Abstract
Background
Adenosine signaling is now an accepted explanation for the therapeutic mechanism of Methotrexate (MTX) in rheumatoid arthritis (RA). Adenosine receptors categorized into four subclasses: adenosine A1 receptor (ADORA1), adenosine 2a receptor (ADORA2a), adenosine 2b receptor (ADORA2B), and adenosine 3 receptor (ADORA3). Our aim is to check the mRNA expression of two adenosine receptors; ADORA2a and ADORA3 in whole blood cell of RA patients and its relation in prediction of MTX clinical response in Egyptian patients.
Results
There was significant correlation between both ADORA2a and ADORA3 gene expression in RA patients as compared with healthy controls. The expression of ADORA2a and ADORA3 was increased in good and moderate response groups compared to no response group. There was significant correlation between both genes in mRNA expression before and after MTX treatment. Matrix metalloproteinase-3 (MMP3) concentration was significantly decreased after treatment in good and moderate response groups in comparison to non-responder group.
Conclusion
The inflammatory and clinical responses in RA patients which is demonstrated by DAS28 and suppression of MMP3 were regulated by ADORA2a and ADORA3. Their level of expression can predict MTX response and their agonists may offer a novel and effective therapeutic option for RA patients.
Similar content being viewed by others
Background
RA is a chronic autoimmune disease characterized by a diffuse and progressive degeneration of joints with synovial proliferation and secretion of high levels of pro-inflammatory mediators as cytokines and growth factors [1].
Early diagnosis and treatment are very important to prevent joint loss and functional disability. Treatment depends mainly on disease modifying anti-rheumatic drug (DMARDs) with or without biological drugs according to patient response to treatment [2]. Despite of the major progress in the treatment of (RA) and the availability of new biologics and targeted therapies still the initial therapy of patients with RA is MTX [3].
It is the drug of choice for RA and is considered as a first line in nearly all patients. The mechanism of action of MTX has been studied throughout the years as an inhibitor of novo synthesis of purines and pyrimidines and proliferation of many different types of cells including malignant and non-malignant cells and that was the postulated mechanism by which suppression of inflammation occurred in RA. Inhibition of transmethylation reactions was another proposed mechanism of action that was the basis for clinical testing of a direct trans-methylation inhibitor, 3-deazaadenosine, as a therapy for RA; however, results proved that it has no effect on the course of RA, now well accepted that one of its main mechanism of action is to increase the extracellular adenosine levels which acts via adenosine receptors expressed on inflammatory cells to suppress inflammation. Adenosine receptors are G-protein coupled receptors categorized into four subclasses: (ADORA1), (ADORA2a), (ADORA2b), and (ADORA3). The adenosine receptors, ADORA2a and ADORA2b are associated with Gs protein inducing cAMP production while ADORA1 and ADORA3 with Gi protein which inhibits cAMP production [4]. It is well known that high concentrations of adenosine are generated from the hydrolysis of ATP in any inflammatory or traumatic condition, adenosine behaves as a “retaliatory metabolite” that has an anti-inflammatory and immunomodulatory properties. Demonstrating this mechanism is clinically difficult because of the rapid reuptake of adenosine by cells and its conversion to inosine by adenosine deaminase present in plasma [5, 6].
Studying the level of adenosine and mRNA expression of adenosine receptors in RA and its relation to MTX therapy become a field of interest in many studies [7,8,9].
Explanation of the variety in expression of ADORA subtypes may be strongly related to the affected tissue [10]. ADORA3 is found in peripheral blood mononuclear cells (PBMNC) of patients with autoimmune inflammatory diseases, such as RA, psoriasis, and Crohn’s disease, while stimulation of ADORA2a and ADORA3 mediated a significant decrease of IL-1b or IL-6 release in RA patients [4, 11].
In our study, we checked baseline ADORA2a and ADORA3 gene expression in whole blood of Egyptian RA patients on MTX therapy and its role in prediction of its therapeutic effect.
