet count did not show significant variance among both groups. AFP level was significantly elevated in group I than in group II (p = 0.012) (Table 3).
Age, sex, and diabetes mellitus (DM) were insignificantly different among both groups. Smokers were more prominent in group I than in group II (p = 0.031). Obese and hypertensive cases were significantly elevated in group II than in group I (p = 0.035 and 0.029, respectively). Ascites and hepatic encephalopathy were significantly elevated in group II than in group I (p = 0.02 and 0.035, respectively). The mean duration between DAA therapy and HCC occurrence was 18.5 ± 8.3 months (Table 4).
As regards treatment regimens of DAAs used were: sofosbuvir plus IFN and ribavirin for 122 patients (6%), sofosbuvir plus ribavirin for 413 patients (20.3%), sofosbuvir plus simeprevir for126 patients (6.2%), sofosbuvir and daclatasvir for 621 patients (30.5%), sofosbuvir plus daclatasvir and ribavirin for 346 patients (17%), sofosbuvir and ledipasvir for 259 patients (12.7%), sofosbuvir plus ledipasvir and ribavirin for 149 patients (7.3%). No significant difference was found between different treatment regimens regarding AFP level the rates of MVI, portal vein invasion, extrahepatic metastasis, grades of differentiation, and the rates of infiltrative lesions (Table 5).
Discussion
Since 2013 Chronic HCV treatment has changed dramatically. DAAs are oral medications with potent antiviral efficacy against persistent HCV infection with high SVR [11]. Controversy around the possible relationship between antiviral treatment and de novo or recurrent HCC and about the behavior of HCC lesions that developed after DAAs. Some studies found increased occurrence or recurrence of HCC post-DAA treatment [12,13,14]. Some studies reported that DAAs are not associated with the increased incidence nor the recurrence of HCC, and referred the high incidence and recurrence to other risk factors such as age, sex, and grade of fibrosis but not due to the treatment itself [15, 16].
Many recent studies reported that HCC in DAAs treated cases observed more aggressive profiles than those who did not receive DAA treatment. These behavioral changes have been registered in some studies regarding HCC occurring post DAA either in new incidence or recurrent lesions [13, 17,18,19].
In our study, there was a significant difference in both groups regarding lesion number, PVI, AFP levels, and tumor metastasis. Also, there was a significant difference regarding the Child–Pugh score, FIB-4 index, and MELD score.
AFP is a fetal-specific glycoprotein, and while its use in the diagnosis of HCC has diminished, it continues to play a role in the monitoring of HCC patients following various treatment modalities and in predicting the prognosis of liver transplantation. In our research, AFP was elevated in group I than in group II, and similar results were observed in other recent studies [17,18,19]. Higher AFP levels are considered independent predictors of aggressive HCC, and lower post-transplant survival and HCC recurrence after LT [20].
The portal vein invasion rate was elevated in group I than in group II, this is also reported in other new studies [17,18,19]. Regarding HCC without portal vein (PV) invasion, HCC with PV invasion is characteristic of disease aggressive course, elevated chance of developing problems associated with portal hypertension presence, and treatment poorer tolerance. Limited outlook and therapeutic options [21].
Local lymph node metastasis and distant metastasis were significantly elevated in group I than in group II. This was also reported in recent studies [17, 19, 22]. Lymph node metastasis and distant metastasis of HCC are associated closely with aggressive tumor behavior, high risk of tumor recurrence, and poor survival [23].
Infiltrative lesions are seen in triphasic CT and dynamic MRI imaging studies in significantly elevated rates in group I than in group II. This was in line with Romano et al., Omar et al., and Khalid et al. as they mentioned that infiltrative lesions were significantly more prominent in HCC patients who received DAAs than those not exposed to DAA treatment [17, 18, 24]. The prognosis for infiltrative HCC cases is poor as upon presentation, the tumor is often quite progressed and frequently coupled with vascular invasion [25]. Multiple hepatic focal lesions were significantly more common in group I than in group II, this is in agreement with similar studies. Multiple hepatic focal lesion is a marker of aggressive behavior of the tumor [17,18,19].