Methods
Inclusion criteria
Sixty-five newly diagnosed RA patients by fulfilling the American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2010 classification criteria and 50 healthy controls matched for age and sex were included in the study [12].
All patients were MTX naïve and not under any other (DMARDs ) with disease activity using Disease Activity Score 28 (DAS28) > 3.2. All patients started a weekly 10 mg MTX, the dose was increased every 2 weeks by 2.5 mg till they achieved DAS28 < 2.6, or reached a maximum dose with upper limit of 25 mg/week.
Patients were classified according to EULAR criteria for response to good, moderate, and non-responders [13].
Exclusion criteria
Patients with any contraindications to MTX therapy, e.g., chronic liver or renal disease, pregnancy, and lactation were excluded from the study.
Ethical consideration
The study was approved by scientific and ethics committees in accordance with the principles of Declaration of Helsinki II. All procedures were explained to RA patients and control subjects before having their written informed consents to participate in this study.
All patients and controls were assessed at the start and after 6 months of treatment by:
-
1-
Clinical assessment
Measurement of disease activity using DAS28 [6].
-
2-
Laboratory assessment
Biochemical assay:
-
1-
Erythrocyte sedimentation rate (ESR in mm/h) was determined using the Westergren method.
-
2-
Serum C-reactive protein (CRP) and rheumatoid factor using nephlometry principle by Beckman coulter AU 480.
-
3-
Anti-cyclic citrullinated peptide antibodies were measured using commercial ELISA plates coated with second generation citrullinated peptides (ELISA, Quanta Lite CCP version 3.1 for IgG/IgA from Inova Diagnostics, San Diego, CA).
-
4-
Complete blood count using SIEMENNS ADVIA 2120I.
-
5-
Liver function tests and kidney function tests: using Beckman coulter AU 480.
-
6-
Serum MMP3 measurement: using quantitative ELIZA sandwich assay, R&D systems. Catalog # DMP300, sensitivity 0.045 ng/ml, assay range 0.2–10 ng/ml. CV% of intra-assay and inter-assay precision is 5.7 and 7.9 respectively. Average % recovery is 90%.
-
7-
Genetic assessment of Adenosine receptor quantification using RT-qPCR
-
8-
Genetic analysis of ADORA2a and ADORA3 mRNA expression. Two milliliters of blood in EDTA tubes obtained from peripheral blood of all subjects and RNA was obtained from samples using QIAamp RNA Blood Mini (QIAGEN GmbH, Hilden, Germany).
All the RNA samples had an optical density 260/280 ratio ranging from 1.81 to 1.875, which confirms the good quality of the RNA. RNA samples with an optical density 260/280 ratio of less than 1.8 were excluded from the study.
The cDNA was reverse transcribed in a 10 μl mixture containing 6 μl of total RNA, 0.5 μl random primers (Promega, Madison, WI), 2 μl of reverse transcriptase buffer (Life Technology, Gaithersburg, MD), 1 μl of deoxyribonucleosid triphosphate (dNTP) mixtures (10 Mm), and 0.5 μl of AMV reverse transcriptase (5 U/μl).
The mixture was incubated at 37 °C for 10 min, 52 °C for 45 min, 95 °C for 5 min, and in ice bath for 5 min. The synthesized cDNA samples were stored at – 20 °C until use [14].
The cDNA is used as a template to amplify the studied genes and it normalized using glyceraldehydes-3-phosphate dehydrogenase ( GAPDH) as housekeeping gene.
The assay identification numbers of target and housekeeping genes are as followed: ADORA2a (Assay ID # Hs00169123_ml) , ADORA3 (Assay ID Hs 04194761_ ml)
and Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) gene (Assay ID # Hs02786624_g1) using the primers and Probes assays listed in TaqMan Gene expression assays from Applied Biosystems.
DNA amplifications of the ADORA2a and ADORA3 genes were carried out by real time PCR using the real time PCR Step One instrument and software (Applied Biosystems). This included an initial denaturation step for 10 min at 95 °C, followed by 40 cycles of annealing and elongation for 15 s at 95 °C and 1 min at 60 °C, respectively.