It is still obscure the mechanisms by which aggressive behavior of the HCC lesions occurs. Many theories suggest that; the rapid decline of viral load induced by DAAs leads to a fast reduction in immune surveillance of cancer due to a sudden decrease in natural killer (NK) cells in liver and virus-specific CD4+ and CD8+ T cells and of their cytotoxic activity and innate immunity reconstitution and down-regulation of type II and III interferon, their receptors and IFN-stimulated genes. IFN activation lack might permit malignant cell growth and loss of its anti-proliferative effects, this may be a possible factor encouraging faster HCC foci progression [13, 25, 26]. Also, an increase in certain growth factors and cytokines like vascular endothelial growth factor and endothelial growth factors and downregulation of cytotoxic T lymphocytes may have a function [27]. Tumor dormant micronodules potentially present might find ideal conditions to become apparent and to grow. Thus, after treatment with DAA, the transitory immunosuppressive phase and tumor cell presence with highly aggressive conduct might be a factor in the fast development of tumors in these cases [28].
Limitations: it is a one-center study; we need larger multicenter studies in different countries to confirm our findings.
Conclusion
Our study showed aggressive behavior in HCC post DAAs treatment regarding the number of lesions, PV invasion, local and distant metastasis, and serum AFP level more than in HCC patients not treated with DAAs. Strict surveillance in cirrhotic cases who received DAA should be done according to the international guidelines for HCC early diagnosis and management.
Availability of data and materials
The datasets used and analyzed during the current study are available upon reasonable request from the first author Mohamed Elgazzar.
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- DAAs:
-
Direct-acting antivirals
- AFP:
-
Alfa fetoprotein
- CHC:
-
Chronic hepatitis C virus infection
- CHB:
-
Chronic hepatitis B virus infection
- NASH:
-
Non-alcoholic steatohepatitis
- NAFLD:
-
Non-alcoholic fatty liver disease
- GT4:
-
Genotype 4
- EASL:
-
European Association for the study of liver disease
- AASLD:
-
American Association for the Study of liver diseases
- CT:
-
Triphasic computed tomography scan
- MRI:
-
Magnetic resonance imaging
- HBV:
-
Hepatitis B virus
- Non-SVR:
-
Not achieved sustained viral response for HCV infection
- IFN:
-
Interferon
- MELD:
-
Model of end stage liver disease score
- DM:
-
Diabetes mellitus
- PV:
-
Portal vein
References
Liu Z, Jiang Y, Yuan H, Fang Q, Cai N, Suo C et al (2019) The trends in incidence of primary liver cancer caused by specific etiologies: results from the Global Burden of Disease Study 2016 and implications for liver cancer prevention. J Hepatol 70:674–683. https://doi.org/10.1016/j.jhep.2018.12.001
Ibrahim AS, Mikhail NNH (2015) The evolution of cancer registration in Egypt: from proportions to population-based incidence rates. SECI Oncol J 3:23–43. https://doi.org/10.21608/secioj.2015.5678
Chhatwal J, Chen Q, Ayer T, Bethea ED, Kanwal F, Kowdley KV et al (2018) Hepatitis C virus re-treatment in the era of direct-acting antivirals: projections in the USA. Aliment Pharmacol Ther 47:1023–1031. https://doi.org/10.1111/apt.14527
Waked I, Esmat G, Elsharkawy A, El-Serafy M, Abdel-Razek W, Ghalab R et al (2020) Screening and treatment program to eliminate Hepatitis C in Egypt. N Engl J Med 382:1166–1174. https://doi.org/10.1056/NEJMsr1912628