Instrument raw data (fluorescence) for all the samples were converted into threshold cycles (Ct) using SDS 1.2 software (Applied Biosystems). ΔCt values were then imported onto an Excel worksheet for relative quantification (RQ) or fold change.
For RQ calculation, the geometrical mean of GAPDH was used as a normalization factor. Calculation of the results was performed by application of the comparative Ct method for RQ = 2−∆∆Ct
∆ Ct; unknown= Ct target-Ct reference
The difference in threshold cycles for target and reference genes is as follows: ∆∆Ct = Ct unknown-Ct calibrator. Where, ∆Ct, calibrator is the mean value for a healthy control human.
RQ = 2−∆∆Ct the fold change, defined as the ratio between the averaged normalized expression levels of targets in patients and corresponding control samples, was calculated.
Normalized RQ values were log 2 transformed for statistical analysis.
Statistical analysis
Data were analyzed using IBM SPSS software package version 20.0. (Armonk, NY: IBM Corp.). The Kolmogorov- Smirnov was used to verify the normality of distribution of variables.
Difference at baseline levels of adenosine receptors between patient group and healthy control group were assessed using Mann–Whitney U test.
Spearman coefficient was used to correlate between quantitative variables (age, duration of disease, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR) and baseline DAS28 at the start with ADORA2a and ADORA3 RQ at baseline. It was also used to correlate between ADORA2a RQ vs. ADORA3 RQ in patients group at baseline and after treatment.
Student’s t test was used for normally distributed quantitative variables, to compare between patient and control group regarding MMP3.
Paired t test was used for normally distributed quantitative variables, to compare MMP3 at baseline and after treatment.
Kruskal-Wallis test was used to compare between good responders (GR), moderate responders (MR), and non-responders (NR) patient groups response to treatment and different parameters of disease activity (CRP, DAS28, MMP) comparison of patient group response with ADORA2a RQ, ADORA3 RQ, and MMP
Wilcoxon signed-ranks test was assessed for comparison between baseline and follow-up for not normally distributed quantitative variables.
Receiver operating characteristic curve (ROC) was used to determine the diagnostic performance of ADORA2a and ADORA3 gene expression, and area that is more than 50% gives acceptable performance and area about 100% is the best performance for the test. Significance of the obtained results was judged at the 5% level. All data are expressed as means.
Results
Patients characteristics at baseline time and after 6 months of MTX treatment 65 patients (63 females and 2 males) were enrolled in the study with mean age of 41.8± 6.6 years, mean disease duration of 15.2± 4.2 months and mean DAS28 4.4 ± 1.2
Baseline level of adenosine receptors in whole blood RNA of patients
The mean delta CT for the expression levels of ADORA2a and ADORA3 in 65 patients, respectively, were – 3 ± 1 and − 3.4 ± 1.2 with GAPDH as reference gene. In the control group mean expression was − 0.5 ± 0.3 for ADORA2a and − 0.6 ± 0.2 for ADORA3. Significant difference was observed between patients and control groups for both gene expression (p < 0.001). The mRNA expression levels of ADORA2a and ADORA3 were upregulated 6 months after MTX therapy (Fig. 1) and (Tables 1 and 2). There was significant difference between MMP3 concentration between patients and control groups.
There was significant correlation between both ADORA2a and ADORA3 gene expression in patients group (Table 3)
The expression levels of ADORA2a and ADORA3 were negatively correlated with baseline DAS28, ESR, CRP, and RF, and there was no correlation between gene expression with age, duration of disease, and ACPA (Table 4).
After 6 months of MTX therapy
Most patients improved with MTX therapy. Out of 65 patients, 32 (49%) were classified as GR, 25 (38%) as MR, and 8 (12%) as NR to MTX (Table 5) showed significant difference between 3 groups regarding DAS28 and CRP. The mean mRNA expression of ADORA2a at baseline was 3.4 and that was up regulated to 6.6 at the follow up in GR, and to 3.6 in MR and down regulated to 1.5 in NR. The mean mRNA expression of ADORA3 at baseline was 4.3 which was up regulated to 8.5 at the follow-up in GR, 4.5 in MR and downregulated to 1.5 in NR. The expression of ADORA2a and ADORA3 was not increased in NR as compared to GR and MR. As shown in Table 5 and Fig. 2, MMP3 concentration was significantly decreased after treatment in GR and MR groups in comparison to NR group.
Table 6 shows ADORA2a mRNA expression cut-of value of less than 1.4 (RQ), with a sensitivity of 75.5% and specificity of 91.2% predicts non-response to MTX The positive predictive value is 54.5% and the negative predictive value is 96.3.
ADORA3 mRNA expression cut-off value of less than 1.6 (RQ), with a sensitivity of 62.5% and specificity of 96.5% predicts non-response to MTX. The positive predictive value is 71.4% and the negative predictive value is 94.8%.
Discussion
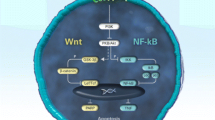
In the management of RA, rapid and effective disease control is the main goal and MTX is frequently the first step in the long-term management. Unfortunately, the response to MTX therapy is not predictable which mandate the study of its possible effects at the pro-inflammatory cytokine level [10]. MTX inhibits inflammation by increasing the extracellular levels of adenosine which stimulates ADORA2a and ADORA3 receptors [15]. ADORA3 overexpression in RA patients induced by pro inflammatory cytokines is able to decrease NF-kB stimulation. ADORA2a and ADORA3 overexpression inhibit NF-ĸB pathway with subsequent decrease in inflammatory cytokines [16].
NF-ĸB regulates genes that encode cytokines, like TNF-a, IL-1b, IL-6, and other inflammatory factors, so this transcription factor could be one of the main regulators of cytokine invention in RA [17]. TNF alpha may be principally involved in the pathogenesis of cartilage destruction. There is considerable attention in developing treatments decreasing NF-kB activation and cytokine production in RA through the involvement of ADOR2A and ADORA3 [18]. Proteases belong to MMP class enzymes mediate the damage of joint cartilage which is a distinctive feature of arthritis like RA. MMP-1, and MMP-3 are of certain interest as they directly destroy the cartilage matrix components, including aggrecan and collagen [19]. NF-kB expression is linked to MMP-1 and MMP-3 concentration proceeding to cartilage damage and articular destruction which in turn explains why adenosine receptor stimulation has an inhibitory effect of on MMP-1 and MMP-3 expression in synovial fibroblasts [20,21,22,23].
The clinical response of MTX is directly linked to its ability to stimulate the expression of adenosine receptors and measuring their gene expression in rheumatoid patients can help in prediction of patient’s response to treatment.
Our results show increasing both ADORA2a and ADORA3 gene expression in newly diagnosed RA patients in comparison to healthy control with a negative correlation between RA disease activity evaluated with DAS28 and expression of both genes. This is in agreement with Varani et al. 2009 [24] who reported ADORA2a and ADORA3 upregulation in lymphocyte and PBMNC membranes from patients with RA as compared to healthy controls.
We examined the mRNA expression of ADORA2a and ADORA3 and its impact on RA disease activity in our patients to predict clinical response at 6 months of MTX therapy. Forty-eight percent of patients were GR with significant increase in ADORA2a from a median of – 3. 3 to 7 and ADORA3 from – 3.4 to 8.5. It has been reported that the presence of a relationship between DAS28 and serum adenosine deaminase (ADA) levels, which subsequently stimulate adenosine receptors, suggests that ADA concentration may predict disease activity in RA patients Zamani 2012 [25]. Many factors including receptor expression, receptor sensitivity and extra-cellular concentration of ligand affect ADORA signaling. Greatest expression of ADOR3 was shown in rheumatoid synovia and control patient response to MTX therapy according to Lisa Stamp et al. 2012 [10].