Hassanin A, Kamel S, Waked I, Fort M (2021) Egypt’s ambitious strategy to eliminate Hepatitis C virus: a case study. Glob Health Sci Pract 9:187–200. https://doi.org/10.9745/ghsp-d-20-00234
Kozbial K, Moser S, Schwarzer R, Laferl H, Al-Zoairy R, Stauber R et al (2016) Unexpected high incidence of hepatocellular carcinoma in cirrhotic patients with sustained virologic response following interferon-free direct-acting antiviral treatment. J Hepatol 65:856–858. https://doi.org/10.1016/j.jhep.2016.06.009
Ravi S, Axley P, Jones D, Kodali S, Simpson H, McGuire BM et al (2017) Unusually high rates of hepatocellular carcinoma after treatment with direct-acting antiviral therapy for Hepatitis C related cirrhosis. Gastroenterology 152:911–912. https://doi.org/10.1053/j.gastro.2016.12.021
European Association for the Study of the Liver (2018) EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 69:182–236. https://doi.org/10.1016/j.jhep.2018.03.019
Heimbach JK, Kulik LM, Finn RS, Sirlin CB, Abecassis MM, Roberts LR et al (2018) AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 67:358–380. https://doi.org/10.1002/hep.29086
Aubé C, Bazeries P, Lebigot J, Cartier V, Boursier J (2017) Liver fibrosis, cirrhosis, and cirrhosis-related nodules: imaging diagnosis and surveillance. Diagn Interv Imaging 98:455–468. https://doi.org/10.1016/j.diii.2017.03.003
Foerster F, Galle PR (2019) Comparison of the current international guidelines on the management of HCC. JHEP Rep 1:114–119. https://doi.org/10.1016/j.jhepr.2019.04.005
Falade-Nwulia O, Suarez-Cuervo C, Nelson DR, Fried MW, Segal JB, Sulkowski MS (2017) Oral direct-acting agent therapy for Hepatitis C virus infection: a systematic review. Ann Intern Med 166:637–648. https://doi.org/10.7326/m16-2575
Reig M, Mariño Z, Perelló C, Iñarrairaegui M, Ribeiro A, Lens S et al (2016) Unexpected high rate of early tumor recurrence in patients with HCV-related HCC undergoing interferon-free therapy. J Hepatol 65:719–726. https://doi.org/10.1016/j.jhep.2016.04.008
El Kassas M, Funk AL, Salaheldin M, Shimakawa Y, Eltabbakh M, Jean K et al (2018) Increased recurrence rates of hepatocellular carcinoma after DAA therapy in a hepatitis C-infected Egyptian cohort: a comparative analysis. J Viral Hepat 25:623–630. https://doi.org/10.1111/jvh.12854
Waziry R, Hajarizadeh B, Grebely J, Amin J, Law M, Danta M et al (2017) Hepatocellular carcinoma risk following direct-acting antiviral HCV therapy: a systematic review, meta-analyses, and meta-regression. J Hepatol 67:1204–1212. https://doi.org/10.1016/j.jhep.2017.07.025
Ide T, Koga H, Nakano M, Hashimoto S, Yatsuhashi H, Higuchi N et al (2019) Direct-acting antiviral agents do not increase the incidence of hepatocellular carcinoma development: a prospective, multicenter study. Hepatol Int 13:293–301. https://doi.org/10.1007/s12072-019-09939-2
Abdelaziz AO, Nabil MM, Abdelmaksoud AH, Shousha HI, Hashem MB, Hassan EM et al (2019) Tumor behavior of hepatocellular carcinoma after hepatitis C treatment by direct-acting antivirals: comparative analysis with non-direct-acting antivirals-treated patients. Eur J Gastroenterol Hepatol 31:75–79. https://doi.org/10.1097/meg.0000000000001264
Khalid J, Umar M, Ur-Rehman T, Ali M, Khan GM (2020) Tumor aggression among hepatitis-C related hepatocellular carcinoma patients: an observational study regarding the impact of anti-HCV therapy. Infect Agent Cancer 15:35. https://doi.org/10.1186/s13027-020-00300-z
El Fayoumie M, Abdelhady M, Gawish A, Hantour U, Abdelkhaleek I, Abdelraheem M et al (2020) Changing patterns of hepatocellular carcinoma after treatment with direct antiviral agents. Gastrointest Tumors 7:50–60. https://doi.org/10.1159/000505326