Varani et al. 2011 [4] also reported inverse correlation between DAS28 and ADORA2a or ADORA3 density, confirming the role of adenosine and its receptors in control of inflammation in RA. They also suggested the use of ADORA2a and/or ADORA3 agonists as novel potential pharmacological treatment combined with the classical therapy in human diseases characterized by a marked inflammatory component as in RA. This can explain significant correlation between ADORA2a and ADORA3 with ESR, DAS28, and CRP.
Our results also showed upregulation of ADORA2a and ADORA3 after 6 months of MTX therapy in MR with lower expression in NR, these results are in agreement with Ochaion et al. 2006 [26] who reported increase in protein level of ADORA3 on 10 weeks of MTX treatment. Varani et al. 2011 also reported upregulation of ADORA2a and ADORA3 after MTX therapy.
Ankita Singh 2019 [27] reported a higher basal expression of ADORA3 and 2a in the whole peripheral blood of RA patients at the beginning of MTX monotherapy. Low expression of ADORA3 mRNA in blood was associated with non-response to MTX and increased expression was associated in moderate and GR. ADORA2a signaling is valuable in decreasing inflammation and may be the signaling pathway of MTX action in treatment of RA [27].
In another cohort using synovial fluid samples, protein level of ADORA2a and ADORA2b but not ADORA3, was more in patients on MTX in comparison with patients not on MTX. So, MTX exerted significant anti-inflammatory role at the main area of rheumatoid inflammation [10].
Many studies magnified the role of adenosine through which MTX acts. ADORA2a may start developing of T regulatory cells (Tregs) so decreasing T cell stimulation [28]. On the other hand, Huang, et al. and Bettelli reported that adenosine decreases T cell initiation, production, and apoptosis, so Tregs act as immunosuppressive agent [29,30,31,32].
ADORA2a is suggested to mediate adenosine clinical action, and ADORA2a suppresses recruitment, adhesion, and formation of superoxide in neutrophils. In macrophages, ADORA2a decreases pro-inflammatory cytokine formation, starts the change from pro to anti-inflammatory response, and stops bone damage by decreasing osteoclast formation. ADORA3 signaling stimulate neutrophil chemotaxis to the inflammation area, and suppression of certain cytokines in macrophages [6].
Our results also revealed significant decrease in serum concentration of MMP3 after 6 months of MTX therapy in comparison with its level before therapy. This is in agreement with Varani et al. 2011 who reported that ADORA2a and ADORA3 stimulation showed a significant decrease in MMP-1 and MMP-3 release in RA patients signifying the role of adenosine in the inhibition of inflammation and cartilage destruction by MMP.
Conclusions
Our results confirm previous reports signifying the role of adenosine receptors 2a and 3 gene expressions as predictors distinguishing RA patients’ response to MTX therapy in Egyptian patients.
Our study suggests a correlation of ADORA2a and ADORA3 up regulation with MTX treatment response in RA and that their increased mRNA can somehow affect the inflammatory pathway. In focus, the upregulation of both receptors is related to improvement of inflammatory markers, MMP3, and DAS28, so stimulation of both receptors lowers the disease activity through the suppression of cellular proinflammatory and degenerative molecules.
However, our result requires more validation in larger patient sample to assess the utility of estimated cut-of value because some overlap between responders and NR was noticed.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ADORA1:
-
Adenosine A1 receptor
- ADORA2a:
-
Adenosine 2a receptor
- ADORA2B:
-
Adenosine 2b receptor
- ADORA3:
-
Adenosine 3 receptor
- MTX:
-
Methotrexate
- RA:
-
Rheumatoid arthritis
- DMARDs:
-
Disease modifying anti-rheumatic drugs
- DAS28:
-
Disease Activity Score 28
- MMP3:
-
Matrix metalloproteinase-3
- PBMNC:
-
Peripheral blood mononuclear cells
- ESR:
-
Erythrocyte sedimentation rate
- CRP:
-
C-reactive protein
- RQ:
-
Relative quantification
- CT:
-
Threshold cycles
- Tregs:
-
T regulatory cells
- GR:
-
Good responders
- MR:
-
Moderate responders
- NR:
-
Non-responders
References
Pal Y, Bandyopadhyay N, Pal RS et al (2019) Perspective and potential of A2A and A3 adenosine receptors as therapeutic targets for the treatment of rheumatoid arthritis. Curr Pharm Des 25:2859–2874
Ishaq M, Muhammad JS, Hameed K et al (2011) Leflunomide or methotrexate? Comparison of clinical efficacy and safety in low socioeconomic rheumatoid arthritis patients. Mod Rheumatol 21:375–380
El Miedany Y, Abu-Zaid MH, El Gaafary M et al (2021) Egyptian consensus on treat-to-target approach for osteoporosis: a clinical practice guideline from the Egyptian Academy of bone health and metabolic bone diseases. Egypt Rheumatol Rehabil 48:5. https://doi.org/10.1186/s43166-020-00056-9
Varani K, Padovan M, Vincenzi F et al (2011) A 2A and A 3 adenosine receptor expression in rheumatoid arthritis: up regulation, inverse correlation with disease activity score and suppression of inflammatory cytokine and metalloproteinase release. Arthritis Res Ther 13:R197
Cronstein BN, Aune TM (2020) Methotrexate and its mechanisms of action in inflammatory arthritis. Nat Rev Rheumatol 16:145–154
Cronstein BN, Sitkovsky M (2017) Adenosine and adenosine receptors in the pathogenesis and treatment of rheumatic diseases. Nat Rev Rheumatol 13:41
Silverman MH, Strand V, Markovits D et al (2008) Clinical evidence for utilization of the A3 adenosine receptor as a target to treat rheumatoid arthritis: data from a phase II clinical trial. J Rheumatol 35:41–48
Kokkonen H, Söderström I, Rocklöv J, et, al. (2010) Up-regulation of cytokines and chemokines predates the onset of rheumatoid arthritis. Arthritis Rheum 62:383–391
Csòka B, Haskò G (2011) Adenosine, inflammation pathways and therapeutic challenges. Joint Bone Spine 78:4–6
Stamp LK, Jody H, Rebecca LR et al (2012) Adenosine receptor expression in rheumatoid synovium: a basis for methotrexate action. Arth Res Ther 14:R138
Ochaion A, Bar-Yehuda S, Cohen S, Barer F et al (2009) The anti inflammatory target A3 adenosine receptor is over-expressed in rheumatoid arthritis, psoriasis and Crohn’s disease. Cell Immunol 258:115–122
Aletaha D, Neogi T, Silman AJ et al (2010) The 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League against rheumatism collaborative initiative. Ann Rheum Dis 62(9):2569–2581
Prevoo ML, Van’t Hof MA, Kuper HH et al (1995) Modified disease activity scores that include twenty-eight–joint counts: development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 38:44–48
Mabrouk MM, Medhat A et al (2016) Evaluation of the diagnostic value of serum cytokeratin-8 as a marker of liver injury in chronic hepatitis C patients. Br J Med Med Res 12(4):1–13
Montesinos M, Desai A, Delano D et al (2003) Adenosine A2A or A3 receptors are required for inhibition of inflammation by methotrexate and its analogue MX-68. Arthritis Rheum 48:240–247
Madi L, Cohen S, Ochayon A et al (2007) Overexpression of A3 adenosine receptor in peripheral blood Mononuclear cells in rheumatoid arthritis: involvement of nuclear factor κB in mediating receptor level. J Rheumatol 34:20–26
Lee YR, Kweon SH, Kwon KB et al (2009) Inhibition of IL1beta-mediated inflammatory responses by the I kappa B alpha super repressor in human fibroblast-like synoviocytes. Biochem Biophys Res Commun 378:90–94