Cillo U, Giuliani T, Polacco M, Herrero Manley LM, Crivellari G, Vitale A (2016) Prediction of hepatocellular carcinoma biological behavior in patient selection for liver transplantation. World J Gastroenterol 22:232–252. https://doi.org/10.3748/wjg.v22.i1.232
Chan SL, Chong CC, Chan AW, Poon DM, Chok KS (2016) Management of hepatocellular carcinoma with portal vein tumor thrombosis: review and update at 2016. World J Gastroenterol 22:7289–7300. https://doi.org/10.3748/wjg.v22.i32.7289
Fouad M, El Kassas M, Ahmed E, El Sheemy R (2021) Tumor characteristics of hepatocellular carcinoma after direct-acting antiviral treatment for hepatitis C: comparative analysis with antiviral therapy-naive patients. World J Hepatol 13:1743–1752. https://doi.org/10.4254/wjh.v13.i11.1743
Xia F, Wu L, Lau WY, Li G, Huan H, Qian C et al (2014) Positive lymph node metastasis has a marked impact on the long-term survival of patients with hepatocellular carcinoma with extrahepatic metastasis. PLoS One 9:e95889. https://doi.org/10.1371/journal.pone.0095889
Romano A, Capra F, Piovesan S, Chemello L, Cavalletto L, Anastassopulos G et al (2016) Incidence and pattern of" de novo" hepatocellular carcinoma in HCV patients treated with oral DAAs. Hepatology 64:10–10
Musa NI, Mohamed IE, Abohalima AS et al (2020) Impact of treating chronic hepatitis C infection with direct-acting antivirals on the risk of hepatocellular carcinoma recurrence. Egypt Liver J 10:26. https://doi.org/10.1186/s43066-020-00035-x
Debes JD, van Tilborg M, Groothuismink ZMA, Hansen BE, Schulze Zur Wiesch J, von Felden J et al (2018) Levels of cytokines in serum associate with development of hepatocellular carcinoma in patients with HCV infection treated with direct-acting antivirals. Gastroenterology 154:515-517.e513. https://doi.org/10.1053/j.gastro.2017.10.035
Kanda T, Matsuoka S, Moriyama M (2018) Early occurrence and recurrence of hepatocellular carcinoma in hepatitis C virus-infected patients after sustained virological response. Hepatol Int 12:90–93. https://doi.org/10.1007/s12072-018-9862-1
Sanduzzi-Zamparelli M, Boix L, Leal C, Reig M (2019) Hepatocellular carcinoma recurrence in HCV patients treated with direct antiviral agents. Viruses. https://doi.org/10.3390/v11050406
Acknowledgements
Many thanks to all the staff members who helped us to finish our study.
Funding
None to be declared.
Author information
Authors and Affiliations
Contributions
All authors co-operated in work design, creation of data, detection of resources, data interpretation, validation, and methodology, writing draft and revision. All authors provided comments on the manuscript at various stages. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
It was approved by the Institutional Review Board of the National Liver Institute (IRB number 00525), Menoufia University. An informed written consent was obtained from the participants. The study was conducted from January 2015 till the end of October 2019.
Consent for publication
All patients included in this study gave informed consent to publish the data contained within this study.
Competing interests
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elgazzar, M., Salman, T., Abdelsameea, E. et al. Hepatocellular carcinoma in hepatitis C virus patients treated with direct acting antivirals (DAAs) and patients not exposed to DAAs: a large center comparative study. Egypt J Radiol Nucl Med 55, 88 (2024). https://doi.org/10.1186/s43055-024-01249-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43055-024-01249-4