Haskó G, Linden J, Cronstein B et al (2008) Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat Rev Drug Discov 7:759–770
Yoshihara Y, Nakamura H, Obata K et al (2000) Matrix metalloproteinases and tissue inhibitors of metalloproteinases in synovial fluids from patients with rheumatoid arthritis or osteoarthritis. Ann Rheum Dis 59:455–461
Ishikawa T, Nishigaki F, Miyata S et al (2005) Prevention of progressive joint destruction in collagen-induced arthritis in rats by a novel matrix metalloproteinase inhibitor, FR255031. Br J Pharmacol 144:133–143
Andreas K, Lübke C, Häupl T et al (2008) Key regulatory molecules of cartilage destruction in rheumatoid arthritis: an in vitro study. Arthritis Res Ther 10:R9
Boyle DL, Han Z, Rutter JL et al (1997) Posttranscriptional regulation of collagenase-1 gene expression in synoviocytes by adenosine receptor stimulation. Arthritis Rheum 40:1772–1779
Noh EM, Youn HJ, Jung SH et al (2010) Cordycepin inhibits TPA-induced matrix metalloproteinase-9 expression by suppressing the MAPK/AP-1 pathway in MCF-7 human breast cancer cells. Int J Mol Med 25:255–260
Varani K, Massara A, Vincenzi F et al (2009) Normalization of A2A and A3 adenosine receptor up-regulation in rheumatoid arthritis patients by treatment with anti–tumor necrosis factor α but not methotrexate. Arthritis Rheum 60:2880–2891
Zamani B, Jamali R, Jamali R (2012) Serum adenosine deaminase may predict disease activity in rheumatoid arthritis. Rheumatol. Int 32(7):1967–1975
Ochaion A, Bar-Yehuda S, Cohn S et al (2006) Methotrexate enhances the anti-inflammatory effect of CF101 via up-regulation of the A 3 adenosine receptor expression. Arthritis Res Ther 8:R169
Singh A, Misra R, Aggarwal A (2019) Baseline adenosine receptor mRNA expression in blood as predictor of response to methotrexate therapy in patients with rheumatoid arthritis. Rheumatol Int 39:1431–1438
Brown PM, Pratt AG, Isaacs JD (2016) Mechanism of action of methotrexate in rheumatoid arthritis, and the search for biomarkers. Nat Rev Rheumatol 12:731
Huang S, Apasov S, Koshiba M et al (1997) Role of A2a extracellular adenosine receptor-mediated signaling in adenosine-mediated inhibition of T-cell activation and expansion. Blood 90:1600–1610
Bettelli E, Carrier Y, Gao W et al (2006) Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441:235–238
Alam MS, Cavanaugh C, Pereira M et al (2020) Susceptibility of aging mice to listeriosis: role of anti-inflammatory responses with enhanced Treg-cell expression of CD39/CD73 and Th-17 cells. Int J Med Microbiol 310(2):151397
Peres RS, Liew FY, Talbot J et al (2015) Low expression of CD39 on regulatory T cells as a biomarker for resistance to methotrexate therapy in rheumatoid arthritis. Proc Natl Acad Sci USA 112:2509–2514
Acknowledgements
Not applicable
Funding
We did not receive any funds for this study.
Author information
Authors and Affiliations
Contributions
NF set the design, collected the data, and performed data analysis. SA collected the data, performed analysis and drafting. NN performed biochemistry analysis and drafting. HA performed biochemistry analysis and drafting. RA performed biochemistry analysis and drafting. SH suggested the study design, performed analysis, interpretation of data shared in drafting. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
The study was implemented in agreement with the ethical principles of Helsinki and was approved by the local Research Ethics Committee of Faculty of Medicine, Tanta University (approval code 32041/03/20). All of the participants gave written informed consent after a full explanation of the study.
Consent for publication
Not applicable
Competing interests
All authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Darwish, N.F., Tabra, S.A.A., Baiomy, N.N. et al. The implication of adenosine receptor expression in prediction of methotrexate clinical response in Egyptian rheumatoid arthritis patients. Egypt Rheumatol Rehabil 49, 5 (2022). https://doi.org/10.1186/s43166-021-00107-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43166-021-00107-9